Abstract
Introduction
Laryngeal cancer ranks as the second most prevalent respiratory neoplasm after lung cancer, with squamous cell carcinomas (SCCs) constituting over 90% of laryngeal tumors.1,2 Treatment of laryngeal carcinoma depends on the staging of the disease as well as patient and surgeon preference. Stage III and IV laryngeal carcinomas are considered advanced. The standard of care for locally advanced laryngeal cancers that are not suitable for organ-preservation techniques, such as larynx-preservation surgery or combined chemoradiation, is total laryngectomy (TL) with neck dissection guided by the status of lymph node involvement.3,4 TL may also be performed in advanced cases of hypopharyngeal SCC, which accounts for around 3% to 7% of head and neck cancers. 5 The current National Comprehensive Cancer Network (NCCN) guidelines for tumors of the larynx and hypopharynx indicate that the choice between TL and conservative laryngeal surgery is made collaboratively by the surgeon and patient. The treatment approach should aim for curative intent, focusing on complete tumor removal and, whenever feasible, preserving laryngeal function. The NCCN also recommends thyroidectomy along with laryngectomy for advanced staged laryngeal and hypopharyngeal carcinomas. The presumed purpose of thyroidectomy in conjunction with TL is due to concern of direct extension or lymphatic spread to the gland. Better visualization of the central neck compartment has also contributed to variability of thyroid removal with advanced disease. 6
Despite the NCCN recommendations to perform thyroidectomy with TL, this surgical management remains controversial. Multiple studies have shown that thyroid gland invasion only occurs in around 8% to 11% of advanced laryngeal carcinomas, with most of these involving direct invasion from tumors with subglottic extension greater than 10 mm. 6 Patients undergoing TL have been shown to have a 31.9% incidence of postoperative hypothyroidism with thyroid lobectomy which increases to 88% incidence with adjuvant radiation.7,8 Lifelong thyroid hormone replacement can potentially have detrimental effects on patients’ health and quality of life (QOL). 9 Considerations must also be given to the direct injury, devascularization, or erroneous removal of the parathyroid glands during thyroidectomy leading to complications such as hypocalcemia. Transient hypocalcemia (lasting 6 months or less) has a reported incidence ranging from 0.3% to 42% post-thyroidectomy, while the incidence of persistent hypocalcemia (lasting longer than 6 months) ranges from 0% to 13%. 10 Hypocalcemia can lead to paresthesia, muscle cramps, tetany, seizures, arrhythmias due to prolonged QT interval, and even death.11-13 It is therefore imperative to preserve the thyroid and parathyroid glands when possible because many patients need thyroid replacement treatment indefinitely, as well as monitoring and supplementation of calcium. 14
The decision to preserve the thyroid during TL is surgeon dependent at our institution. We therefore sought to explore this unique dataset comparing disease outcomes between patients undergoing TL with or without thyroidectomy. The goal of this study was to investigate the rate of recurrence and survival outcomes in patients who received thyroidectomy with TL compared to patients with preserved thyroid glands. We also sought to investigate factors significantly associated with thyroid invasion to add to existing literature. These data can serve as an aid in considerations for surgical management of the thyroid in advanced staged laryngeal and hypopharyngeal SCC in the future.
Methods
Patient Selection
All research was conducted at a single tertiary referral center after obtaining approval by the University of Arkansas for Medical Sciences Institutional Review Board (IRB: 274369). Our electronic medical record was queried for patients who underwent TL between 2014 and 2022. Patients with laryngeal or hypopharyngeal SCC were included for retrospective chart review. Patients who underwent partial laryngectomy or functional laryngectomy were excluded. Patients were stratified based on their treatment into 4 distinct groups: hemithyroidectomy (HT), total or completion thyroidectomy (TT), isthmusectomy (I), and no thyroid resected (NT). The comparison among these groups was conducted utilizing Kruskal-Wallis and Jonckheere-Terpstra tests. Furthermore, data analysis was conducted between the NT cohort and those with any form of thyroid resection (T). A comparison between NT and T was performed using Mann-Whitney and Kolmogorov-Smirnov tests for independent samples. Patient demographics including age, sex, race, medical comorbidities, and modified frailty index were collected for each cohort.
History of prior head and neck cancer treated with surgery or radiation therapy was also noted. Information was gathered on preoperative imaging modalities and their interpretation of tumor location and subsites involved, including thyroid gland invasion. Furthermore, patients were categorized based on whether they presented with thyroid gland invasion or not, and their surgery-specific information such as resection of pharynx, neck dissection, and type of thyroidectomy performed was recorded. Pathologic staging, margin status, and detailed involvement of specific subsites were gathered. Postoperative information that was collected included adjuvant therapies recommended and completed, recurrence, and last known follow-up.
Statistical Analysis
Statistical analysis was performed using SPSS Statistics v29.0 (IBM Corp). Means and standard deviations (SDs) as well as minimums and maximums were calculated for continuous variables. Chi-square analysis was used for comparison of categorical variables with use of Fisher’s exact test in cases of low numbers. Kruskal Wallis, Jonckheere-Terpstra, Mann-Whitney, and Kolmogorov-Smirnov tests were performed for ordinal variables. A P value of <.05 considered statistically significant.
Results
A total of 147 patients underwent TL, with or without thyroidectomy, for laryngeal or hypopharyngeal SCC and were included in the study. Of these 147 patients, the majority were male (83%, N = 122) and white (82%, N = 120). The supraglottis was the most common tumor site (46%, N = 67), and the majority of the patients were stage IVA (60%, N = 88). Ninety-nine patients (67%) had thyroid resected during TL while 48 patients had NT (33%). Of the 99 patients with thyroid resected, 60 patients underwent HT, 35 underwent TT, and 4 had an I. Data were run between the following 4 patient groups: HT, TT, I, and NT. Data were also run between the cohort of NT versus any thyroid resected (T). See Table 1 for demographic information. There was no significant difference of recurrence rates between the 4 groups (P = .087) or between NT and T (P = .614). There was no significant difference of deaths from recurrent disease between the 4 groups (P = .156) or between NT and T (P = .112). The average follow-up for the entire 147 patients was 519 days (±SD 542). There was no significant difference of follow-up between the 4 groups (P = .612).
The Baseline Demographic and Clinical Characteristics for Each Group.
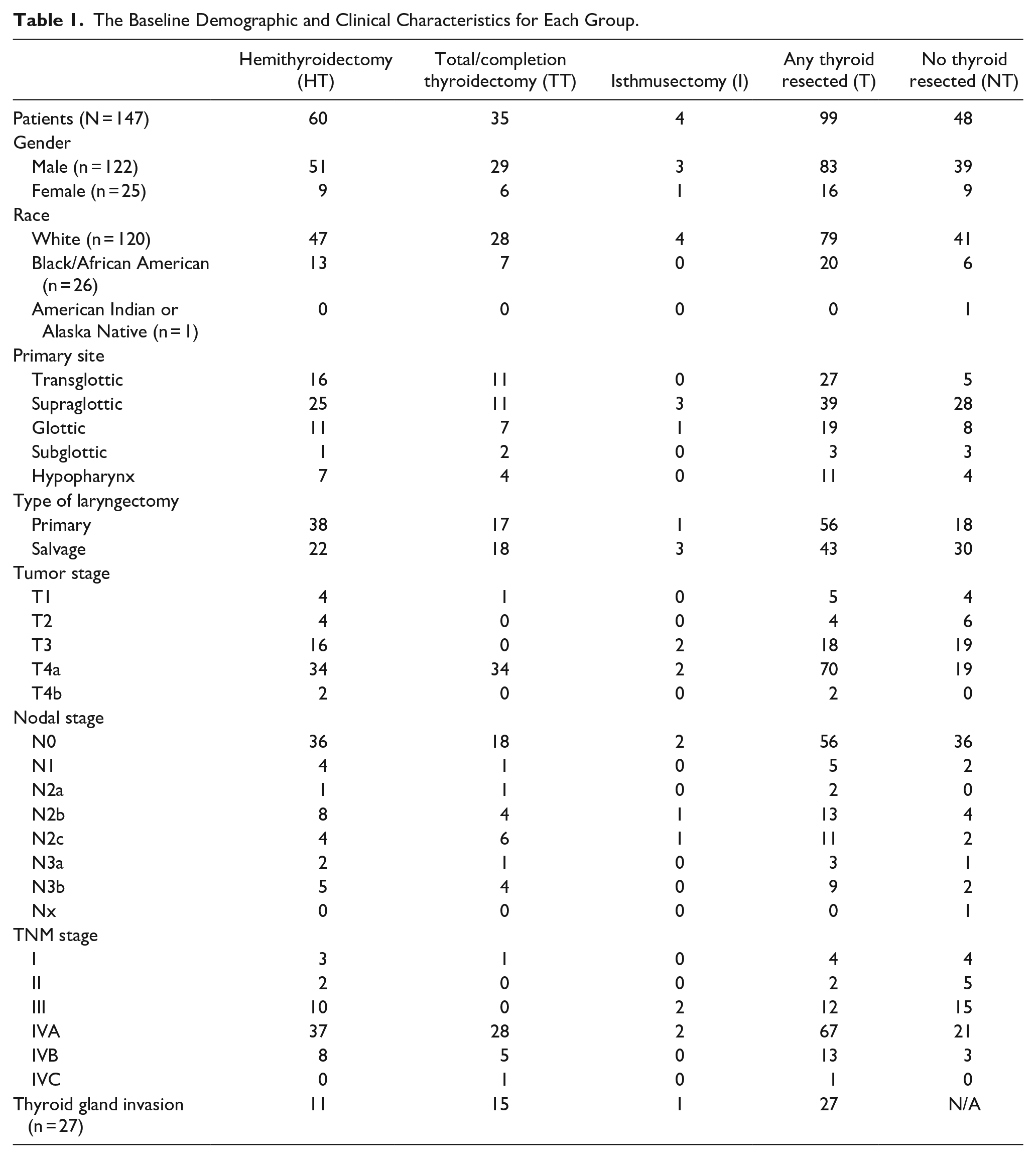
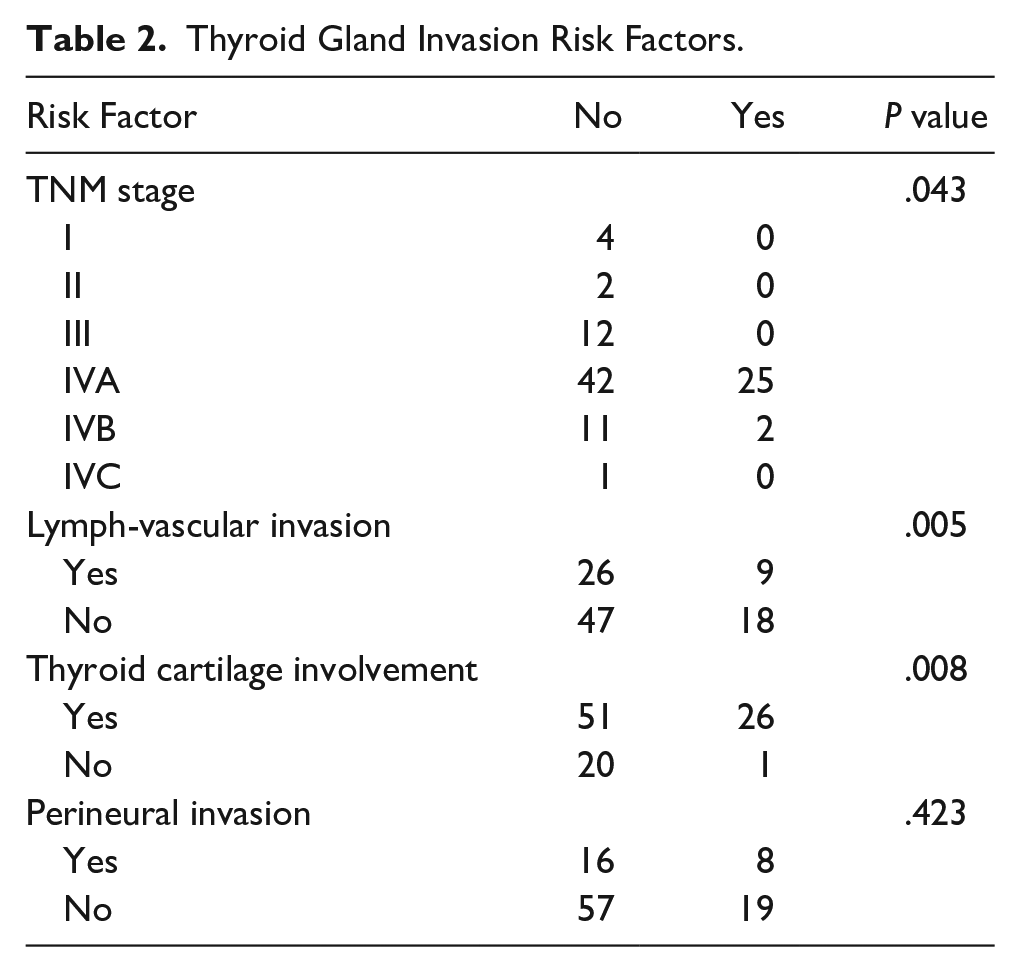
There was a significant difference between T staging and TNM prognostic staging between the 4 groups (P = .001 and P = .013) and between NT versus T (P = .003 and P = .004). The cohorts TT and T were more likely to be tumor stage IV and TNM staging IVA. Of the 99 patients who had thyroid resected (T), 27 of them had positive gland involvement, 26 of which were from direct extension of disease. Factors associated with thyroid gland involvement included TNM stage IV (P = .043), T4 tumor (P = .003), lymphovascular invasion (P = .005), and thyroid cartilage involvement of the tumor (P = .008). See Table 2 for thyroid gland invasion risk factors.
Thyroid Gland Invasion Risk Factors.
Discussion
Total laryngectomy for advanced stage laryngeal and hypopharyngeal SCC is the standard of care, although management of the thyroid gland in these cases remains clinically controversial.15,16 In 1955, Pogura was the first to describe thyroid gland involvement as a metastatic feature of laryngeal cancer, recommending its routine removal. 17 Certain studies indicate a low incidence of thyroid gland invasion, advocating for thyroid preservation in numerous cases placing emphasis on the potential risks associated with thyroidectomy.16,18-20 Other studies reported incidence of thyroid gland involvement in laryngopharyngeal cancer ranges from 0% to 23%, a variation comparable to the 27% observed in this study.17,21,22 These findings have led some to advocate for prophylactic thyroidectomy to reduce the chance of recurrence and to resect clinically occult thyroid gland involvement.8,23 Several authors have advocated for selective thyroidectomy during TL.24,25 Brennan et al proposed selective criteria, such as tumorous involvement of the anterior commissure, subglottic space, and T3 to T4 transglottic laryngeal cancer. 25 Sparano et al recommended HT for patients with preoperative fixation of the ipsilateral true vocal cord or a subglottic extension of the tumor greater than 15 mm. 24 Our study aims to provide further insight for the management of the thyroid in patients with laryngeal and hypopharyngeal SCC.
The extent of thyroidectomy also remains a topic of contention among authors as well. Some suggest performing ipsilateral HT when indicated for patients with laryngopharyngeal cancer, with the decision based on factors like frozen section examination or intraoperative evidence of thyroid gland involvement. They caution against routine total thyroidectomy, citing concerns about lifelong thyroid hormone replacement and the risk of hypocalcemia, arguing that it may be excessive treatment for these patients.21,26,27 Conversely, some studies suggest total thyroidectomy in patients with laryngopharyngeal cancer to ensure adequate surgical margins, given the possibility of microcarcinoma in the contralateral thyroid lobe. 28
Similar to what is found in the literature, our study found that factors significantly associated with thyroid gland invasion were TNM stage IV, T stage of T4, lymphovascular invasion, and thyroid cartilage involvement of the tumor. A common theme among other studies is the correlation between subglottic extension of the primary tumor and thyroid gland involvement. While our study did not demonstrate this finding, it is important to recognize data surrounding these findings. In a retrospective review of 9 years of data by Pasha et al, 12 of 130 (9.2%) of excised thyroid glands were involved by SCC and 7 of these 12 (58.3%) also had subglottic involvement. 29 Another study by Gaillardin et al in 2012 demonstrated that subglottic extension greater than or equal to 10 mm was statistically correlated with histological thyroid gland invasion (P < .01). 30 Attention to subglottic extension on preoperative imaging and histology can help guide management of the thyroid gland.
Laryngopharyngeal cancer can involve the thyroid gland either through direct extension or indirectly via lymphatic or vascular structures. The isthmus of the thyroid gland typically lies anterior to the second, third, and fourth tracheal rings. Consequently, direct invasion of the thyroid gland occurs due to proximity.23,31 Histopathologic studies indicate that cancer spread appears to follow collagen bundles where connective tissue membranes attach to the cartilage. As the cancer progresses, it enlarges the collagen bundles, creating a direct pathway for cancer spread through the perichondrium. 32 Typically, the perichondrium serves as an excellent protective barrier against the spread of carcinoma. However, once the carcinoma breaches this barrier, it may propagate in a subperichondrial plane within the cartilage, thereby increasing the likelihood of extralaryngeal spread. There is a defect in the inner perichondrium at the anterior angle of the thyroid cartilage, where Broyles’s ligament penetrates, offering a pathway for tumor spread.29,33 Kim et al reported that the anterior commissure was involved in all cases where laryngeal cancer invaded the thyroid gland. Due to its rich vascularization, the thyroid gland possesses an extensive lymphatic network, and involvement through lymphatic invasion, though less common, still presents significant risk. 23 Brennan et al discovered that the majority of thyroid gland invasion by laryngeal SCC (78%) occurred via direct extension, with only 10% (11/107) of cases attributed to lymphatic spread. 25
Thyroid cartilage involvement has also been regarded as a potential risk factor for thyroid gland involvement, leading to consideration for thyroid lobectomy. However, it has not been confirmed as an independent risk factor.24,27,34 In our cohort, we noted a significant association between tumor involvement of the thyroid cartilage and thyroid gland involvement (N = 26, P = .011). In Buckley et al’s histopathologic study of laryngeal cancer spread, they found that 65% (19/29) of cases with invasion of the laryngeal framework occurred through the thyroid cartilage. 35 Biel et al discovered that 100% (11/11) of cases with thyroid gland involvement also exhibited involvement of the thyroid cartilage. 22 Harrison et al reported that thyroid cartilage invasion is believed to occur in areas where cartilage has undergone ossification. Furthermore, Olszewski et al injected blood vessels with silicone rubber during laryngectomy to map their behavior. They concluded that cancer primarily spread through neo-ossified cartilage, with minimal involvement of nonossified avascular cartilage. 36 Therefore, in the aging larynx with ossification changes, these centers serve as potential routes for the direct extension of laryngeal tumors.
Few studies discussing the prognosis of cases with thyroid gland invasion suggest a dismal outlook for these patients. 37 Gilbert et al mentions that the survival in a group of patients with thyroid gland involvement was poor, with (18/23) patients dying of their disease within 3 years. 37 Nayak et al reported a 5 year survival rate of 30% for T4a cases, which includes those with thyroid gland invasion. 20 However, they found that the difference in survival between patients with thyroid gland invasion and those with invasion of other adjacent structures was statistically insignificant. The relatively high rate of thyroid gland invasion in our cohort and poor prognostic factor associated with involvement of thyroid gland suggests that thyroidectomy may be a necessary component of TL to ensure complete tumor resection and to potentially reduce the risk of recurrence. This finding is particularly relevant for patients with advanced disease or specific tumor characteristics that increase the risk of thyroid gland involvement.
When surgeons decide whether or not to perform thyroidectomy, considerations must also be given to outcomes associated with thyroid gland removal. Thyroidectomy can lead to hypothyroidism as well as hypoparathyroidism secondary to incidental parathyroid gland removal.38,39 A study on hypothyroidism after treatment for nonthyroid head and neck cancer found that the incidence of hypothyroidism after TL and radiotherapy was 61%. They demonstrated that the incidence of hypothyroidism after TL is significantly higher than the incidence in other treatments for head and neck cancer (P < .001). 40 It is important to consider the possibility of hypothyroidism as a consequence of thyroid removal because it can lead to constipation, fatigue, cold intolerance, depression, weight gain, hair loss, and cardiovascular disease. Untreated hypothyroidism may increase morbidity and mortality, and can even lead to coma and death. 41 Rates of postoperative hypoparathyroidism following thyroid surgery has been shown to vary between 0% and 46%, though a study by Edafe et al demonstrated that transient hypocalcemia can occur in up to 100% of patients after TL. 42
In a meta-analysis of 25 studies exploring thyroid gland invasion in TL, only 8% of patients had thyroid gland invasion. Of note, they suggested that subglottic primaries, T4 classification, subglottic extension, and thyroid cartilage invasion were risk factors for thyroid invasion. 43 Similar to what is found in the literature, our study found that factors significantly associated with thyroid gland invasion were TNM stage IV, T stage of T4, lymphovascular invasion, and thyroid cartilage involvement of the tumor. In a randomized controlled trial by El-Sebai Ali et al, the incidence of thyroid gland invasion was only 2.5%. 16 In our study, thyroid gland invasion occurred in 27.3% of patients who underwent thyroid removal, suggesting that thyroid gland invasion in patients with laryngeal and hypopharyngeal SCC varies. Another critical factor to consider is the impact of thyroidectomy on patients’ QOL and emotional well-being. Research indicates that the risk of parathyroid injury can significantly affect patients’ well-being, leading to heightened psychological stress.9,44 Furthermore, Nickel et al reported that patients diagnosed with thyroid cancer who underwent total thyroidectomy reported a wide range of QOL issues compared to HT. Their findings demonstrated that the risk of poorer QOL is 1.5 times higher in the case of total thyroidectomy. 45 Common symptoms of hypothyroidism and hypoparathyroidism, such as insomnia, fatigue, weakness, weight gain, muscle cramps, constipation, irritability, memory loss, and abnormal menstrual cycles, have been reported to contribute to the deterioration of the QOL in patients undergoing thyroidectomy.46,47 Given the findings of this study indicating no significant difference in oncological outcomes, the decision to perform thyroidectomy during TL should take into account the potential risks and benefits it entails.
One limitation of the study is its retrospective nature, patients who underwent TL for laryngeal and hypopharyngeal malignancies may not be representative of all patients with such malignancy. Another limitation of this study is that it was conducted at a single center, which may limit the generalizability of our results to other surgical centers with different patient demographics, protocols, and resources. Future studies should aim to address these limitations, potentially through a prospective multicenter design.
Conclusion
The decision regarding thyroidectomy during TL for advanced laryngeal and hypopharyngeal SCC is multifaceted, and our retrospective study has provided insights into this complexity. At our institution, we found no significant difference in recurrence rates or mortality from recurrence between patients who did not undergo thyroid removal during TL and those who underwent any type of thyroidectomy. However, those without thyroid removal often had lower T stages. Among patients who underwent thyroid removal, 27% tested positive for gland invasion, suggesting a continued role for thyroid removal in TL. Further research is necessary to offer more definitive guidance on this matter and to establish more precise, evidence-based guidelines for managing the thyroid gland during TL.
Footnotes
Authors’ Note
This study was presented at the American Academy of Otolaryngology Meeting in Nashville, Tennessee, on October 3, 2023. There are no prior published, posted, or submitted articles from this same study.
Author Contributions
A.C.G.: contributed to the conception, to the design and implementation of the research, to the analysis of the results, and to the writing of the manuscript and presentation of the research.
M.C.: contributed to the conception, to the design and implementation of the research, to the analysis of the results, and to the writing of the manuscript and presentation of the research.
S.F. contributed to writing, editing, and submission of the manuscript.
D.K.: contributed to the design and implementation of the research, to the analysis of the results and to the writing of the manuscript.
K.D.: contributed to the conception, design and implementation of the research, to the analysis of the results and to the writing of the manuscript.
E.V.: contributed to the conception, design and implementation of the research, to the analysis of the results, writing of the manuscript and presentation of the research.
J.S.: contributed to the design and implementation of the research, to the analysis of the results and to the writing of the manuscript.
M.M.: contributed to the design and implementation of the research, to the analysis of the results and to the writing of the manuscript.
Data Availability
The data presented in this study are available on request from the corresponding author.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Approved by the University of Arkansas for Medical Sciences Institutional Review Board: IRB# 274369.
