Abstract
Keywords
Introduction
Adult vascular anomalies differ from those in infants in that they are less likely to resolve spontaneously. Approximately 60% of all vascular anomalies occur in the head and neck region; however, adult hypopharyngeal vascular anomalies (AHVAs) are extremely rare. The first documented case of AHVA was reported by Hayden in 1925. 1 There have been limited publications on these lesions, and all existing articles focusing on AHVAs are case reports. Currently, there is a dearth of dedicated research studies specifically examining AHVAs.2-7
The International Society for the Study of Vascular Anomalies (ISSVA) classifies vascular anomalies into 2 groups based on the classification of Mulliken and Glowacki. Lesions characterized by abnormal cell proliferation are termed “vascular tumors,” while relatively stable anomalies are classified as “vascular malformations.” This classification system provides a framework for distinguishing between these 2 types of vascular anomalies. 8
Due to the rarity of AHVAs, systematic studies are currently lacking. Various treatment methods have been suggested for adult AHVAs, including carbon dioxide (CO2) lasers, low-temperature plasma, ultrasonic scalpels, neodymium-doped yttrium aluminum garnet (Nd:YAG) lasers, and potassium-titanyl-phosphate (KTP) lasers, as reported in previous studies.3-7 However, it remains unclear which laser modalities are most suitable for AHVAs. Furthermore, for AHVAs of varying shapes and sizes, the optimal surgical methods and laser treatment options have not been clearly defined. The treatment of AHVAs lacks systematic investigation, and effective surgical approaches for these anomalies remain uncertain. This study aims to evaluate treatment methods for AHVAs by reviewing the outcomes of patients affected by these anomalies.
Patients and Methods
The study received approval from the research ethics board of our hospital, and the requirement for informed consent was waived due to its retrospective nature. The inclusion criteria were as follows: (1) outpatient diagnosis of AHVAs based on laryngoscopy and enhanced CT or MRI findings, with confirmation through postoperative pathology when available; (2) patients aged 18 years or older; (3) surgical procedures involving either bleomycin injection or excision surgery; (4) availability of pre-and postoperative laryngoscopy results. In cases where patients underwent multiple surgical procedures, each surgery and its perioperative data were treated as separate instances for inclusion in the study. One patient was excluded due to the lack of postoperative follow-up data. From September 2013 to November 2022, our department admitted a total of 23 adult patients who underwent 30 distinct procedures.
Certain patients received multiple treatments. The diagnosis of AHVAs was based on the patient's medical history, laryngoscopic examination, and imaging findings, with enhanced computed tomography or magnetic resonance imaging utilized for evaluation. Postoperative pathology, when accessible, was employed to confirm the diagnosis.
Classification
Based on enhanced CT or MRI results, AHVAs were categorized into 2 groups: vascular tumors and vascular malformations. In some cases, pathological diagnosis was also used to differentiate between these 2 categories. In our study, all vascular tumors were classified as hemangiomas according to enhanced CT, MRI, and pathological diagnoses.
Selection of Surgical Procedures
Before surgery, patients underwent a comprehensive otolaryngological evaluation that included assessments such as chest radiography, renal function tests, electrocardiograms, blood glucose levels, and routine clinical examinations. These evaluations were conducted to rule out any concurrent systemic diseases. We have initially chosen the surgical method according to Figure 1.
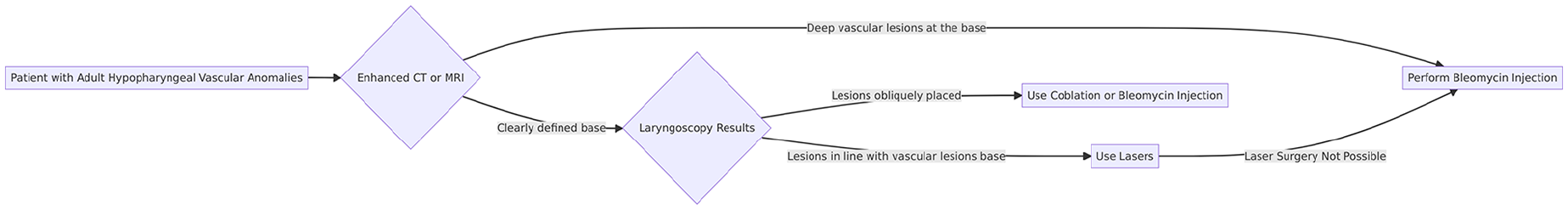
Flowchart for surgical method selection for AHVAs.
The cases were divided into 2 groups: the bleomycin injection group and the excision group. In the bleomycin injection group, patients received diluted bleomycin (1.5 million units per bottle) from Tianjin Tai-He Pharmaceutical (Tianjin, China) in a 5 mL normal saline solution. Bleomycin was injected into AHVAs using a 23-gauge needle until a slight pallor appeared on the surface of the hemangioma. Multiple injections at different sites were recommended, with a maximum dose of 24 mg for a single injection in our study.
The excision group employed various surgical techniques, including KTP laser, CO2 laser, low-temperature plasma, and Holmium laser. After administering general anesthesia, a laryngoscope was inserted through the patient’s mouth to visualize and expose the vascular anomalies. Multiple surgical instruments were utilized during the procedures, including specific devices such as the KTP/532 laser® quartz fiber (Laserscope, San Jose, USA), operated in contact mode at a power of 6 W; the WILD Heerbrugg M 655 microscope (Jena, Germany) equipped with a 400-mm focal lens coupled with a LUMENISTM 40 model CO2 laser (Yokneam, CA, ISRAEL); the Holmium laser (VersaPulse PowerSuite, Lumenis, Yokneam, Israel) utilizing a 550-micron end-firing fiber for laser delivery; and the Coblator II low-temperature plasma radiofrequency system (ArthroCare Corporation, Austin, TX, USA) with coblation plasma wands 7070#.
Excision Methods
The excision of AHVAs was performed using various methods under a micro-laryngoscope. The procedures are detailed as follows:
CO2 Laser Excision: The procedure commenced with the insertion of a laryngoscope to visualize the AHVAs. A CO2 laser, emitting 270-micron spots and operating in super-pulse mode, was utilized with a power setting between 5 and 7 watts. Microlaryngeal forceps were employed to elevate the AHVAs, after which the CO2 laser was used to meticulously remove the lesions at their base.
KTP Laser Excision: Once the laryngoscope was successfully positioned, the fibers of the KTP laser were manipulated. The laser was applied in direct contact with the tissue at an energy setting of 6 watts. Similar to the CO2 laser method, microlaryngeal forceps were used to lift the AHVAs prior to gradual removal with the KTP laser.
Holmium Laser Excision: The process for the Holmium laser was analogous to that of the KTP laser. The fibers of the Holmium laser were managed, and the excision of AHVAs was performed using a contact approach.
Low-Temperature Plasma Excision: The Coblator device was positioned beneath the base of the AHVAs with the laryngoscope in place. Its beveled tip allowed for a wider excision area using plasma. However, achieving precision in tissue removal posed challenges, occasionally leading to the inadvertent removal of adjacent normal tissue.
Evaluation of the Therapeutic Effect
The therapeutic effect was assessed using a four-point scale developed by Achauer, which evaluated changes in volume.9,10 The scoring system was as follows:
- Score “1” indicated no change in the size of the lesion.
- Score “2” indicated a decrease in the size of the lesion by ≤50%.
- Score “3” represented a decrease in the size of the lesion by ≥51% but ≤100%.
- Score “4” indicated a complete cure, defined as the disappearance of the lesion without recurrence for at least 1 year.
Additionally, ImageJ imaging processing and analysis software was utilized to measure the changes in the size of AHVAs via laryngoscopy, both preoperatively and postoperatively. The ImageJ software enabled the analysis of the area of a selected region. By using the mouse to choose a specific area, the numerical value of the selected region was displayed on the screen, facilitating an approximate calculation of the volume change in vascular malformations before and after the procedure.
Statistical Analysis
To identify factors influencing treatment outcomes, an ordinal logistic regression analysis was performed. Volumes between groups were compared using Student’s t-test, with statistical significance determined at P < .050. Data analysis was conducted using SPSS® 20.0 software (SPSS Inc., Chicago, IL, USA).
Results
In this study, we treated a cohort of 23 patients, comprising 8 males and 15 females. The ages of the patients at the time of treatment ranged from 30 to 77 years, with a mean age of 53.6 years. Fourteen patients reported a globus sensation during swallowing, 1 patient experienced recurrent hemoptysis, and 8 patients were asymptomatic, diagnosed through physical examination or gastroscopy. The AHVAs were located in the pyriform sinus in 11 patients, the postcricoid area in 3 patients, and the posterior pharyngeal wall in 6 patients. Additionally, 3 patients had multiple laryngeal subzones affected by AHVAs. Among the participants, 8 were diagnosed with hemangiomas, while 15 were diagnosed with vascular malformations (Figure 2).
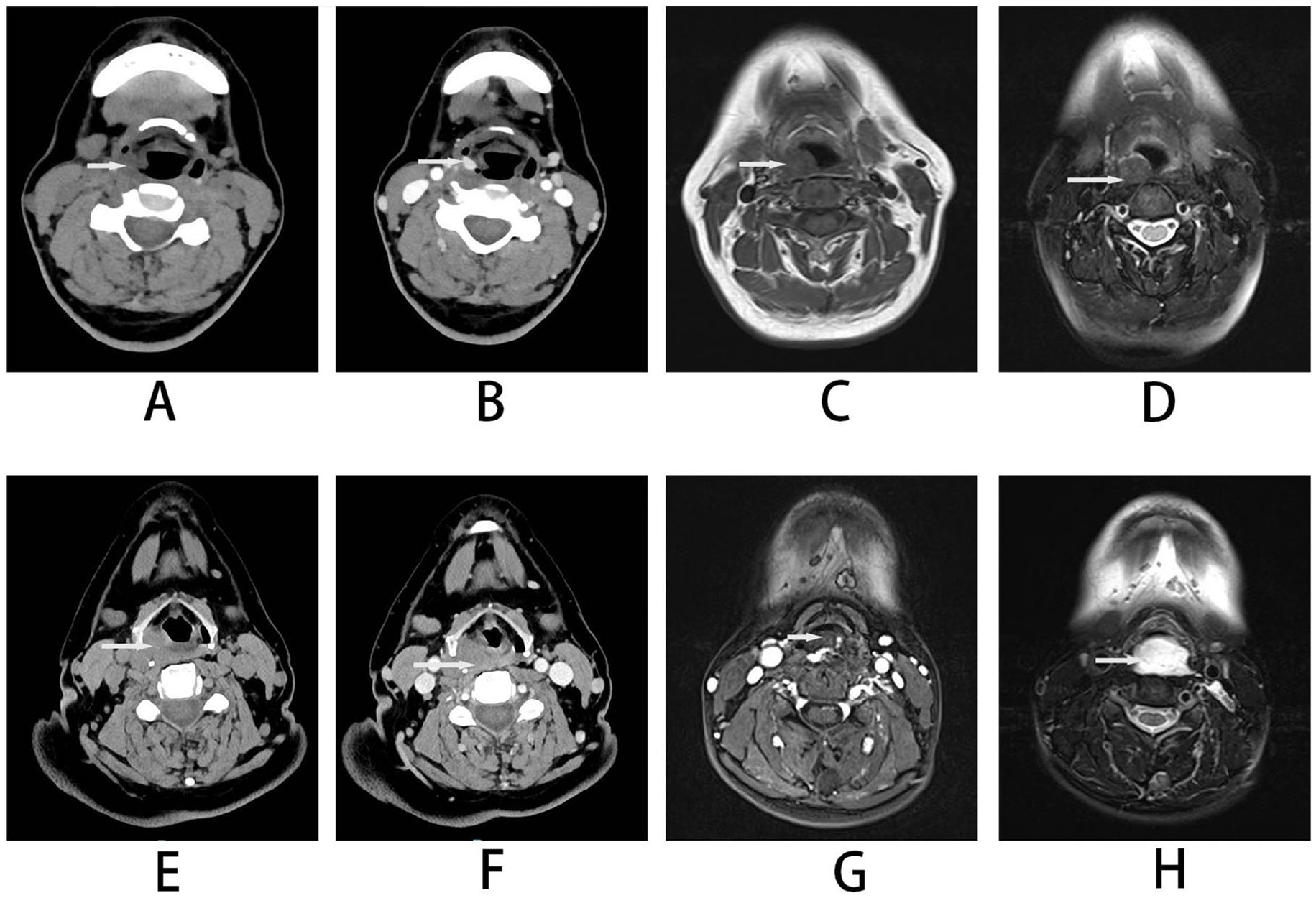
CT and MRI images of adult hypopharyngeal vascular anomalies (AHVAs). On the CT axial image of the hemangioma, it appears as an isodensity lesion (A) with significant enhancement in iodine contrast imaging (B). The lesions on both T1WI (C) and T2WI fat-suppressed (D) images were isointense. Venous malformations appear as isodense lesions on axial CT images (E) that do not enhance on iodine contrast imaging (F). T1WI (G) showed isointense, while T2WI fat-suppressed (H) images showed hyperintense lesions.
According to the “4-point scale evaluation” employed in our study, all patients were followed for 1-year post-surgery. If the treatment outcome was deemed unsatisfactory after 1 year, a reoperation could be considered. All patients underwent successful surgical intervention. A score greater than 2 on the scale was deemed effective, and all treatment modalities yielded effective results (Figure 3).
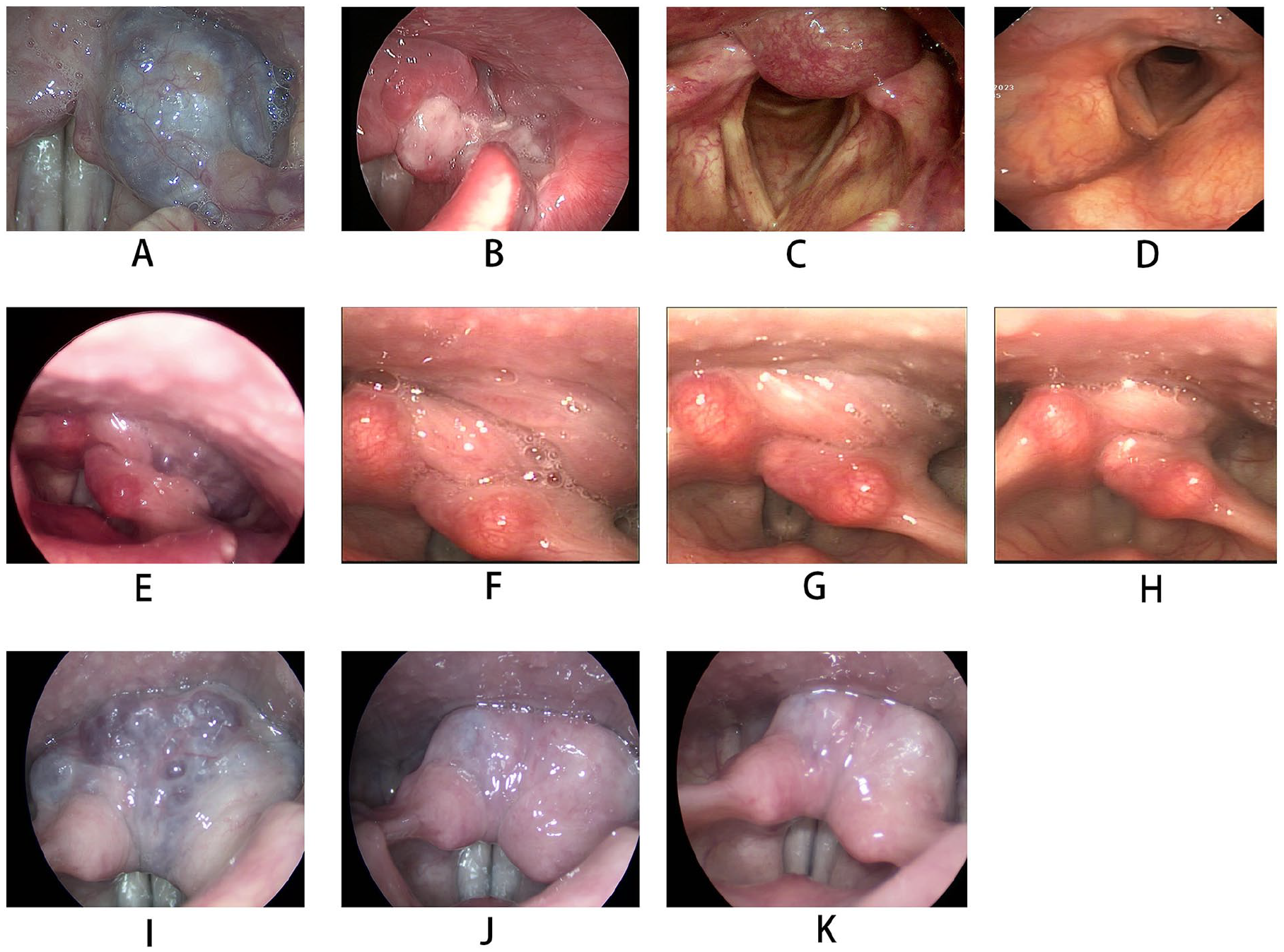
Results of laryngoscopy before and after AHVAs surgeries. (A) The AHVA was mainly located at the left pyriform sinus by laryngoscope before the KTP laser treatment. (B) 1 month after the KTP laser treatment of AHVA. (C) The AHVA was mainly located at the posterior pharyngeal wall before the low-temperature plasma treatment. (D) 1 month after the low-temperature plasma treatment. (E) The AHVA was mainly located at the left pyriform sinus and posterior pharyngeal wall by laryngoscope before the bleomycin injection. (F) 1 month after the bleomycin injection. (G) 4 months after the bleomycin injection. (H) 6 months after the CO2 excision following treatment with bleomycin injection (E, F, G, and H are images of the same patient). (I) The AHVA was mainly located at the both arytenoid and the postcricoid region by laryngoscope before the bleomycin injection. (J) 1 month after the bleomycin injection. (K) 1 year after the bleomycin injection.
The volume of the AHVAs ranged from 0.0066 cm³ to 20.8792 cm³, with a mean volume of 4.7416 cm³. A total of 23 adult patients who underwent 30 distinct procedures were enrolled in the study. Three patients underwent 2 surgeries: the first involving bleomycin injection and the second comprising complete surgical excision. Four patients each received 2 bleomycin injections. Twenty-one cases were treated with bleomycin injection, while excision methods were employed in 9 cases. Among the 9 AHVA patients who underwent excision, 1 was treated with Holmium laser, 1 with KTP laser, 2 with low-temperature plasma, and 5 with CO2 laser. The mean lumen constriction score for the chemotherapy group was 2.14. In the excision group, the tumors were completely removed without recurrence. The choice of excision method significantly influenced therapeutic outcomes, as indicated by the ordered conditional regression model. However, neither the volume nor the classification of the AHVAs affected the therapeutic effect (Table 1, Figure 3).
Results of Ordinal Logistic Regression of the Factors Affecting the Therapy Results of Adult Hypopharyngeal Vascular Anomalies (AHVAs).
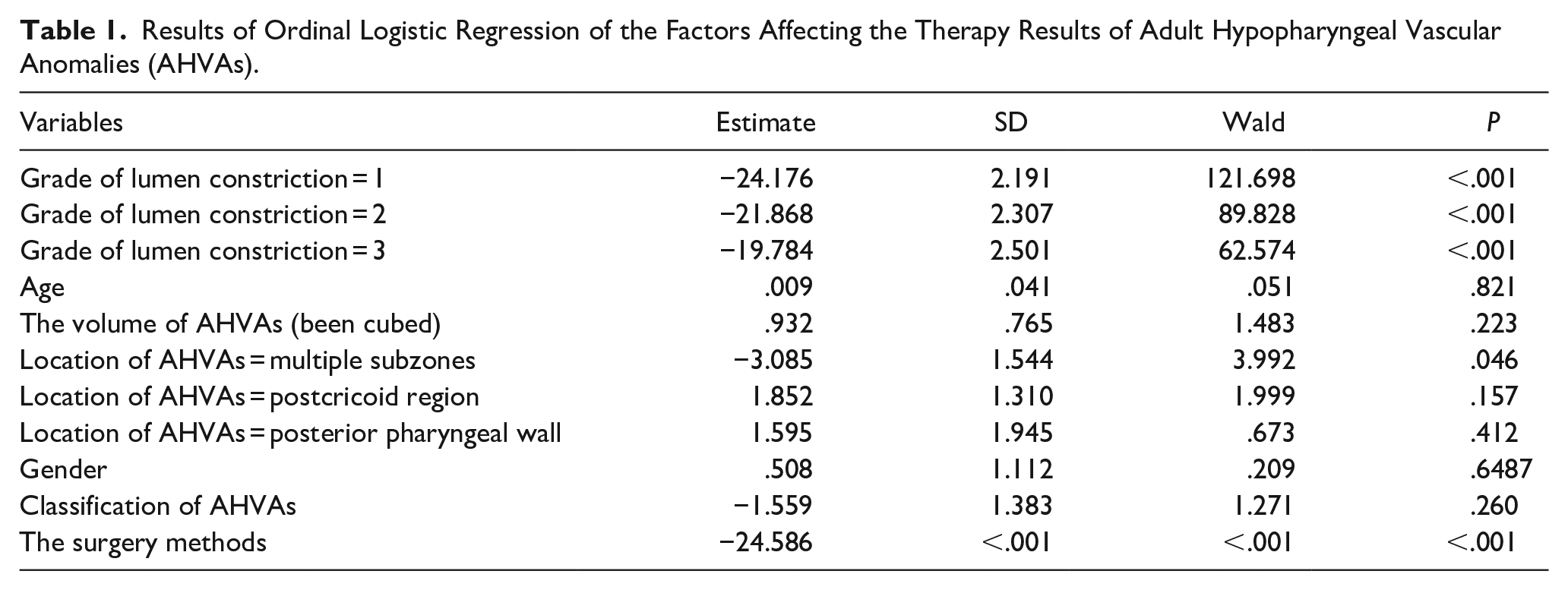
It was observed that the therapeutic effect was less favorable for AHVAs involving multiple subzones compared to those with single subzone invasion (Table 1). The excision group demonstrated smaller volumes of AHVAs in contrast to the bleomycin injection group, as illustrated in Table 2. Due to the non-normal distribution of volume values, the cube root of the volume was utilized for statistical analysis. Notably, 3 patients underwent 2 surgeries each: the first involving bleomycin injection and the second consisting of complete surgical excision. This study reported no occurrences of surgical side effects, such as tumor rupture or ulceration.
Comparison of Baseline Characteristics, Locations, Classification, and AHVAs Size in 2 Groups.
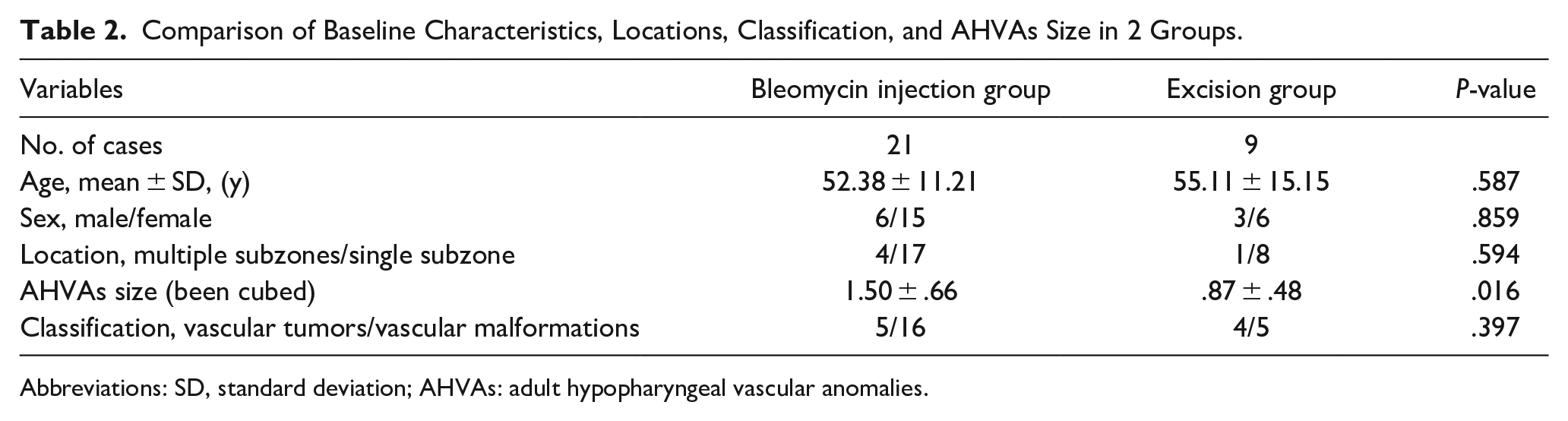
Abbreviations: SD, standard deviation; AHVAs: adult hypopharyngeal vascular anomalies.
Discussions
Currently, there are no systematic studies on the treatment of AHVAs due to their rarity, with only a few cases reported in the literature.2-7 All articles focusing on AHVAs as a distinct research topic are case reports. Generally, the effectiveness of a treatment is assessed based on the number of patients involved. In this article, we evaluated therapeutic effects based on the number of cases. For individual patients, the morphology of the hemangioma may change after just 1 treatment, influencing potential surgical approaches. Therefore, it is more scientifically rigorous to evaluate treatment effects through specific case examples.
In this study, we treated 23 patients using various methods to establish clearer guidelines for the management of AHVAs. Accurately distinguishing between non-malignant vascular tumors and malformations is critically important due to significant variations in prognosis, treatment strategies, and long-term management requirements. Following the guidelines set forth by the ISSVA, precise diagnosis of vascular anomalies, which includes both non-malignant tumors and malformations, necessitates the use of MRI or contrast-enhanced CT scans.
Based on findings from contrast-enhanced CT or MRI scans, AHVAs were categorized into vascular tumors and vascular malformations. In the excision group, pathological examination was also performed to differentiate between vascular tumors and vascular malformations. On MRI, hemangiomas typically exhibit heterogeneous parenchyma within the mass and have ill-defined borders. They present as lobulated masses with isointensity or hypointensity on T1-weighted images and relatively homogeneous hyperintensity on T2-weighted images. Additionally, T2-weighted images and fat-suppressed T2-weighted images may reveal flow voids, indicating arterial blood flow. Contrast-enhanced MRI demonstrates early and persistent enhancement, which continues during the delayed phase, resulting in intense enhancement of the lesion.11,12
MRI sequences are routinely employed for imaging vascular malformations and serve as the gold standard for evaluating slow-flow vascular malformations. Vascular anomalies appear as single or multiple lobulated or cavernous masses, occasionally displaying infiltrative growth patterns. They typically show isointensity or hypointensity on T1-weighted images and hyperintensity on T2-weighted and Short Tau Inversion Recovery (STIR) images. For hemangiomas, enhanced CT scans can reveal intense contrast accumulation during the arterial phase. 13 In adult vascular tumors, common types include congenital hemangiomas, tufted angiomas, and kaposiform hemangioendotheliomas. 14 In our study, based on enhanced CT or MRI scans, all vascular tumors identified were hemangiomas.
Fourteen patients with AHVAs experienced a noticeable globus sensation during swallowing, which was the most common symptom. Only one patient suffered from recurrent hemoptysis, while 8 patients were asymptomatic. This finding is inconsistent with conventional wisdom, as the proportion of patients experiencing throat bleeding was relatively low. Further research is needed to understand the underlying reasons for this phenomenon.
Previous studies have reported varying treatments for AHVAs, particularly in cases involving larger anomalies.3-7 Our study found that surgical excision yielded better results compared to bleomycin injection treatment. Therefore, based on safety considerations, complete excision of the tumor is recommended. The AHVA region should be evaluated using enhanced CT or MRI. However, for large AHVAs that cannot be completely resected, bleomycin injection or other procedures, such as non-contact KTP laser irradiation, are advised. When the AHVAs diminish in size, and the boundaries become clearer, surgical excision is recommended. Frequent bleomycin injections proved insufficient for completely curing AHVAs in our study. It is crucial to avoid the indiscriminate removal of tumors in pursuit of therapeutic effects.
Bleomycin has been extensively utilized in numerous studies due to its ability to target the G2 and S phases of rapidly dividing cells, inducing DNA degradation in undercoiled chromatin regions. It promotes the regression of vascular lesions by selectively damaging proliferating endothelial cells.15,16
When surgical excision of AHVAs is not appropriate, bleomycin injection may be one of the available treatment options. Based on experience, achieving satisfactory results for larger AHVAs may require 2 to 3 treatment sessions, while smaller AHVAs may need 1 to 2 sessions, which is consistent with previous reports. 17 Currently, there is no clear consensus on the time interval between bleomycin injections. Observations indicated that the reduction in the size of the AHVAs remained fairly stable 1 month after treatment. For large AHVAs that are expected to require multiple treatments, if the results after the first bleomycin injection are not significant, a second injection or an alternative treatment can be administered 1.5 months later. If the initial bleomycin injection shows good results, the interval before the second treatment can be appropriately extended. Patients undergo regular laryngoscopic examinations postoperatively, and the degree of lumen reduction is assessed using laryngoscopy and ImageJ software. If the base of the lesion is not clearly identifiable, enhanced CT and MRI scans may be conducted before any subsequent surgical intervention is performed.
Unlike common adult laryngeal vascular anomalies, excision surgery for AHVAs demonstrated superior outcomes in this study. The hypopharyngeal area is anatomically divided into 3 subregions: the postcricoid region, pyriform fossa, and posterior pharyngeal wall. The orientation of AHVAs in these regions aligns parallel to the surgeon’s visual field during direct laryngoscopic surgery, thereby justifying their surgical excision. After proper traction, if the bases of AHVAs are well exposed, CO2 laser excision is a suitable, simple, and rapid surgical procedure. If the bases of AHVAs are well exposed but the horizontal line is angled relative to the visual field, lasers with fibers, such as KTP or Holmium lasers, are recommended. Low-temperature plasma is also suggested; however, the operating space around the AHVAs must be sufficiently large. The administration of bleomycin injection is considered a safe and effective approach in cases where AHVAs are not adequately exposed during excision.
Conclusions
In conclusion, selecting the appropriate procedure for AHVAs is crucial. When surgical excision is clearly feasible, it is more effective for patients with AHVAs. For these patients, enhanced CT or MRI should be performed. If the vascular anomaly deeply invades the tissue at the base and the condition is complex, bleomycin injection might be the first choice. If the base is relatively clear, the decision should be based on the results of laryngoscopy. If the lesions are in line with the tumor base, lasers might be used. If the lesions are obliquely placed, coblation or bleomycin injection should be used. If laser surgery cannot be performed, timely bleomycin injection should be employed, prioritizing surgical safety. However, for patients who are unsuitable for surgical excision, after reducing the size of AHVAs with bleomycin injection, it may be advisable to consider surgical excision.
Footnotes
Acknowledgements
None.
Data Availability Statement
The datasets generated and analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by the Science and Technology Commission of Shanghai Municipality (CN) (18411965600).
Ethical Statement
The research ethics board of our hospital approved the study and waived the requirement for informed consent due to its retrospective nature.
