Abstract
A 24-year-old man with von Recklinghausen’s disease presented with complaints of difficulty in swallowing for 6 months and change of voice for 3 months. He also had recent-onset difficulty in breathing. Telelaryngoscopy and subsequent contrast-enhanced computed tomography scan revealed a well-defined, smooth submucosal mass in the oropharynx (attached to the posterior pharyngeal wall, superior to the level of left aryepiglottic fold), obscuring the ipsilateral pyriform fossa, and nearly blocking the pharyngeal lumen. The mass was removed with endoscopic coblation-assisted laryngeal surgery, and subsequent histopathology revealed it to be neurofibroma. Neurofibromas are rare neoplasms to be encountered in the oropharynx. However, in the setting of von Recklinghausen’s disease (neurofibromatosis type 1), one or more well-demarcated, submucosal nodular lesions in the upper aerodigestive tract may be considered as neurofibromas, and workup and treatment should be directed accordingly based on this clinical presumption. Endoscopic coblation during laryngeal surgery can effectively be used as a surgical tool to excise such lesions. It provides a relatively bloodless field compared to the conventional cold steel excision, and reduces the risk of complications at surgery and during the follow-up period. This clinical record illustrates the presentation and management of a solitary, isolated oropharyngeal neurofibroma in a man suffering from von Recklinghausen’s disease. It further emphasizes the role of endoscopic coblation-assisted laryngeal surgery in this setup, and the need to maintain a low threshold of suspicion in having a provisional clinical diagnosis of such lesions.
Keywords
Introduction
Approximately 4% to 5% of all benign soft tissue neoplasms in the body are neurofibromas, which are benign nerve sheath tumors in the peripheral nervous system arising from Schwann cells or perineural fibroblast cells. 1 It may manifest as a single, isolated swelling in individuals in their third decade of life, or it may be linked to generalized neurofibromatosis, also known as von Recklinghausen’s disease.2,3 Although neurofibromas are commonly seen in the different subsites of the oral cavity, it is not usual to encounter a neurofibroma in the oropharynx in everyday otolaryngology practice.4,5 To date, only 5 examples of oropharyngeal neurofibroma, 4 in the soft palate and 1 in the palatine tonsil, have been documented in the literature. 2 The present report illustrates an isolated, solitary neurofibroma in a young man with von Recklinghausen’s disease that originated from the oropharynx causing mass effect due to luminal obstruction.
Case Report
A 24-year-old man with neurofibromatosis type 1 (Figure 1) arrived at the otolaryngology outpatient department complaining of difficulties in swallowing for 6 months, along with a change of voice and foreign body sensation in throat for the previous 3 months. There was also difficulty in breathing for the last 3 weeks.
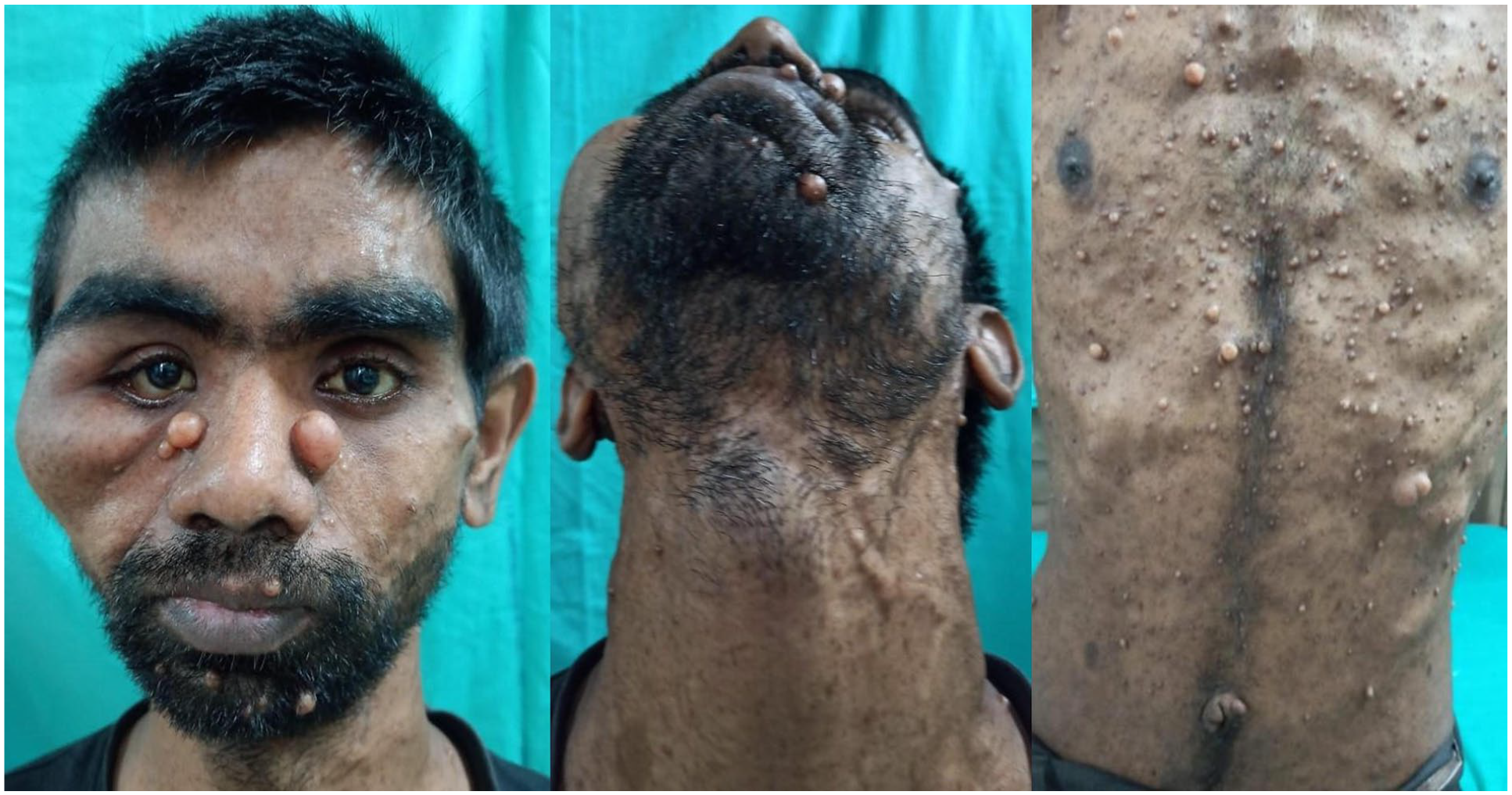
This 24-year-old man had von Recklinghausen’s disease (neurofibromatosis type 1). Note the large café-au-lait patches on his neck and chest.
Rigid fiber-optic telelaryngoscopy revealed a smooth, well-defined submucosal mass lesion in the oropharynx. It appeared to originate from the posterior pharyngeal wall of the oropharynx at a level superior to the left aryepiglottic fold, that overlooked the left pyriform sinus nearly blocking the pharyngeal lumen (Figure 2). Contrast-enhanced computed tomography (CECT) showed an overall homogeneous lesion in the oropharynx overlooking and extending inferiorly along the pyriform fossa (Figure 3). A provisional diagnosis of the mass being a neurofibroma was considered given that the patient had von Recklinghausen’s disease. The high index of clinical suspicion also followed the authors’ own experience in managing a similar patient diagnosed to have laryngeal neurofibroma. 6
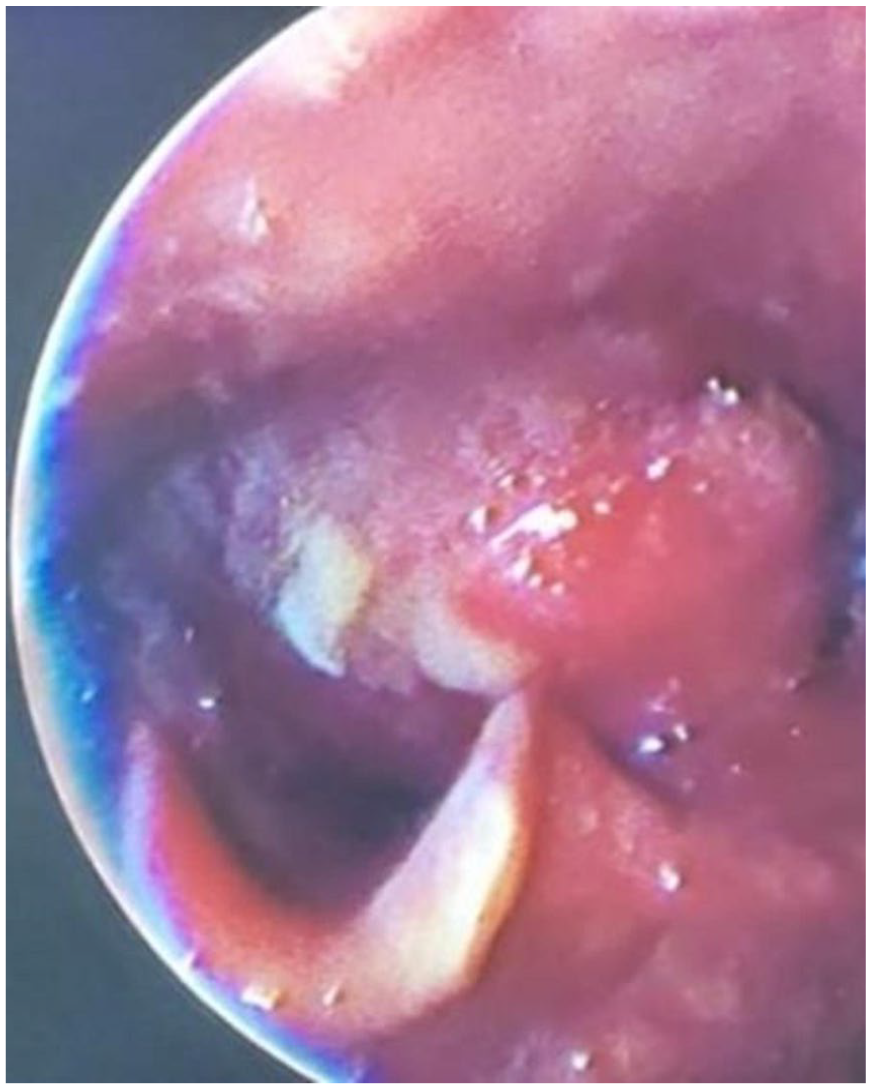
Rigid fiber-optic endoscopy (telelaryngoscopy) revealed a large globular submucosal swelling involving the posterior pharyngeal wall of the oropharynx superior to the level of the left aryepiglottic fold, extending inferiorly and overlooking the left pyriform sinus.
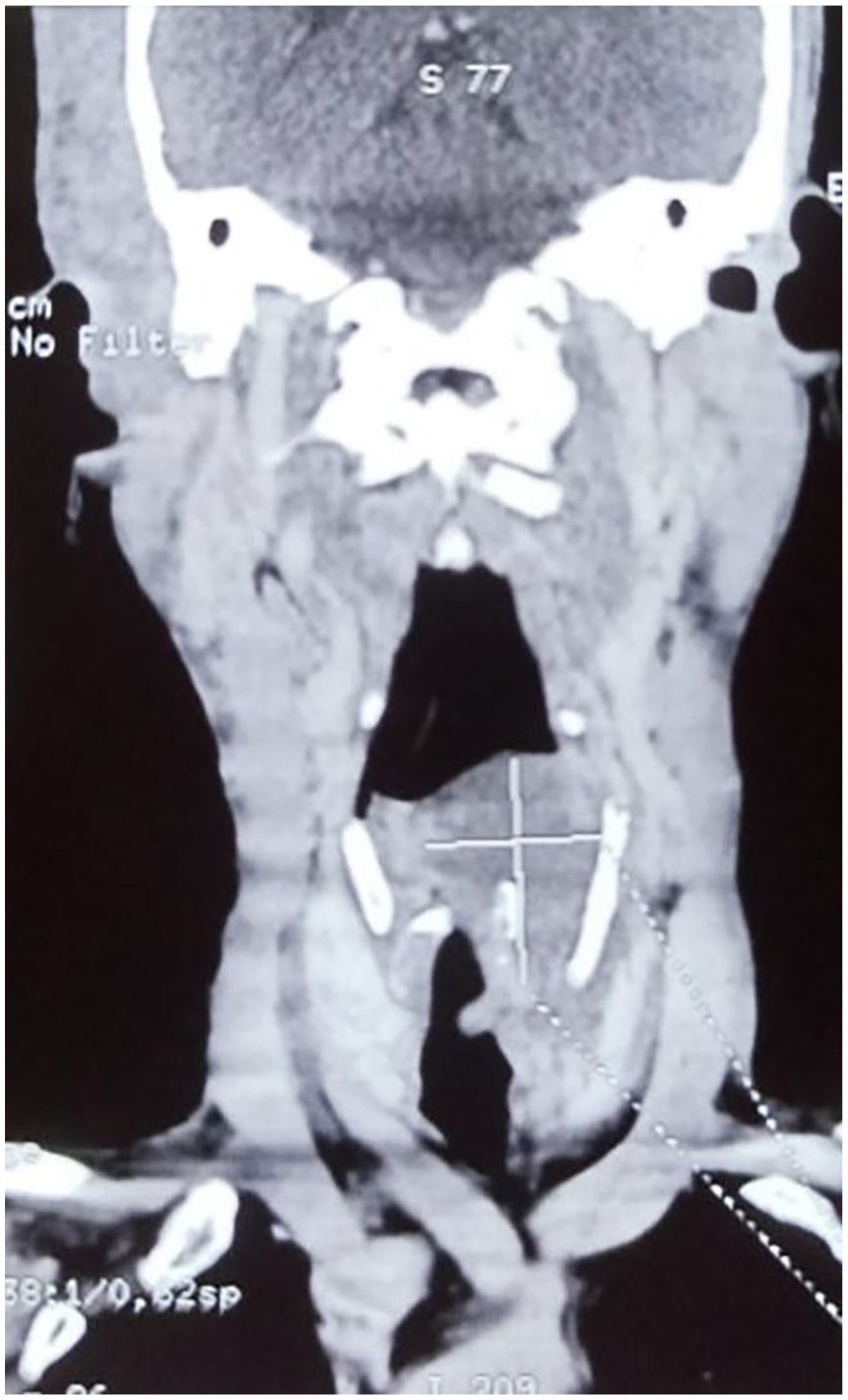
Contrast-enhanced computed tomography scan (coronal reconstruction) showing a homogenous globular mass in the oropharynx overlooking and extending caudally up to the level of left pyriform sinus.
The patient underwent routine preanesthetic investigations and checkup, and the results were unremarkable. After obtaining informed consent from the patient, a temporary tracheostomy was performed in anticipation of a difficult endotracheal intubation since the mass obscured a major portion of the pharyngeal lumen. The lesion was excised through endoscopic coblation-assisted laryngeal surgery (Figure 4). Immediate postoperative period was uneventful. Biopsy revealed the characteristic histopathologic features of a neurofibroma (Figure 5).
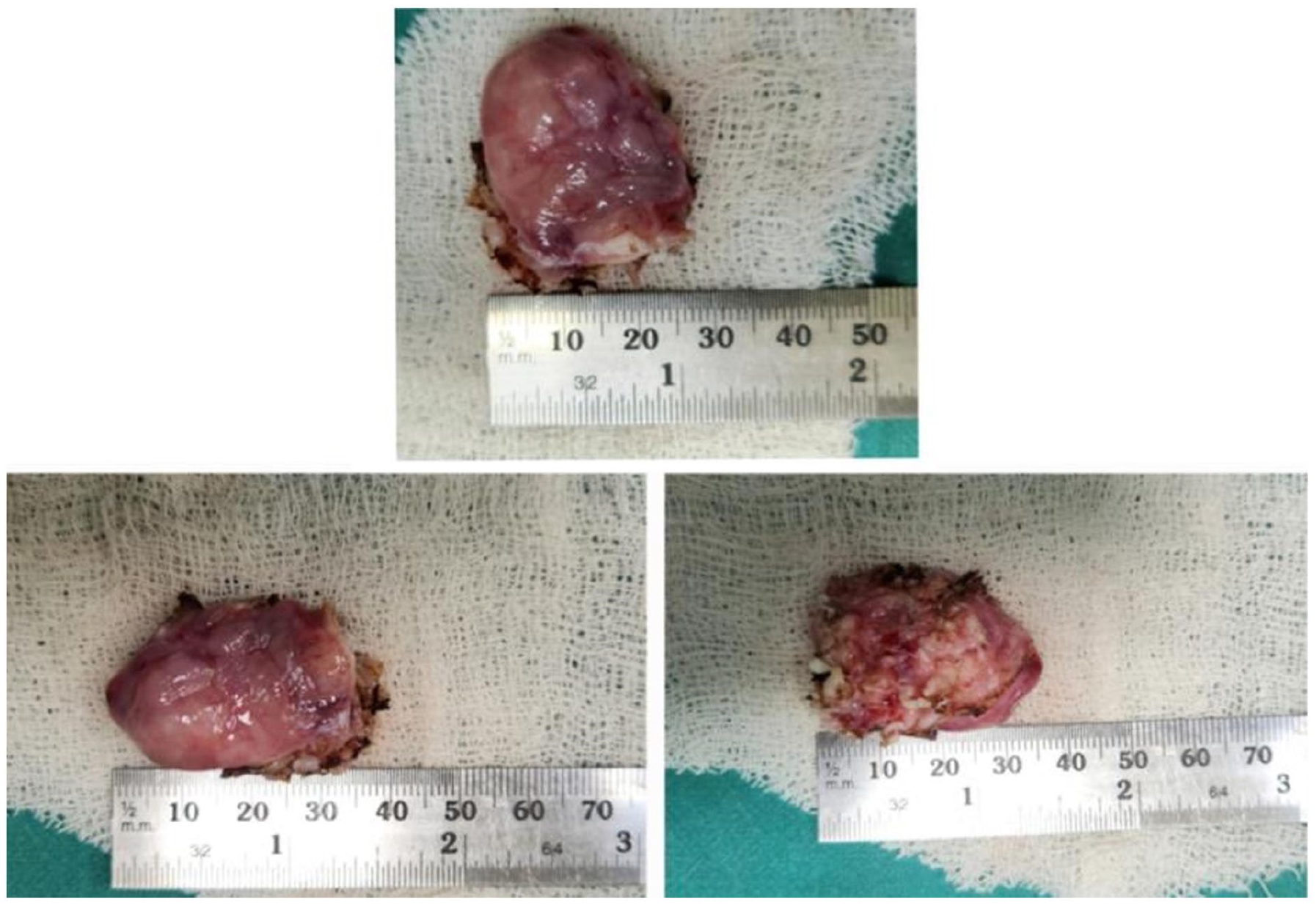
Gross specimen after excision through endoscopic coblation procedure.
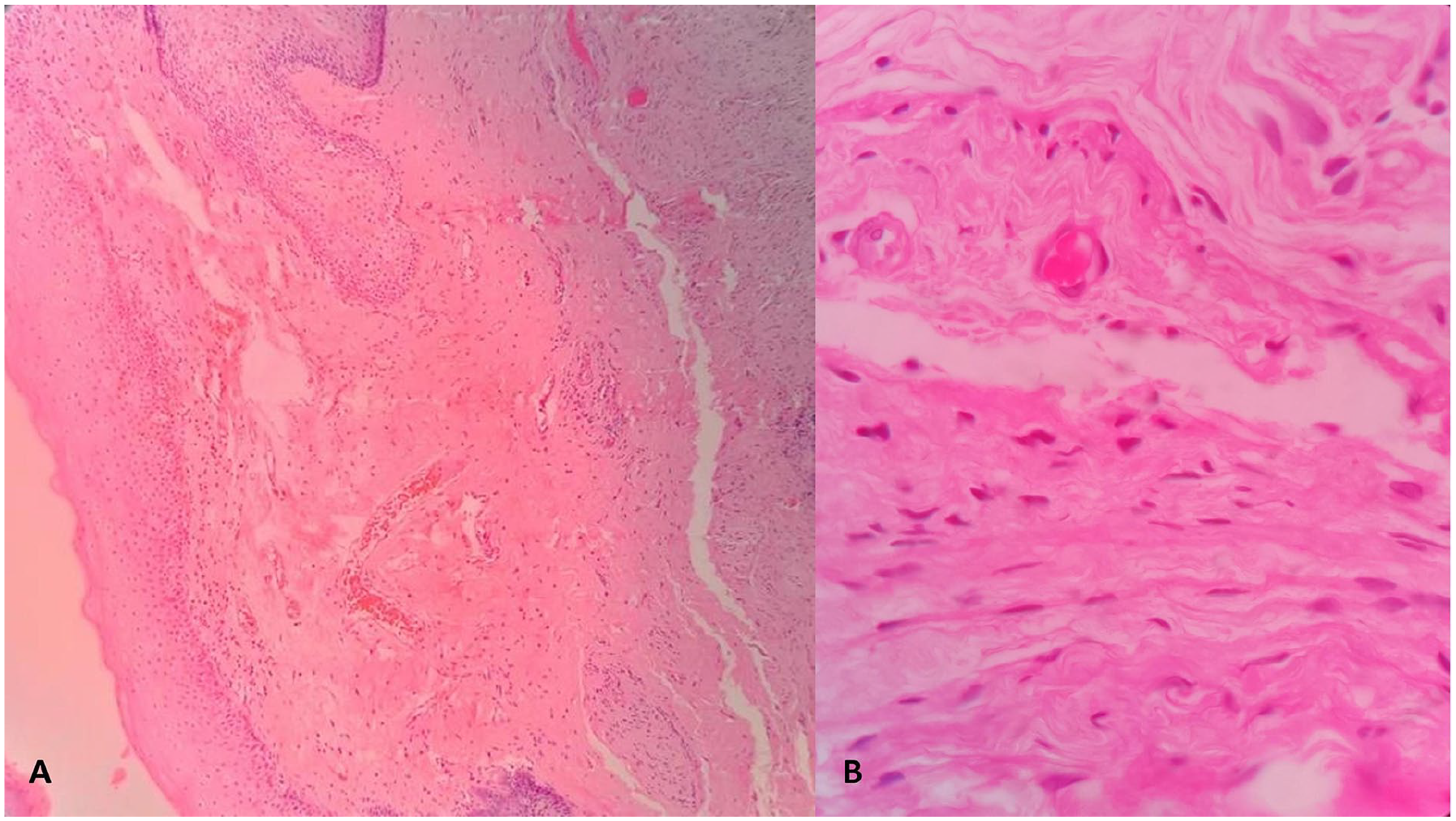
Microscopic examination of the oropharyngeal mass revealed wavy fascicles of short spindle cells (A; hematoxylin-eosin, 100×), which had dark wavy nuclei closely associated with collagen strands, and were without cellular atypia (B; hematoxylin-eosin, 400×). The features were suggestive of neurofibroma.
After 6 months of regular follow-up visits, the patient was found to be asymptomatic without having any evidence of recurrence.
Discussion
Neurofibroma is a benign Schwann cell nerve sheath tumor that is sometimes described as a hyperplastic hamartomatous deformity as opposed to an actual neoplastic disease. 7 The condition affects patients primarily in their third decade of life, although it is also encountered in the extremes of life—in patients as young as 10 months and as old as 70 years.2,8 There is no sex predilection for a neurofibroma and no known predisposing factors.2,8 Although isolated solitary neurofibromas in the head and neck region are fairly rare, oropharyngeal involvement is further uncommon.4,5,9,10 Clinical signs and symptoms are primarily due to the mass effect, which could range from dysphagia to stridor depending on the location and extent of the lesion. Although it is a slow-growing benign tumor, it may cause pain if it compresses a nerve. It may present as a smooth, mucosa-covered globular lesion in one of the several subsites of the oro- and hypopharynx. In addition, a thorough correlation between radiological (CECT) impression and clinical examination is essential to determine the type and size of the lesion prior to surgical excision.
As shown in our patient, airway obstruction has always been a risk with oropharyngeal neurofibroma; therefore, a tracheostomy was performed prior to surgical removal.
It is generally advisable to rule out solitary neurofibroma from the most prevalent differential diagnosis, that is, schwannoma. An encapsulated tumor of the nerve sheath, schwannomas are derived from differentiated Schwann cells, which are in charge of neural myelination. On the other hand, when a neurofibroma grows, it integrates itself with the surrounding tissues, and it becomes challenging to completely remove the tumor while still separating the nerve. The current gold standard for treating neurofibroma is surgical removal, particularly in patients where the condition results from the mass effect, like dysphagia and breathing difficulties. 5 A similar case of a patient was reported by Kodiya et al who underwent a tracheostomy and successful removal of a soft palate neurofibroma. 9 Seth et al have reported a similar clinical situation in which a patient with von Recklinghausen’s disease developed respiratory difficulties due to laryngeal neurofibroma. 6 However, the patient was not a good candidate for surgery owing to various skeletal abnormalities, cardiovascular problems, and an unpredictable course of the disease with aging.
Histopathology of the family of spindle cell neoplasms, including neurofibroma, is characterized by large, unencapsulated fascicles of small spindle cells that are without cellular atypia and typically have dark, wavy nuclei closely linked to the collagen strands. However, the disease is infiltrative, and complete surgical excision may never be easy for the surgeon, leaving with a potential risk of recurrence.11,12 To minimize recurrence or avoid leaving behind residual tumor, the surgeon may become relatively aggressive and overzealous, which may inadvertently lead to loss of important adjacent structures, and the resultant functional deficits and morbidity. The use of coblation effectively addresses this problem.
Coblation, a safer substitute for cold knife, microdebrider, or laser in laryngeal surgeries, uses bipolar waves to create a plasma field that dissociates the molecular bond within the soft tissues in a saline environment.13,14 This allows early wound healing and hemostasis with minimal thermal damage to the neighboring structures. This considerably reduces the chance of leaving behind residual lesion that may cause the symptoms to worsen, recur, or persist. This potentially obviates the need for a revision surgery. In the present illustration, the oropharyngeal neurofibroma was completely removed with the aid of endoscopic coblation.
There has been a recent upsurge in interest on coblation-assisted endo-/microlaryngeal surgery that ensures precision and minimal contiguous tissue damage. This preserves the delicate and functional laryngeal structures from scarring, thereby minimizing potential compromise with functions like airway protection, respiration, and phonation. With this primary aim, endolaryngeal coblation has been popularized in cordotomy in bilateral vocal fold immobility, arytenoidectomy for vocal cord paralysis, excision of adult and childhood respiratory papillomatosis, excision of laryngeal hemangioma in adults, laryngomalacia, and so on.15-21 However, addressing an oropharyngeal and pyriform sinus neurofibroma is the first of its kind application of endoscopic coblation-guided laryngeal surgery, and is a major perspective of interest in this clinical report.
The present illustration underlines the importance of maintaining a low threshold of suspicion in diagnosing an oro-/laryngopharyngeal mass lesion in a setting of a preexisting systemic disorder. Likewise, a patient with clinical evidence of von Recklinghausen’s disease presenting with features of obstruction in the upper aerodigestive tract and with endoscopic evidence of a submucosal mass lesion in and around the laryngeal inlet should indicate a provisional diagnosis of a neurofibroma. This clinical insight, aided by imaging and other related clinical investigations, provides with a logical methodology of management akin to a benign laryngeal lesion and a suitable plan of intervention can be decided. This article highlights the importance of holding on to this clinical acumen and the need for thorough systemic examination in a patient presenting with dysphagia with/without hoarseness. The suspicion of a nonmalignant oro-/laryngopharyngeal lesion was strong enough in this patient to opt for coblation-assisted laryngeal excision so that the mass could be excised in its entirety and no residual tissue was left behind.
To conclude, this article presents a rare event of oropharyngeal neurofibroma in a patient with von Recklinghausen’s disease that was successfully removed with endoscopic coblation. Such patients need a high index of suspicion for a benign lesion given the fact that von Recklinghausen’s disease may have neurofibromas spread throughout the body, and oro-/laryngopharynx and the laryngeal airway, albeit uncommon, may also be the sites for such lesions. In this patient, the neurofibroma in the upper aerodigestive tract was isolated and solitary, and it would have been difficult to predict a provisional clinical diagnosis had the patient not suffered from von Recklinghausen’s disease.
Footnotes
Data Availability
The clinical data for this patient are available from the authors and can be reproduced on request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
The clinical and surgical principles adopted for their management complied with the ethical standards of relevant national and institutional guidelines on human experimentation, as laid down in the Declaration of Helsinki, 1975, as revised in 2008.
Informed Consent
Written informed consent has been obtained from the patient for the publication of this clinic document.
Grant Number
Not applicable.
