Abstract
Follicular dendritic cell sarcoma (FDCS) is an extremely rare malignancy arising from follicular dendritic cells accounting for less than 0.4% of all soft tissue sarcomas. While it can manifest in various anatomical locations, its occurrence in the nasopharynx is particularly uncommon with only 10 reported cases in the English literature. This case report describes a 45-year-old woman who complained of persistent neck swelling, nasal obstruction, and left ear fullness. Imaging showed a nasopharyngeal process and cervical lymphadenopathy. Biopsies confirmed FDCS. Treatment included chemotherapy and radiation, resulting in a favorable outcome with no recurrence during 2 years of follow-up.
Introduction
Follicular dendritic cell sarcoma (FDCS) is a rare malignancy arising from follicular dendritic cells, accounting for less than 0.4% of all soft tissue sarcomas.
First described by Monda et al in 1986, 1 FDCS was later classified by the World Health Organization (WHO) in 2008 as a type of histiocytic and dendritic cell tumor characterized by low-grade malignant sarcomas with follicular dendritic cell differentiation. 2 Only a handful of cases of FDCS arising from the nasopharynx have been reported in the English literature.
In this article, we present a case of FDCS arising from the nasopharynx and provide insights into its clinical presentation, diagnostic complexities, histopathologic features, and management strategies.
Case Presentation
A 45-year-old woman was referred to our ENT department for the management of neck swelling that persisted for 1 year, accompanied by nasal obstruction and a sensation of fullness in the left ear. No relevant clinical history of a previous malignancy was obtained from the patient. The patient had no prior consumption of raw milk or exposure to tuberculosis in her environment. In addition, she did not experience other related symptoms such as hoarseness, difficulty swallowing, weight loss, or nosebleeds. Furthermore, there were no reported neurological or ocular symptoms.
On examination, the patient’s general condition was preserved and she was apyretic. Cervical examination revealed a left upper cervical jugulo-carotid lymphadenopathy, measuring 3 cm long axis, firm, painless, and fixed in both planes. The thyroid, submandibular, and parotid glands were clear. Nasal endoscopy showed the presence of a non-bleeding budding formation in contact with the left lateral wall of the nasopharynx.
A CT scan of the nasopharynx showed the presence of an enlarging tissue process in the left posterolateral wall of the nasopharynx after contrast injection partially filling the homolateral Rosen-Muller fossa associated with a 42 × 32 mm left jugulo-carotid adenopathy (Figure 1A and B).
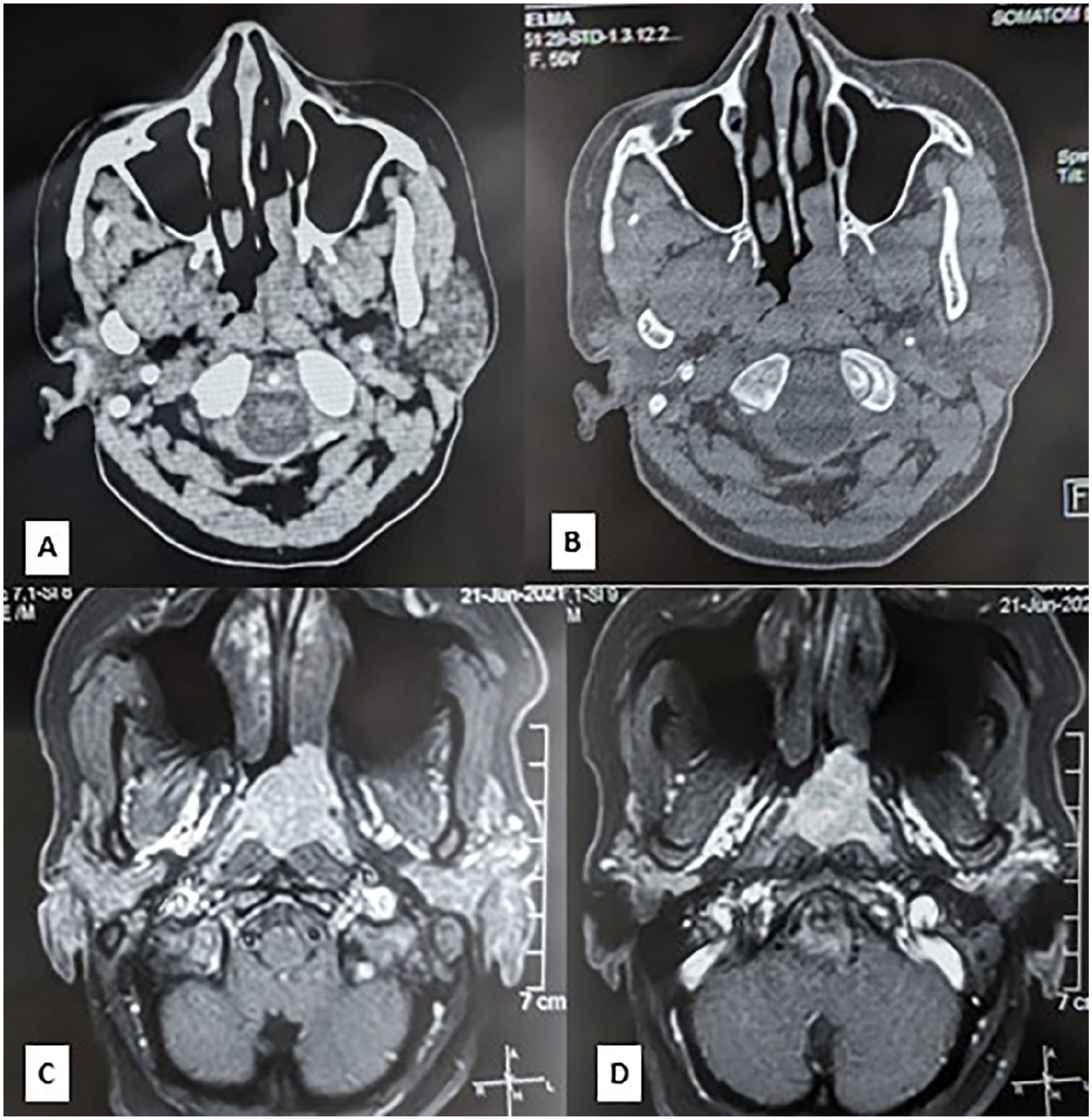
(A, B) Axial CT scan of the nasopharynx showed the presence of an enlarging tissue process in the left posterolateral wall of the nasopharynx after contrast injection partially filling the homolateral Rosen-Muller fossa. (C, D) The MRI of the nasopharynx depicted axial T1-weighted fat-saturated gadolinium-enhanced images with fast saturation: a left-sided nasopharyngeal process characterized by increased signal intensity on T1-weighted images that did not enhance after gadolinium injection, filling the Rosen-Muller fossa and reaching the left choana.
Additional MRI of the nasopharynx revealed a left-sided nasopharyngeal process characterized by increased signal intensity on T1-weighted images and decreased signal intensity on T2-weighted images of 27 × 28 × 23 mm, which did not enhance after gadolinium injection, filling the Rosen-Muller fossa and reaching the left choana with integrity of the deep facial spaces (Figure 1C and D).
Two nasopharyngeal biopsies performed under local anesthesia yielded inconclusive results, necessitating adenectomy and a third nasopharyngeal biopsy under general anesthesia.
Microscopically, the lymph node and nasopharynx shared the same histologic aspect (Figure 2A and B): They were massively invaded by uniform, epithelioid cells harboring diffuse architecture and regularly admixed with small lymphocytes and plasmocytes. There was a small area of necrosis. The neoplastic cells had abundant eosinophilic cytoplasm with undefined limits, ovoid nuclei, delicate chromatin, and small nucleoli (Figure 2C). Immunohistochemical stains were performed on both lymph node and nasopharynx biopsies showing identical immunohistochemical profiles: tumors cells were positive for CD23 (Figure 2D), CD21 (Figure 2E), cycline d1, EMA, vimentin, and CD99 (focal expression). They were negative for PS100, CD68, CD45, Pancytokeratin, P63, PS100,CD20, CD3, CD30, CD15, CD10, CD68, CD138, CD5, CD25, CD35, MUM1, Bcl2, Bcl6, ALK, LMP1, and Tdt, Myélopéroxydase, Chromogranine. The estimated value of Ki67 staining was 80%. The diagnosis of a FDCS of the nasopharynx with lymph node involvement was established. The positron emission tomography (PET) scan showed significant localized metabolic activity Standardized Uptake Value (SUVmax = 11.04) in the thickened tissue of the posterolateral wall of the nasopharynx. This hypermetabolic focus suggests increased metabolic processes in this area (Figure 3). Importantly, there were no other areas of hypermetabolism detected elsewhere in the body that would raise suspicion of malignancy. The tumor was staged as T2N1M0. The patient underwent 4 cycles of chemotherapy followed by external radiotherapy at a dose of 66 Gy. The course of the treatment was favorable, with no recurrence of the tumor locally or in nearby lymph nodes and no evidence of distant metastasis observed during the 2-year follow-up period.
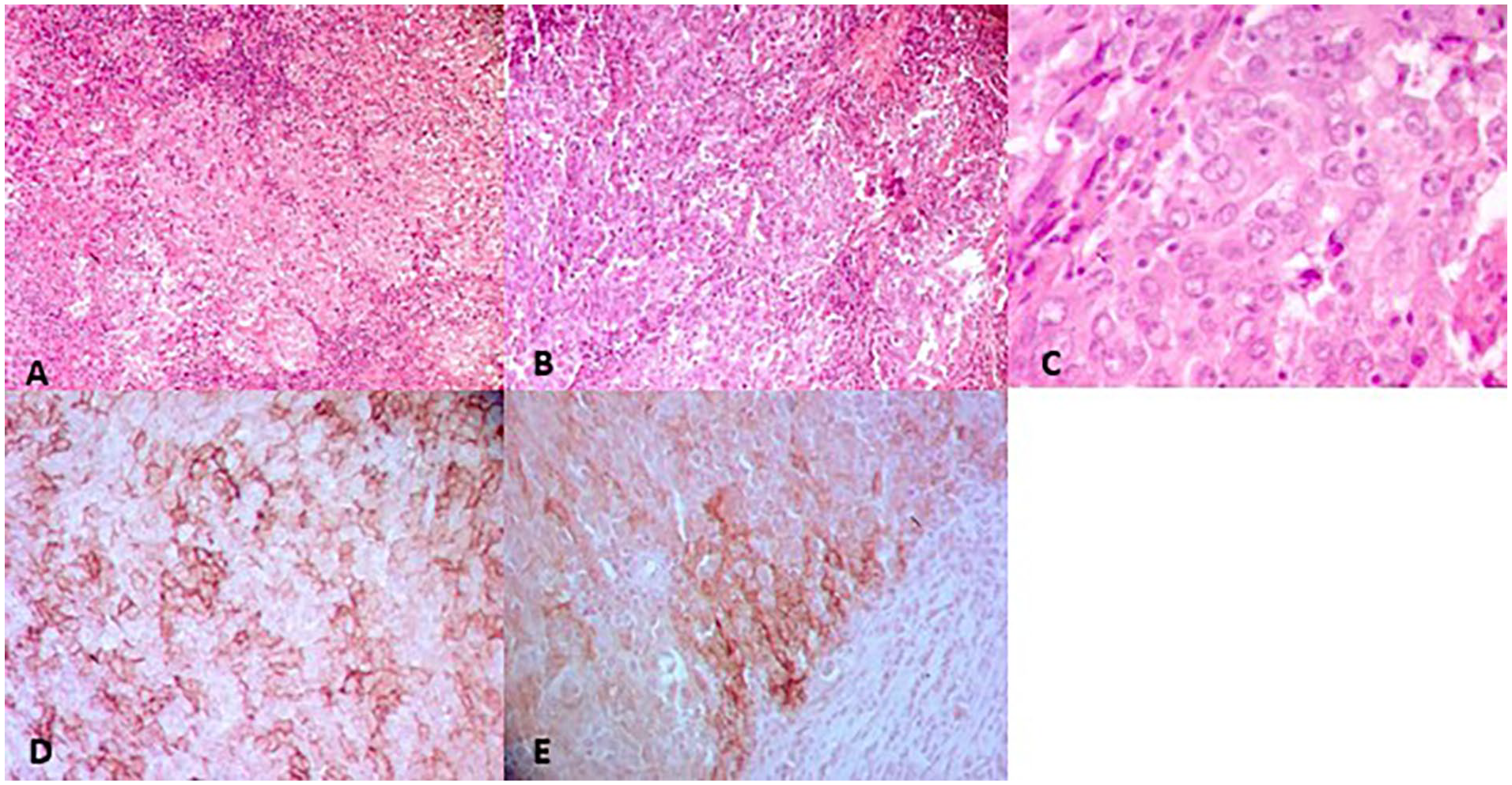
(A) Nasopharyngeal biopsy entirely occupied by a diffuse neoplasm made of epithelioid cells (Hex200). (B) Lymph node metastasis presenting with the same histologic aspect (Hex20). (C) Epithelioid cells with ovoid nuclei, delicate chromatin, and small nucleoli (HE ×40). (D) Diffuse expression of CD23. (E) More heterogeneous expression of CD21.
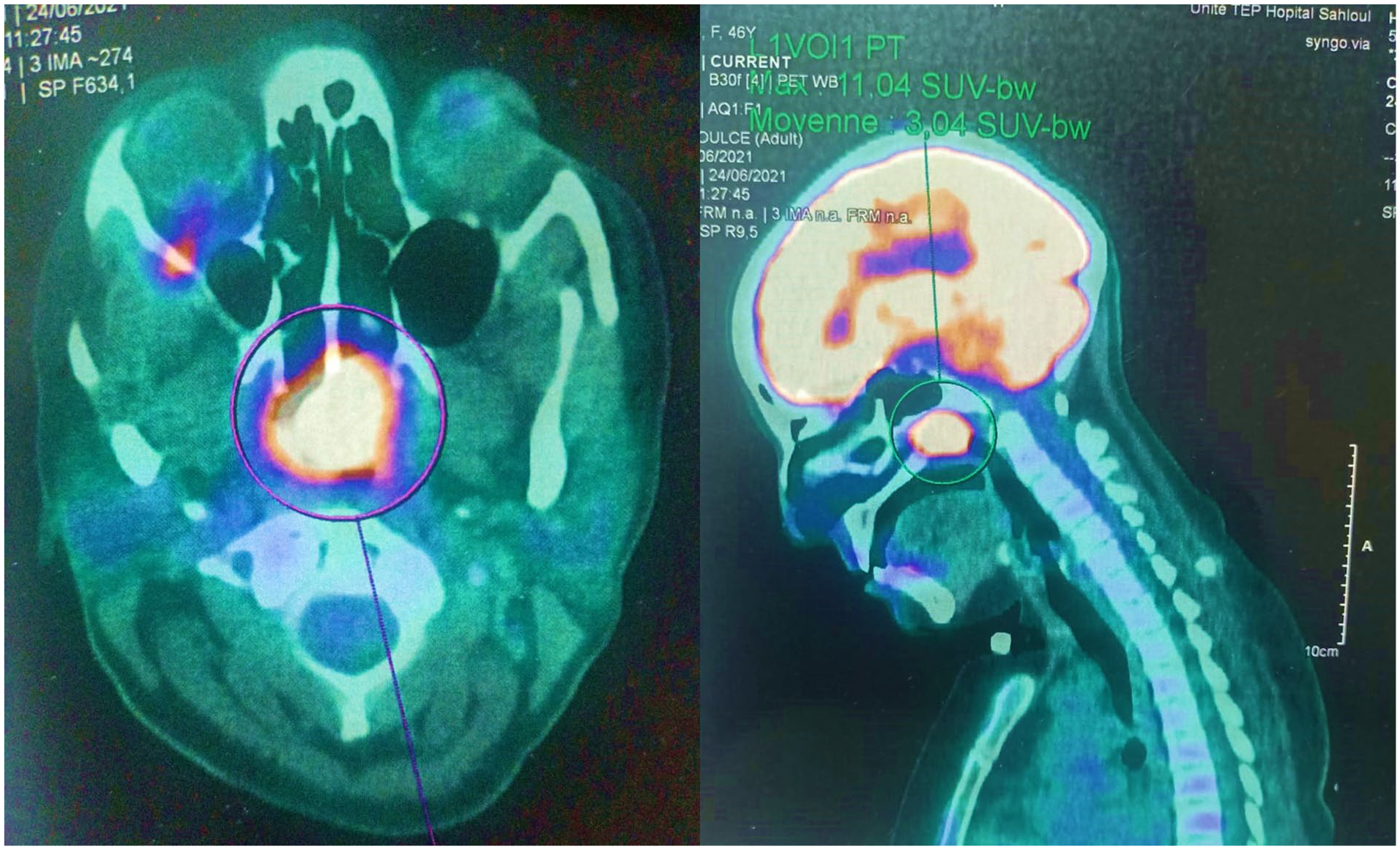
The positron emission tomography scan showed significant localized metabolic activity (SUVmax = 11.04) in the thickened tissue of the posterolateral wall of the nasopharynx. This hypermetabolic focus suggests increased metabolic processes in this area.
Discussion
FDCS is a rare low-grade sarcoma, which presents as intranodal or, more commonly, extranodal disease at a wide variety of sites. To the best of our knowledge, since the first case reported by Beham-Schmid in 1998, 3 only 10 cases of FDCS originating from the nasopharynx have been documented. Our case would represent the 11th reported case in the literature. Table 1 provides a summary of these individual cases.3-11 A male predominance has been observed, with 6 affected men and 4 women. Among these cases, FDCS manifested in adults with ages ranging from 23 to 70 years and a median age of 42 years. Two patients were treated for Castleman’s disease.4,6
Summary Table Including Cases of FDCS Reported in the English Literature.
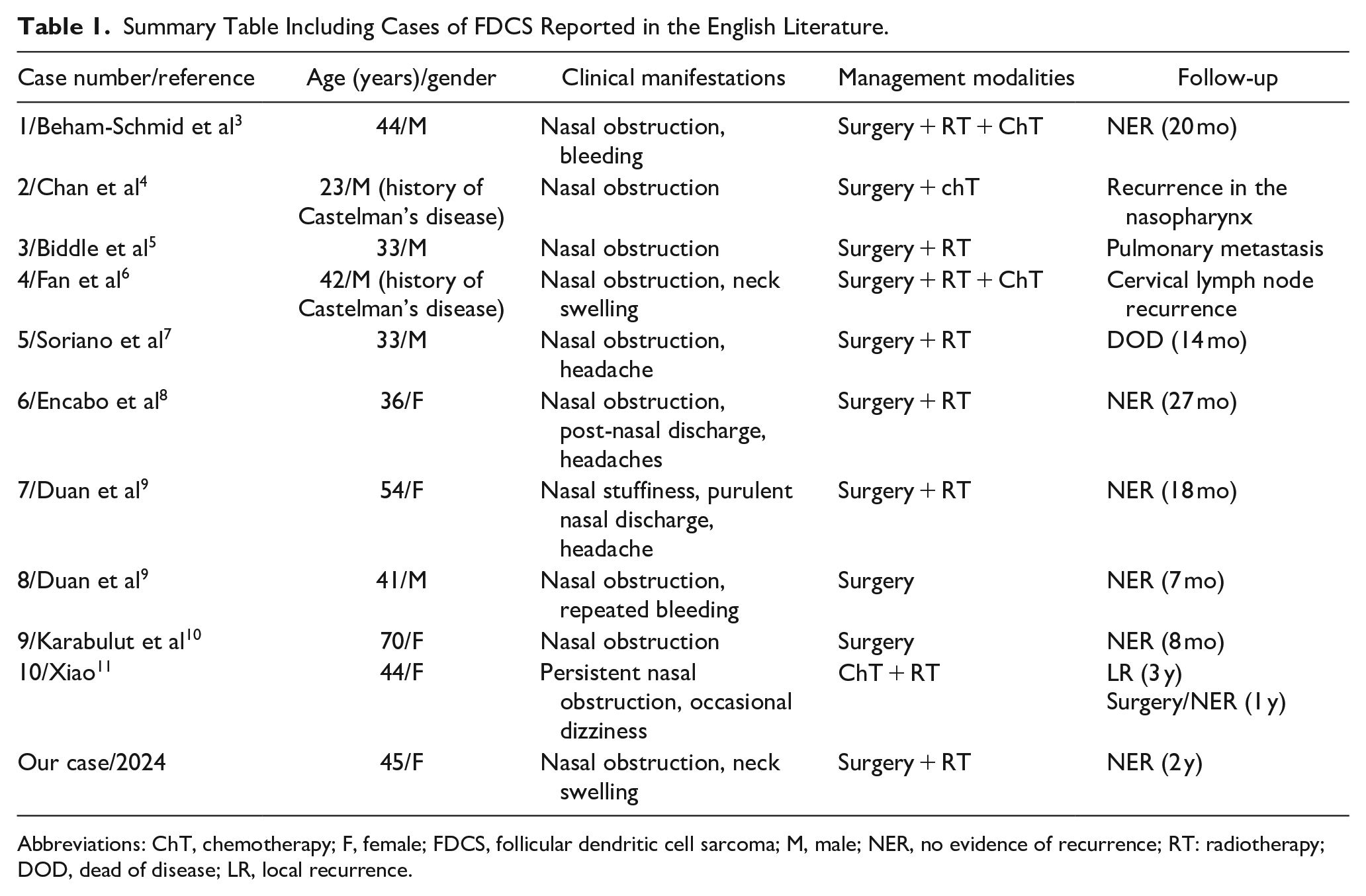
Abbreviations: ChT, chemotherapy; F, female; FDCS, follicular dendritic cell sarcoma; M, male; NER, no evidence of recurrence; RT: radiotherapy; DOD, dead of disease; LR, local recurrence.
This tumor is a low-grade to intermediate-grade sarcoma, that is classified under mesenchymal dendritic cell neoplasms in the current (5th edition) WHO classification of hematolymphoid neoplasms. 12 Its diagnosis is highly challenging regarding its rarity and its common morphologic features that may recall many histologic entities, including carcinoma, particularly undifferentiated nasopharyngeal carcinoma, carcinosarcoma, and mesenchymal tumors such as myofibroblastic inflammatory tumors.11,13 FDCS should be suspected whenever the tumor exhibits a spindled, ovoid, or epithelioid with syncytial morphology, that is negative for epithelial (Pancytokeratin AE1-AE3), melanocytic (MelanA, PS1OO) lymphoïd (CD45), and histiocytic (CD68, CD1a) markers, especially when it is associated with an inflammatory background.12,13 Expression of follicular dendritic markers (CD21, CD23, CD35, clusterin, and CXL13) is mandatory for diagnosis.11-13 Moreover, it is important to mention that FDCS is positive for vimentin and it may occasionally express CD68, CD4, CD30, CD20, and EMA.12,13 Until now, there is no relevant molecular diagnostic tool, but molecular investigations are showing the implication of NF-κB pathway genes in 40% to 60% of cases, and tumor suppressor genes like RB1, CDKN2A, and TP53. 12 Treatment options include surgical excision, if possible, along with radiation and chemotherapy. Due to the rarity of this condition in the nasopharyngeal region, there is not a standardized protocol.3-11
The FDCS typically progresses slowly. However, local recurrence is common in 40-50% of cases. Pulmonary metastasis has been reported in one case. 5
Conclusion
In conclusion, we present a rare case of FDCS arising from the nasopharynx. Despite its rarity, this entity should be considered in the differential diagnosis of the nasopharyngeal process especially when histologic features suggestive of follicular dendritic cell differentiation are encountered. Prompt diagnosis and appropriate treatment including surgery, chemotherapy and radiation, can lead to favorable outcomes as demonstrated in our case. However, given the limited understanding of this rare malignancy, further research is warranted to elucidate its pathogenesis, optimal treatment strategies, and long-term outcomes.
Footnotes
Acknowledgements
None.
Authors’ Note
All the authors have read and agreed to the final manuscript.
Data Availability Statement
Data will be made available on request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Our institution Faculty of Medicine of Sousse’s Ethics Committee does not require ethical approval for reporting individual cases or case series since we anonymously reported clinical and imaging information concerning our patient’s case.
Statement of Informed Consent
A written informed consent was obtained from the patient for his anonymized information to be published in this article.
