Abstract
Pleomorphic adenoma (PA) is primarily found in the salivary glands, and is extremely rare in the subglottic region. Here we present a subglottic PA that presented with symptoms of dry cough and dyspnea. A submucosal mass was found in the subglottic region under laryngoscopy, occluding approximately 40% of the lumen. The patient underwent the transoral endoscopic CO2 laser microsurgery under high-frequency jet ventilation for mass resection, and the pathology report supported the diagnosis of PA. At the 2-year follow-up, there was no evidence of recurrence, and the patient is currently under regular long-term monitoring. Dyspnea and dry cough are nonspecific respiratory symptoms. When no findings discovered in the regular site, it should be noted that the subglottic area is often a blind spot for both pulmonologists and otolaryngologists, and as such, requires careful examination. Transoral endoscopic CO2 laser microsurgery under high-frequency jet ventilation was found to be an effective and less invasive method for treating subglottic PA. This approach helped avoid tracheostomy and resulted in better postoperative recovery.
Introduction
Pleomorphic adenoma (PA) is the most common epithelial tumor of the salivary glands. 1 About 75% of PAs develop in the parotid gland, 15% in the submandibular gland, and only 10% of PAs originate from the minor salivary glands.2 -4 PAs originating from the larynx are rare, and only 8 cases of PAs originating from the subglottic region have been reported. Dry cough and dyspnea are the most common symptoms of respiratory diseases. Medical attention is usually focused on lesions occurring in the trachea, bronchi, and lungs. However, the subglottic region, which forms a part of the transition between the upper and lower airways, can sometimes be overlooked. Here we present a case of PA originating in the subglottic region in a 51-year-old female patient with complaints of dyspnea and dry cough for a year. The clinical features, diagnostic challenges, and treatment strategies of subglottic PA are discussed in this case. Moreover, a comprehensive analysis of all the available cases was conducted to provide additional evidence for the diagnosis and therapeutic strategies of PAs.
Case Report
A 51-year-old woman presented to our hospital with a 1-year history of dry cough and post-activity dyspnea. The symptoms were relieved by rest. She reported no hoarseness or dysphagia and had no history of asthma, cardiovascular disease, or trauma. Antitussives and bronchodilators had been administered but had no effect and chest computed tomography (CT) scan was negative. The patient was thus referred to an otorhinolaryngologist by her pulmonologist for further evaluation. Physical examinations were unremarkable. The laryngoscopy was performed and showed a submucosal mass with a smooth surface in the subglottic region that measured 1.8 × 0.8 × 0.5 cm and occluded approximately 40% of the lumen (Figure 1A and B). CT scan of the neck showed (Figure 1D) that the mass was round, with a smooth edge, clear boundary, uniform density, and no calcification. And the lesion was located at the mid posterior wall of the subglottic area and extended from the upper border of the cricoid cartilage to 0.7 cm below the glottis. There was no evidence of lymphadenectasis. Therefore, a diagnosis of subglottic tumor was made.
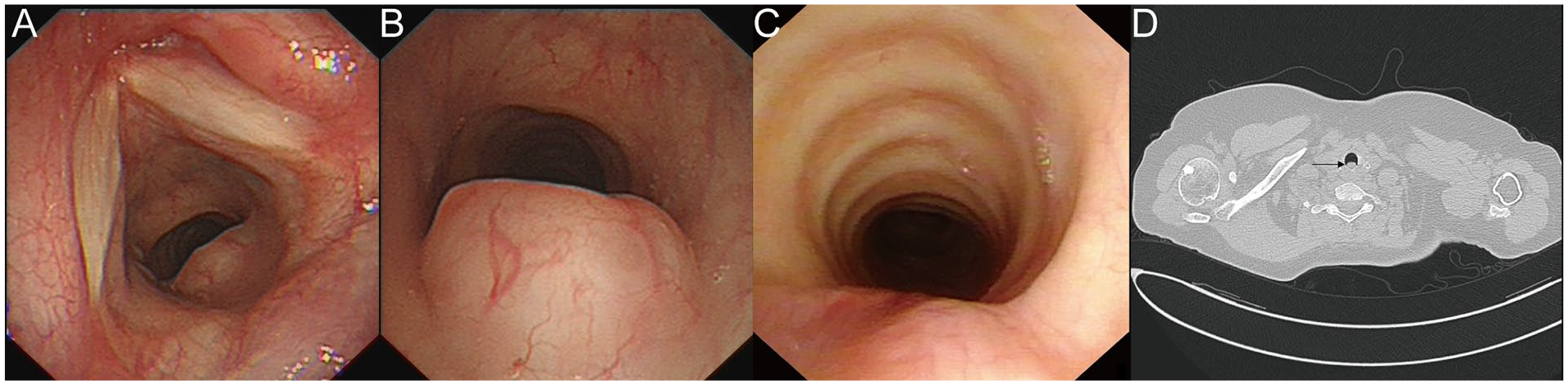
Laryngoscope view and computed tomography (CT) scan result of the subglottic pleomorphic adenoma (PA). (A and B) Preoperative laryngoscope view of the subglottic PA. (C) 2-Year postoperative laryngoscope view of the subglottic PA. (D) Preoperative CT scan of the subglottic PA.
Transoral endoscopic CO2 laser microsurgery was performed for resection of the subglottic tumor. The tumor was en bloc resected with negative margins, which was confirmed by intraoperative frozen biopsy, and no malignancy was detected. Both postoperative pathology and immunohistochemistry (IHC) results were supportive for a diagnosis of PA. Histopathology (Figure 2A) showed that the tumor consisted mainly of a glandular structure. Immunohistochemical staining (Figure 2B and C) was positive for S-100 and Smooth Muscle Actin which is consistent with the characterization of PA.
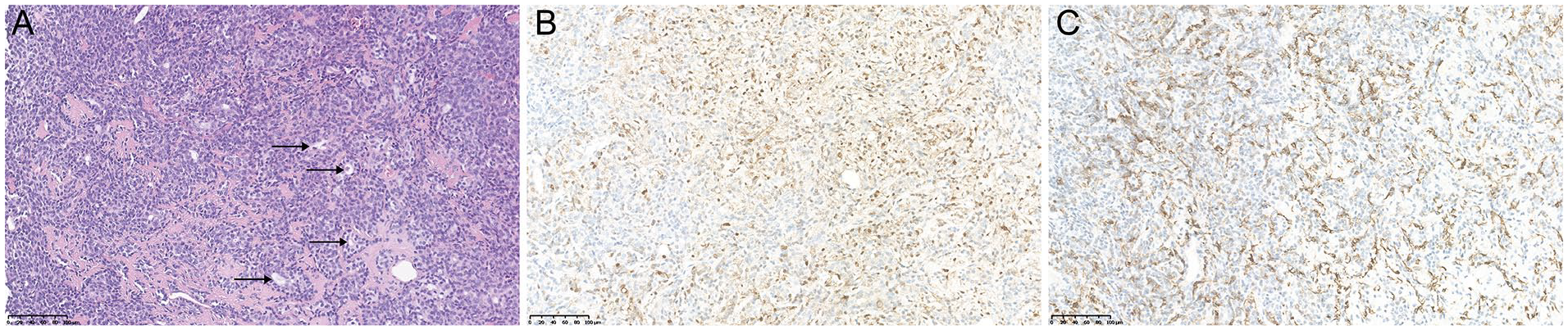
Pathological results of the subglottic pleomorphic adenoma (PA). (A) Hematoxylin and eosin (H&E) stained section showing tumors mainly composed of glandular structure. (H&E, ×20). (B) Immunohistochemistry indicates that the myoepithelial cells are focally positive for S-100 protein and (C) the myoepithelial cells cytoplasm is positive for Smooth Muscle Actin (A: ×20; B: ×20).
After surgery, the patient received prophylactic antibiotics and inhaled triamcinolone acetonide. Postoperative recovery was uneventful, and she was discharged on the fourth day after surgery. Her clinical symptoms improved significantly. Two years after surgery, there is no evidence of recurrence according to the history taking and postoperative laryngoscope (Figure 1C). The patient is currently under regular long-term follow-up.
Discussion
Laryngeal PA is a benign tumor arising from the mucous glands of the larynx. Although primary PA (mixed tumor) is typical in the major salivary glands, it is rare in the larynx, and only 43 cases have been reported. An additional file demonstrates this in detail (see Supplemental Additional file 1). The mucous glands scattered within the submucosa in the larynx could be an explanation for the occurrence of the above cases. The discovery of laryngeal PA was mostly incidental.5 -8 Among all laryngeal PA, the occurrence of subglottic PA is even exceedingly rare, and the median waiting time for the diagnosis of subglottic PA is 1 year. Because of the limited available information about this situation, it is difficult to gain a thorough understanding of the clinical manifestations, diagnosis, and treatment options. A comprehensive analysis of all available subglottic PA cases is performed, and a flowchart outlining the diagnostic and therapeutic strategies for this condition is developed (Figure 3 and Table 1).
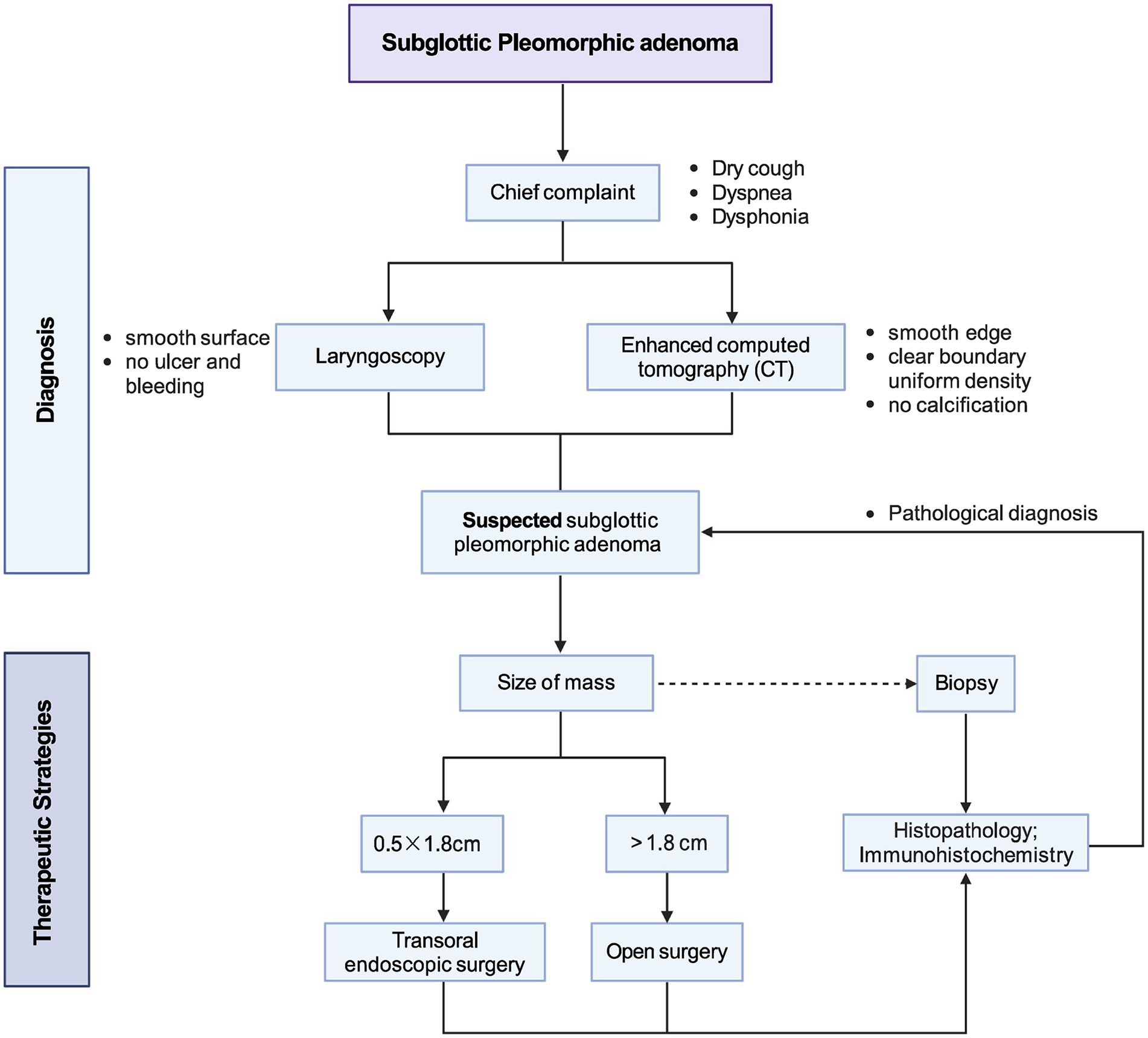
Workflow diagram of diagnosis and therapeutic strategies for subglottic pleomorphic adenoma.
Reported Subglottic Pleomorphic Adenoma in the Literature (up to 2023).
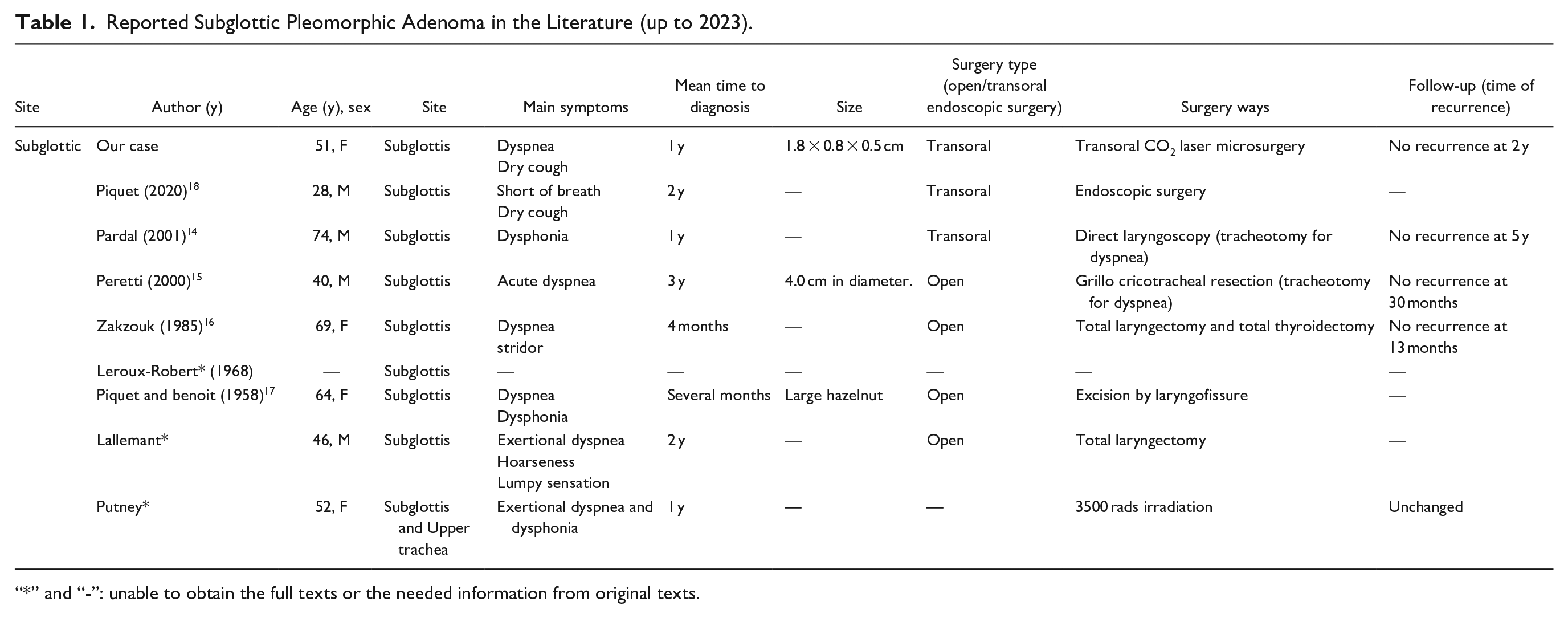
“*” and “-”: unable to obtain the full texts or the needed information from original texts.
When presented with a patient complaining of dry cough and dyspnea, healthcare providers are more likely to consider upper respiratory tract infection, allergic reaction, upper airway obstruction, or even ignore the symptoms when they occur. 9 The nonspecific symptoms and the resulting incomplete examinations are the main reasons for delayed diagnosis.
Most complains of laryngeal PA center on respiratory symptoms, including dyspnea, hoarseness, lumpy sensation, and so on (see Supplemental Additional file 1). The type and severity of the symptoms depend on the size and location of the mass. The most common sites of the laryngeal PA are the epiglottis and vocal cords. For epiglottic PA, almost all the patients complain primarily of a lumpy feeling in the throat and occasionally choking when eating. For PA located on the vocal cords, nearly all the patients complain of hoarseness and may have dyspnea when the tumor reaches the posterior commissure.10 -13
As for subglottic PA, only 8 cases have been reported so far, all of which were associated with dyspnea, including our case (Table 1).14 -18 The median time to diagnosing subglottic PA is 1 year, and the delayed diagnosis might be correlated with the size of the mass. Moreover, otorhinolaryngologists mainly focus on the supraglottic and glottic areas in their examinations, whereas pulmonologists prefer to focus on the areas below the trachea. Hence, the subglottic area naturally becomes a blind area for detection, which explains the delayed diagnosis and the resulting severe complications.
A thorough upper airway examination is necessary for diagnosis, especially based on the tumor size, to determine the severity of dyspnea. When the PA fills 90% of the lumen, the patient could suffer from severe dyspnea and cyanosis. 19 The modalities for diagnosing PA mainly include laryngoscopy and CT scan. Laryngoscopy is a convenient technique to exclude neoplasms in the larynx without ionizing radiation, providing detailed information on tumor-originating location and its appearance. However, this method does not reveal the tumor’s internal structure and whether it is infiltrating. Then contrast-enhanced CT scan could make up for this defect, and sometimes it can even exhibit mucosal changes in millimeter level.5,20 Biopsy, as the gold standard for tumor diagnosis, should be taken in overall consideration for laryngeal PA evaluation due to its unknown characteristics. Sometimes the biopsy results can lead to a more radical surgical approach, most commonly tracheostomy. 11 Although the methods for diagnosing laryngeal PA are limited, the main reasons for delayed diagnosis are still the ignorance of nonspecific symptoms and unthorough examinations. Differential diagnoses include angioma, fibroma, lymphoma, aberrant thyroid, vestigial cyst, and internal laryngocele, which are rare laryngeal neoplasms with smooth, intact, and overlying mucosa. Considering that carcinomas usually ulcerate or have an irregular surface of the tumor or overlying mucosa, the probability of malignancy in our case is low. 8
Like other benign laryngeal neoplasms, surgical resection is the first choice of treatment for PA, and the extent depends on its size and location. The approaches reported for laryngeal PA excision mainly include 2 categories: transoral endoscopic surgery and open surgery (Table 1). The former mainly includes transoral endoscopic CO2 laser surgery and suspension laryngoscopy excision, which are feasible for small benign tumors with a maximum diameter of 2 cm, as shown in our literature review. This minimally invasive approach is widely adopted because of its lower morbidity, shorter hospital stays, and preservation of better postoperative function.21,22
Based on the limited information, the surgical approaches for subglottic PA including open and transoral endoscopic surgery, differ significantly. Notably, the approach chosen by otorhinolaryngologists can be a determining factor for the time of recurrence.
However, what we cannot ignore are the difficulties in proper positioning and complete exposure that could hinder the use of endoscopic surgery. 23 Open surgery of the larynx PA usually involves lateral pharyngotomy, partial epiglottectomy, and laryngofissure. In the cases of all laryngeal PA reported to date, open surgery was performed in more than two-thirds of the cases in which treatment details were accessible. This approach inevitably resulted in more complications, hospitalization, and medical costs. 24 But when patients have obvious airway obstruction symptoms, such as severe dyspnea and stridor, timely tracheostomy and open surgery are preferred.14,15 And it should be noted that open surgery provides better visualization and ensures complete removal of the large tumor. 25 Overall, the choice of surgical procedures was largely attributed to the oversized mass and unfamiliarity with laryngeal PA. A deeper understanding of laryngeal PA could avoid most delayed diagnoses and provide more options for the best treatment modality.
In our case, local excision was performed with laryngeal endoscopic surgery, preserving laryngeal function. The high-frequency jet ventilation device created a nearly tubeless field for excision and made it possible not to perform endotracheal intubation, but to maintain stable ventilation under general anesthesia. Microscopy allows us to clearly see the peripheral and deep margins of the tumor, ensuring complete excision.26,27 The CO2 laser in the incision minimized the amount of normal tissue removed or destroyed, and the high-precision resection can also help pathologists evaluate the margin of the surgical specimen.
In most cases, recurrence occurred 18 months after surgery. And the time of relapse depends almost entirely on the adequacy of the primary excision. 11 In our case, the use of microsurgery makes it possible to remove the tumor with less trauma and shorter hospital stays. Considering the characteristics of PA, long-term follow-up with laryngoscopy is required.
This case and the summary from the literature review outline the diagnosis and therapeutic strategies of the unusual subglottic PA. In diagnosis, it reminds us that when a patient comes in with an untypical but prolonged history of respiratory symptoms, we sometimes need to search for pathologies of uncommon sources in the airway. At the onset of laryngeal PA, symptoms may only be dry cough and mild dyspnea as our case. Upper airway examination with laryngoscope or bronchoscope is necessary, and bronchial provocation test and esophageal lipiodol angiography are required for differential diagnosis. For the therapeutic strategies of subglottic PA, this case recommends microsurgical treatment using a high-frequency jet ventilator for complete resection of the mass without tracheostomy.
Supplemental Material
sj-docx-1-ear-10.1177_01455613231181456 – Supplemental material for Diagnosis and Therapeutic Strategies of Unusual Subglottic Pleomorphic Adenoma: A Rare Case Report and Literature Review
Supplemental material, sj-docx-1-ear-10.1177_01455613231181456 for Diagnosis and Therapeutic Strategies of Unusual Subglottic Pleomorphic Adenoma: A Rare Case Report and Literature Review by Xiaoru Sun, Dan He, Qiao Wen, Shi He, Yong Jiang, Shixi Liu, Haiyang Wang and Jian Zou in Ear, Nose & Throat Journal
Footnotes
Acknowledgements
The author thanks BioRender for assistance with figure development.
Presentation
The International Federation of ORL Societies (IFOS), Dubai, Jan 17–21, 2023.
Authors’ Contributions
XS drafted the manuscript. DH, HW, and JZ studied the concept and designed the report. SL, YJ, and JZ diagnosed and treated the patients. QW, XS, YJ, and JZ followed up the patients. SH provided useful suggestions on the article. All authors have read and approved the article.
Data Availability Statement
The raw data supporting the conclusions of this article will be made available by the author(s), without undue reservation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Science & Technology Department of Sichuan Province (Grant Nos. 2020YFS0114 and 2021YFS0134). In this study, laboratory analyses and cover publication costs were supported by the fundings.
Ethics Statement
This case report is in accordance with the Code of Ethics of the World Medical Association (Helsinki Declaration). The patients/participants provided their written informed consent to participate in this study. Written informed consent was obtained from the patient for publication of this case report and accompanying images. A copy of the written consent is available for review by the Editor-in-Chief of this journal on request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
