Abstract
Introduction
Schwannomas, neuromas, neurofibromas, and granular cell tumors represent peripheral, benign, nerve sheath tumors, which are relatively uncommon in the head and neck region.1,2 Neurilemmoma, commonly referred to as a schwannoma, is a benign tumor originating from the nerve sheath, and characterized by slow growth. Comprising Schwann cells evident in the myelin sheath surrounding the nerve, these tumors can arise from any cranial, peripheral, or autonomic neuron, except the optic and olfactory nerves. Approximately 25% to 40% of all schwannoma cases affect the head and neck regions.1-3 Sinonasal involvement occurs in less than 4% of head and neck schwannomas, making it a rare occurrence.1,2 These tumors demonstrate no significant gender or racial predilection, and typically present in middle-aged adults. Clinically, they manifest as slowly growing masses, leading to symptoms such as headaches, hyposmia, epistaxis, and progressive nasal obstruction. The clinical appearance of the tumor, typically solitary, depends on its location and size.2,4,5
In addition to nasal endoscopy, radiological imaging techniques, such as computed tomography (CT) and magnetic resonance imaging (MRI), aid in diagnosis. 3 Choosing the most appropriate surgical technique for mass removal can be challenging due to critical and close areas around the nasal cavity. Surgical removal is the most effective method of treatment. 4 It may be guided by preoperative biopsy for differential diagnosis from other nasal neoplasms, or may be performed as excisional biopsy for definitive histopathological diagnosis if the mass is located in a favorable location and relatively small in size.2,6 Histopathological examination typically reveals a biphasic pattern with alternating zones of compact and spongy cellular space, characterized by spindle cells containing elongated wavy nuclei and homogeneous cells.2,6 Additional features such as bleeding, necrosis, and cystic degeneration may also be observed. Immunohistochemistry for S-100 is positive in this benign nerve tumor.3,7
This article presents a single-center experience of schwannomas located in the nasal region, a rare presentation, providing up-to-date clinical, radiological, and pathological information. Our diagnostic and treatment processes are presented in the context of the existing literature.
Case Report
All Patients
Over a 10 year period, 5 patients with schwannoma of nasal cavity origin and surgically treated were identified and included (Table 1).
Demographic, Clinical, and Histopathological Data of Schwannoma Cases.
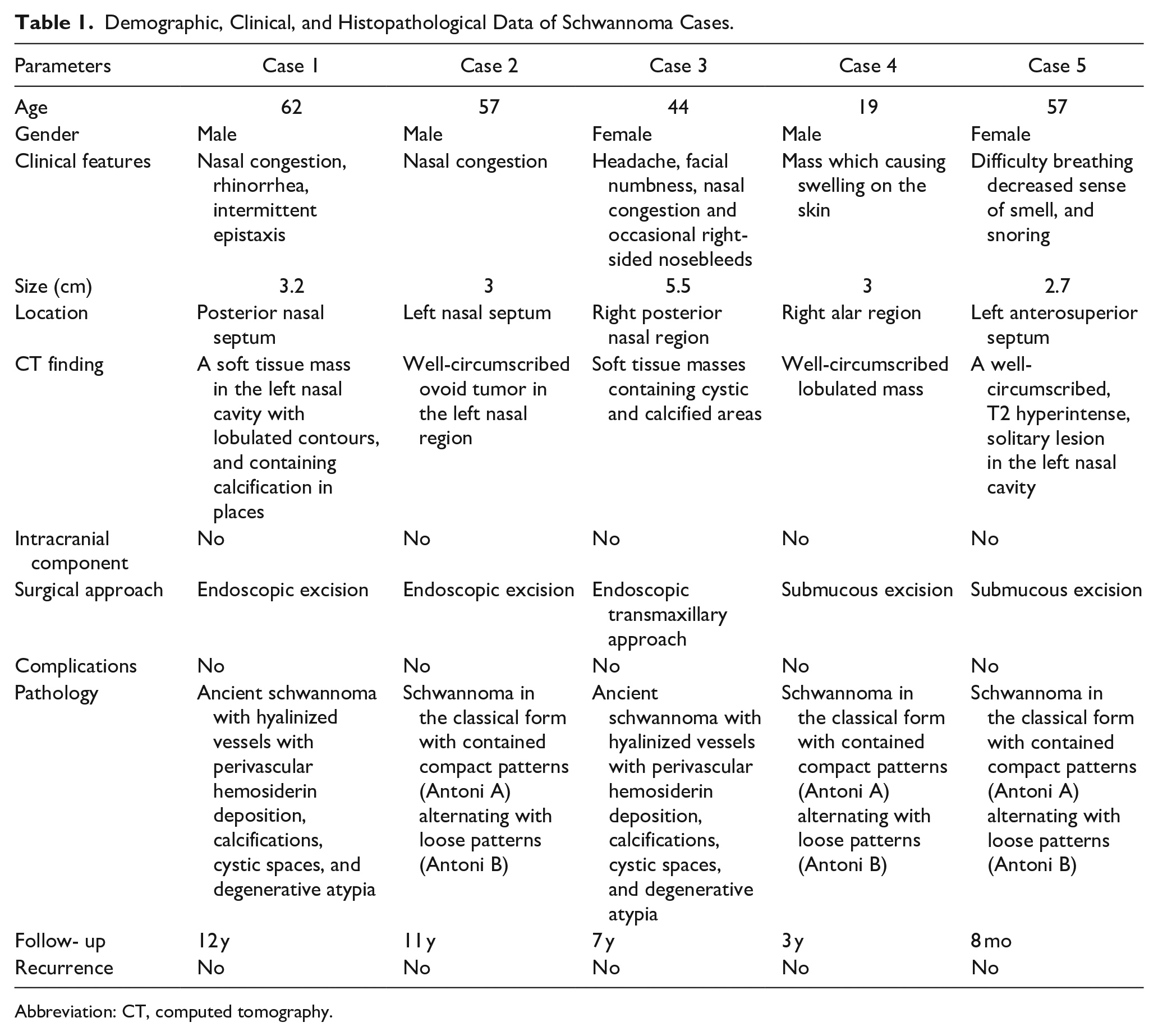
Abbreviation: CT, computed tomography.
Demographic Information: The patients were adults, aged between 19 and 64 years with a mean age of 47.8 years. Three were male, and two were female.
Clinical Features: All patients, except for one who complained of a small swelling visible in the alar region, presented with unilateral nasal congestion, which was the most common symptom. The second most common symptom encountered was unilateral intermittent nosebleeds, evident in two patients, and milder in one other patient. In addition, one patient presented with rhinorrhea, one with headache and facial numbness, and one with loss of sense of smell. A 19-year-old male patient presented with a small papillomatous swelling in the alar region of the nasal vestibule, which he could also see, but had no other complaints. No orbital symptoms were observed in any of the 5 patients. The duration of symptoms ranged from 2 to 12 months, with an average of 6.8 months. Endoscopic examination usually revealed a nonspecific mass lesion with a lobulated surface, obstructing the nasal lumen.
Radiology: Radiologically, well-circumscribed, lobulated, non-infiltrative mass lesions without any associated destruction of the surrounding tissues or bone but exerting mass effects on surrounding tissues were observed (Figure 1). The largest dimension of the masses ranged from 3 to 55 mm, with an average of 29.4 mm. While calcified areas were observed in 2 of 4 patients who underwent CT examination, they were not observed in the remaining 2 patients (Figure 2a). While heterogeneous contrast enhancement was seen in the patient with the 55 mm tumor, in the other 4 patients, the mass was homogeneous or had no CT contrast. On MRI, the masses were isointense or hypointense on T1, showing intense enhancement with contrast; at T2, they were heterogeneously hyperintense. Areas of cystic degeneration were observed in 2 patients with tumors of 32 and 55 mm in size (Figure 2b).
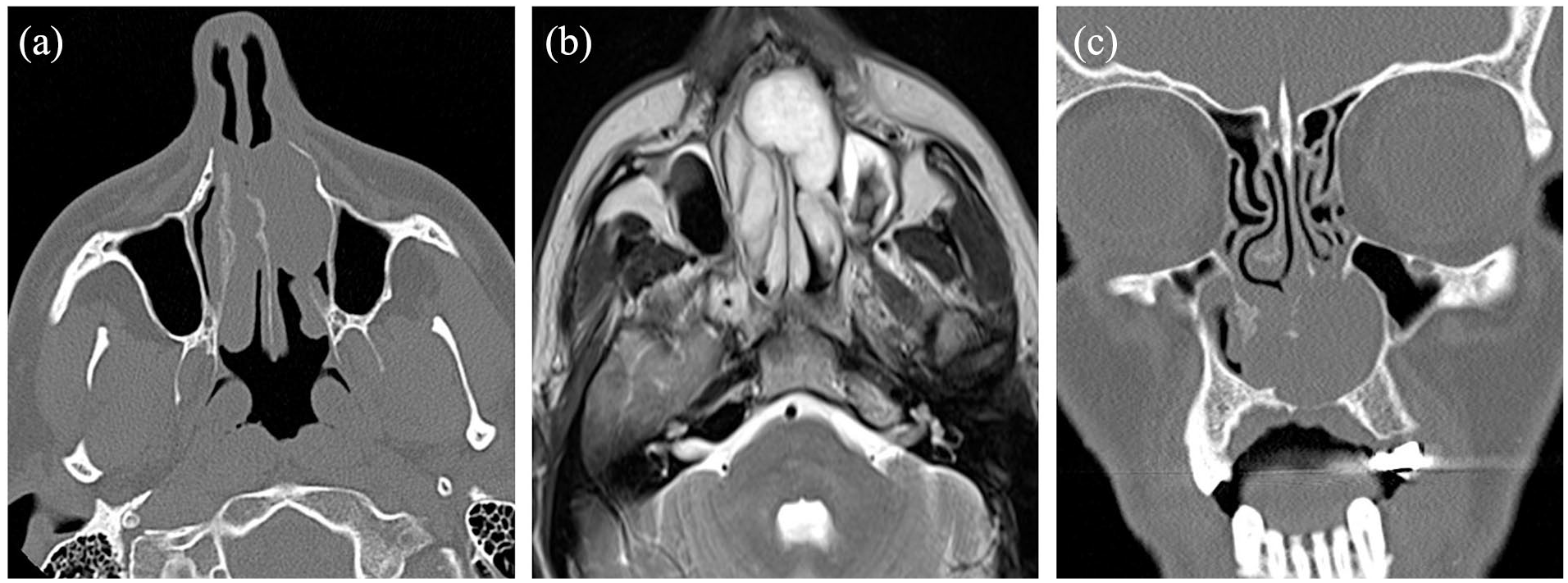
A left nasal cavity mass lesion which is well-circumscribed, lobulated, non-infiltrative, not destructive, but pushing the surrounding tissues and bone on axial CT (a), axial MRI (b), and coronal CT (c) images. CT, computed tomography; MRI, magnetic resonance imaging.
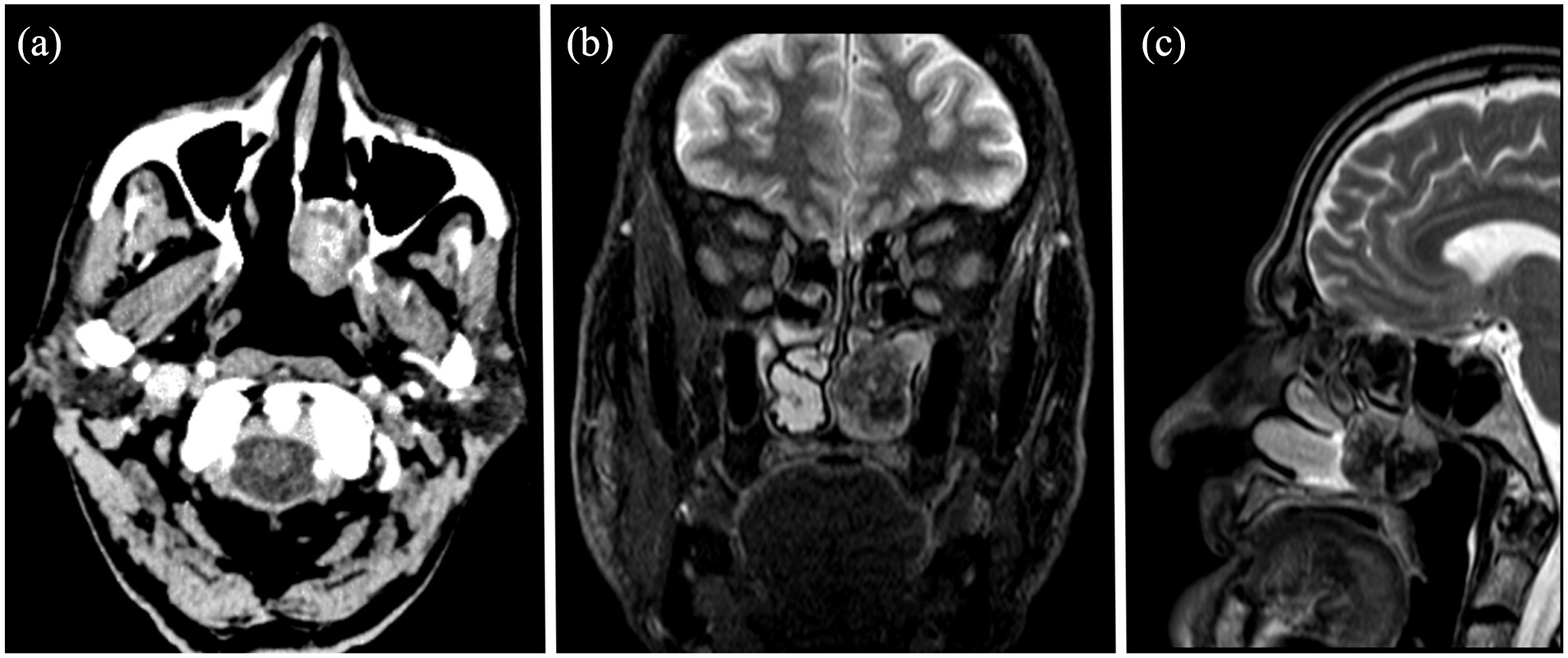
(a) Calcified areas on CT image, (b) cystic degenerative areas on T2-weighted MRI. (c) inflammatory findings just anterior to the mass on T2-weighted MRI. CT, computed tomography; MRI, magnetic resonance imaging.
Surgical Treatment: Surgeries were performed transnasally in all patients, and no facial incision was used. While there was no need to use an endoscope in 2 cases with anterior nasal septum and vestibular location, all other surgeries were performed endoscopically. Septal cartilage was protected as much as possible in all cases. During surgery, it was observed that the masses generally caused mild bleeding. In terms of location, two originated from the nasal septum, one from the middle conchae, one from the lateral nasal wall, and one from the alar mucosa. Three of the masses were excised in en bloc fashion, while the remaining two were excised by piecemeal resection. No perioperative or postoperative complications were observed. The follow-up period ranged from 10 to 122 months, with an average of 61 months, and no recurrence was observed in any patient.
Pathology: Macroscopically, most of the solid masses had a light, skin-colored appearance surrounded by a fibrous capsule. Yellow coloration was often seen due to lipid content or lipid-laden macrophages. Three cases had cysts of variable size and exhibited hemorrhagic changes. Morphologically, the excised masses contained compact patterns (Antoni A) alternating with loose patterns (Antoni B) seen in schwannomas in the classical form (Figure 3a-d). Antoni A regions were characterized by increased cellularity and spindle nuclei. Verocay bodies, which are clumps of cellular aggregates encountered in palisade patterns, were observed in 1 case. Antoni B domains were irregular arrangements that were hypocellular and contained a variable degree of macrophage infiltration. Common histological findings included hyalinized vessels with perivascular hemosiderin deposition, calcifications, cystic spaces, and degenerative atypia (“ancient change”; Figure 3e). No malignant transformation or accompanying malignancy was observed in any of the cases.
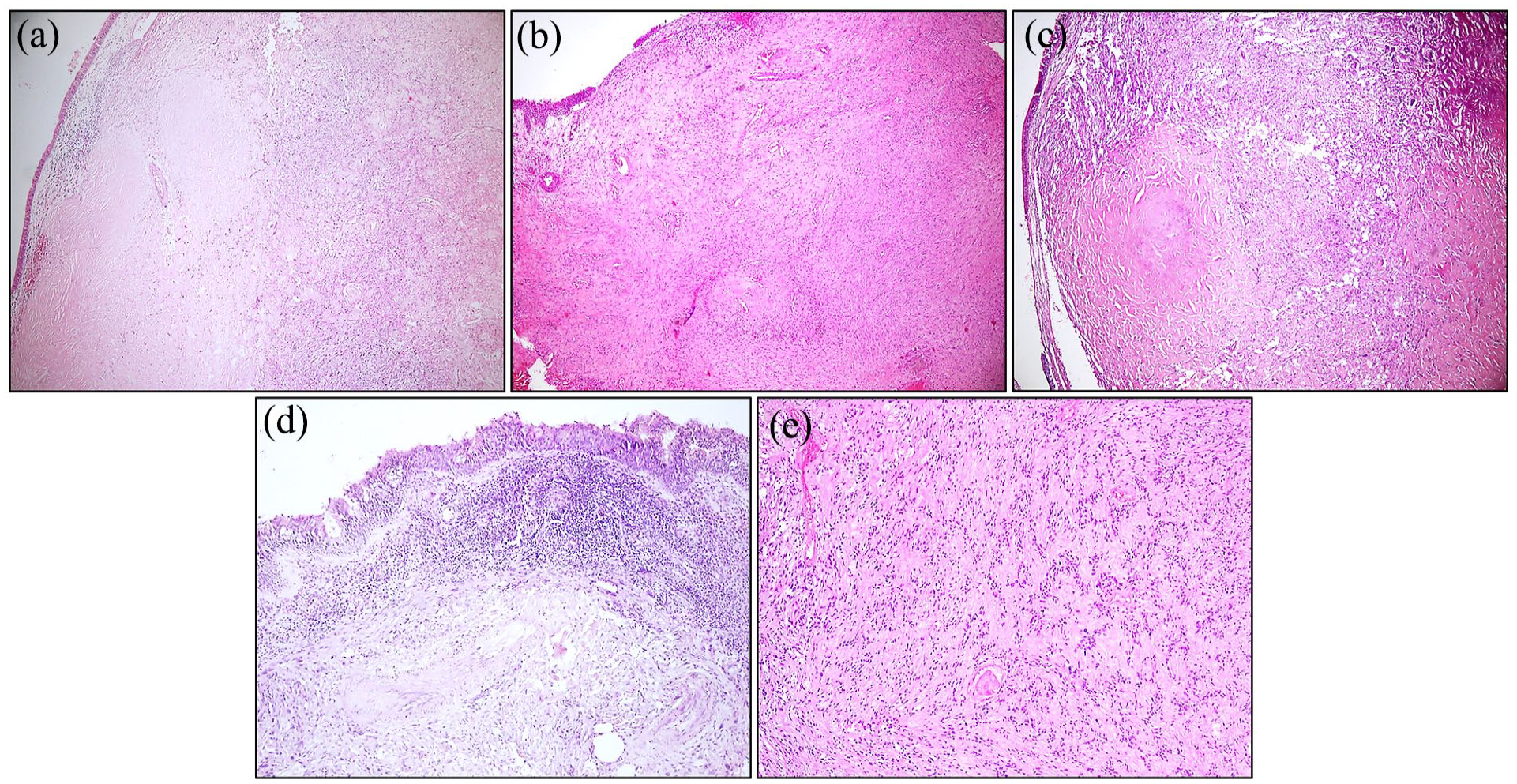
(a-d) Case examples of spindle cell mesenchymal neoplasia, located just below the respiratory epithelium, with widespread hyalinized and vascular areas, without a specific alignment pattern (H&E, ×40 and ×100), (e) spindle cells with elongated nuclei with palisade and ill-defined pale eosinophilic cytoplasm in classic schwannoma (H&E, ×100). H&E, hematoxylin and eosin.
Case 1
A 62-year-old male patient presented with complaints of nasal congestion, rhinorrhea, and intermittent epistaxis that had been gradually worsening for a year. He had hypertension and diabetes mellitus. Nasoendoscopy revealed a mass with a lobulated surface, and an obstructed lumen in the left nasal passage and posterior middle meatus. MRI of the patient showed inflammatory findings just anterior to the mass that resulted in relatively bigger dimensions on CT images (Figure 2c). The resected tumor was reported as “ancient schwannoma” (Figure 3a).
Case 2
A 57-year-old male patient was admitted to the outpatient clinic due to congestion in the left nasal cavity that had been present for the past 6 months. This patient had no significant comorbidities. An excisional biopsy procedure was performed to this fragile septal mass, and histopathology revealed spindle tumor cells, organized in parallel and flowing patterns on histological examination. There were also occasional hyalinized areas (Figure 3b). There was no evidence of cytological atypia, mitosis, or necrosis. The spindle cells showed positive immunostaining for S-100 (Figure 3a), SOX10, and PGP9.5.
Case 3
A 44-year-old female patient attended because of headache, facial numbness, nasal congestion, and occasional right-sided nosebleeds that had been present for the preceding 10 months. Nasoendoscopy revealed a semisolid mass originating from the right posterior nasal region and lateral wall, and extending to the nasal turbinates. CT revealed a soft tissue mass, 55 mm in diameter, containing cystic and calcified areas, originating from the right nasal region, compressing the maxillary sinus, and extending to the pterygopalatine fossa. Excision of the tumor was performed endoscopically with a medial maxillectomy and transmaxillary pterygopalatine fossa approach. The histopathological report described a benign, mesenchymal, spindle cell neoplasia, which was reported as “ancient schwannoma” (Figure 3c).
Case 4
A 19-year-old male patient was admitted due to a mass that had been noticed 2 months earlier. The mass was located on the mucosa of the right alar region, and caused swelling visible on the skin. Anterior rhinoscopy revealed a smooth-surfaced, noninfected papillomatous lesion. With a small mucosal incision, the mass was excised in a submucous resection fashion, and histopathological examination confirmed the diagnosis of schwannoma (Figure 3d).
Case 5
A 57-year-old female patient attended because of difficulty breathing through the left nasal passage, a decreased sense of smell, and snoring, which had started 4 months previously. She had hypertension and hypercholesterolemia as comorbidities. Nasoendoscopy showed a solid mass originating from the anterosuperior septum in the left nasal passage, and adhering inferiorly to the medial surface of the inferior turbinate. It was thought that this adhesion may have occurred secondary to a previous biopsy procedure. The MRI reported a possible nasal polyp, but the mass was excised to exclude possible malignancy. The tumor contained Antoni A and Antoni B areas, characteristics of classical schwannoma (Figure 3e). Since immunostaining for SOX10, S-100, and PGP9.5 was positive, the diagnosis of schwannoma was confirmed.
Discussion
The primary objective of the current study was to present a single-center case series of schwannomas in the nasal region, which are known to be a rare finding. Through these cases, the aim was to provide comprehensive clinical, radiological, and pathological information about nasal schwannomas. In addition, we wished to highlight the diagnostic and treatment approaches performed in these cases, and discuss them within the broader framework of the existing literature concerning nasal schwannomas.
Sinonasal schwannomas are rare in the paranasal sinuses and nasal cavity, and typically, these two are evaluated together in this area. 8 In the current study, we specifically present only nasal cavity schwannoma as a distinct entity, due to the common clinical problem of masses encountered during nasal endoscopic examinations in the outpatient clinic setting. The diagnosis and treatment approaches can be challenging depending on their anatomic location and clinical findings, which usually mimic benign diseases of this region. It is clinically difficult to distinguish schwannoma from other, more common, unilateral nasal masses. However, endoscopy may not be very helpful in the differential diagnosis. Although calcification on CT examination is not specific to schwannomas, it can aid in the differential diagnosis. In our series, calcification on CT was observed in 2 patients. This amounts to 40% of the cases, which is a greater proportion than has previously been reported, 8 but as the case series only consisted of 5 patients, this finding may be a result of small group size. MRI is regarded as the main auxiliary tool for the differential diagnosis of schwannomas. 9 The main expected findings are isointense or hypointense enhancement on T1, intense enhancement with contrast, and heterogeneously hyperintense enhancement on T2 images.8-10 Cystic degenerative areas may be present, especially in larger tumors, and were observed in 2 with the largest tumors out of 5 patients in this study. Hemorrhagic changes may also occur, but we did not encounter any. Other useful signs include the split-fat sign, target sign, and fascicular sign, which are not always present.8,11-13 None of these signs were observed in this case series. Calcified and cystic areas, recognized as radiologically useful findings, are typically observed in larger masses. In the present study, the masses that showed these findings were the largest ones encountered, measuring 32 (Case 1) and 55 mm (Case 3) in diameter.
MRI examination may also be required in most patients to evaluate inflammatory changes, the actual size of the mass, and intracranial extensions, if any.14,15 In our series, the true size and extent of the masses was better evaluated with MRI. Therefore, we consider MRI useful for both differential diagnosis and to provide accurate information about the size and extent of the mass. We observed that the average tumor diameter at the time of diagnosis was around 30 mm. This suggests that these masses generally become symptomatic after reaching this size in the nasal cavity. Simultaneously, we believe that patients need to consult a physician on experiencing these symptoms. One of our patients presented when the mass was much smaller in size because the lesion was in the vestibular region of the nose and was evident to the patient himself.
Surgery for these neoplasms, which tend to have increased vascularity relative to other soft tissue tumors, may cause intraoperative bleeding, either from the tumor itself or because of inflammatory changes in surrounding tissues.15-17 Although we did not observe excessive bleeding in any of our cases, surgical teams should be prepared for bleeding control in these patients. We believe that it is safer to perform these surgeries in centers experienced in endoscopic tumor surgery, as often as possible. Depending on the location of the mass, access to the mass may be direct, or it may be necessary to pass through the mucosa or a thin bone wall. In cases where the mass remains behind bone, excision can be performed by carefully removing the anterior bony structures, to expose the capsule of the mass. Small masses can be removed in en bloc fashion, while larger ones may be successfully removed in a piecemeal fashion. For functional and cosmetic reasons, treatment of patients with an endoscopic approach is optimal, and none of our patients required an approach with a transfascial external incision. With careful and appropriate surgery, recurrence is not expected, and although there were cases in the present series with a relatively short follow-up period, we did not encounter any recurrences.
Morphologically, in classical schwannoma, 2 primary patterns, Antoni A and Antoni B, are observed when examined under a microscope. The arrangement of nuclei around a core mass of cytoplasm, known as Verocay bodies, is seen in Antoni A areas. The stroma in Antoni B regions is loose and lacks any discernible cell or fiber patterning. It is also possible for both traits to be mixed together. Schwannomas are frequently positive for mature Schwann cell markers on immunohistochemistry, including S100, SOX10, and PGP9.5 proteins.3,5,11 However, ancient schwannoma is a somewhat uncommon type. Atypia and enhanced cellularity are its defining features. Diffuse fibrosis and areas of hyalinization with nuclear pleomorphism and hyperchromasia but without atypia are further histologic features. 2 These alterations are thought to be the outcome of aging and long-term tumor growth. These cellular alterations may be unclear and challenge a precise pathological diagnosis. Atypia and enhanced cellularity may resemble malignant characteristics. Extreme caution must be used when differentiating between malignant schwannoma and fibrosarcomas. 2 Four of the cases in the present series were diagnosed with ancient schwannoma, and mature Schwann cell markers were positive in all cases on immunohistochemical examination.
Conclusion
Schwannomas that originate in the nasal cavity are uncommon tumors, which grow slowly and became symptomatic after attaining a size that causes effects noticeable to each individual patient. Schwannoma should be included in the differential diagnosis of unilateral nasal masses, and diagnostic imaging may be particularly helpful when there is preoperative suspicion. However, the definitive diagnosis is histopathological, and to achieve that and relieve the patients’ complaints, an endoscopic approach to the nasal cavity is very helpful and appropriate. An external approach may be avoided in many cases. It should be kept in mind that increased cellularity and atypia may occur in ancient schwannoma. With the advances in endoscopic technologies and additional research, the best possible outcomes may be achieved for these particularly rare lesions.
Footnotes
Author Contributions
B.Y.B., A.Y., and M.O. made substantial contributions to conception and design. B.Y.B. and A.Y. contributed for acquisition of data, analysis, and interpretation of data. B.Y.B. and M.O. were involved in drafting this manuscript and revising it critically for important intellectual content. B.Y.B. and M.O. gave final approval of the version to be published.
Data Availability Statement
All data generated or analyzed during this study are included in this published article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
This study was conducted according to the Helsinki principles, the patient signed the consent for the participation and nothing offensive was done against the patient’s privacy. Written informed consent was obtained from the patient for publication of this case report and any accompanying images. A copy of the written consent is available for review by the editor of this journal.
Informed Consent
Written informed consent to publish this case report has been obtained from the patient’s legal guardian.
