Abstract
Atrophic rhinitis (AR) is a chronic nasal disease characterized by atrophy of the nasal mucosa and turbinates. Occasionally, nasal myiasis complicates AR. This case illustrates an uncommon complication of nasal myiasis, palatal perforation. A 35-year-old South Asian woman presented with recurrent bilateral epistaxis and expulsion of maggots from the nose. She had a history of nasal obstruction, foul-smelling nasal discharge, and anosmia. On examination, the patient had atrophied nasal turbinates and later developed 2 perforations in the palate. She was offered conservative management for AR. Besides, she underwent manual and endoscopic removal of maggots in multiple settings and was isolated in a mosquito net to prevent reinfestation. Nasal myiasis can complicate AR in tropical regions. Early diagnosis of AR and simple prophylactic measures, such as nasal douching and use of mosquito nets while sleeping, can prevent complications such as palatal perforation and intracranial extension caused by nasal myiasis.
Background
Atrophic rhinitis (AR) is a chronic nasal disease characterized by atrophy of the nasal mucosa and turbinates. It leads to thick greenish-black crusts over nasal mucosa. 1 AR-induced mucosal changes create favorable conditions for fly larvae growth. Thus, nasal myiasis can complicate AR uncommonly. 2 Larval activity in the nasal cavity presents as epistaxis, mucopurulent nasal discharge, malodor, anosmia, and discomfort in the nasal and facial areas.3,4 Maggots can burrow into deeper tissues, destroying deeper structures such as the stomatal complex, medial and lateral walls of the nasal cavity, the tympanic membrane, and palate.5,6 Severe cases can lead to intracranial or intraorbital extension. 4 Therefore, prompt removal of maggots is essential to prevent complications and ensure a favorable outcome.
This article presents an unusual complication of nasal myiasis, palatal perforation, in a patient with AR. Furthermore, we discuss the management of and preventive measures for AR and nasal myiasis, as there is no general consensus on the management of these diseases.
Case Description
A 35-year-old South Asian farmer presented with recurrent bilateral epistaxis for 3 days and expulsion of maggots from the nose for 1 day. She had a history of bilateral nasal obstruction, yellowish nasal discharge, anosmia, and headache, and her family members reported foul-smelling breath. The patient neither had any chronic medical conditions such as diabetes mellitus, hypertension, or systemic lupus erythematosus (SLE), nor had any history of tropical infections such as tuberculosis or leprosy. She was a nonsmoker and did not consume alcohol. In addition, there was no family history of AR or other chronic diseases.
On examination, the patient appeared cachectic and pallid, which is indicative of heavy epistaxis and nutritional deficiency. Her nasal cavities were spacious, with atrophied bilateral middle and inferior turbinates and active bleeding from the nasal mucosa. Maggots were swarming over the nasal lining and burrowing into deeper structures. She had poor oral hygiene with dental crowding and had a red bulge measuring 0.5 cm in diameter on the hard palate. Within 24 hours, the bulge progressed to a perforation of 0.6 cm × 0.6 cm in the hard palate, with a new perforation of 1.2 cm × 0.7 cm in the soft palate (Image 1). Subsequently, the patient experienced difficulty with mastication, nasal regurgitation of food and fluids, and speech disturbances. The otology and laryngology examinations were unremarkable.
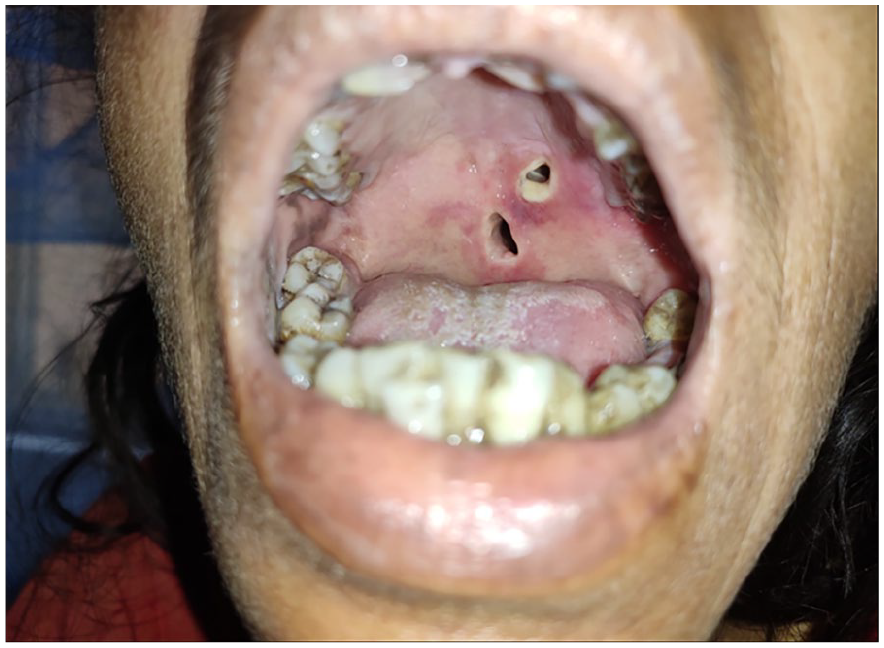
Perforation on the hard palate (0.6 cm × 0.6 cm) and soft palate (1.2 cm × 0.7 cm) and poor oral hygiene.
The endoscopic evaluation confirmed multiple maggots in the nasal cavity (Image 2). Due to financial constraints, an imaging of the nose and paranasal sinuses was not performed. The patient was diagnosed with palatal perforation caused by nasal myiasis secondary to AR. She was admitted to an isolation ward, placed inside of a mosquito net to prevent reinfestation, and treated with intravenous antibiotics and a single dose of oral albendazole. Furthermore, turpentine oil (4-5 drops) was administered nasally every 2 hours, along with a few drops of 25% dextrose-glycerin solution every 4 hours. Alkaline nasal douching (with solution made up of 28.4 g of sodium bicarbonate, 28.4 g of sodium diborate—an antiseptic and buffers—56.7 g of sodium chloride—maintains osmolarity of solution—and 280 ml of warm water) was performed twice daily. Over 5 days, approximately 90 to 100 live maggots were removed by endoscope and manually. Iron and vitamin supplements were prescribed.
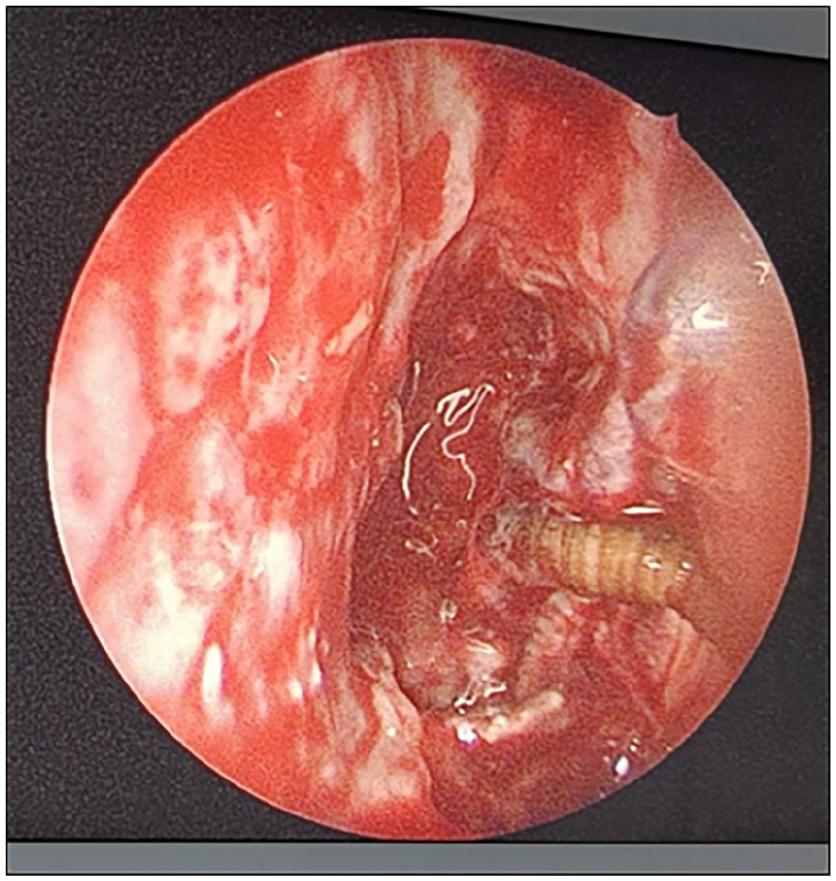
Image showing maggot on nasal endoscopy.
The patient improved gradually over a week. She was discharged after 2 days without maggots with oral medications and instructions for daily nasal douching. She was encouraged to sleep under mosquito nets to prevent reinfestation. Although she was advised to follow up for evaluation of AR and closure of the oroantral fistula, she did not follow up.
Discussion
AR is a chronic condition characterized by gradual degeneration of the nasal mucosa and turbinates, with symptoms including nasal blockage, crusting, unpleasant breath, and occasional nosebleeds. AR can be primary or secondary. Both forms share clinical characteristics but differ in their etiological factors and underlying pathogenesis. Primary AR is idiopathic, commonly seen in females and residents of tropical and subtropical regions, and is often associated with nutritional deficiencies and poor socioeconomic conditions. Secondary AR occurs as a result of underlying conditions, such as infections (eg, syphilis, leprosy, tuberculosis), trauma, previous surgeries, or chronic granulomatous diseases.1,7 In this case, no definitive risk factor for secondary AR was identified, suggesting AR of primary origin, probably due to nutritional deficiency.
Several studies report an association between AR and nasal myiasis.4,8 AR-induced mucosal changes create conditions favorable for the growth of fly larvae, with nasal crusting and atrophy providing a feeding substrate. Altered anatomy and impaired mucociliary clearance in patients with AR further facilitate larvae retention.7,8 Therefore, timely identification and management of AR can prevent complication of nasal myiasis.
Nasal myiasis presents as epistaxis, mucopurulent nasal discharge, malodor, anosmia, discomfort in the nasal and facial areas, and expulsion of maggots.3,4 Maggots may burrow into deeper tissues, destroying deeper structures such as the stomatal complex, medial and lateral walls of the nasal cavity, the tympanic membrane, and the palate. Additionally, severe cases can lead to intracranial or intraorbital extension and even death.2,4-6
The passage of maggots through the nostrils with corroborative symptoms and/or visualization of the parasite in the nasal cavity confirms the diagnosis of nasal myiasis. When maggots burrow in the mucosa, granulation tissue, or debris, nasal endoscopy aids in visualization and extraction. Further details of nasal endoscopy in the management of nasal myiasis are discussed in a later paragraph. Imaging, especially computed tomography scans, helps to determine maggot location, extent of tissue damage, and surgical needs. Identification of maggot species can be performed by microscopic examination, and pupa culture can be performed. 3 In our case, the insect species was not identified due to the inaccessibility to an entomology laboratory.
AR can be managed with a conservative and/or surgical approach. Conservative measures include maintaining proper hygiene, consumption of a balanced diet, prompt detection and treatment of nasal abnormalities, alkaline nasal douching, 25% glucose-glycerin nasal drops, and nasal submucosal injections. Additionally, according to the nasal culture of purulent discharge, systemic antibiotics, such as tetracycline, aminoglycosides, and ciprofloxacin, should be directed from time to time. However, due to compliance problems, patients generally have a relapse of the disease. In such cases, surgical interventions (such as Young’s and the modified Young’s operation) reduce the size of the nasal cavity to retain moisture and regenerate mucosa. 1
Currently, there is no consensus on a treatment standard for nasal myiasis. However, the primary objective in the treatment of nasal myiasis is to minimize tissue damage by ensuring complete removal of maggots, achieved by manual and/or endoscopic approaches. Various agents, such as turpentine oil, a chloroform-turpentine oil mixture (1:4 ratio), or liquid paraffin, can be applied to the nasal cavity before manual extraction.5,6 Turpentine oil acts as an irritant, driving maggots to migrate from deeper tissues, while liquid paraffin creates an anaerobic environment that induces larval death. 5 Victoria et al proposed the usage of topical ivermectin for the treatment of nasal myiasis. Following topical ivermectin, there was immediate decrease in pain and parasite death of parasites within 24 hours. 9 Additionally, the administration of oral antiparasitic agents, such as ivermectin and pyrantel pamoate, is presented in multiple case reports/series.10,11
With advances in medical technology, the endoscopic approach of maggots removal has become increasingly favored over manual methods. Endoscopy offers several advantages: It is quicker, requires fewer sessions, allows for precise removal of maggots from otherwise inaccessible areas, is less traumatic, and effectively reduces the risk of complications. 12 However, its widespread adoption is often limited by financial and technical constraints.
In our case, financial limitations precluded the repeated use of endoscopic procedures for maggot extraction. Consequently, a combined approach was adopted. An initial endoscopic procedure was performed to extract a significant number of maggots. This was followed by 1 to 2 sessions per day of manual removal, supplemented with occlusion of the nasal cavity with gauze soaked in turpentine oil to facilitate the extraction of maggots. This combined approach aims to achieve effective management while addressing resource limitations.
The treatment of nasal myiasis in patients with AR is incomplete without addressing the prevention of recurrence. Due to the altered nasal anatomy and the high risk of fly infestation associated with AR, there is a significant likelihood of recurrence. Therefore, patients should prioritize proper nasal hygiene, use mosquito nets, maintain cleanliness in their surroundings, implement fly control measures, and adhere to basic sanitation practices. We strongly recommend the use of mosquito nets, as they protect against infestation during the most susceptible time, while sleeping. Additionally, using mosquito nets is one of the most affordable and plausible methods to prevent nasal myiasis, which particularly affects people of lower socioeconomic status.
The palatal fistula is an abnormal opening between the nasal and oral cavities, occurring in the hard palate, soft palate, or uvula. It may be congenital or caused by trauma, infections (syphilis, leprosy, tuberculosis), collagen vascular diseases (SLE, Wegener’s granulomatosis), granulomatous diseases (Sarcoidosis, Crohn’s), neoplasia, or drug abuse (cocaine, heroin). 13 In nasal myiasis, as in the above case, maggots may invade the palate, forming an oronasal fistula. These fistulas may be asymptomatic or may cause nasal regurgitation, hypernasal resonance, malodor, and poor oral hygiene. 14 Treatment depends on location and size, with options including prostheses or surgical reconstruction using flaps such as the tongue, buccal, temporalis, or free flaps. 15
Conclusions
Nasal myiasis can complicate AR in tropical regions. Therefore, early diagnosis of AR and simple prophylactic measures, such as nasal douching and the use of mosquito nets while sleeping, can prevent complications such as palatal perforation and intracranial extension caused by nasal myiasis.
Footnotes
Acknowledgements
None.
Author Contributions
D.P. conceptualized the study, was associated with patient care, and reviewed the report. S.P. and A.A. performed background research, reviewed literature, and prepared the manuscript. K.N.S. was associated with patient care.
Availability of Data and Materials
No data sets were generated or analyzed during the study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval and Consent of the Patient
The patient and the patient party were explained about the use of their clinical scenario and images in a depersonalized form, for report writing and publication purposes, both verbally and followed by obtaining written consent for the same.
Consent for Publication
Written consent obtained from the patient covered consent for publication.
