Abstract

Significance Statement
Laryngeal neurofibromas are rare lesions that can present with hoarseness and respiratory distress. Differential diagnoses of submucosal globular swelling that appears cystic on contrast-enhanced computed tomography (CECT) should be discussed while managing such lesions in the laryngeal framework. However, when associated with von Recklinghausen’s disease (neurofibromatosis type 1), such a lesion should be considered neurofibroma, unless otherwise proven. Knowledge of this dictum should eliminate misdiagnoses and would help with definitive management planning so that the lesion can be adequately removed and the need for future interventions is minimized.
Text Proper
A 67-year-old woman with von Recklinghausen’s disease (neurofibromatosis type 1; NF-1) (Figure 1) presented with progressing hoarseness and dysphagia, along with occasional respiratory distress. She was frail, had kyphoscoliosis, and a stunted stature aggravated by pseudoarthrosis of the lower extremities. There were no comorbidities, but she had a long-term history of smoking and tobacco-chewing. This 67-year-old woman had von Recklinghausen’s disease (neurofibromatosis type 1). Note the large cafe-au-lait patches on her back and the evident scoliosis.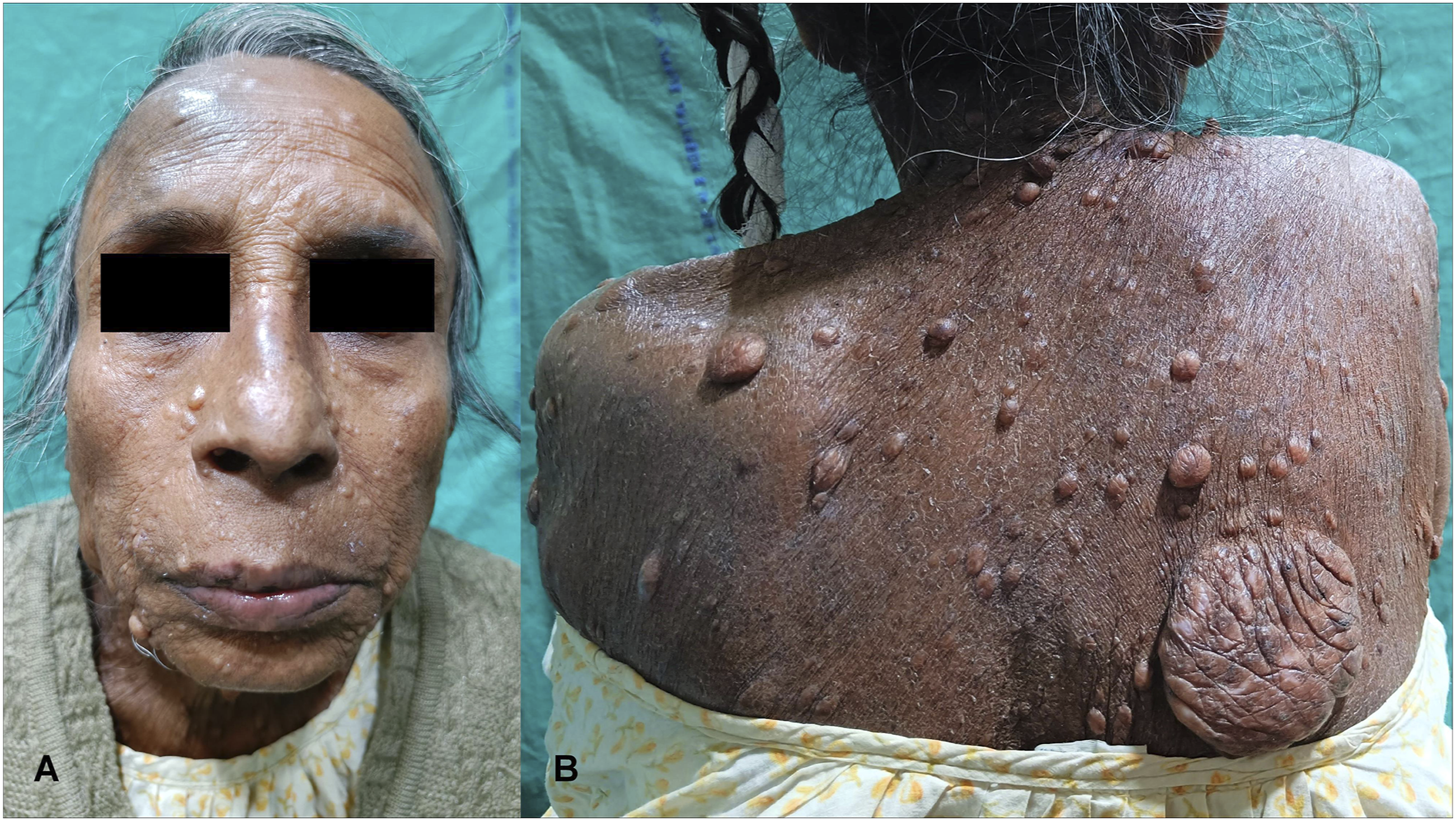
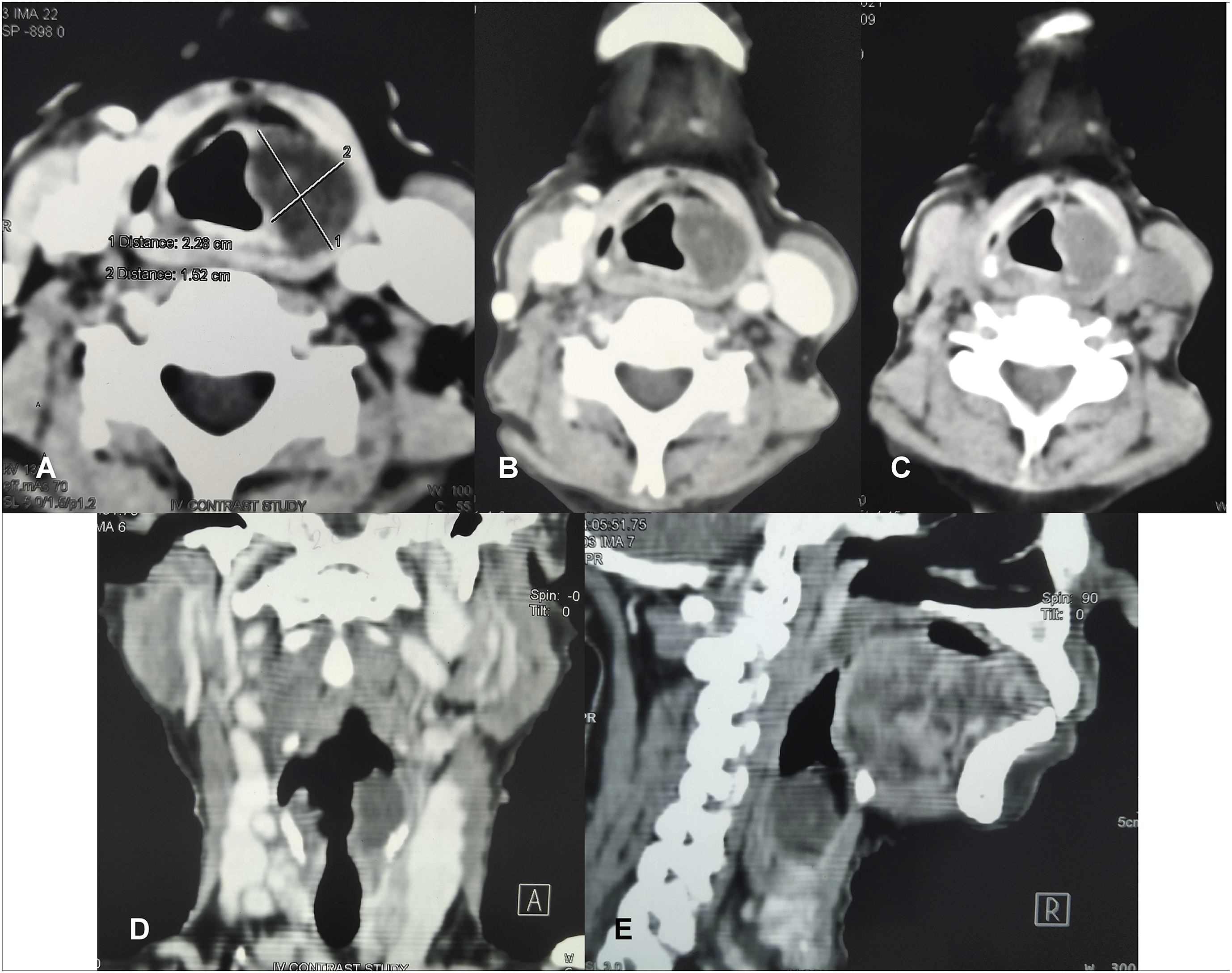
Fiber-optic laryngoscopy revealed a smooth, globular, submucosal swelling involving the left aryepiglottic fold, partly compromising the airway and obscuring the corresponding pyriform fossa (Figure 2). CECT showed a homogeneous mass in the medial wall of the left pyriform fossa, extending caudally beyond the level of glottis (Figure 3). The mass itself did not take up contrast, but there was rim-enhancement for which it appeared cystic. Few close differentials were considered. Because the patient had NF-1, laryngeal neurofibroma was a distinct possibility. Other probabilities were aryepiglottic fold cyst, saccular cyst, benign minor salivary gland neoplasms, and, with smoking as a risk factor, a remote chance of submucosal malignancy. A microlaryngoscopic evaluation under general anesthesia was planned for deciding the next steps in intervention. Fiber-optic laryngoscopy revealed a large globular submucosal swelling involving the left aryepiglottic fold, extending inferiorly. The lesion almost obscured the corresponding pyriform sinus and partly compromised the laryngeal airway. Contrast-enhanced computed tomography of the neck (soft tissue windows; A–C: serial axial cuts, from superior to inferior; D: coronal reconstruction; E: parasagittal reconstruction) revealed a homogeneous radiolucent globular mass involving the left aryepiglottic fold that extended caudally up to a level beyond the glottis. Mild rim enhancement was observed in the axial cuts for which the mass appeared cystic.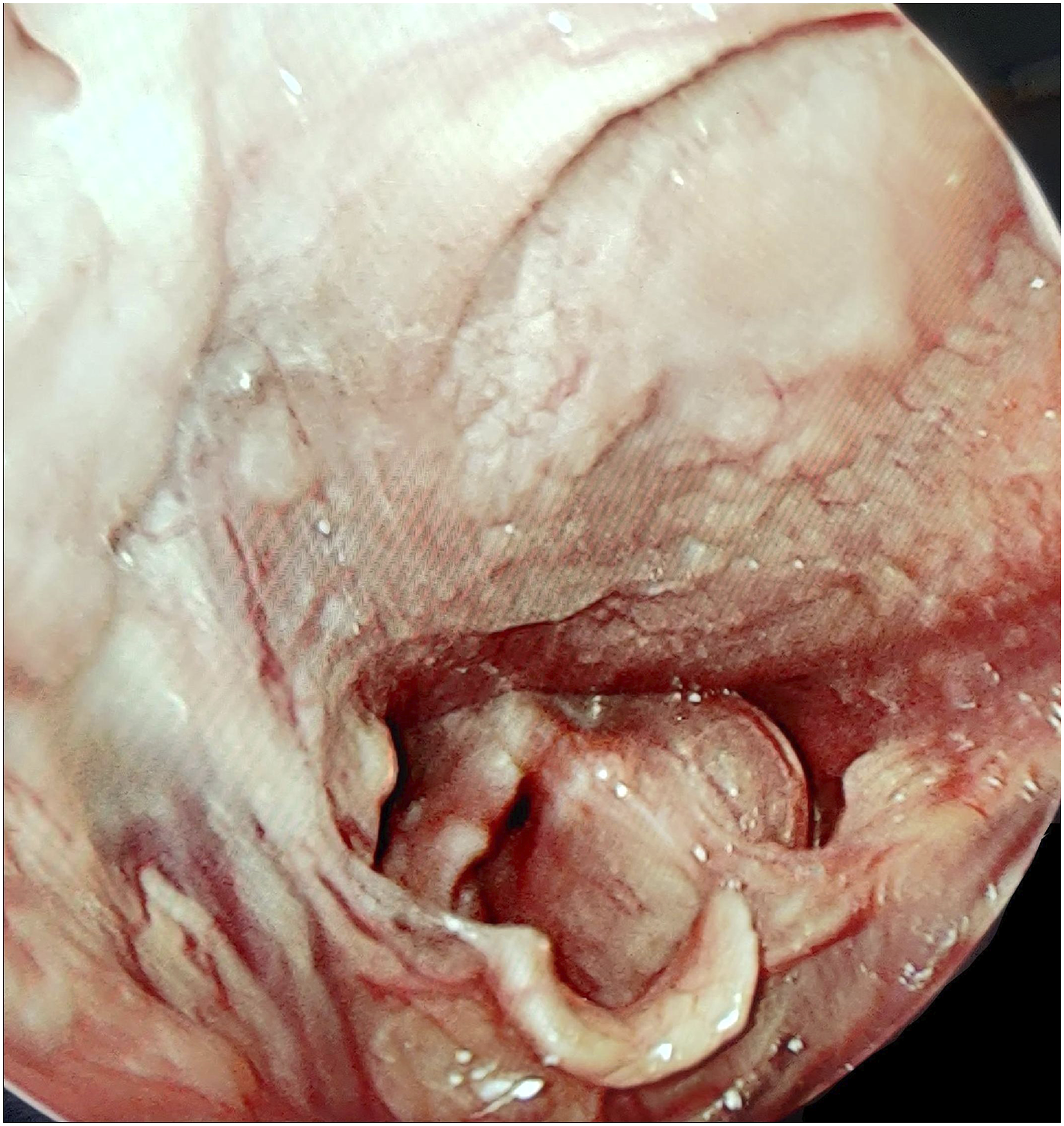
A difficult endotracheal intubation was anticipated considering the patient’s stature and skeletal abnormalities. However, the anesthetists successfully intubated her without the need for tracheostomy. The microlaryngoscopic view confirmed the findings at fiber-optic laryngoscopy. A careful incision was made in the mucosa covering the swelling close to the false vocal fold, just superior to the area that appeared most bulged. A submucosal plane was created on the luminal side. A globular mass was exposed which was soft but did not feel cystic at this time. However, its full extent could not be uncovered. It was adherent to the overlying mucosa with significant lateral and inferior extent. It was not possible to excise the entire lesion through microlaryngoscopic approach, although most of it was accessible and could be removed. Histopathology revealed spindle cells and collagen fascicles arranged in wavy arrays in an eosinophilic mucinous stroma, suggesting neurofibroma (Figure 4). Histopathology photomicrograph showed spindle cells and collagen fascicles arranged in wavy arrays in an eosinophilic mucinous stroma, suggesting neurofibroma (Hematoxylin-Eosin; x100).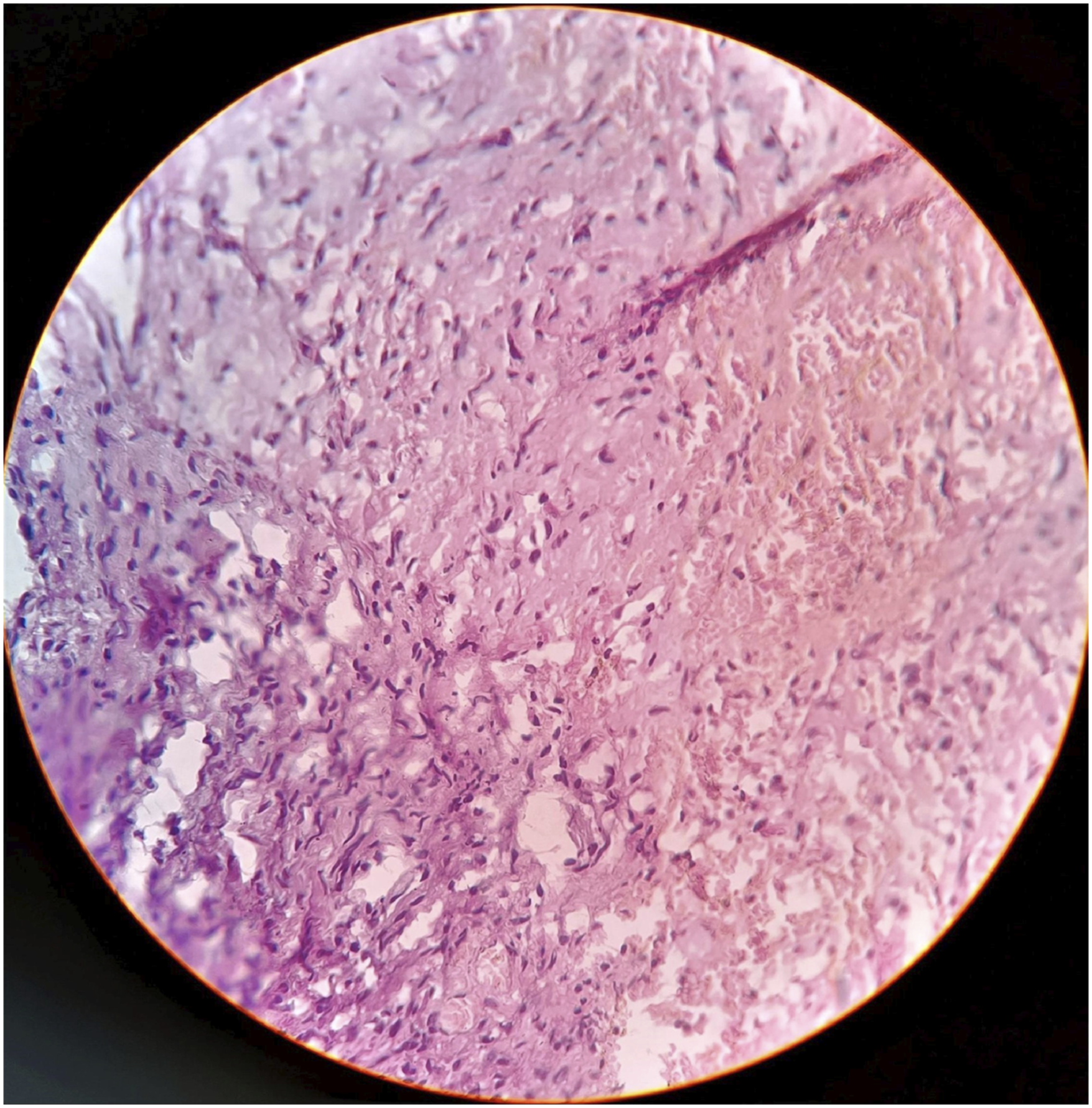
The patient and her daughter (her primary caregiver who also had NF-1) were informed about the incomplete removal of the mass and were advised for long-term follow-up and for a possible revision surgery through an external approach if respiratory distress returns and aggravates. At 4 months, the patient has been doing well, although she still has a hoarse voice.
Neurofibroma of the larynx is seldom encountered in routine otolaryngology practice. To date, less than 40 patients have been reported, and the overall incidence is .03–.1% of the benign tumors of the larynx.1,2 Most patients with laryngeal neurofibroma have NF-1, although isolated laryngeal neurofibromas could be more common than those associated with NF-1.3,4 Such observations are mainly from the Orient (Japan), indicating that there may be geographic and/or ethnic factors determining the association between laryngeal neurofibroma and NF-1.3,4
Here, the terms “solitary” and “isolated” should be distinguished. The former refers to a single laryngeal neurofibroma associated with NF-1, as seen in the present patient. However, the term “isolated” connotes laryngeal neurofibroma as a discrete entity not associated with NF-1, although the reported isolated laryngeal neurofibromas are actually single in number. 5
When associated with NF-1—a well-defined and relatively common disorder (1 in 2500–3000 births) 6 —the sparse reporting of laryngeal neurofibromas opens up avenues for discussion. Under-reporting is possible because smaller neurofibromas in the laryngeal framework can remain asymptomatic throughout life. A definitive way to estimate true incidence of laryngeal neurofibromas associated with NF-1 could be cadaver studies. Although routine laryngeal examination might not be warranted in patients with NF-1 having no laryngeal symptoms, it can be deduced that any benign laryngeal neoplasm associated with NF-1 should be considered neurofibroma until proven otherwise. With this knowledge, misdiagnoses could be avoided and management can be planned methodically. On retrospection, magnetic resonance imaging seems to be a more appropriate imaging modality when a laryngeal lesion is associated with NF-1.
In the present patient, documented rarity of laryngeal neurofibroma and the cystic appearance in CECT made the authors consider other diagnoses and adopt an open mind regarding the management. Since patients with NF-1 can have multiple skeletal abnormalities, 6 cardiovascular problems, 6 unpredictable progression of disease with age, 6 and may present with respiratory distress or stridor, 7 endotracheal intubation can be challenging and can risk the provision for a second look surgery without a tracheostomy. Large laryngeal neurofibromas are often not possible to excise completely. In fact, partial endoscopic resection as a minimally invasive method is currently preferred over complete resection with more aggressive approaches (open transcervical). 8 Nevertheless, a low threshold of suspicion would guide surgeons to consider more decisive planning and adopt a definitive approach (laser, cold steel [open/endolaryngeal]), allowing complete resection. This would obviate possible revision surgery or a tracheostomy in potential situations when symptoms may re-appear, persist, or aggravate due to the residual mass.
Footnotes
Author’s Note
This Clinic paper is dedicated to the patient’s 40-year-old daughter who has inherited von Recklinghausen’s disease following her family’s pedigree. She has decided to donate her body after death for reasons she believes would advance research to understand the pathobiogenesis of von Recklinghausen’s disease.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Statement
Written informed consent has been obtained from the patient for the publication of this Clinic paper. The clinical and surgical principles adopted for her management complied with the ethical standards of the relevant national and institutional guidelines on human experimentation, as laid down in the Declaration of Helsinki, 1975, as revised in 2008.
Data Availability Statement
The clinical data pertaining to this patient are available with the authors and can be reproduced on request.
