Abstract
Carcinoma showing thymus-like differentiation (CASTLE) is a rare tumor occurring in the thyroid and soft tissues of the neck. Treatments and prognosis for CASTLE significantly differ from those associated with squamous cell carcinoma and anaplastic thyroid carcinoma. To date, fewer than 100 cases have been reported. Given the lack of a standardized treatment protocol and the diagnostic complexity, documenting these cases is essential. A 74-year-old male patient was admitted to the hospital due to hoarseness lasting for 1 month and thyroid nodules for over 2 weeks. The ultrasound revealed a grade 4 nodule in the left thyroid, and the needle biopsy reported the presence of glandular epithelial cells of varying sizes, some arranged in a papillary pattern, and was negative for BRAF mutation. Thyroid surgeons performed a left partial thyroidectomy and a left cervical lymph node biopsy, and the immunohistochemical staining led to a diagnosis of intrathyroid thymus carcinoma. Follow-up on his postoperative recovery shown no signs of local recurrence or distant metastasis during this period. This paper conducts a systematic review of the literature to enhance understanding, diagnosis, treatment, and prognosis of this condition. The goal was to raise clinical awareness and guide appropriate treatment strategies.
Keywords
Introduction
Carcinoma showing thymus-like differentiation (CASTLE) is a rare tumor found in the thyroid gland and soft tissues of the neck. 1 It was first described by Miyauchi 2 and Chan, 3 who coined the term “CASTLE” to suggest its potential origin from ectopic thymic tissue or residual branchial pouch remnants within the thyroid gland. The tumor retains the differentiation capabilities of the thymus, typically occurring at the lower pole of the thyroid gland. In 2004, the World Health Organization recognized CASTLE as having distinct clinicopathological features, classifying it as a type of thyroid cancer. 4 Despite its typically-slow growth, reports of invasion into adjacent soft tissues, metastasis to nearby lymph nodes, and local recurrence exist. 5 CD5 staining of the tumor is a critical marker for differential diagnosis. For surgical management, total thyroidectomy combined with selective neck dissection is recommended, 6 and radiotherapy is advisable for patients with positive cervical lymph nodes.
Case Report
A 74-year-old male patient was admitted to the hospital due to “hoarseness lasting for 1 month and thyroid nodules for over two weeks.” On examination, a palpable mass measuring 6 × 4 cm was identified in the left thyroid gland, described as firm and moving with swallowing. The patient had a history of well-controlled hypertension but denied any prior treatment or family history of thyroid disorders.
His medical history included a Thyroid Color Doppler ultrasound, which showed nonuniform echoes in both thyroid glands and suggested further examination of thyroid function. The ultrasound revealed a nodule in the right thyroid gland classified as Thyroid Imaging Reporting and Data System (TIRADS) grade 3, and a grade 4 nodule in the left thyroid. The needle biopsy reported the presence of glandular epithelial cells of varying sizes, some arranged in a papillary pattern, and was negative for BRAF mutation. An enlarged left lateral cervical lymph node with abnormal morphology was also noted, prompting an ultrasound-guided puncture for further evaluation.
Upon admission, enhanced preoperative examinations were conducted. A thyroid computed tomography (CT) scan, both unenhanced and contrast-enhanced, suggested a high probability of left thyroid cancer, accompanied by bilateral enlargement of multiple lymph nodes (Figure 1). The anteroposterior and lateral views of the cervical spine (tracheal M-V film) showed that the trachea was compressed and shifted to the right. Contrast-enhanced magnetic resonance imaging of the thyroid displayed a mass in both the left and right lobes (with greater prominence in the left lobe), leading to a preliminary diagnosis of papillary thyroid carcinoma with bilateral cervical lymph node metastases. Additionally, the left lateral wall of the trachea was compressed and slightly displaced to the right. A chest CT showed no abnormalities, and the serum concentrations of thyroid-stimulating hormone (TSH), free thyroxine (FT4), thyroglobulin (TG), calcitonin, and antithyroid peroxidase antibodies (ATPO) were all within normal ranges.
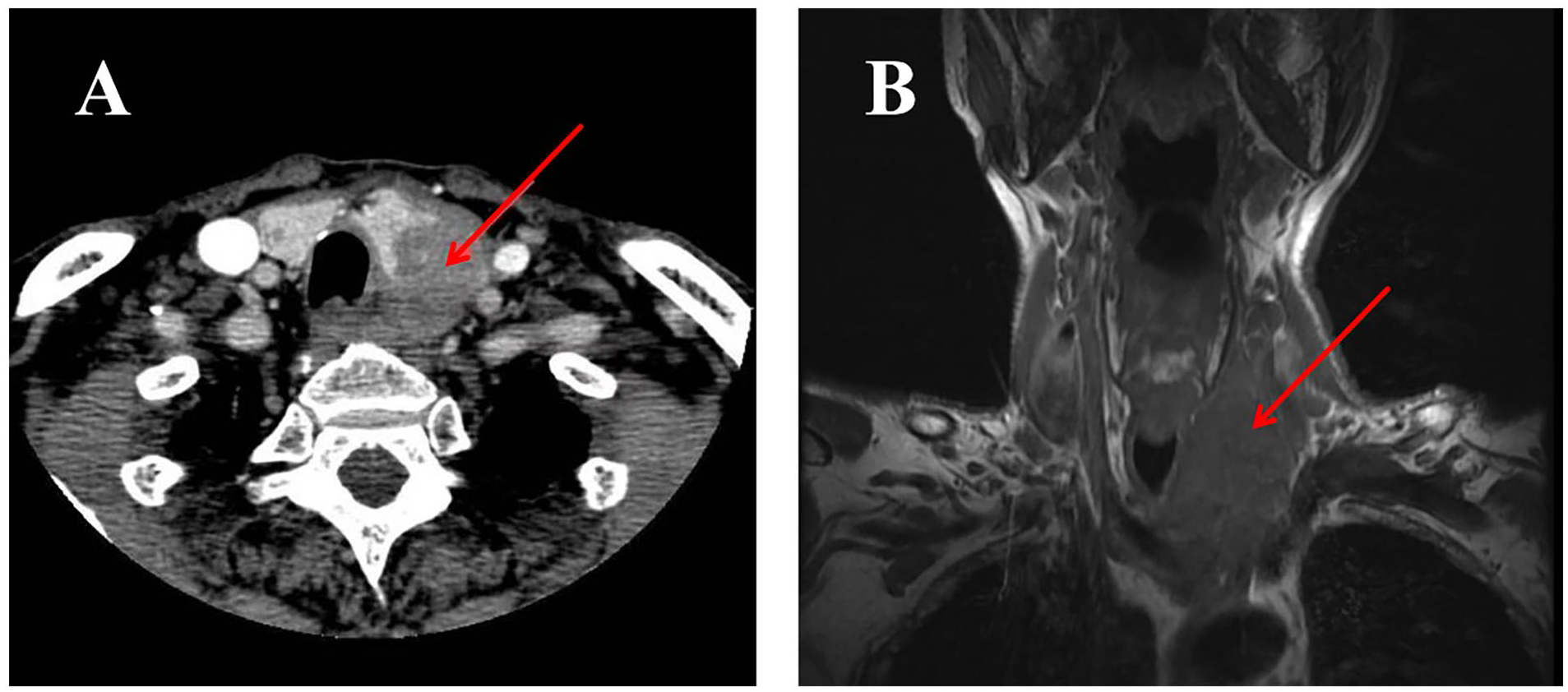
(A) Contrast-enhanced neck computed tomography scan showing a hypodense mass in the thyroid gland, with irregular borders, compressing the trachea. (B) Head and neck MRI showed the inferior pole of the mass extends into the superior mediastinum (arrow).
Thyroid surgeons performed a left partial thyroidectomy and a left cervical lymph node biopsy. The surgery proceeded without complications. During the procedure, the left thyroid nodule was found to have penetrated its capsule, invaded the surrounding soft tissue, and was adherent to the left recurrent laryngeal nerve and the trachea. Given the tumor’s locally-advanced stage, a rapid pathological examination identified malignant cells in some left thyroid nodules. Cancer metastasis was detected in 3 of 4 lymph nodes in the third region on the left side, pending confirmation by immunohistochemistry. Routine pathology postsurgery revealed a poorly-differentiated carcinoma with thymus-like differentiation in the left thyroid lobe (Figure 2), classified as intrathyroid thymic carcinoma according to the 2017 WHO classification. Subsequent immunohistochemical staining showed positive reactions for CD5, CD117, and P63 (Figure 3), leading to a diagnosis of intrathyroid thymus carcinoma. Unfortunately, the patient and his family declined further treatment for financial reasons. Follow-up on his postoperative recovery has been ongoing, and he has shown no signs of local recurrence or distant metastasis during this period.
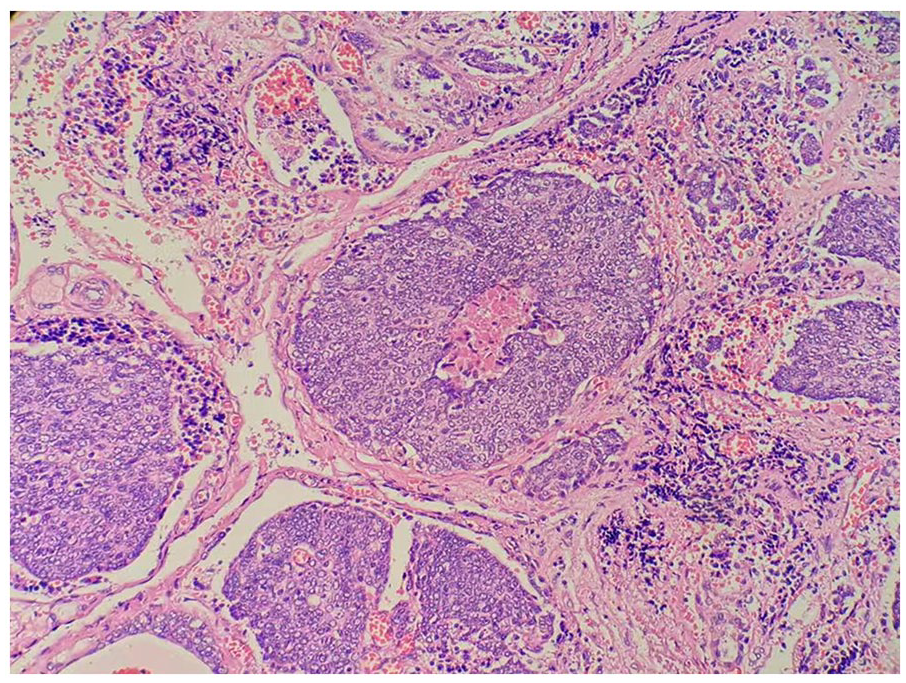
Histologic features of carcinoma showing thymus-like differentiation (×200).
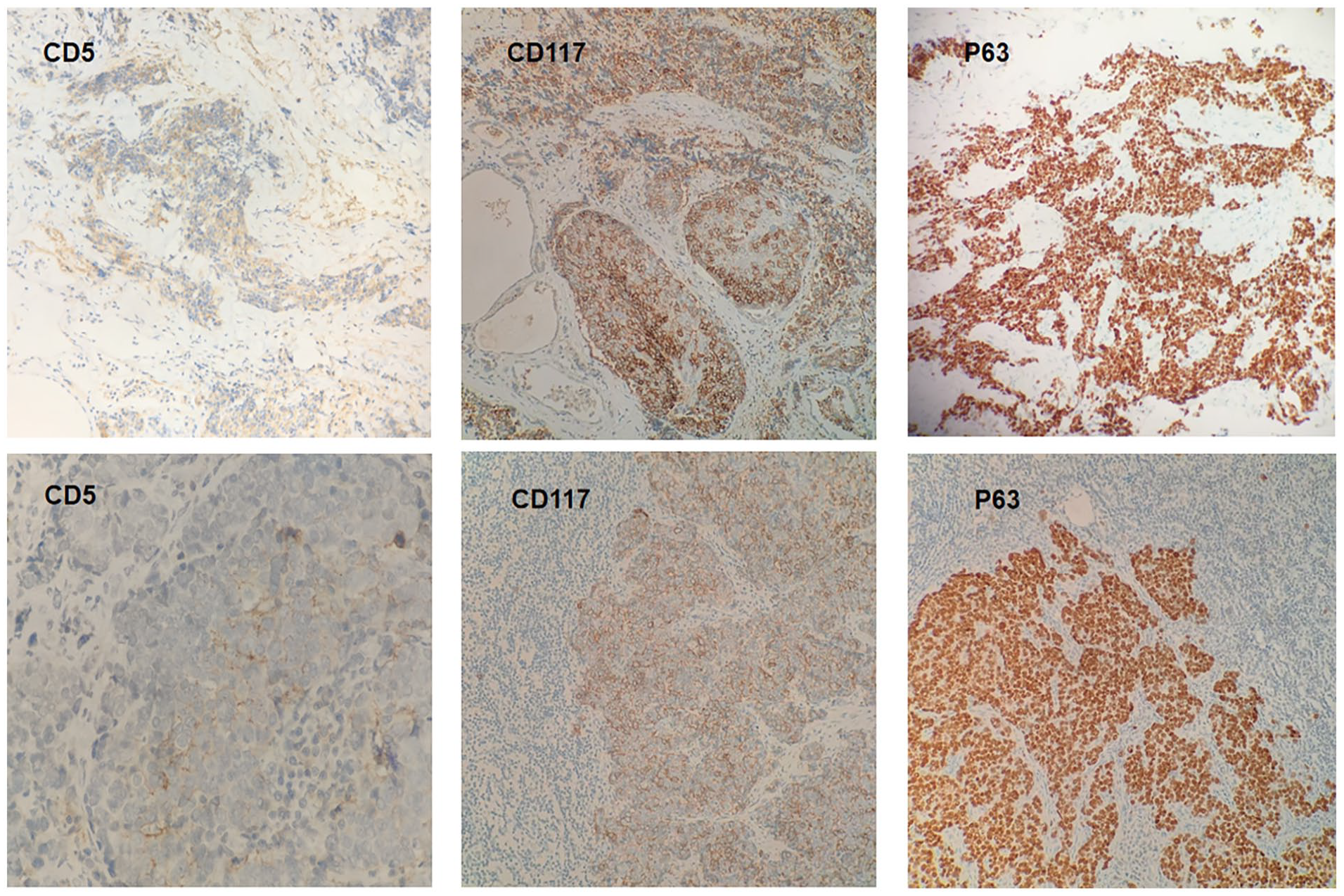
Subsequent immunohistochemical staining demonstrated positivity for CD5, CD117, and tumor protein (P63); the upper 3 images are of thyroid tissues and the lower 3 of lymph node tissues (×100). CD, cluster of differentiation.
Discussion
In the literature, CASTLE is more commonly observed in middle-aged individuals and exhibits a slight predominance in women compared with that in men (M:F = 1:3). 7 The current observations suggest that the tumor may originate from ectopic thymic tissue or residual branchial pouch remnants within the thyroid gland. 3 It primarily manifests in the lower pole of the thyroid gland 8 and is classified as an extrathymic tumor in the neck, typically along the embryonic migration path of the thymus, extending from the mandibular angle to the retrosternal region. These tumors exhibit differentiation resembling the thymus, attributed to the presence of thymic epithelial cells that form keratin pearls. 9 Although the lesions predominantly occur in the thyroid, cases have been reported in extrathyroidal locations such as the lateral trachea 10 and parotid gland. 11 Patients typically present with a painless goiter, followed by symptoms of tracheal compression such as hoarseness and other related symptoms. 12 The clinical manifestations in our patient were consistent with those reported in the literature.
Through a series of literature studies, Wu B noted that CASTLE does not exhibit significant distinguishing features from thyroid nodules on CT or MRI scans, except for its unique location in the lower neck—usually situated between the lower pole of the thyroid gland and the upper mediastinum. 13 Fine needle aspiration (FNA) biopsy plays an important role in the early diagnosis of isolated thyroid nodules. However, it is not highly specific for CASTLE. A retrospective analysis by Dong et al 14 of 6 patients diagnosed with CASTLE indicated that preoperative ultrasound, CT scans, and FNA biopsy were insufficient for diagnosis. Instead, a definitive diagnosis required pathological examination and immunohistochemistry, particularly for CD5-positive staining. CASTLE typically shows a strong positive reaction for CD5, P63, and CD117.15,16 The sensitivity and specificity of CD5 for CASTLE diagnosis were found to be 82% and 100%, respectively, and importantly, CD5 is negative in other head and neck tumors such as squamous cell carcinoma, poorly-differentiated carcinoma, follicular carcinoma, and most papillary thyroid carcinoma. Additionally, Hassle’s corpuscle, which are unique to the thymus medulla, are often identified in CASTLE tumors and serve as a histological marker that confirms the carcinoma’s thymic differentiation. 17
The distinction between CASTLE and other head and neck malignancies lies in their treatment and prognosis. CASTLE is potentially invasive, capable of invading the soft tissues around the neck, including muscles, trachea, esophagus, and nerve, necessitating procedures such as combined tracheotomy or laryngotracheal resection.2,5,14,18,19 While there is no established gold standard for CASTLE treatment, systematic reviews indicate that the prognosis is generally favorable with indolent behavior and a long survival time, although local recurrence is common. Clinical reports suggest that the disease can also exhibit more aggressive behavior, leading to distant metastasis.20-22 Most surgeons prefer surgical resection, with or without adjuvant radiation or chemotherapy.3,15,16
A retrospective analysis by Wei Ge involving 82 CASTLE patients found that postoperative radiotherapy did not reduce the recurrence rate. 1 Case reports also suggest that conservative treatment may be an acceptable option for patients experiencing recurrence after total resection. 23 However, Kovářová 24 posited that due to the radiosensitivity of the tumor, systematic adjuvant radiotherapy could be beneficial, as evidenced by cases where patients received only radical radiotherapy and achieved complete remission. 25 Patients without lymph node metastasis still face a potential for local recurrence. Therefore, for those with lymph node involvement, adjuvant radiotherapy is recommended after surgical treatment. 14 Tsutsui reported a case where a patient, who refused surgery and received only radiation therapy, showed no signs of local recurrence for at least 7 years. However, 10 years post-radiotherapy, the patient died of an unresectable recurrent tumor, indicating that while the tumor may respond to radiotherapy, it can still exhibit aggressive characteristics. 26
Complete surgical resection is crucial for achieving local control of the disease and ensuring long-term survival. 27 Since CASTLE tumors do not originate from thyroid follicular cells, radioiodine suppression therapy is unsuitable. 28 Chemotherapy is typically reserved for patients with metastatic or unresectable CASTLE tumors. 29 Moreover, Lin et al 30 performed whole exome sequencing on patients diagnosed with CASTLE, identifying unique tumor-driving gene mutations that may inform potential therapeutic pathways. Nonetheless, the efficacy of adjuvant or palliative chemotherapy remains uncertain due to limited data.
Conclusions
In summary, complete tumor removal significantly improves the prognosis for patients with CASTLE compared to other more invasive thyroid malignancies. Although presurgical diagnosis of CASTLE is challenging, CD5-positive immunologic staining serves as a useful diagnostic indicator. The most effective treatment involves complete excision of the lesion and assessing the necessity for lymph node dissection. Additionally, the potential need for adjuvant radiotherapy should be considered. The efficacy of chemotherapy has not been definitively reported, and close follow-up is essential for managing this condition.
Footnotes
Acknowledgements
The authors thank the patients and their families for their assistance, as well as all clinical departments.
Author’s Note
We declare that this manuscript is original, has not been published before, and is not currently being considered for publication elsewhere. We confirm that the manuscript has been read and approved by all named authors and that there are no other persons who satisfied the criteria for authorship but are not listed. We further confirm that the order of authors listed in the manuscript has been approved by all of us. We understand that the corresponding author is the sole contact for the editorial process. He is responsible for communicating with the other authors about progress, submissions of revisions, and final approval of proofs.
