Abstract
Despite the availability of multiple treatments for solid tumors, the prognosis of advanced solid tumors remains poor. Antibody–drug conjugates, a novel type of antitumor drugs, which are created by conjugating cytotoxic small-molecule drugs with monoclonal antibodies using well-designed linkers, can selectively deliver effective cytotoxic drugs to tumor cells. Currently, antibody–drug conjugates targeting multiple therapeutic targets for solid tumors are being constantly developed, optimized, and improved, opening up a broad-spectrum antitumor treatment model. However, the use of antibody–drug conjugates is still associated with some limitations that need to be urgently overcome such as poor permeability in solid tumors, notable toxic adverse reactions, and drug resistance. This review mainly discusses the functionality, iteration history, and research progress regarding the use of antibody–drug conjugates in the treatment of common solid tumors and explores possible optimization strategies for the future based on their toxicity, efficacy, and drug resistance, which will ultimately benefit more patients with cancer.
Keywords
Introduction
Current treatment modalities for solid tumors, including conventional chemotherapy, radiotherapy, and certain targeted therapies, are associated with several fundamental limitations in clinical practice. Although traditional chemotherapy exhibits cytotoxicity, its lack of tumor specificity causes significant systemic toxic adverse effects and achieves limited penetration into the tumor microenvironment. 1 Radiotherapy is a localized treatment method 2 that cannot address distant metastases; furthermore, it lacks tumor specificity, and the curative dose is constrained by the tolerance of surrounding normal tissues. 3 Many targeted therapies are further limited by target mutations and endogenous or acquired drug resistance. 4 Antibody–drug conjugates (ADCs) are complexes in which cytotoxic small-molecule drugs are conjugated to monoclonal antibodies through a reasonably constructed linker that can selectively deliver effective cytotoxic drugs into tumor cells. 5 This targeted therapy is more selective than traditional systemic chemotherapy and can reduce damage caused to normal cells. 6 The high-efficiency payload carried by ADCs can overcome drug resistance and exert potent cytotoxic effects on cells resistant to targeted therapy. 7 Additionally, ADCs can kill adjacent heterogeneous tumor cells through the bystander effect. 8 They can also induce tumor cell death via oxygen-independent cytotoxic payloads. Therefore, when combined with radiotherapy, they can synergistically overcome radiotherapy resistance induced by the hypoxic microenvironment, thereby improving tumor control. 9 Over the past decade, ADCs have been refined via the selection of better cytotoxic drugs, use of biological coupling methods, better targeting of antigens, and optimized antibody engineering.
The process of ADC development can be divided into three stages. The first stage involves the concept proposal and early exploration of ADCs and use of mouse-derived antibodies (1910–1980). In 1913, German physician and scientist, Paul Ehrlich, first proposed the concept of ADCs and coined the term “magic bullet” to describe the method of selectively delivering cytotoxic drugs by targeting tumor cells.
10
In 1958, Mathe et al. coupled anti-mouse leukocyte immunoglobulins with methotrexate to treat leukemia, marking the beginning of ADC research.
11
Owing to the strong immunogenicity of initial ADCs and the limitations of antibody-preparation technology, initial efforts showed limited success in translating laboratory findings to clinical use. ADC development was slow in the subsequent decades, and none of the ADCs developed during this period demonstrated sufficient efficacy and safety to be established as therapeutic drugs. However, the advent of monoclonal antibody technology in 1975 facilitated the translation of ADC research from the laboratory to clinical application, and the first modern version of ADCs was developed in the same year.12,13 This was followed by the second stage of ADC development, which was characterized by technological breakthroughs and rapid development (1980–2000). The development of humanized antibody technology resolved the problems associated with antibody preparation and purification. Subsequently, ADC development progressed through stages using mouse-derived monoclonal, human mouse chimeric, humanized, and fully humanized antibodies, and the first human clinical trials of ADCs were performed.14–18 At present, most ADCs are prepared using humanized or fully humanized antibodies. The third stage is characterized by the maturation and market-oriented development of ADCs (from 2000 to the present). In 2000, gemtuzumab ozogamicin (Mylotarg) received accelerated approval from the US Food and Drug Administration (FDA) for the treatment of CD33-positive acute myeloid leukemia,
19
becoming the world’s first commercially available ADC and marking the beginning of the era of ADC-targeted cancer therapy. Supported by rapid advancements in biotechnology, ADCs have undergone three generations of updates and iterations. Currently, 15 ADCs are approved for clinical use,20,21 with 13 still on the market, targeting multiple antigens such as human epithelial growth factor receptor 2 (HER2) and trophoblast cell surface antigen 2 (TROP2).
21
Key milestones in their development include the following:
The first ADC drug, gemtuzumab ozogamicin, was approved in 2000. China’s first independently developed ADC drug, disitamab vedotin (Aidixi), was approved for marketing in 2021.
21
ADCs have been successfully used to treat various solid tumors, including HER2-positive/low-expressing breast cancer (BC), gastric cancer (GC)/gastroesophageal junction cancer, urothelial carcinoma, cervical cancer (CC), ovarian cancer (OC), and non-small cell lung cancer (NSCLC). 21 The above three stages of ADC development before entering the commercialization period are summarized in Figure 1.

The three stages of ADC development before entering the commercialization period. ADCs: antibody–drug conjugates; FDA: Food and Drug Administration.
ADCs exhibit transhistological activity, meaning that their therapeutic efficacy is not confined to cancer types originating from a single tissue; the core mechanism of action relies on the antibody component to recognize specific targets on the surface of tumor cells, and an increasing number of cancer patients are expected to be suitable for ADC treatment in the future. 22 The treatment lines for ADCs are gradually moving forward from advanced cancers to cover early-stage solid tumors. However, the use of ADCs is still associated with limitations such as poor permeability in solid tumors, substantial toxic adverse effects, and drug resistance, which are key challenges that need to be overcome for the development of ADCs. This review mainly discusses the functionality, iteration history, and research progress on ADCs for the treatment of common solid tumors and explores possible future optimization strategies to address their toxicity, efficacy, and drug resistance, ultimately benefiting more cancer patients. Additionally, we performed a systematic literature search across multiple databases, including PubMed using predefined keywords, applied stringent inclusion criteria, and performed strict methodological quality assessment to include recent studies. Data were synthesized narratively and analyzed by solid tumor type to ensure objective interpretation.
ADCs functionality and iteration history
ADCs constitute a specific drug-delivery system 23 that precisely introduce chemotherapeutic drugs into solid tumors through the targeting effects of antibodies. 24 By combining antibodies, payloads, and a linker, they combine the advantages of the highly specific targeting ability of antibodies with the efficient killing action of traditional cytotoxic drugs. 23 Most contemporary ADCs utilize humanized or fully human monoclonal antibodies that selectively recognize the surface antigens of tumor cells, while the payloads are drugs responsible for strong cytotoxicity in tumor cells.14,25 The linker is responsible for connecting the antibody to the payload; influencing the toxicity, specificity, stability, and potency of ADCs; and controlling the release of the payload into specific tumor cells.26,27 The key steps required for ADCs to exert their antitumor effects include targeted selection, payload endocytosis, and payload release. 28 Briefly, after ADCs enter the body, their antibodies specifically recognize and bind to the surface targets of tumor cells and enter these cells via endocytosis. Under the action of lysosomes, the linker is cleaved, releasing the payload and producing cytotoxic effects that trigger payload-mediated apoptosis.29–31 In addition, ADCs can produce a bystander effect, and after acting on target antigen-positive cells, the released cytotoxic payload can kill the surrounding cells (including target antigen-negative and normal cells).32–34 In addition, they can exert effector functions mediated by monoclonal antibody fragments.29,31 Although this feature enhances the effectiveness of antitumor treatments, it also produces off-target adverse reactions. The detailed mechanisms underlying the effects of ADCs in solid tumors is presented in Figure 2.

The mechanism diagram of ADCs’ role in solid tumors. ADCs: antibody–drug conjugates; HER1/2/3: human epithelial growth factor receptor 1/2/3; TF: tissue factor; FRα: folate receptor-alpha; TROP2: trophoblast cell surface antigen 2.
As outlined above, ADCs have developed through three stages, from concept proposal to commercialization, and have benefited from advancements in monoclonal antibody technology and recombinant-protein preparation technology as well as the transition from mouse-derived antibodies to contemporary humanized antibody structures. Since the first mature ADC was launched in 2000, ADCs have undergone continuous iteration and upgrades in terms of drug composition and technical characteristics over three generations, each of which has improved drug specificity, stability, and efficacy (Table 1). The representative drug of the first-generation ADCs is Mylotarg, which consists of a recombinant humanized anti-CD33 monoclonal antibody conjugated to the cytotoxic antibiotic, calicheamicin. 19 However, Mylotarg showed limitations such as low drug payload, unstable linkers, and susceptibility to trigger immune responses in the body, which resulted in low therapeutic efficacy and high toxicity. 35 These factors were responsible for the failure of Mylotarg treatment; the drug was withdrawn from the market in 2010 and re-approved for marketing in 2017 after specification adjustments and supplementary clinical evidence.19,35,36 Subsequently, the development of ADCs slowed down in the next decade. The representative drugs for second-generation ADCs include ado-trastuzumab emtansine (T-DM1) 37 and Adcetris. 38 In comparison with first-generation ADCs, second-generation ADCs used improved monoclonal antibody technology, carefully selected humanized/fully humanized monoclonal antibodies, and cytotoxic drugs with higher activity. Although these ADCs showed improved targeting of tumor antigens coupled with the effectiveness of cytotoxic small molecules, 39 they still employed traditional nonsite conjugation methods, resulting in poor distribution uniformity, presence of unconjugated antibodies, and aggregation or rapid clearance of drugs with a high drug–antibody ratio (DAR). In addition, the stability of the linkers was low, and they could be easily cleaved to release the payload, causing serious off-target toxic adverse reactions.40,41 Representative third-generation ADCs include trastuzumab deruxtecan (T-DXd), sacituzumab govitecan (SG), polatuzumab vedotin (Polivy), and enfortumab vedotin (EV). On the basis of the insights gained from the first- and second-generation ADCs, third-generation ADCs adopted site-specific conjugation technologies, with a uniform payload and DAR reaching 4–8, which enhanced the ability to target antigens and significantly improved drug stability, showing smaller off-target toxicity and better pharmacokinetic efficiency.42–44 Additionally, third-generation ADCs used more hydrophilic and cleavable linkers conjugated to cytotoxic drugs with higher activity, such as monomethyl auristatin E (MMAE) and topoisomerase I inhibitors, which produced potent bystander effects through antigen internalization and linker cleavage.45–47 Overall, third-generation ADCs had lower toxicity, stronger anticancer activity, and higher stability. However, they exhibit potential for further optimization. For example, the payload is mainly composed of cytotoxic compounds with limited selection, easy drug resistance, insufficient linker stability, and a high drug molecular weight with low permeability. 48 Currently, several new ADCs are under clinical research. Through the continuous optimization of antibodies, cytotoxic drugs, linkers, and conjugation methods, the overall performance of ADCs can be improved, benefiting more cancer patients.
The evolution of ADCs in the marketization stage.

ADCs: antibody–drug conjugates; T-DM1: ado-trastuzumab emtansine; T-DXd: trastuzumab deruxtecan; Polivy: polatuzumab vedotin; SG: sacituzumab govitecan; EV: enfortumab vedotin; PBD: pyrrolobenzodiazepine; CPT: camptothecin; DAR: drug–antibody ratio.
Clinical application of ADCs in solid tumors
Over the past decade, ADCs have developed rapidly and gradually emerged in the field of solid tumor treatment. After the FDA approved the first ADC drug, Mylotarg, in 2000, the HER2-targeting T-DM1 was approved for marketing in 2013 and became the first ADC to be used for treating solid tumors. 49 At present, 15 approved ADCs are available worldwide, including eight for solid tumor treatment, including T-DXd, disitamab vedotin (also called RC48-ADC), T-DM1, cetuximab sarotalocan sodium, SG, EV-ejfv, tisotumab vedotin-tftv (TV), and mirvetuximab soravtansine (MIRV), whose targets included human epithelial growth factor receptor 1 (HER1, also called EGFR), HER2, Nectin-4, tissue factor (TF), folate receptor-alpha (FRα), and TROP2.20,21,50 In addition, many ADCs targeting the aforementioned targets and other targets are in the clinical research stage. Detailed classification is provided in Figure 3 and Supplementary Table S1.
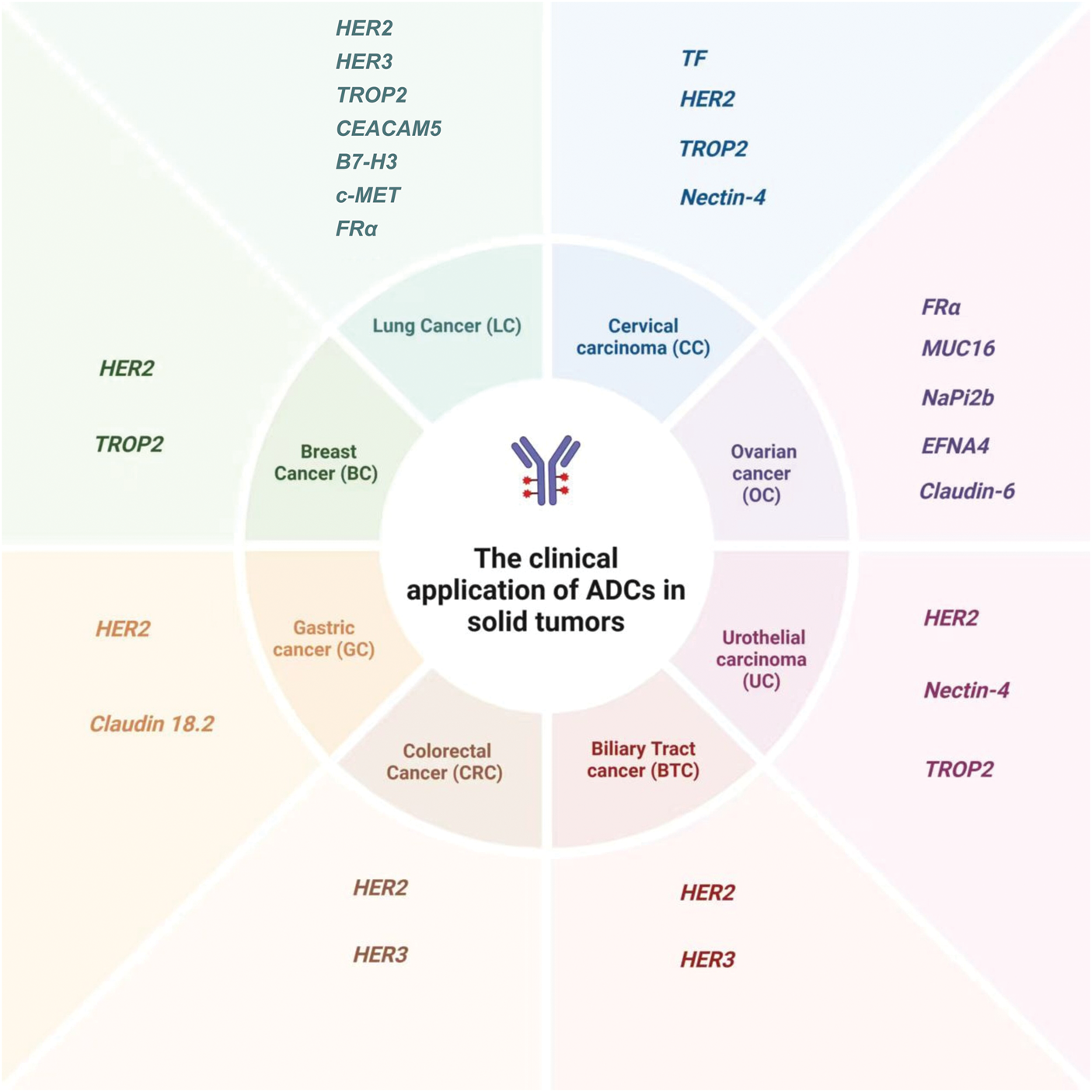
Clinical application and involved targets of ADCs in different solid tumors. ADCs: antibody–drug conjugates; HER1/2/3: human epithelial growth factor receptor 1/2/3; TF: tissue factor; FRα: folate receptor-alpha; TROP2: trophoblast cell surface antigens 2; EGFR: epidermal growth factor receptor.
ADCs are essentially targeted chemotherapeutic agents. The detailed overview presented in this review describes the functions, mechanisms of action, and drug design characteristics of ADCs, which will help us better understand the differences in the efficacy and safety of ADCs in different solid tumors and the influence of factors such as the selection and expression level of tumor cell target antigens, design and preparation of antibodies, combination of linkers, and changes in effective payloads. We also explores the applications of ADCs in different, common solid tumors (Table 2).
Targets and ADCs in common solid tumors.

ADCs: antibody–drug conjugates; LC: lung cancer; HER2/3: human epithelial growth factor receptor 2/3; T-DXd: trastuzumab deruxtecan; T-DM1: ado-trastuzumab emtansine; HER3-DXd: patritumab deruxtecan; TROP2: trophoblast cell surface antigen 2; Dato-DXd: datopotamab deruxtecan; SG: sacituzumab govitecan; CEACAM5: carcinoembryonic antigen-related cell adhesion molecule 5; TUSA: tusamitamab ravtansine; B7-H3: B7 homolog 3 protein; I-DXd: ifinatamab deruxtecan; c-MET: cellular–mesenchymal to epithelial transition factor; Teliso-V: telisotuzumab vedotin; FRα: folate receptor-alpha; MORAb-202: farletuzumab ecteribulin; BC: breast cancer; RC48-ADC: disitamab vedotin; GC: gastric cancer; UC: urothelial cancer; Nectin-4: nectin cell adhesion molecule 4; EV: enfortumab vedotin; OC: ovarian cancer; MIRV: mirvetuximab soravtansine; MUC16: mucin 16; SV: sofituzumab vedotin; NaPi2b: sodium-dependent phosphate transport protein 2B; LIFA: lifastuzumab vedotin; EFNA4: ephrin A4; CC: cervical cancer; TF: tissue factor; TV: tisotumab vedotin.
Lung cancer (LC)
HER2
ADCs targeting various types of LCs are currently under clinical research, and they involve targets such as HER2, human epithelial growth factor receptor 3 (HER3), EGFR, TROP2, carcinoembryonic antigen-related cell adhesion molecule 5 (CEACAM5), B7 homolog 3 protein (B7-H3), cellular–mesenchymal to epithelial transition factor (c-MET), 20 and FRα. 51 The DESTINY-Lung01 study has shown that T-DXd exhibited durable anticancer activity in patients with metastatic HER2-mutant NSCLC that was refractory to standard treatment. Among the 91 patients with HER2-mutant NSCLC treated with T-DXd, the objective response rate (ORR) was 55%. One patient exhibited complete response (CR) and 49 exhibited partial response (PR); the median progression-free survival (mPFS) duration was 8.2 months, and the median overall survival (mOS) duration was 17.8 months. 52 Researchers further explored the efficacy and safety of different doses of T-DXd in the HER2-overexpressing cohort in the DESTINY-Lung01 study. The results showed that the ORRs of the high-dose group (6.4 mg/kg) and low-dose group (5.4 mg/kg) were 26.5% and 34.1% (both PR), respectively; the median durations of overall response (mDORs) were 5.8 and 6.2 months, respectively; the disease control rates (DCRs) were 69.4% and 78.0%, respectively; the mPFS durations were 5.7 and 6.7 months, respectively; and the mOS durations were 12.4 and 11.2 months, respectively. These findings indicated that T-DXd exhibits strong anticancer potential in HER2-overexpressing NSCLC and that the low-dose group experienced more benefits in terms of efficacy and safety. 53 Subsequently, the DESTINY-Lung02 study enrolled 152 patients with HER2-mutant NSCLC to investigate the therapeutic efficacy and safety of two different doses of T-DXd (5.4 mg/kg and 6.4 mg/kg). The results further confirmed that both doses of T-DXd showed clinically significant responses and controllable safety, with the dose of 5.4 mg/kg exhibiting better safety. 54 The DESTINY-Lung05 study was designed to evaluate the efficacy and safety of T-DXd in Chinese patients with HER2-mutant metastatic NSCLC who had received first-line treatment. In that study, the ORR confirmed by the investigator’s assessment was 58.3%, mDOR was 9 months, the DCR was 93.1%, the mPFS duration was 10.8 months, and the 12-month progression-free survival (PFS) rate was 39.7%. Moreover, T-DXd (5.4 mg/kg) showed better clinical significance and durable remission, with a manageable safety profile. 55 In addition to T-DXd, another ADC targeting HER2, T-DM1, has been subjected to a series of clinical studies for LC. Results from a phase II basket trial has shown that T-DM1 treatment in 18 patients with advanced HER2-mutant lung adenocarcinoma (median number of prior systemic therapies, two) achieved an ORR of 44% and mPFS duration of 5 months, reaching the primary endpoint with good tolerability. This was the first positive result for T-DM1 in LC research. 56 Subsequently, the research team expanded the sample and found that among 49 patients with HER2-amplified or HER2-mutant advanced LCs, the ORR of T-DM1 treatment was 51% and mPFS duration was 5 months, further confirming the findings of the previous study. In addition, the anticancer activity of T-DM1 can be enhanced by cotreatment with irreversible pan-HER inhibitors or by switching the ADC to T-DXd. 57
HER3
Patritumab deruxtecan (HER3-DXd) is an ADC that targets HER3. HERTHENA-Lung01, a phase II study, aimed to evaluate the efficacy and safety of HER3-DXd in advanced EGFR-mutant NSCLC patients who had previously undergone EGFR tyrosine kinase inhibitor (TKI) treatment and platinum-based chemotherapy (PBC). The results showed that 225 patients who received HER3-DXd (5.6 mg/kg once every 3 weeks) had a confirmed ORR of 29.8% determined by blinded independent central review (BICR), mDOR of 6.4 months, mPFS duration of 5.5 months, and mOS duration of 11.9 months, indicating that HER3-DXd showed clinically significant efficacy and persistent response in patients with EGFR-mutant NSCLC. 58 A phase III trial of EGFR-mutant NSCLC following EGFR-TKI progression is currently underway (HERTHENA-Lung02). BL-B01D1 is an ADC that targets the HER3xEGFR dual target. A recent phase I study has shown that BL-B01D1 has good efficacy in patients with advanced EGFR-mutant NSCLC, with an ORR of 63.2% and DCR as high as 89.5%. 59
TROP2
Next, we explored the application of ADCs targeting TROP2 in LC. Datopotamab deruxtecan (Dato-DXd), a novel TROP2-targeting ADC, was the only ADC that showed positive results in a phase III registration study on LC. Shimizu et al. evaluated the safety, tolerability, and antitumor activity of Dato-DXd in patients with locally advanced or metastatic NSCLC in a first-in-human dose-escalation and dose-expansion study. The results showed that the ORR, mDOR, mPFS duration, and mOS duration were 26%, 10.5 months, 6.9 months, and 11.4 months, respectively, and the toxic reactions were tolerable. Importantly, a therapeutic response was observed, irrespective of TROP2 expression. 60 Subsequently, the phase III TROPION-Lung01 study yielded positive results, showing that, in comparison with docetaxel, Dato-DXd significantly improved the survival benefits in patients with advanced NSCLC who had previously undergone immunotherapy (for populations without actionable genomic alterations (AGAs)) or targeted therapy (for populations with AGAs), especially in the non-squamous NSCLC subgroup. The detailed results of the TROPION-Lung01 study for non-squamous NSCLC subgroups were announced at the 2024 European Lung Cancer Congress (ELCC) and may further support Dato-DXd as a new treatment option for non-squamous NSCLC patients (without AGAs) who have received immunotherapy. 61 The TROPION-Lung02 study further confirmed that Dato-DXd with or without the PD-1 inhibitor, PBC, exhibited promising antitumor activity in patients with NSCLC without AGAs. The data has shown that the ORR in the dual-drug group (Dato-DXd + pembrolizumab) and the triple-drug group (Dato-DXd + pembrolizumab + PBC) was 38% and 49%, respectively; the DCR was 84% and 87%, respectively; and the mPFS durations were 8.3 and 7.8 months, respectively. 62 The phase II TROPION-Lung05 study revealed the efficacy of Dato-DXd in patients with advanced NSCLC with AGAs. The study included 137 patients, of whom 72% received ≥3 lines of treatment. The overall population had a confirmed ORR, mDOR, DCR, and mPFS duration of 35.8%, 7 months, 78.8%, and 5.4 months, respectively. Subgroup analysis showed better efficacy in patients with EGFR-mutant NSCLC, with a confirmed ORR, mDOR, DCR, and mPFS duration of 43.6%, 7 months, 82.1%, and 5.8 months, respectively. 63 In addition to Dato-DXd, another drug targeting TROP2, SG, has shown anticancer potential in NSCLC. In a single-arm multicenter trial (n = 54), in the study and intent-to-treat (ITT) populations, the ORRs of SG treatment for pretreated NSCLC patients were 19% and 17%, respectively, with a manageable safety profile. 64
CEACAM5/B7-H3/c-MET/FRα
Other ADCs explored for LC treatment include tusamitamab ravtansine (TUSA), which targets CEACAM5; ifinatamab deruxtecan (I-DXd, also known as DS-7300), which targets B7-H3; telisotuzumab vedotin (Teliso-V), which targets c-MET; and farletuzumab ecteribulin (MORAb-202), which targets FRα. The phase II open-label CARMEN-LC05 study determined the efficacy and safe dose of TUSA. Sanofi conducted the phase III CARMEN-LC03 study to evaluate second-line TUSA treatment of CEACAM5-positive metastatic non-squamous NSCLC. Mid-term analysis data showed that, in comparison with docetaxel, although TUSA monotherapy improved overall survival (OS) trends, it did not reach the dual primary endpoint of PFS. 65 Therefore, Sanofi announced the termination of TUSA’s global clinical development plan. In addition to Sanofi, Bolt Biotherapeutics also failed to achieve this target, terminating further development of preclinical CEACAM5-targeting immunostimulatory antibody conjugates owing to off-target toxicity. 66 Researchers have suggested that CEACAM5 is a feasible target; however, a more selective antibody is required. A phase I/II clinical trial aimed at evaluating the efficacy and safety of I-DXd in the treatment of advanced solid tumors was presented at the 2023 World Conference on LC. Among the 21 small cell lung cancer (SCLC) patients who received I-DXd treatment (median treatment lines 2 (1–7)), the ORR, mPFS duration, mOS duration, and mDOR were 52.4%, 5.6 months, 12.2 months, and 5.9 months, respectively. Moreover, among the 13 patients with squamous NSCLC (median treatment lines, 3 (1–12)), the ORR, mDOR, mPFS duration, and mOS duration were 30.8%, 4.1 months, not reached, and not reached, respectively. Overall, the safety and tolerability were good. 67 Based on data from the 2022 American Society of Clinical Oncology (ASCO) conference, Teliso-V has received breakthrough therapy recognition from the FDA and can be used to treat patients with advanced or metastatic non-squamous NSCLC showing wild-type EGFR, high c-MET expression, and progression after PBC. The presented study had determined the ORR of 122 patients; the ORR was 36.5% in the EGFR wild-type overexpressing c-MET non-squamous group and slightly lower in the EGFR-mutant group (11.6% in the non-squamous group and 11.1% in the squamous group). 68 MORAb-202 is an ADC formed by conjugating an FRα-targeting antibody to the microtubule inhibitor, eribulin. Early clinical data have demonstrated its antitumor activity in FRα-positive solid tumors (including LC) with controllable toxicity. A first-phase human study (NCT03386942) has shown that MORAb-202 exhibits encouraging antitumor activity, with a DCR of approximately 81.82% (1 patient exhibited CR, 9 PR, and 8 stable disease); the overall safety was manageable. This provides a potential therapeutic direction for selected FRα-positive LC patients.51,69
BC
HER2
BC is the first solid tumor to be selected for treatment with ADCs and currently has the most approved indications for ADCs. 70 The existing ADCs with approved indications for BC include T-DM1 (targeting HER2), T-DXd (targeting HER2), and SG (targeting TROP2); other ADCs are being evaluated in different clinical trials. The EMILIA study has shown that T-DM1 significantly prolongs the PFS and OS durations in HER2-positive patients with advanced BC who were previously treated with trastuzumab and taxane and was less toxic than lapatinib plus capecitabine. Among the 991 patients evaluated after receiving T-DM1, the mPFS duration (9.6 vs. 6.4 months), mOS duration (30.9 vs. 25.1 months), and ORR (43.6% vs. 30.8%), as assessed in an independent review, were all favorable. 71 In the phase III open-label KATHERINE study, among the HER2-positive early-stage BC patients showing residual invasive disease after completing neoadjuvant therapy, 88.3% and 77.0% patients with 3-year noninvasive diseases were treated with T-DM1 and trastuzumab, respectively. The risk of invasive disease recurrence or death after T-DM1 treatment was 50% lower than that after treatment with trastuzumab alone. 72 Based on a phase III clinical study, T-DM1 has become the standard treatment for patients with HER2-positive advanced BC following treatment with anti-HER2 antibodies combined with a taxane. DESTINY-Breast03 is a phase III, multicenter, open-label, randomized trial comparing the efficacy and safety of T-DXd and T-DM1 in the posterior-line treatment of HER2-positive metastatic BC. The results of this trial have shown that the mPFS duration of the T-DXd group was 4 times longer than that of the T-DM1 group (28.8 vs. 6.8 months, p < 0.0001). Although neither group reached mOS, the T-DXd group showed a significant trend toward prolongation and a significantly reduced mortality risk (hazard ratio (HR) = 0.64, p = 0.0037). In terms of safety, the proportion of adverse events in the two groups was similar and controllable. Therefore, in comparison with T-DM1, T-DXd is a better choice for patients with HER2-positive metastatic BC who exhibit disease progression after receiving trastuzumab combined with chemotherapy.73,74 The DESTINY-Breast04 study further confirmed that in comparison with chemotherapy, T-DXd treatment for unresectable/metastatic BC with low HER2 expression could significantly prolong the mPFS duration (9.9 vs. 5.1 months, p < 0.001) and mOS duration (23.4 vs. 16.8 months, p = 0.001), and subgroup analysis showed consistent benefits in both hormone receptor (HR)-positive and HR-negative populations. 75 Other ADCs targeting HER2 have also been used in BC, including RC48-ADC and SHR-A1811. In a study conducted by Qu et al., RC48-ADC (2.5 mg/kg) treatment every 2 weeks in HER2-positive and HER2-low expression patients resulted in an overall ORR of 38.3% and an mPFS duration of 5.7 months. In subgroup analyses, RC48-ADC showed consistent efficacy in HER2-positive and HER2-low expression populations. 76 Li et al. reported that RC48-ADC exhibited good efficacy and controllable adverse events in patients with metastatic HER2-positive BC who received trastuzumab treatment. Their study reported an mPFS duration of 5.9 months and ORR of 29.6%, with one patient achieving CR and 23 achieving PR. 77 SHR-A1811 is a novel HER2-targeting ADC that binds to the HER2 protein on the surface of tumor cells, is endocytosed into tumor cells, and is then cleaved by lysosomes to release toxins and induce cell apoptosis. A global phase I, multicenter, first-in-human study has shown that SHR-A1811 demonstrates good clinical efficacy in posterior-line treatment of patients with advanced HER2-expressing BC. The confirmed ORR in HER2-positive patients was 76.3%, and in patients with low HER2 expression, the ORR was 60.4%. 78
TROP2
Metastatic triple-negative breast cancer (TNBC) lacks specific therapeutic targets and is associated with limited treatment efficacy and poor prognosis, representing a challenging subtype in the treatment of BC. 79 In recent years, SG, which targets TROP2, has shown good clinical efficacy against TNBC. The phase III ASCENT study has shown that in patients with unresectable, recurrent, or refractory locally advanced/metastatic TNBC (who have undergone at least second-line chemotherapy), the mPFS duration (5.6 vs. 1.7 months, HR = 0.41, p < 0.001) and mOS duration (12.1 vs. 6.7 months, HR = 0.48, p < 0.001) were significantly longer with SG treatment than with monotherapy using eribuline, vinorelbine, capecitabine, or gemcitabine. The ORR of the SG group was 35%, whereas that of the chemotherapy group was 5%. The incidence of myelosuppression and diarrhea was higher after SG treatment; however, these adverse events were controllable. 80 In a phase II NeoSTAR trial involving patients with locally advanced/unresectable TNBC, among the 50 patients who received single-agent SG, the pathological CR (pCR) rate was 30%, and the ORR was 64%. With a median follow-up duration of 18.9 months, the 2-year event-free survival (EFS) was 95%, and the 2-year EFS for patients achieving pCR was 100%. This trial validates the clinical utility of SG in the neoadjuvant setting for TNBC, offering a low-toxicity, targeted neoadjuvant strategy for locally advanced TNBC and addressing the unmet need for targeted therapy in this clinical context. 81 The Ib/II phase trial MORPHEUS-pan BC, which targeted patients with inoperable locally advanced or metastatic TNBC, has shown that the ORR of the SG + atezolizumab group (30 patients) reached 76.7%, with a mPFS duration of 12.2 months and a mDOR of 14.0 months. These results were significantly better than those of the atezolizumab + albumin paclitaxel control group (ORR = 67.7%, mPFS duration = 5.9 months, and mDOR = 7.1 months). The clinical benefit rate (CBR) was 83.3% vs. 66.7%, providing a more efficient targeted + immune synergistic strategy for the first-line treatment of inoperable locally advanced or metastatic TNBC. 81 Similarly, in patients with endocrine therapy-resistant metastatic HR+/HER2-, SG showed very good efficacy, significantly prolonging the patients’ PFS and OS duration. The phase III TROPiCS-02 trial has shown that compared with chemotherapy, SG exhibited significant improvements in the mOS duration (14.4 vs. 11.2 months, HR = 0.79, p = 0.020), ORR (21% vs. 14%, OR = 1.63, p = 0.035), median time to deterioration of global health status and quality of life (4.3 vs. 3.0 months, HR = 0.75, p = 0.0059), and median fatigue (2.2 vs. 1.4 month, HR = 0.73, p = 0.0021), with good overall safety and controllability. In addition, the survival benefits in the TROP2 expression level subgroups were consistent. These data support SG as a new treatment option for patients with endocrine therapy-resistant HR+/HER2- metastatic BC. 82
GC
HER2
Currently, only RC48-ADC and T-DXd, which target HER2, have been approved for the treatment of locally advanced or metastatic GC or gastroesophageal junction (G/GEJ) adenocarcinoma. A single-arm phase II clinical study evaluated the efficacy and safety of RC48-ADC posterior-line therapy for patients with advanced G/GEJ cancer exhibiting HER2 overexpression. Among the 125 patients who received RC48-ADC (2.5 mg/kg) treatment every 2 weeks, the ORR, DCR, mPFS duration, and mOS duration were 24.8%, 42.4%, 4.1 months, and 7.9 months, respectively. The major adverse reactions were hematologic toxicity; no serious adverse reactions were observed, and no treatment-related deaths were reported. These data indicate that RC48-ADC demonstrates anticancer efficacy and controllable safety in patients with advanced G/GEJ cancer exhibiting HER2-overexpression who have previously received at least two lines of chemotherapy. 83 Although RC48-ADC monotherapy has shown clinical efficacy in HER2-overexpressing advanced G/GEJ cancers, the effectiveness of monotherapy is limited. Subsequently, an open-label, multicenter, phase I trial was conducted in three hospitals in China to evaluate the safety and efficacy of RC48-ADC plus toripalimab as a second-line and higher treatment for patients with HER2-expressing G/GEJ cancer. The results showed that in patients with G/GEJ cancer (n = 30), the confirmed ORR, mPFS duration, and mOS duration were 43%, 6.2 months, and 16.8 months, respectively. Clinical benefits were observed in both HER2-positive and low HER2-expression populations, with ORRs of 56% and 46% and mPFS durations of 7.8 and 5.1 months, respectively. The HER2-positive population did not reach mOS, while the mOS duration in the low HER2-expression group was 14.0 months. Adverse events were controllable. Thus, the combination therapy of RC48-ADC and toripalimab showed encouraging efficacy in pretreated patients with HER2-positive and low HER2-expression G/GEJ cancer. 84 An open-label randomized phase II trial (DESTINY-Gstric01) compared T-DXd and chemotherapy in patients with HER2-positive advanced GC undergoing posterior treatment. In this trial, 125 and 62 patients received T-DXd and monotherapy, respectively. The ORR of the T-DXd group was 51%, whereas that of the chemotherapy group was 14%. The mOS of the T-DXd group was also higher (12.5 vs. 8.4 months, HR = 0.59, p = 0.01). However, the toxic adverse effects of T-DXd treatment were severe and mainly manifested as myelosuppression and interstitial lung disease. 85 The DESTINY-Gstric01 study included patients from Japan and South Korea with HER2-positive locally advanced or metastatic G/GEJ cancers. The DESTINY-Gstric02 study is a single-arm phase II study that included patients with G/GEJ from 24 study sites in the United States and Europe. According to the updated data cutoff for the analysis (median follow-up duration = 10.2 months), 33 of the 79 patients (42%) reported confirmed objective responses, including 4 (5%) with CR and 29 (37%) with PR, as assessed by an independent central review. These results support the use of T-DXd as a second-line therapy for patients with HER2-positive advanced G/GEJ cancer. 86 ARX788 is a novel HER2-targeted ADC that binds to AS269 as a cytotoxic payload. In preclinical studies, in vitro and in vivo experiments have confirmed that ARX788 exerts a significant inhibitory effect on HER2-positive GC cells that are sensitive or resistant to T-DM1 as well as in mouse transplanted tumors. 87 A phase I multicenter dose-expansion clinical trial that included patients with HER2-positive advanced G/GEJ cancer (standard treatment failure) showed a confirmed ORR of 37.9% and DCR of 55.2%. The median follow-up duration was 10 months, and the mPFS and mOS durations were 4.1 and 10.7 months, respectively. The mDOR was 8.4 months. These data indicate that ARX788 exhibits good tolerability and promising anticancer activity in patients with HER2-positive advanced G/GEJ cancer. 88
Claudin 18.2
ADCs targeting claudin 18.2 are still in the exploratory stage of clinical trials. SYSA1801 is an ADC targeting claudin 18.2 and consists of a monoclonal antibody targeting claudin 18.2 and an MMAE payload. The results of the first-in-human dose-escalation and expansion study of SYSA1801 were announced at the 2023 ASCO conference, according to which, in patients with advanced solid tumors expressing claudin 18.2, the ORR was 38.1%, and the DCR was 57.1%. Among patients with GC, the ORR was 47.1%, while the DCR was 64.7%, indicating that SYSA1801 is more effective in GC treatment. 89 CMG901 is another ADC targeting claudin 18.2 that has exhibited strong preclinical anticancer activity through the MMAE-mediated bystander effect, antibody-dependent cytotoxicity, and complement-dependent cytotoxicity. A phase I dose-escalation trial evaluated the preliminary clinical efficacy and tolerability of CMG901 in patients with claudin 18.2-positive advanced G/GEJ cancer. Among the 13 patients with claudin 18.2-positive G/GEJ cancer, the ORR and DCR were 75.0% and 100%, respectively, with an ORR of 100% in the 2.6-, 3.0-, and 3.4-mg/kg Q3W dose groups. 90
Colorectal cancer (CRC) and biliary tract cancer (BTC)
HER2
Existing studies on the use of ADCs in other malignant tumors of the digestive system have mainly focused on CRC and BTC with HER2 and HER3 as the targets. These investigations are still in the clinical research stage. Approximately 2%–3% of patients with CRC experience HER2 amplification; however, HER2-targeted therapy has not yet been approved for CRC treatment. DESTINY-CRC01 is an open-label phase II study evaluating the anticancer activity and safety of T-DXd in patients with HER2-positive metastatic CRC (mCRC) after standard treatment failure. This study recruited 78 patients with HER2-expressing mCRC from 25 centers worldwide (RAS wild-type; median number of previous treatments = 4). Preliminary results from HER2-positive patients (immunohistochemistry (IHC)3+/IHC2+ and in situ hybridization (ISH)+) showed an ORR of 45.3% and an mPFS duration of 6.9 months after T-DXd treatment (6.4 mg/kg). Anticancer activity has also been observed in patients receiving anti-HER2 therapy. The safety data were consistent with those in previous reports, and caution and timely intervention are required for managing interstitial lung disease and pneumonia. 91 Although the two dose levels of T-DXd (5.4 mg/kg and 6.4 mg/kg) have shown clinical efficacy in multiple cancer types, the 5.4-mg/kg dose has not been evaluated in patients with HER2-overexpressing mCRC. DESTINY-CRC02 is a multicenter, randomized, double-blind, dual-arm, parallel phase II study that aimed to evaluate the efficacy and safety of different doses of T-DXd in patients with HER2-positive (IHC3+/IHC2+ and ISH+), RAS wild-type, and RAS mutant mCRC. The results of this trial have shown that the ORR of the 5.4-mg/kg group reached 37.8%, while that of the 6.4-mg/kg group reached 27.5%. The mOS duration of the 5.4-kg/mg group was 13.4 months, while the 6.4-mg/kg group did not reach mOS. In terms of safety, no unexpected adverse reactions were noted, which is consistent with known safety data. These findings indicate that both 5.4- and 6.4-mg/kg doses of T-DXd exert anticancer effects on HER2-positive mCRC; however, the efficacy risk ratio of the low-dose group was better than that of the high-dose group. 92 DESTINY-PanTumor02 is an open-label phase II clinical study that aimed to evaluate the efficacy and safety of T-DXd (5.4 mg/kg) for the treatment of patients with locally advanced or metastatic solid tumors with HER2 expression who have received at least one systemic therapy or no alternative therapy. The results showed that in patients exhibiting HER2 expression (IHC2+ or 3+) in BTC, the overall ORR was 22%; in the IHC3+ group, the ORR was 45.5%, with controllable safety. 93
HER3
HER3-DXd has shown promising clinical efficacy in advanced EGFR-mutant NSCLC resistant to EGFR-TKI treatment and has been explored in preclinical studies on CRC. In murine xenograft models, HER3-DXd significantly inhibited the growth of the DiFi cell line (high HER3 expression and KRAS wild-type) and SW620 cell line (high HER2 expression and KRAS mutant) xenograft tumors,; however, it exerted no effect on the Colo320DM cell line (low HER3 expression). Moreover, in comparison with the control group, HER3-DXd significantly inhibited the growth of SW620 tumors and demonstrated greater anticancer activity than the irinotecan group. These data indicate that the anticancer activity of HER3-DXd depends on HER3 expression rather than the KRAS mutation status. 94 Based on the preclinical results for HER3-DXd, we will conduct well-designed clinical studies in the future to further explore its clinical efficacy in patients with CRC and HER3 expression, potentially benefiting a larger patient population.
Urothelial cancer (UC)
HER2
The ADCs currently approved for UC include RC48-ADC targeting HER2, EV targeting Nectin-4, and SG targeting TROP2. A preclinical study has shown that RC48-ADC exerts anticancer effects by inducing G2/M phase arrest and caspase-dependent apoptosis and that its anticancer effect is positively correlated with HER2 expression in bladder cancer cell lines and organoid models. In an orthotopic bladder cancer model, intravesical instillation of RC48-ADC significantly inhibited tumor growth in comparison with disitamab, MMAE, epirubicin, or phosphate-buffered saline control. In addition, the potential toxicity of RC48-ADC intravesical instillation was evaluated by dose-escalation in normal nude mice. Intravesical instillation may be safe within the effective therapeutic dose range. 95 An open-label, multicenter, single-arm phase II clinical trial evaluated the efficacy and safety of RC48-ADC in patients with HER2-positive (IHC3+ or 2+) locally advanced or metastatic UC (mUC) after standard treatment failure. The results showed that the confirmed overall ORR was 51.2%, with mPFS and mOS durations of 6.9 and 13.9 months, respectively. Subgroup analysis suggested that RC48-ADC exhibited good clinical efficacy in patients with liver metastases and those who had previously received immunotherapy. No severe treatment-related adverse reactions (TRAEs) were observed in the study. 96 A combination of HER2-ADCs and PD-1 inhibitors is currently being investigated in clinical trials. In 2022, ASCO announced the preliminary research results for the combination of RC48-ADC with toripalimab for the treatment of locally advanced UC or mUC. This open-label, multicenter, phase Ib/II trial showed a confirmed ORR of 76.7% (10% CR) and a DCR of 96.7% at a median follow-up of 8.0 months. The ORRs for HER2 (IHC2+ or 3+) and PD-L1 (+) patients were 100%. The ORRs for HER2 (IHC2+ or 3+) and PD-L1 (-) patients were 92.3%. The ORRs for HER2 (0 or 1+) and PD-L1 (+) patients were both 50%. The ORRs for HER2 (0 or 1+) and PD-L1 (-) patients were 50%. 97 Taken together, HER2-ADC-targeted therapy has become the standard treatment for mUC, and its therapeutic effect is positively correlated with HER2 expression levels. The combination of RC48-ADC and PD-1 inhibitors can improve the efficacy of HER2-targeted therapy and is expected to replace chemotherapy as the first-line treatment for mUC.
Nectin-4
Nectin-4 is highly expressed in UC, 98 and EV is an ADC that targets Nectin-4. Effective treatment options for patients with locally advanced UC or mUC who have previously failed to respond to platinum therapy and immunotherapy are currently lacking. EV-101 is a phase I dose-escalation/expansion study that recruited 155 patients with mUC, of whom 96% had previously received PBC, and 29% had received ≥3 lines of treatment. The recommended phase II dose of EV was determined to be 1.25 mg/kg, and the most common TRAEs were of grade 1–2. In patients with mUC receiving monotherapy with EV, the confirmed ORR, DOR, mOS duration, and 1-year OS rate were 43%, 7.4 months, 12.3 months, and 51.8%, respectively. Similar ORRs and estimated mOS durations were observed in patients aged ≥75 years, regardless of whether they had received anti-PD-1/L1 therapy or had liver metastasis. 99 A global phase II single-arm study, EV-201, included 125 patients with locally advanced mUC that had progressed after PBC and anti-PD-1/L1 treatment. The confirmed ORR and mDOR in this study were 44% (CR = 12%) and 7.6 months, respectively. Subgroup analysis showed that patients with liver metastasis and no response to previous anti-PD-1/L1 therapy both had clinically significant response rates and showed tolerable safety. 100 A recent phase III global, open-label, randomized trial, EV-302, has confirmed that the use of EV combined with pembrolizumab for patients with locally advanced UC or mUC who had not previously received treatment yielded significantly better results than PBC, and its safety was consistent with that in previous reports. The study included 886 patients, and the median survival follow-up duration was 17.2 months as on 8 August 2023. In comparison with the chemotherapy group, the EV-pembrolizumab group demonstrated a longer mPFS duration (12.5 vs. 6.3 months, HR = 0.45, p < 0.001) and mOS duration (31.5 vs. 16.1 months, HR = 0.47, p < 0.001). Furthermore, 55.9% of those in the chemotherapy group and 69.5% of those in the EV-pembrolizumab group experienced grade ≥3 TRAEs. 101
TROP2
In addition to HER2 and Nectin-4, another important target in UC is TROP2, which is highly expressed in UC tissues. 102 The ADC drug, SG, which targets TROP2 has shown promising anticancer activity in early clinical trials. Research has shown that among six extensively pretreated patients with metastatic, platinum-resistant UC treated with SG, three exhibited clinically significant responses (PFS duration =6.7–8.2 months and OS duration = 7.5–11.4 months), with good treatment tolerability. 103 Based on these results, a multicohort, open-label, phase II clinical study, TROPHY-U-01, was subsequently initiated, in which cohort 1 evaluated the efficacy and safety of SG treatment for patients with locally advanced or mUC who exhibited disease progression after platinum and/or immune checkpoint inhibitor therapies. The results showed that at a median follow-up of 10.5 months, the ORR, mPFS duration, and OS duration of 113 patients were 28%, 5.4 months, and 10.9 months, respectively. The safety data were consistent with the known toxicity characteristics of SG, and overall, treatment with SG was safe and controllable. 104
OC
FRα
The ADC currently approved for OC is MIRV, which targets FRα. FRα is usually overexpressed in OC. 105 In preclinical studies, MIRV was developed by screening the best anti-FRα antibody and the most effective conjugate IMGN853. Its significant antitumor activity, which was proportional to the FRα expression level on the surface of OC cells, was confirmed in in vitro and in vivo experiments. 106 A phase Ib escalation study confirmed that MIRV combined with carboplatin is a well-tolerated and highly active treatment option for recurrent platinum-sensitive OC with FRα positivity. The study recruited 18 patients who received combination therapy, 13 of whom continued to receive MIRV maintenance therapy after carboplatin discontinuation. Among all the evaluable patients (n = 17), the confirmed ORR was 71% (CR in three patients and PR in nine patients), with an mPFS duration of 15 months. The treatment response was mild. 107 Another phase Ib study has confirmed that the combination of MIRV and bevacizumab also exhibits good tolerability and encouraging efficacy in patients with FRα-positive, recurrent platinum-resistant OC. In total, 66 patients received full-dose MIRV and bevacizumab, and the confirmed ORR and mPFS duration were 39% (CR in five patients and PR in 21 patients) and 6.9 months, respectively. Subgroup analysis showed that in the population that underwent initial treatment with bevacizumab, received few lines of treatment, and showed moderate/high FRα expression (n = 16), the ORR could reach 56%, and the mPFS duration was 9.9 months. Adverse events were mostly of grade 1–2. 108 SORAYA is a single-arm phase II study that recruited 106 patients with platinum-resistant OC that showed high FRα expression who had been treated with 1–3 therapies in the past; all patients had received bevacizumab treatment. The median follow-up duration was 13.4 months, and the ORR with MIRV monotherapy was 32.4% (CR in five patients and PR in 29 patients), with an mDOR of 6.9 months, indicating consistent antitumor activity and good tolerability of MIRV in patients with platinum-resistant OC. 109 The Phase III MIRASOL (GOG 3045/ENGOT-ov55) study compared the clinical efficacy of MIRV and standard chemotherapy in patients with recurrent platinum-resistant OC showing high FRα expression. The results showed that MIRV treatment achieved statistically significant results for mPFS duration (5.62 vs. 3.98 months, HR = 0.65, p < 0.0001), ORR (42.3% vs. 15.9%, p < 0.0001), and mOS duration (16.46 vs. 12.75 months, HR = 0.67, p = 0.0046) in comparison with the chemotherapy group. The adverse events associated with MIRV were consistent with those described in previous reports and were mostly mild. The excellent efficacy and safety data of MIRV make it a new standard treatment for platinum-resistant OC with high FRα expression. 110
MUC16/NaPi2b/ephrin-A4 (EFNA4)/Claudin-6
The application of ADCs targeting other antigens in OC remains in the active preclinical and clinical exploration stages. MUC16 is a tumor-specific antigen overexpressed in OC. 111 Sofituzumab vedotin (SV) is an ADC formed by targeting MUC16 and conjugating to MMAE. A phase I trial evaluated the efficacy and safety of SV treatment in patients with advanced recurrent platinum-resistant OC. The results showed that confirmed responses were only observed in patients with OC who tested positive for MUC16 (IHC2+ or 3+), with a duration of >6 months and acceptable safety. 112 Lifastuzumab vedotin (LIFA) is an ADC that targets NaPi2b and exerts its antitumor effects by conjugating with MMAE. In one clinical study involving 95 patients with platinum-resistant OC who were randomly assigned to receive LIFA or PEGylated liposomal doxorubicin (PLD) treatment, in the ITT population, the mPFS durations in the LIFA and PLD groups were 5.3 and 3.1 months, respectively, and the ORRs were 34% and 15%, respectively. In patients showing high expression of NaPi2b, the mPFS in the LIFA and PLD groups was 5.3 months and 3.4 months, respectively, with ORRs of 36% and 14%, respectively. Thus, the safety of the treatment process was controllable. The results showed that in comparison with PLD, although LIFA had good tolerance and improved ORR in platinum-resistant OC, the mPFS duration did not indicate a statistically significant prolongation, and high expression of NaPi2b did not improve the therapeutic effect of LIFA. 113 PF-06647263 is a novel ADC targeting EFNA4. The first-in-human phase I study preliminarily confirmed that PF-06647263 had limited antitumor activity in patients with advanced OC, and no correlation was observed between tumor response and EFNA4 expression levels. 114 Claudin-6 expression is elevated in OC, whereas little or no detectable expression is observed in normal adult tissues. A novel ADC, CLDN6-23-ADC, developed on the basis of this target, has shown potential antitumor activity in preclinical studies and has significantly inhibited tumor growth in a mouse model of human OC. It is currently being evaluated in a phase I study. 115
CC
TF
TF is usually highly expressed in CC and is associated with a poor prognosis, 116 making it a potential target for CC treatment. TV is an ADC that targets TF and has been approved for the treatment of recurrent or metastatic CC (r/mCC). In the innovaTV201 phase I/II study, 55 patients with r/mCC received TV treatment with a BICR-confirmed ORR of 22%, mDOR of 6.0 months, and 6-month PFS rate of 40%. The treatment safety was controllable. Although TF expression is observed in most cases of CC, it showed no significant association with response. 116 Subsequently, a multicenter, open-label, single-arm, phase II study (innovaTV204/GOG-3023/ENGOT-cx6) conducted in 35 centers in Europe and the US further confirmed the clinically meaningful and persistent antitumor activity, with controllable and tolerable safety, of TV in patients with r/mCC who had previously received standard treatment. The study included 102 CC patients receiving TV treatment, demonstrating a median followup duration of 10.0 months, confirmed ORR of 24% (7% CR and 17% PR), mPFS duration of 4.2 months, and mOS duration of 12.1 months. 117 In addition, a single-arm, open-label phase I/II trial evaluated the safety and efficacy of TV use in Japanese patients with r/mCC. The results showed that the confirmed ORR, mDOR, and median response time were 29.4%, 7.1 months, and 1.2 months, respectively, indicating that in comparison with the findings in European and USA populations, TV also showed manageable and tolerable safety and efficacy characteristics in Asian r/mCC patients. 118 TV not only demonstrates good safety and efficacy in posterior-line monotherapy for r/mCC, but also shows controllable safety and encouraging antitumor activity when used in combination with bevacizumab, carboplatin, or pembrolizumab in treatment-naive and previously treated r/mCC. InnovaTV 205/GOG-3024/ENGOT-cx8, an open-label, multicenter Ib/II study, aimed to evaluate the efficacy and safety of TV in combination with bevacizumab, pembrolizumab, or carboplatin for the treatment of r/mCC. In total, 142 patients were included in this study. In dose-escalation trials (n = 101), the ORR for TV combined with carboplatin as the first-line treatment was 54.5% (18/33), that for TV combined with pembrolizumab as the first-line treatment was 40.6% (13/32), and that for TV combined with pembrolizumab as the second- or third-line treatment was 35.3% (12/34). The mDOR in these three groups was 8.6 months, not reached, and 14.1 months, respectively, indicating a lower incidence of TRAEs. 119 A phase III, multicenter, open-label trial (innovatTV 301) compared the efficacy of TV and chemotherapy as second- or third-line therapies for patients with r/mCC. The results showed that in comparison with the group receiving chemotherapy, the TV group showed significantly better mOS duration (11.5 vs. 9.5 months, HR = 0.70, p = 0.004), mPFS duration (4.2 vs. 2.9 months, HR = 0.67, p < 0.001), and ORR (17.8% vs. 5.2%, OR = 4.0, p < 0.001); the adverse reactions were tolerable. 120
HER2/TROP2/Nectin-4
In addition to TF, many targets are being explored in clinical trials for CC, including HER2, TROP2, and Nectin-4. The preliminary results of the C018 study (a single-arm, multicenter, open-label, phase II study) were orally reported at the 2024 ESGO. Among the 22 HER2-expressing r/mCC patients who had previously received immunotherapy or PBC, the mPFS duration of the patients who received RC48-ADC monotherapy was 4.37 months, mDOR was 5.52 months, mOS was not achieved, 12-month OS rate was 66%, and ORR was as high as 36.4% (1 CR and 7 PR), with controllable safety, indicating that RC48-ADC performed fairly well in terms of efficacy and safety as a single-agent therapy in HER2-expressing r/mCC patients with disease progression after treatment. 121 TROP2 is expressed in various solid tumors, and a previous study has reported an expression rate of >90% in CC. 122 Therefore, ADCs targeting TROP2 are expected to become new targets in CC therapy. Preclinical studies have shown that SG targeting TROP2 exhibits significant antitumor activity both in vitro in CC cell lines and in vivo in transplant tumor trials. 123 The mid-term analysis results of a multicenter, single-arm, multicohort clinical phase II “Basket Trial” (EVER-132-003 study) with a CC cohort were presented orally at the 2024 SGO. In patients with r/m CC who were resistant or intolerant to platinum and paclitaxel drugs, SG monotherapy demonstrated good antitumor activity, and no new safety signals were found. The median followup duration was 10.5 months, with an ORR of 50% as evaluated by the researchers; the DCR was up to 94%, mDOR was 9.2 months, mPFS duration was 8.1 months, and PFS rate was 58% at 6 months. In addition, among patients who had received immunotherapy in the past, the therapeutic benefits of SG were similar to those in the entire study population. 124 Similarly, a multicenter, open-label, phase I/II study of a novel ADC 9MW2821 targeting Nectin-4 for the treatment of patients with r/m CC was reported at the 2024 SGO. Among 37 Nectin-4-positive r/m CC patients with assessable efficacy, the median follow-up duration was 5.60 months, ORR was 40.54%, and DCR was 89.19%. One patient achieved CR (2.70%) and 14 achieved PR (37.84%). The median PFS duration, OS duration, and DOR were not reached. Based on the good antitumor activity and acceptable safety demonstrated by 9MW2821 monotherapy in previously treated patients with r/m CC, further clinical research is necessary. 125
Toxicity, drug resistance, and optimization strategies of ADCs
The use of ADCs in the treatment of solid tumors is becoming increasingly widespread. 20 Whether used as a monotherapy or in combination with other drugs, they play an important role in both first-line and posterior-line therapy, 126 which is another important antitumor therapy after radiotherapy, chemotherapy, targeted therapy, and immunotherapy. However, ADCs still face many challenges, such as off-target drug toxicity, drug resistance, poor permeability in solid tumors, and suboptimal performance. The toxic reactions of ADCs mainly include allergic reactions, hepatotoxicity, hematopoietic myelosuppression, neurological disorders, gastrointestinal reactions, fatigue, and rash.28,34,127–134 Drug resistance to ADCs is complex and diverse, and its potential mechanisms remain unclear. Existing evidence suggests that tumor cells develop ADC resistance through various mechanisms, such as decreased antigenic expression intensity, increased antigenic expression ratio, and enhanced cytotoxic metabolism and clearance rates of the payload.135,136 In addition, the antibody components of ADCs may trigger immune responses, accelerating drug clearance and promoting the emergence of drug resistance. 137
An ADC is a precise drug-delivery system consisting of highly targeted antibodies, well-designed linkers, and highly active payloads. 138 Owing to technological changes, new-generation ADCs have superior specificity and cytotoxicity than those from previous generations. However, the complex composition of ADCs poses certain challenges for their development and optimization (Figure 4). Future ADC-optimization strategies may focus on the following aspects.

Challenges and optimization strategies involved with the used of ADCs. ADCs: antibody–drug conjugates.
First, targeting of mutant proteins. Studies have shown that ADC internalization and intracellular transport pathways play crucial roles in cytotoxicity. 139 In comparison with wild-type proteins, mutant proteins typically have higher levels of ubiquitination and are easier to internalize and degrade. 140 Therefore, ADCs targeting oncogenic mutant proteins (such as certain EGFR mutants) can maximize treatment specificity and reduce off-target possibilities. 141 A preclinical study conducted in 2024 developed the ADC PD0721-DOX, which targets EGFRvIII. EGFRvIII is overexpressed in 80% of human malignancies, including glioblastoma, LC, and GC, but is not abnormally expressed in normal tissues. This ADC utilizes a specific single-chain antibody as a carrier conjugated to doxorubicin (DOX). Experimental evidence has demonstrated that the ADC efficiently internalizes into tumor cells through specific binding to the EGFRvIII antigen on the tumor cell surface, releases DOX after lysosomal degradation, and selectively kills tumor cells expressing EGFRvIII without significant toxicity to cells not expressing this mutant, showcasing the high precision therapeutic potential for mutant proteins. 142
Second, use of dual epitopes or dual targets. Advancements in bispecific antibody technology have introduced more possibilities for the innovation of ADCs, which can improve antibody internalization and enhance tumor specificity. Bispecific ADCs that target different sites on the same antigen can improve receptor aggregation and accelerate target internalization. 143 ZW49 is a bispecific ADC in clinical development that simultaneously targets two nonoverlapping epitopes of the HER2 receptor. This design aims to enhance the endocytotic efficiency and lysosomal delivery of the ADC. An ongoing Phase I clinical trial (NCT03821233) for the treatment of HER2-positive solid tumors has demonstrated its favorable antitumor activity. 144
Third, conjugating two different payloads. Dual-payload ADCs can achieve greater therapeutic effects by precisely controlling the ratios of the two drugs. 145 In addition, synergistic effects of the two payloads can significantly reduce drug resistance. 146 A 2023 study focused on HER2-positive cancer. The researchers conjugated trastuzumab with two distinct microtubule inhibitors, MMAE and DM1, forming dual-drug-loaded ADCs that demonstrated synergistic cytotoxicity in vitro, outperforming both monotherapy and single-drug ADCs. 145
Fourth, develop peptide–drug conjugates (PDCs). We abandoned the traditional monoclonal antibody (mAb) structure and conjugated the payload to peptide fragments with lower molecular weights to develop PDCs. The penetration efficiency was further improved by reducing the molecular weight. 147 BT8009 is a “bicycle toxin conjugate” targeting Nectin-4, belonging to the class of PDCs. It consists of a bicyclic peptide that binds to coagulation peptide 4, a cleavable linker, and a cytotoxic payload, MMAE. Preclinical studies have demonstrated that BT8009 can effectively penetrate tumor tissues and exhibit antitumor activity. 148
Fifth, overcoming ADC barriers. Owing to the antigen barrier, monoclonal antibodies cannot easily diffuse into solid tumor tissues. 149 To solve this problem, noninternalized antibodies can be developed to allow the payload to be released directly outside the tumor microenvironment under reducing conditions and then spread to the inside of cancer cells. In addition, the stromal barrier can be overcome by enhancing stromal penetration and cancer stromal targeting therapy. Studies have shown that inhibiting Hedgehog signaling or matrix cross-linking enzymes can reduce the extracellular matrix and enhance the penetration of anticancer drugs.150,151 Owing to the enhanced permeability and retention effect, ADCs targeting the extracellular matrix accumulate in the cancer stroma and then bind to specific antigens in the stroma to form scaffold structures, effectively and continuously releasing free anticancer drugs.152–154 A pivotal preclinical study has developed an ADC targeting tumor-specific fibronectin splice variants (extra domain B splice variant of fibronectin). The study has demonstrated that this ADC exhibits significant antitumor activity in multiple tumor models, with a mechanism of action distinctly different from those of traditional ADCs dependent on internalization. The feasibility of a noninternalization strategy was validated through optimization of the antibody affinity, linker, and payload. 155
Sixth, selection of appropriate payloads. The currently used cellular toxins have certain limitations. As research progresses, newer cellular toxins with higher activity and fewer adverse effects will be identified, which will further improve the efficacy and tolerability of ADCs.
Finally, to overcome the toxicity and resistance of ADCs, combined use with other therapies such as immunotherapy, targeted therapy, and radiation therapy can be considered, which can not only reduce toxicity by reducing the dosage of ADCs, but can also attack tumor cells at multiple levels, improving treatment efficacy and reducing drug resistance.133,156 In urothelial carcinoma, the combination therapy of EV and the immunotherapy drug, pembrolizumab has demonstrated remarkable efficacy. This regimen has been approved by the FDA as a first-line treatment for locally advanced or metastatic urothelial carcinoma patients who are ineligible for cisplatin chemotherapy, establishing it as a benchmark case for the combined use of ADC and immunotherapy. 157 A preclinical study has shown that the targeted drug, adavosertib, can enhance the antitumor activity of T-DXd in HER2-expressing cancer models, highlighting the superior efficacy of ADC therapy combined with targeted therapy. 158 In the context of ADC combined with radiotherapy, a preclinical study has demonstrated that ADCs can increase tumor cell sensitivity to radiotherapy; thus, radiosensitizer-ADCs can enhance radiation efficacy and improve survival rates in various tumor models. 159
Discussion
Compared with other published reviews on ADCs, most studies tend to focus on ADCs targeting a single specific antigen,30,160–162 or the combination therapy of ADCs and other treatment modalities.159,163 By contrast, very few reviews, such as ours, have comprehensively explored the clinical applications of commonly used ADCs across a broad range of prevalent solid tumors. Our review adopts a much broader and more expansive scope and provides clear, detailed illustrations of representative ADCs corresponding to specific targets in relevant malignancies rather than employing simple lists. This narrative style renders the content more accessible and intuitive, better demonstrates the cross-tissue activity of ADCs, and endows this review with stronger integrative characteristics.
Nevertheless, this review also has certain limitations. Due to manuscript length constraints, we could not include all potential targets and their corresponding ADCs. Furthermore, as a narrative review, our literature screening process may be subject to a certain degree of selection bias. We mainly included rigorously screened clinical trials, resulting in an inaccurate representation of the complexity of real-world clinical settings. Consequently, we did not discuss the performance of ADCs in patients with comorbidities and other complex clinical conditions. Future studies should focus on integrating patient conditions within complex clinical settings.
Summary and prospects
ADCs are a new and effective method for treating malignant tumors following chemotherapy, targeted therapy, and immunotherapy. With their high targeting ability and wide therapeutic window, ADCs open up a broad-spectrum antitumor treatment model that is expected to lead to a new era of precision therapy. However, the use of ADCs is also associated with many challenges, such as toxicity, inadequate efficacy, and drug resistance. In this review, we systematically analyzed and discussed the functions, development history, and clinical applications of ADCs in the treatment of common solid tumors and proposed future optimization strategies based on the toxicity, efficacy, and drug resistance of ADCs.
Although ADCs have achieved significant results in clinical applications, their clinical benefits and objective response durations are limited by several factors. As a novel drug-coupling therapy, PDCs offer advantages over ADCs in many aspects, including pharmacokinetic properties, duration of drug efficacy, molecular safety, and efficacy. In comparison with ADCs, PDCs exhibit smaller molecular weights, faster internalization rates, improved tumor permeability, and lower immunogenicity, which endow them with stronger anticancer activity and better safety in the treatment of solid tumors. In the near future, with advancements in medical technology and clinical research, ADCs and PDCs will expand the therapeutic options for cancer treatment, benefiting more cancer patients in clinical practice.
Supplemental Material
sj-xlsx-1-imr-10.1177_03000605261434386 - Supplemental material for Clinical application and optimization strategies for antibody–drug conjugates in solid tumors
Supplemental material, sj-xlsx-1-imr-10.1177_03000605261434386 for Clinical application and optimization strategies for antibody–drug conjugates in solid tumors by Jie Qu, Xiaoyue Zhao, Linzhu Zhang, Wuyou Cai, Minfang Shen, Xiaofan Liu, Xiaowei Wei and Fei Fei in Journal of International Medical Research
Footnotes
Acknowledgments
We are grateful for the professional language editing services provided by Elsevier.
Author contributions
JQ, JW, LZ, and WC researched the data and wrote the original manuscript. MS and XL designed the figures and tables. XW and FF designed the concept, oversaw the project, and provided expert revisions to the manuscript. All authors made substantial contributions to content discussions and reviewed and/or edited the manuscript before submission.
Data availability statement
All data supporting the study’s conclusions are available in the article, and no new undisclosed experimental data were generated or analyzed herein.
Declaration of competing interests
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Ethics approval and consent to participate
Not applicable.
Funding
This study was supported by the National Natural Science Foundation of China (82203182), Project of Future Technology Star of Nanjing First Hospital, and General Project of Zhejiang Shuren University (Grant No. 2020A11001).
Human and animal rights and informed consent
This article does not report any studies involving human or animal participants performed by any of the authors.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
