Abstract
Epithelial–myoepithelial carcinoma (EMC) is a rare tumor that occurs mainly in the major salivary glands. Cases occurring in the nasal cavity are rarely reported. The patient was a 48-year-old woman with a postoperative pathological diagnosis of EMC. The patient recovered well after surgery. We consulted and summarized all previous cases of nasal EMC. We also discuss the clinical presentation, treatment, and prognosis of EMC of the nasal cavity and paranasal sinuses.
Keywords
Introduction
Epithelial–myoepithelial carcinoma (EMC) occurs predominantly in the major salivary glands and rarely in the minor salivary glands of the inferior turbinate. The diagnosis is mainly based on pathology. Adequate resection of the tumor is the main treatment. Close follow-up is required to prevent tumor recurrence and metastasis.
Case presentation
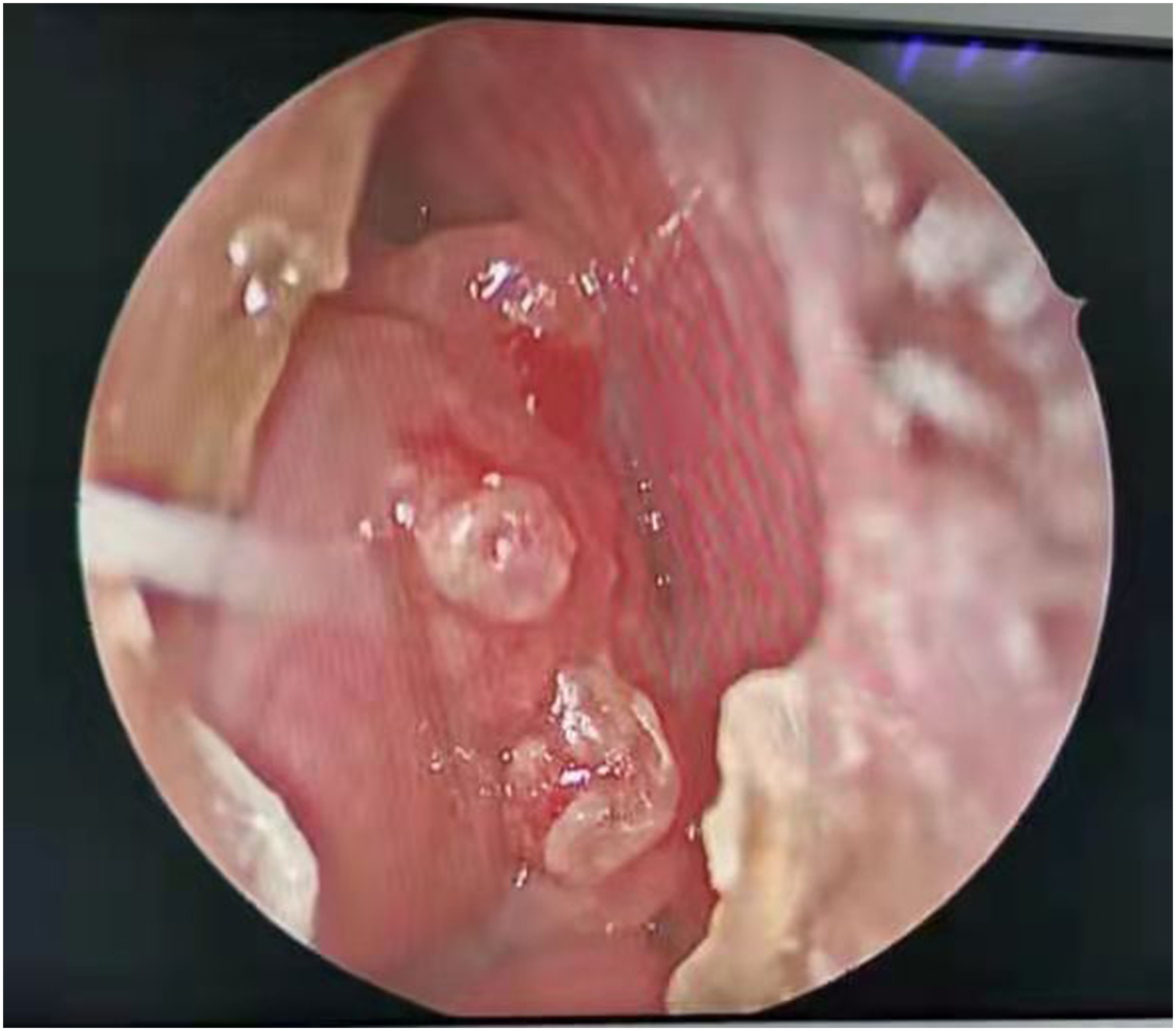
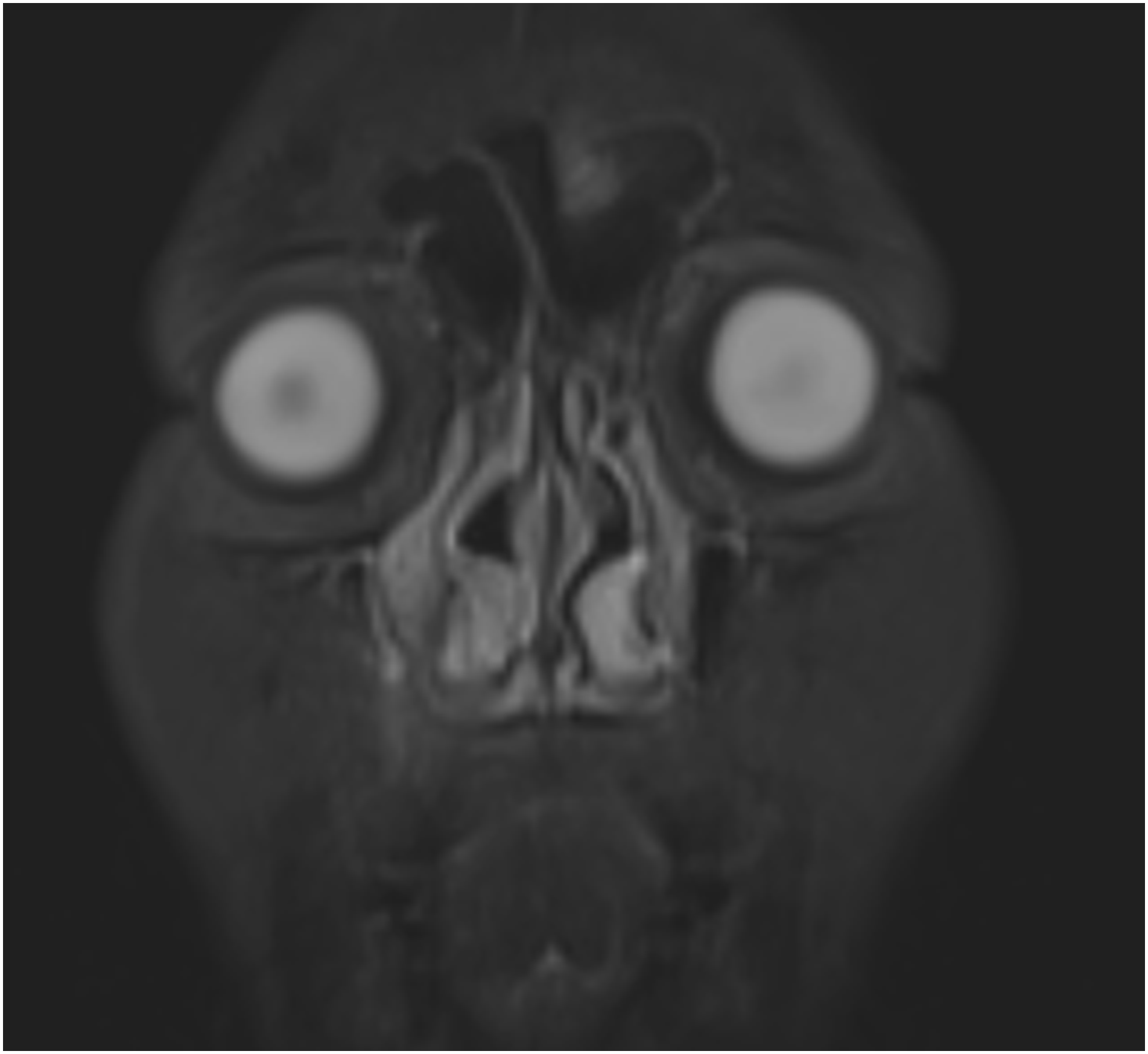
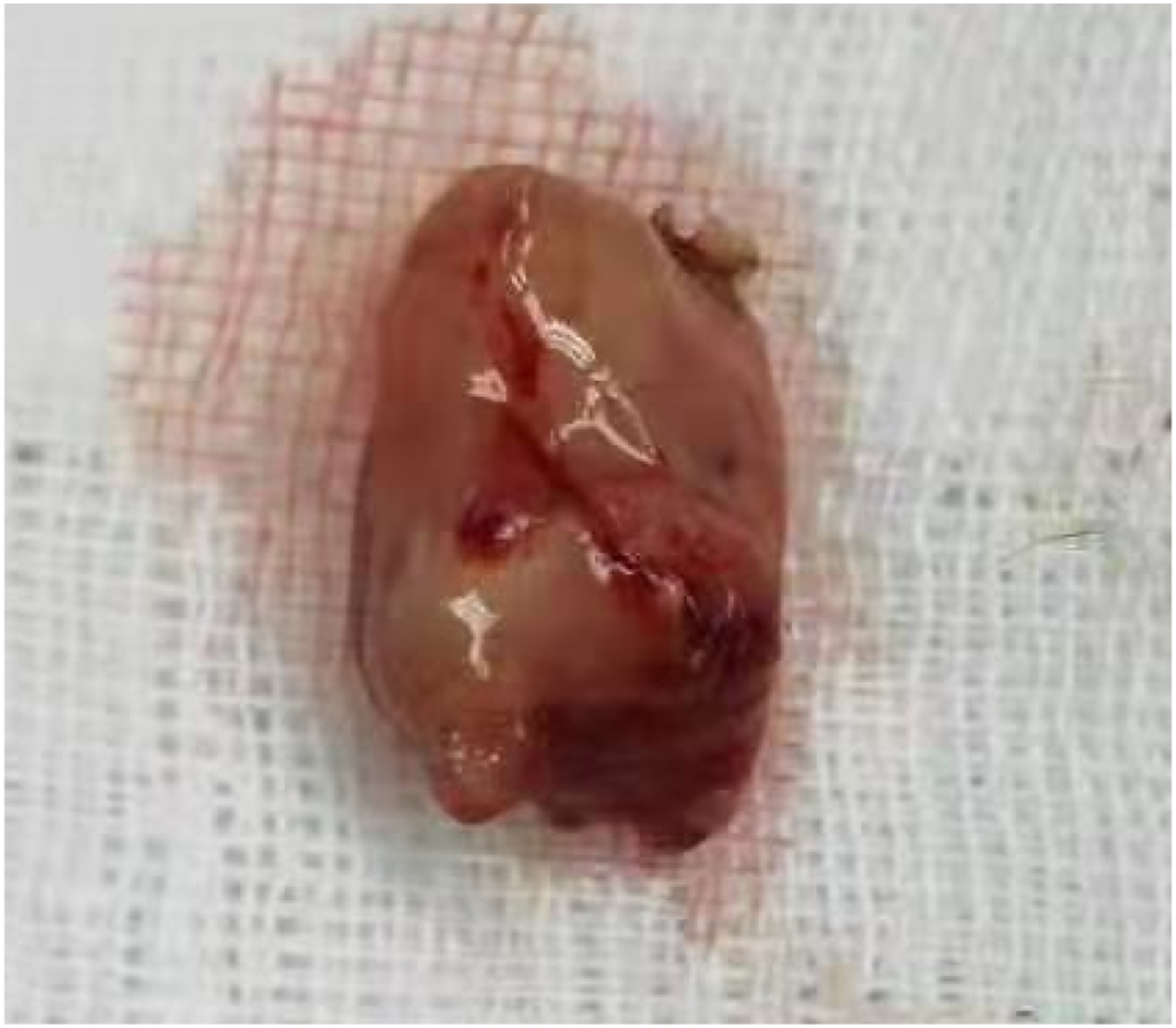
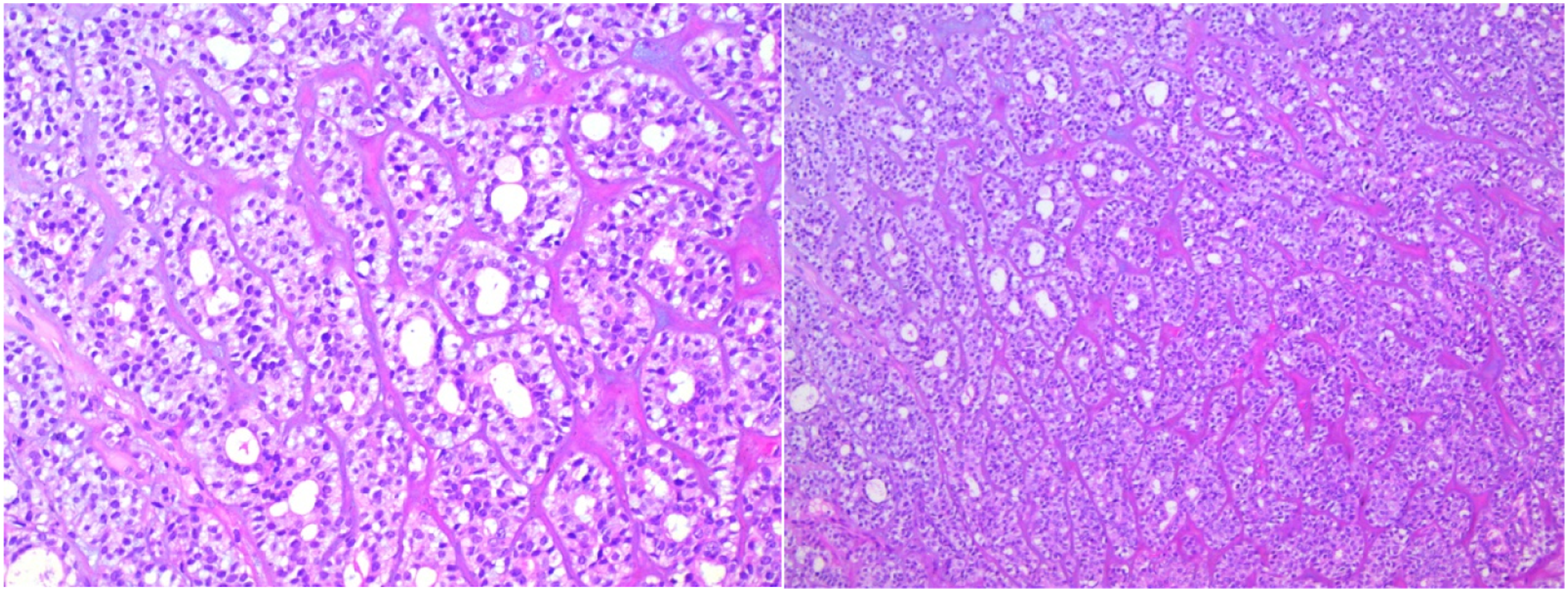
A 48-year-old woman as admitted to our department due to repeated right epistaxis for 4 mo and nasal obstruction for 20 d. Four months ago, the patient had right epistaxis without obvious cause, which stopped spontaneously. Twenty days ago, she developed right nasal obstruction, which was persistent. The right inferior turbinate showed a neoplasm, with a little secretion; the left nasal cavity was smooth; and the middle and inferior turbinates were hypertrophic, without significant nasal septum deviation. No abnormality was found on routine blood test, biochemical examination, electrocardiography, and chest radiography. Computed tomography (CT) (Figure 1) showed normal density of the maxillary, frontal, ethmoid and sphenoid sinuses, no abnormality of sinus wall bone, thickening of middle and inferior turbinate mucosa, and left deviation of the nasal septum. Magnetic resonance imaging (MRI) showed a long T2 signal at the edge of the right maxillary sinus, with moderate enhancement on enhanced scanning (Figure 2). The remaining sinuses showed no abnormalities. The inferior turbinate was enlarged and homogeneously enhanced on contrast-enhanced scans. There was no obvious mass in the nasal cavity. We performed preoperative local pathology. However, the results showed only inflammatory tissue proliferation. Under general anesthesia, the patient underwent resection of the right nasal mass under nasal endoscopy. A neoplasm with an unsmooth surface was seen at the anterior end of the right inferior turbinate (Figures 3 and 4). The diameter was about 1.5 cm. The tumor was resected .5 cm from the safe edge of the tumor. Postoperative pathology showed epithelial–myoepithelial carcinoma (Figure 5). Immunohistochemistry: CK (+), calponin (myoepithelium +), CK5/6 (myoepithelium +), SMA (myoepithelium +), SMA (myoepithelium +), P63 (myoepithelium +), CK7 (+), CK8 (+), S100 (+), and Ki-67 (30% +) CT Magnetic resonance imaging. Nasal endoscopy. Excised tumor. Epithelial-myoepithelial carcinoma (hematoxylin and eosin stain, ×100 (left), ×200 (right).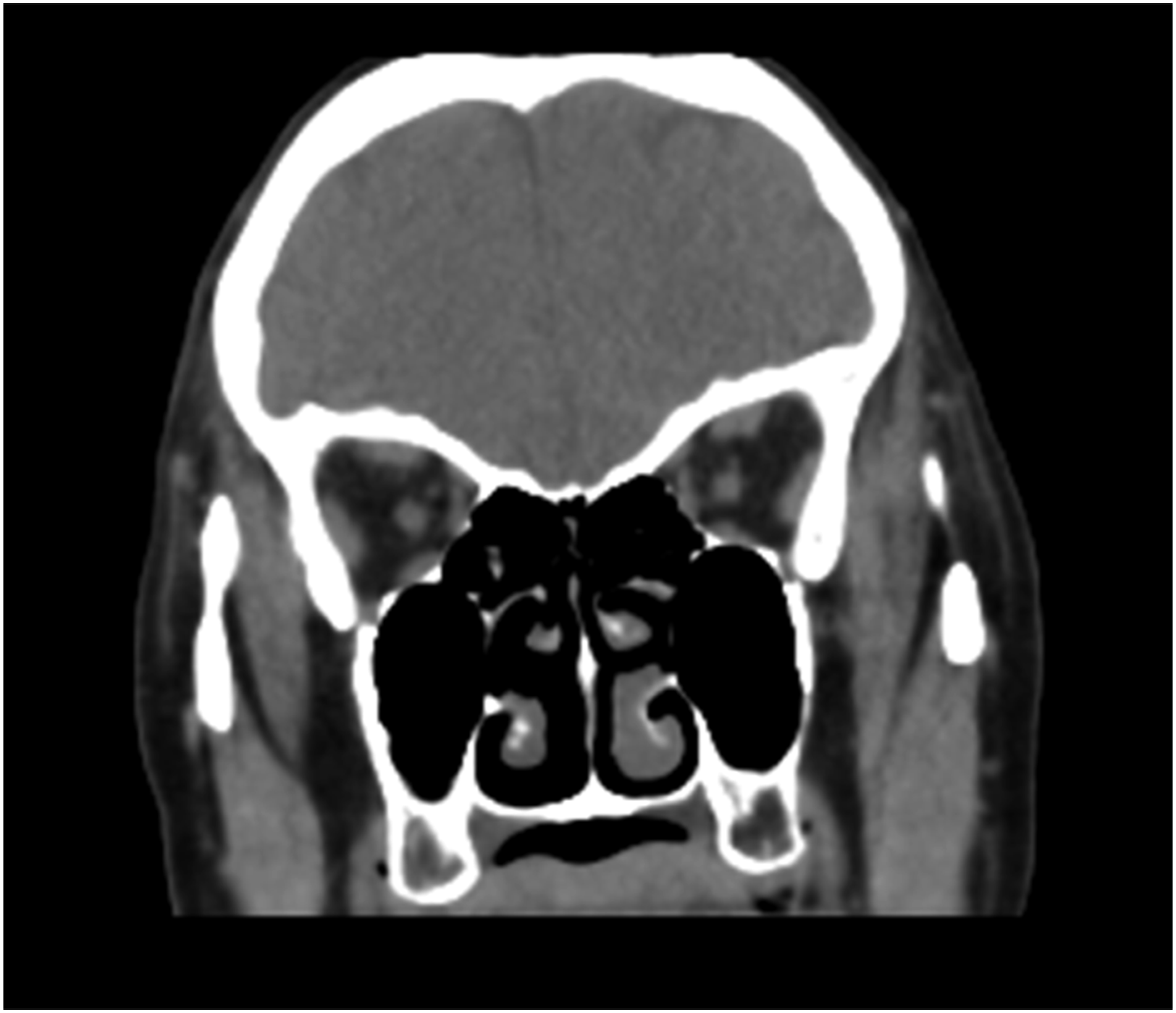
Outcome and follow-up
One week after surgery, the patient was transferred to the Department of Oncology for 6000 cGy adjuvant radiotherapy. The patient had no postoperative nasal obstruction, epistaxis, or other discomfort. Nasal endoscopic re-examination until 6 mo after surgery revealed no significant recurrence.
Discussion
EMC is a rare malignancy. It was included in the World Health Organization (WHO) classification of salivary gland tumors in 1991 and accounts for < 1% of salivary gland tumors. 1 EMC has been reported under different names, such as adenomyoepithelioma, glycogen-rich adenoma, glycogen-rich carcinoma, clear-cell adenoma, and clear-cell carcinoma. 2 EMC is a typical biphasic histological tumor consisting of double-layered arrangement of tubule cells composed of varying proportions of inner intraductal cells and outer myoepithelial cells. 3 It presents 4 different histological patterns, namely, solid, tubular, cribriform, and papillary. 4 Most EMCs arise de novo, but a few arise from malignant transformation of pleomorphic adenomas. 5
Sixteen reported cases of EMC of the nasal cavity and paranasal sinuses.
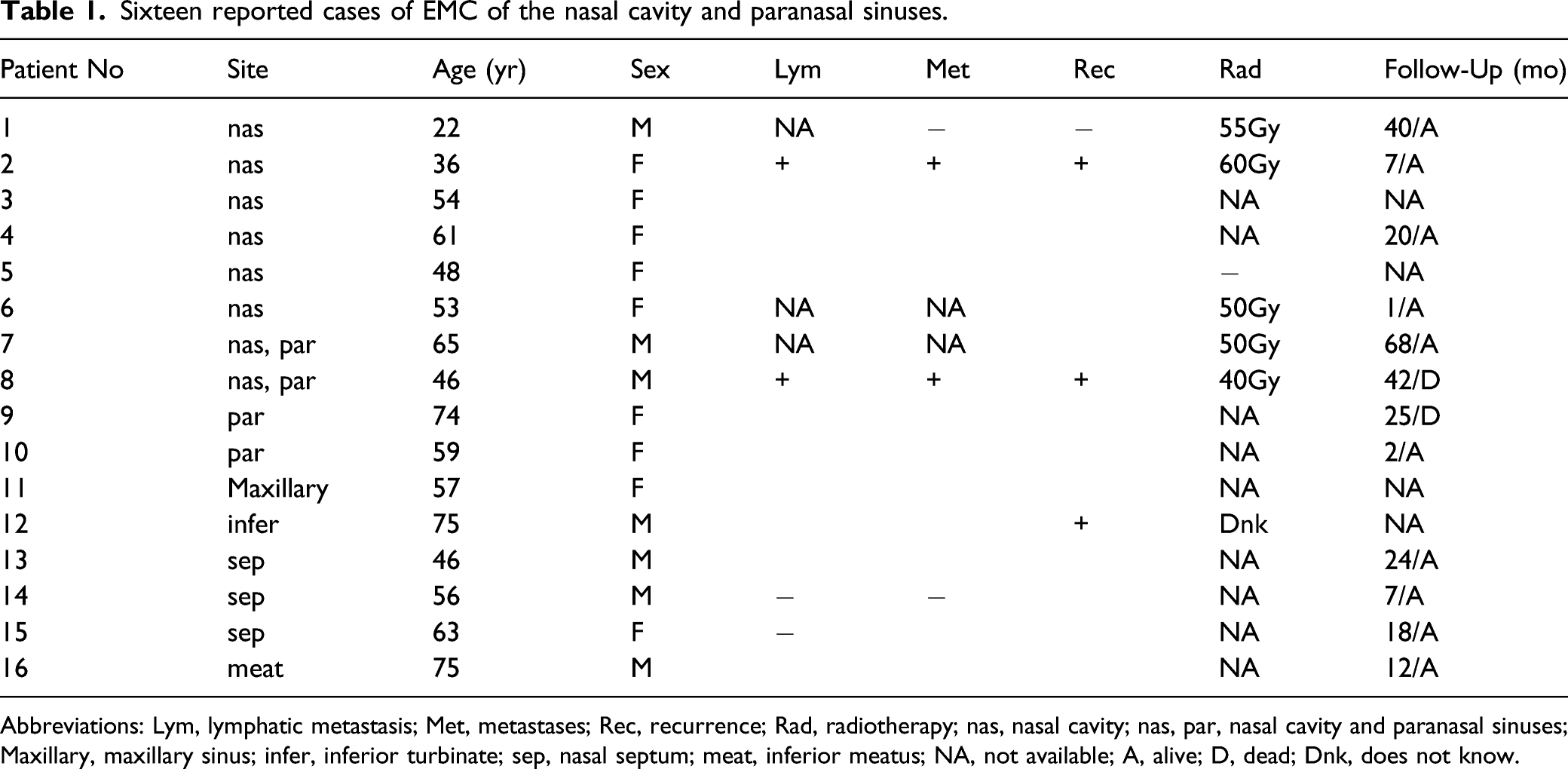
Abbreviations: Lym, lymphatic metastasis; Met, metastases; Rec, recurrence; Rad, radiotherapy; nas, nasal cavity; nas, par, nasal cavity and paranasal sinuses; Maxillary, maxillary sinus; infer, inferior turbinate; sep, nasal septum; meat, inferior meatus; NA, not available; A, alive; D, dead; Dnk, does not know.
The CT findings of EMC in the nasal cavity and paranasal sinuses are nonspecific, and are mainly soft-tissue masses and bone destruction when the tumor enlarges; CT showed worm-like changes in the surrounding bone and compression and absorption and thinning of the bone. Because CT showed soft tissue density shadow, in addition to the fact that the mass grew in the inferior turbinate and the mass was small in this patient, it was easy to mistake the mass for inferior turbinate hypertrophy, and no obvious mass was found by CT and MRI. This demonstrates the advantages of nasal endoscopy for the diagnosis of nasal tumors, including intuitiveness, clarity, convenience, and low cost. Nasal EMC detected by nasal endoscopy is mainly characterized by a smooth surface, no significant ulcer necrosis, friability, and easy bleeding, which has also been described in the literature as a nasal polypoid mass6,8,19 The present case presented as a nasal mass with an uneven surface, ulceration, and the surface albuginea was attached differently from previously reported cases. It is worth noting that some nasal EMCs present as polypoid masses, combined with CT and MRI findings similar to those of soft tissues, which increases the difficulty of diagnosis and it is easily misdiagnosed as nasal polyps. It can be speculated that nasal EMC incidence may be higher than previously reported. There may be patients misdiagnosed with nasal polyps or other diseases, and some patients with nasal polyps may refuse pathological examination, resulting in insufficient surgical resection and insufficient frequency of postoperative re-examination. This may result in shorter survival. Doctors must be more careful if they find polyps in uncommon parts of the nasal cavity, which are hard in consistency and prone to bleeding by touch. Preoperative pathological diagnosis can be made if necessary, which can be performed using fine-needle aspiration, local pathological examination, and intraoperative frozen sections. However, in our case, the preoperative pathological diagnosis also appeared wrong. This may be due to the small size of the nasal mass and unsatisfactory sampling. I think a large enough extent of surgical resection should be better. Some masses are prone to bleeding, so local pathological examination should be undertaken with caution. Fine-needle aspiration is minimally invasive, but cases of misdiagnosis of EMC as adenoid cystic carcinoma using fine-needle aspiration have been reported.20,24
The treatment is based on surgery with extended resection margins, but local recurrence is high at 30%–50%.14,24 There are some different opinions on whether to perform chemoradiotherapy. An EMC survival analysis of 246 patients found no survival benefit in patients who received radiotherapy. 25 A trend toward higher survival after radiotherapy was observed when high tumor grade and tumors > 4 cm were excluded, but this was not statistically significant. 22 Zhang Jiangqian conducted a retrospective survival analysis of 18 patients with EMC, which showed that the 5-year, cumulative recurrence-free survival rate in the adjuvant radiotherapy group was higher than that in the surgery alone group, but there was no significant difference. 26 It has also been reported in the literature that a case of EMC of the nasal cavity and paranasal sinuses, which lost the chance of surgery, achieved a better outcome after receiving an irradiation dose of DT50 Gy. 14 Radiotherapy was performed in 6 of 16 cases collected by our review, and the dosage varied from 40 to 60 Gy. However, due to the limited follow-up time, it was not possible to establish whether radiotherapy was beneficial to survival. However, radiotherapy may be a good option for patients who have lost the chance of surgery or have positive tumor margins. Targeted therapy may be effective for EMC. Mäkelä Rami found that mTOR and MEK inhibitors are potential therapeutic options that can be regarded as alternatives for patients with metastatic EMC. 27 However, this targeted drug therapy was only an in vitro experiment, and clinical studies are needed to verify it.
EMC is considered to be a low-grade malignancy, with only 4.5% of cases showing distant metastasis to the kidney, lungs, brain, and bone marrow.22,25 The overall 5-, 10-, and 20-year survival rates are 72.7%, 59.5%, and 38.3%, respectively. 22 However, some researchers believe that it is a moderately malignant tumor. Fonseca reported that 41% of 22 patients developed metastasis, and 40% of the cases died of tumor-related diseases. 14 Pathological features associated with high recurrence rates are positive surgical margins, intralobular necrosis, loss of capsule (infiltrating margin), > 20% cellular anaplasia, and angiolymphatic origin. 23 Of the 16 cases we reviewed, the distant metastasis rate was 15%, the recurrence rate was 23%, and the distant metastasis rate was slightly higher than previously reported. This may be related to the narrower nasal space and easier occurrence of metastasis, which may also be related to the small number of cases. Factors associated with poor prognosis are older age at diagnosis, race (black survival lower than other races), lymph node or distant metastasis, nonsurgical treatment, mass size > 4 cm, solid growth pattern, high mitotic index, and DNA aneuploidy.7,22 In addition, high-grade translational EMC seems to be more aggressive, with a tendency to lymph node and distant metastasis and a worse prognosis, wider extent of resection, cervical lymph node dissection, and adjuvant radiotherapy should be selected.7,28 Primary tumor site and patient sex are not significantly associated with prognosis. The overall prognosis of EMC is good, but prediction of prognosis needs to consider the different condition of each patient.
Conclusion
We report a rare case of EMC of the inferior turbinate, diagnosed mainly by pathology. We summarized the clinical presentation, treatment, and prognosis of sinonasal EMC.
Footnotes
Author contributions
Li L compiled the patient’s data, reviewed the literature, and wrote the manuscript; Long Y-L was the patient’s surgeons and provided the patient’s information; Wang S-F was responsible for the review and revision of the manuscript; and Su L-L helped perform the analysis with constructive discussions. All authors have given final approval of the submitted version of the manuscript
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Statement of human and animal rights
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
