Abstract
Sinonasal angiomatous polyp (SAP) is a benign pseudoneoplastic lesion rarely reported in the literature. It may be misdiagnosed as a malignant neoplasm due to its aggressive features of bone erosion clinically or on imaging. We report the case of a 43-year-old woman with a 3 month history of unilateral nasal obstruction with recurrent epistaxis. Nasal endoscopy showed polypoid nasal mass occupying the left nasal cavity. Imaging was suggestive of malignant vascular tumor. The patient underwent surgical excision after embolization. Histopathology concluded to the diagnosis of SAP.
Introduction
Sinonasal angiomatous polyp (SAP) is a benign and nonneoplastic lesion rarely reported in the literature. 1 It is a rare entity of inflammatory sinonasal polyps, characterized by extensive vascular proliferation and ectasia, constituting only 4% to 5% of all nasal polyps.2,3 There are few detailed reports within the literature of SAPs, their management, and outcome. This entity is often misdiagnosed as a malignant sinonasal tumor due to its aggressive clinical and radiological features mimicking a neoplastic lesion.
We report a rare and aggressive case of SAP and we discuss through this case with review of literature, its clinicopathological features, and its therapeutic management.
Case Report
A 43-year-old woman presented to our department with a 3 month history of unilateral nasal obstruction, associated with recurrent minor epistaxis from left side of the nasal cavity. There was no history of systemic bleeding, blood transfusion, or nasal packing, and the hemostasis biological assessment was correct.
Nasal endoscopy revealed a red left-sided polypoid nasal mass obstructing the left nasal cavity (Figure 1A’).
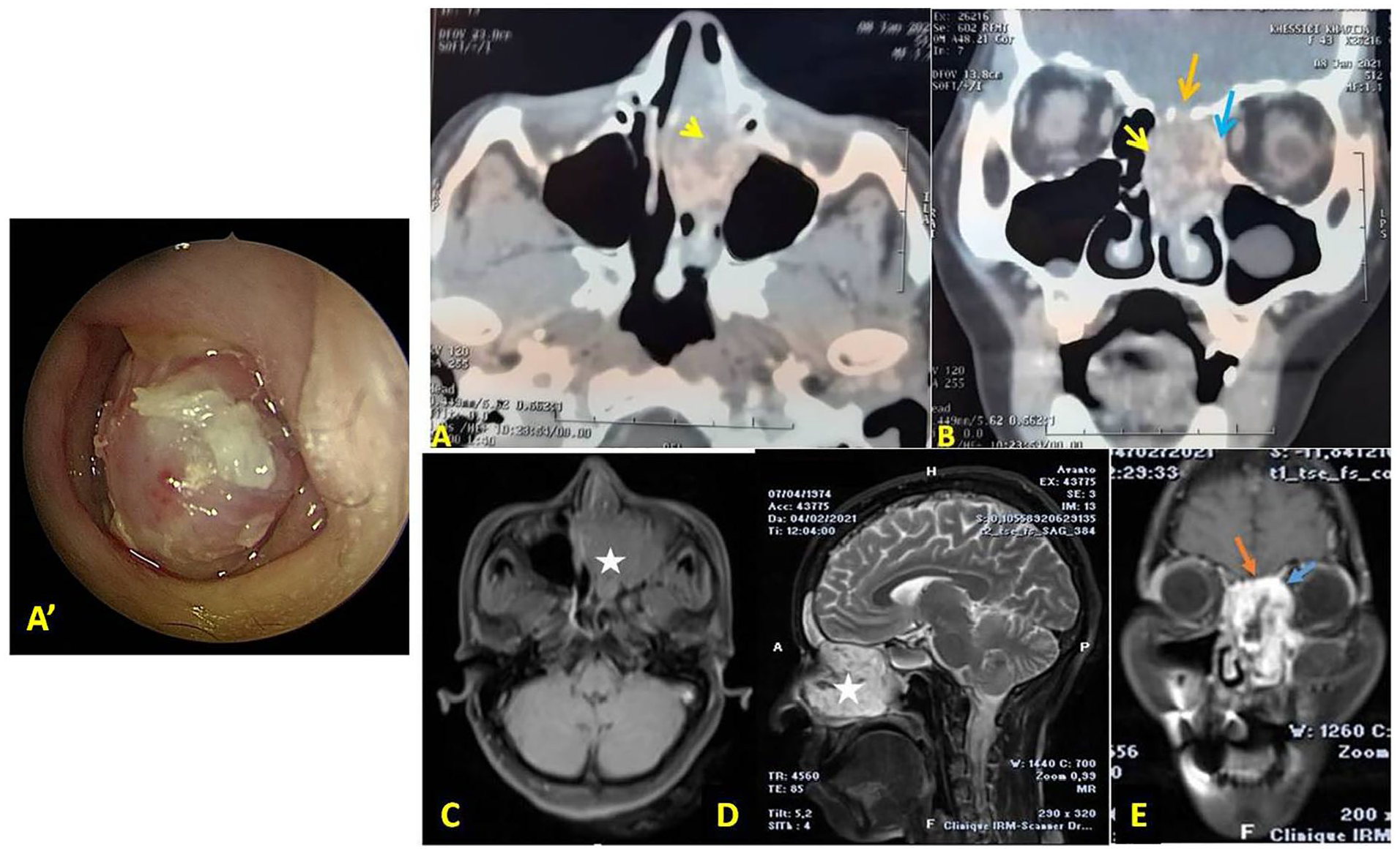
(A’) Endoscopic view showing polypoid mass in the left nasal cavity. Axial (A) and coronal (B) contrast enhanced CT scan and axial T1 (C), sagittal T2 (D), and coronal T1 gado MRI (E) showing soft tissue lesion centered in the left nasal fossa and maxillary sinus with heterogeneous enhancement with erosion of the medial wall of maxillary sinus and the lamina papyracea (blue arrow) of left orbit without intraorbital involvement. There was also bone erosion in base of the skull without evidence of intracranial involvement (orange arrow). CT, computed tomography; MRI, magnetic resonance imaging.
The origin of the mass could not be confirmed. The right nasal cavity and the nasopharynx were normal. No clinical cervical adenopathy was described.
A biopsy of the mass was performed at another institution, and was suggestive of nasal angiofibroma.
A contrast-enhanced computed tomography (CT) realized showed a huge well-circumscribed, soft tissue lesion centered in the left nasal fossa and maxillary sinus with heterogeneous enhancement. There was an erosion of the medial wall of maxillary sinus and the lamina papyracea of left orbit without intraorbital involvement. There was also bone erosion in base of the skull without evidence of intracranial involvement (Figure 1A and B).
The mass expands to the posterior part of the right nasal cavity.
On magnetic resonance imaging (MRI), the sinonasal lesion appears hypointense on T1 and hyperintense on T2 with homogeneous, rapid, and intense enhancement. There was no orbital or intracranial extension (Figure 1C, D, and F).
According to these findings, a primary diagnosis of sinonasal vascular tumor was made and the patient was planned for surgical resection.
Embolization was performed 48 hours preoperatively to reduce operative blood loss. The whole tumor was removed using paralateronasal approach. Intraoperatively, there was a huge, friable, necrotic mass arising from the left nasal cavity extending to maxillary sinus, nasopharynx, and ethmoid sinus with diffuse hemorrhagic aspects. The anterior and medial maxillary sinus wall was eroded. There was also destruction of lamina papyracea without intraorbital involvement. All the attachments of the mass were cauterized to reduce the vascularity and blood loss during surgery. A 4 cm bony defect was identified in the base of the skull without a breach. The bone defect was repaired using septal cartilage, abdominal fat, and biological glue. Using this approach, the margin of the mass was identified. Blood loss was minimal and the patient had no complications during and after the surgery.
Final histopathologic examination of the lesion concluded to an SAP (Figure 2).
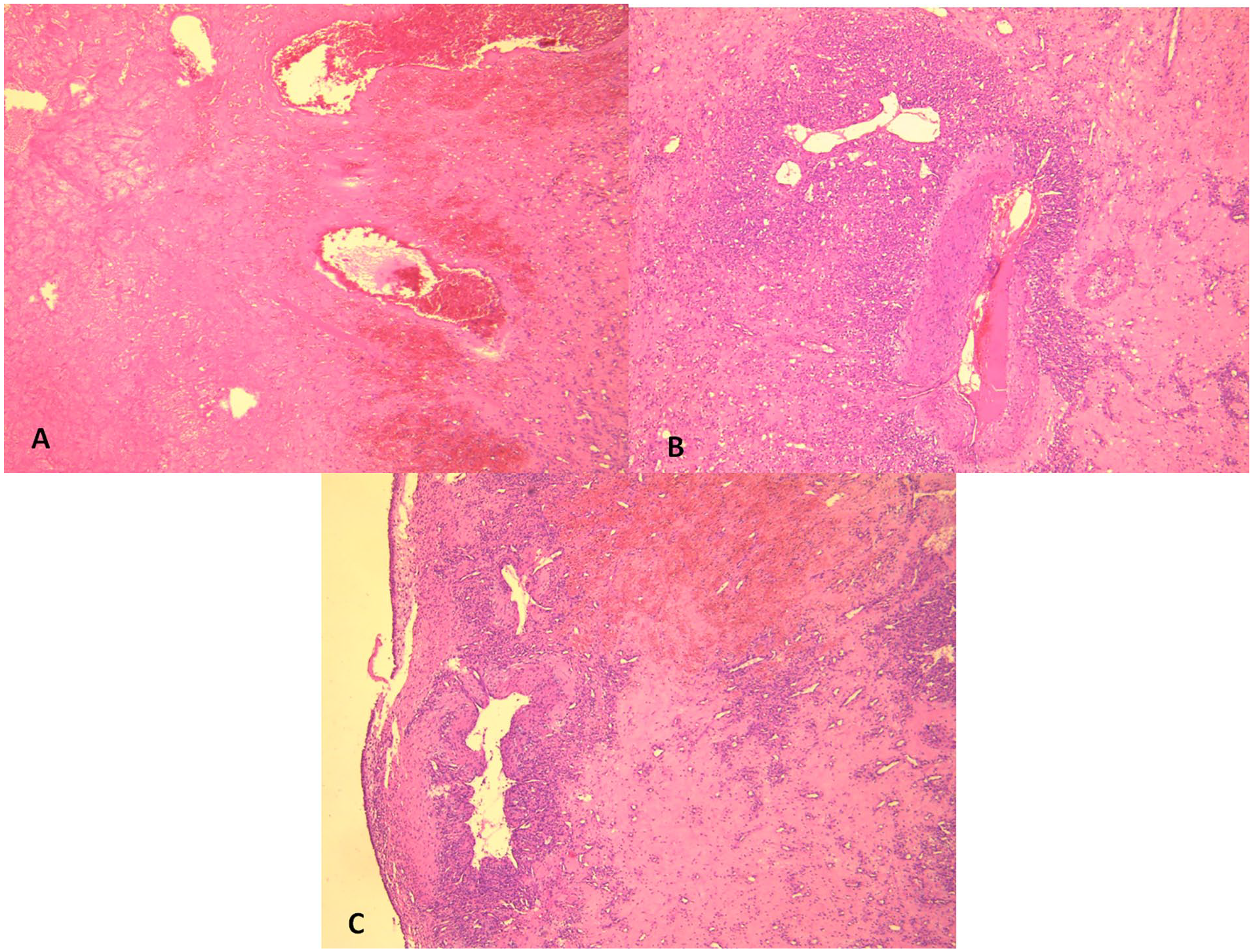
(A) Polypoid mass lined by respiratory epithelium overlying a fibrous stroma with neovascularization and areas of hemorrhage (hematoxylin eosin ×40). (B and C) Stroma containing thick- and thin-walled vascular channels (B) and areas of hemorrhagic necrosis (C) (hematoxylin eosin ×40).
After 12 months of follow-up, the patient presented a minor recurrence on endoscopy, which was revised surgically with good subsequent evolution.
Discussion
SAP is a rare benign and pseudoneoplastic lesion.1,5 It only accounts for about 4% to 5% of all nasal polyps.3-6 It is characterized by extensive vascular proliferation and ectasia. 2 It is often confused with sinonasal malignant tumor due to its aggressive presentation. 3 This entity is described in the literature in different terms, which share the same clinicopathological and imaging features. These lesions include organized hematoma, cavernous hemangioma, angioectatic nasal polyp, and angiomatous polyp.2,4,6 The pathological mechanism of SAP remains poorly understood. Several hypotheses for the pathogenesis of SAPs have been reported in the literature.3,5,7,8
Clinical characteristics of SAP are varied and nonspecific. 4 It usually occurs from the age of 12 to 72 years. The most common symptoms are progressive nasal obstruction, spontaneous epistaxis, and facial pain. 3 They present clinically as soft, gelatinous translucent polypoidal, painless mass filling up the maxillary sinus or nasal cavity. Locally, SAP may cause erosion, displacement of the adjacent bony structures, facial deformity, and proptosis, which can evoke malignant sinonasal tumor. 6
CT examination is essential for the evaluation of enhancement patterns and vascularity of sinonasal tumors. 4
The typical features of this tumor are expansible mass in the sinus with bony wall destruction and remodeling.4,9 Thus, CT findings for SAP are not characteristic and may be similar to those of malignant or other sinonasal tumor. As CT findings lack specificity to identify this lesion, Ding et al have reported that the vessel-like marked enhancement and progressive enhancement in both early and delayed phase scans are typical of SAP, which could be a useful tool to make more confirmative diagnosis before surgery.1,9
On MRI, the lesion is hypointense on T1, heterogeneously hyperintense on T2 with characteristic peripheral hypointense indicating hemosiderin deposition due to old hemorrhage.5,7,9 Dynamic contrast examination is recommended as the lesion will show gradual enhancement pattern typical of a vascular tumor. The neovascularized areas within the lesion demonstrated hyperintensity on T2-weighted image, hypointensity on T1-weighted image, and strong enhancement on postcontrast MR images. The high signal intensity area surrounded by peripheral hypointense is a characteristic finding of SAP.5,9
Differential diagnosis between SAP and other nasal masses, including juvenile angiofibroma, hemangioma, inverted papilloma, and malignant sinonasal tumor, can be challenging. In fact, SAP can present clinical and radiological characteristics often associated with malignancies, such as rich vascularization, erosive behavior toward the lateral nasal wall, or it is contrast enhancing.6,10
Histopathological examination is required for the final confirmatory diagnosis.11,12 Microscopically, SAP presented with irregularly shaped and thin-walled blood vessel. The stroma is hypocellular and expanded by edema and amorphous eosinophilic material. Areas of hemorrhagic necrosis are also seen with thrombus formation and neovascularization. 3 Although atypical mesenchymal cells can raise the suspicion of a soft tissue sarcoma, they should be considered in the context of the reactive changes they are associated with. 7 Sometimes pseudoepitheliomatous hyperplasia of the surface epithelium of SAP may raise suspicion of squamous cell carcinoma which can be easily ruled out on histopathology.
In our case, the diagnosis of nasal angiofibroma was initially made. Final histological examination rectified the diagnosis. Therefore, deep biopsy is warranted to confirm SAP and to exclude malignancy. 3
Currently, the treatment of choice of SAP is surgical resection. Preoperative embolization to reduce the vascularity of the tumor and facilitate complete resection is also an option if the tumor is highly vascularized.
A number of surgical approaches have been described for accessing sinonasal lesions. Actually, there are no established guidelines for management. In most of the reported cases, recent advances in endoscopic surgical techniques allowed endoscopic surgical resection of sinonasal hemangiomas.6,10,13,14 In our case, in view of the extensions of the tumor and its large size, we opted for an open approach with preoperative tumor embolization.
The recurrence rate after surgery is very low as revealed by Dai et al, who do not show any recurrence observed during at least in 60 months period follow-up postoperatively. 3
Conclusion
The SAPs are uncommon pseudoneoplastic lesions that can pose interesting pathophysiological questions and present a difficult diagnostic problem. A correct preoperative diagnosis is important for patients with SAP to avoid unnecessary extensive surgery. The treatment of choice is to completely remove the polypoid mass and restore the drainage system of all sinus cavities. The recurrence is rare.
Footnotes
Acknowledgements
None.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable (no data or figures that compromise the identity of the patient).
Informed Consent
Not applicable (no data or figures that compromise the identity of the patient).
