Abstract
Introduction
The parapharyngeal space (PPS) lies at the posterior aspect of the infratemporal fossa and above the inferior border of the medial pterygoid muscle. 1 The PPS is divided into the pre- and retro-styloid compartments by the styloid process and the attached stylopharyngeal aponeurosis. 2 Adipose tissue, lymph node, and the deep lobe of the parotid gland are the main structures within the pre-styloid space; whereas the retro-styloid compartment contains the parapharyngeal internal carotid artery (pICA), internal jugular vein, and lower cranial nerves (CN IX to XII).3,4 Due to the complex neurovascular relationships of the PPS, extirpation of tumors arising from the PPS still carry great challenges. 5
Multiple minimally invasive approaches have been adopted to address lesions arising from the PPS, among which the endoscopic transnasal and transoral corridors have gained popularity during the past 3 decades.6-8 Using these and other approaches for the resection of benign lesions in the PPS yield satisfactory outcomes with a low incidence of complications or sequelae.9,10 Of note, various anatomical structures, such as the foramen ovale, posterior trunk of V3, lateral pterygoid plate, and tensor veli palatini muscle, have been advocated as landmarks to guide surgical procedures within the PPS and the infratemporal fossa.11-13
Identification and protection of the pICA is critical for any procedure in the PPS, 11 especially when using minimally invasive corridors in a medial to lateral trajectory (ie, transnasal and transoral approaches). 14 Prior studies have shown that the ascending pharyngeal artery (APA) accompanies the pICA as it traverses in the PPS,1,3 and these studies identified its course and anatomical variations, as well as variable correlations with the pICA.15,16
The APA supplies the lower cranial nerves (CN IX to XII) and connects the vertebrobasilar system through collateral communications.17,18 Moreover, the APA may contribute to the blood supply the tumors arising from the PPS. 19 Therefore, intraoperative identification of the APA may provide an additional reference to locate the pICA and to avoid its inadvertent injury. A strategy for intraoperative identification and protection of the APA in this context has not been sufficiently explored and proposed.
This study hypothesized that the APA may serve as an additional landmark for tumor removal in the PPS. Therefore, this study aims to assess anatomical variations of the APA, its correlation with the pICA, and to elucidate surgical implications for the extirpation of benign tumors arising in the PPS.
Materials and Methods
Cadaveric Dissection
The APA in PPS was dissected and assessed on 10 cadaveric heads (20 sides). Course variations of the APA to the pICA were recorded. The cadaveric dissections were conducted at the Anatomy Laboratory Toward Visuospatial Surgical Innovations in Otolaryngology and Neurosurgery (ALT-VISION) at the Wexner Medical Center of The Ohio State University. ALT-VISION and all coauthors were certified by local regulatory agencies dealing with the use of human tissues and cadaveric studies.
Visualization was achieved using rigid rod-lens endoscopes (4-mm diameter, 18-cm length) with 0° scope (Karl Storz Endoscopy; Karl Storz, Tuttlingen, Germany), coupled to a high-definition camera and video monitor. Both video and standard digital images were recorded during dissections using the AIDA recording system (Karl Storz Endoscopy; Karl Storz, Tuttlingen, Germany). A high-resolution CT scan was performed before the dissection and the data were exported to a surgical navigation system (Stryker, Kalamazoo, MI, USA).
Clinical Data
The study comprised a retrospective analysis of patients who underwent an endoscopic transoral extirpation of PPS tumors from January 1, 2014 to December 31, 2018. Informed consent was obtained from all patients and the study protocol was approved by the Ethics Committee of Beijing Tongren Hospital, Capital Medical University in China (no. TRECKY2016-036). During the tumor removal, the course of the APA, its relationship to the pICA, and the tumor in the PPS were assessed and recorded.
Results
Cadaveric Dissection
Technical nuances of surgical access into the PPS have been previously described in detail. 1 Following removal of the adipose tissue in the pre-styloid compartment, the stylopharyngeal aponeurosis was exposed (Figure 1A). The stylopharyngeal aponeurosis was subsequently peeled in a medial to lateral fashion, and the APA was encountered prior to exposing the pICA (Figure 1B). The APA was located at the medial aspect of the tensor veli palatini muscle in all 20 sides (Figure 1C).
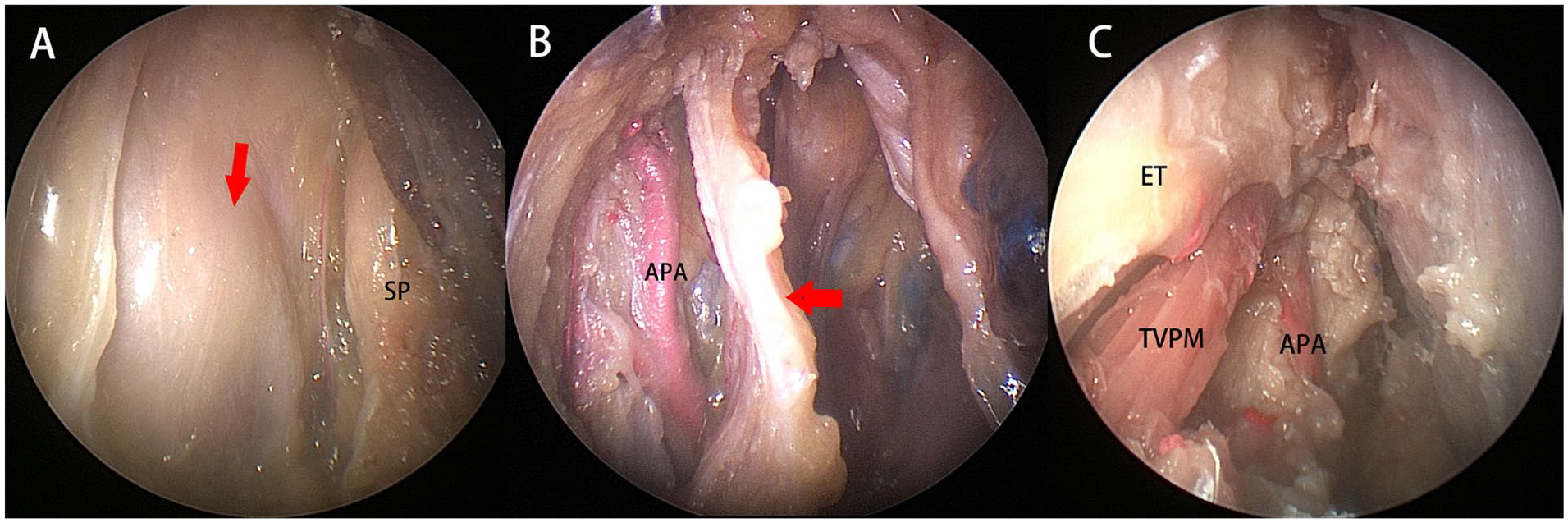
Left side of the specimen, viewed by 0° scope. (A) The stylopharyngeal aponeurosis of the left side was exposed (arrow); (B) after peeling of the stylopharyngeal aponeurosis (arrow), the APA was encountered; and (C) the APA located at the lateral aspect of the TVPM. APA, ascending pharyngeal artery; ET: Eustachian tube; TVPM, tensor veli palatini muscle.
The APA was found to travel at the medial (Figure 2A) and posteromedial (Figure 2B) aspects of the pICA in 12 (60%) and 4 (20%) sides, respectively. In the remaining 4 sides (20%), the APA branched into several subcategory arteries lying at the medial and lateral aspects of the pICA (Figure 2C), respectively.
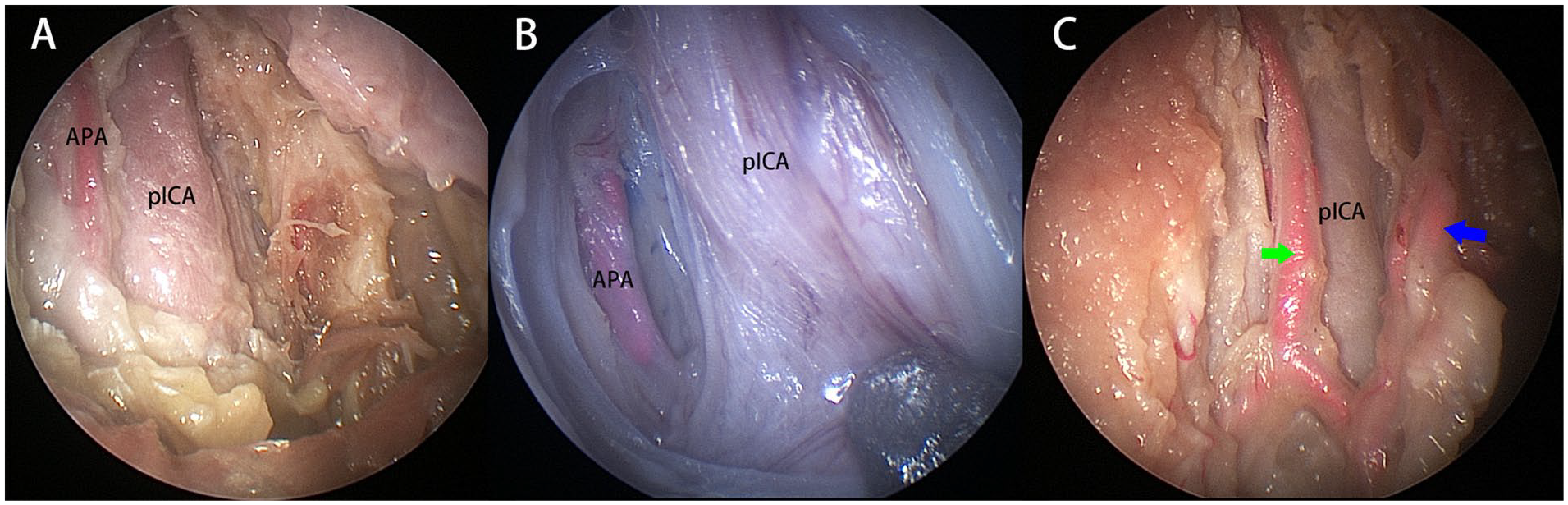
Left side of the specimen, viewed by 0° scope. The APA (left side) courses vertically upward at the medial (A) or posteromedial (B) aspect of the pICA; (C) the branches of APA course at the medial (green arrow) and lateral (blue arrow) aspect of the pICA. APA, ascending pharyngeal artery; pICA, parapharyngeal internal carotid artery.
In 13/20 sides of the specimens (65%), branches of the APA were identified (Figure 3A). Two branches were identified in 9 sides and 3 branches in 4 sides, respectively. At the superior aspect of the PPS close to the skull base, the APA coursed vertically at the medial or posteromedial aspect of the pICA (Figure 3B). Branches of the APA passing through the hypoglossal canal and the jugular foramen could also be observed (Figure 3C).
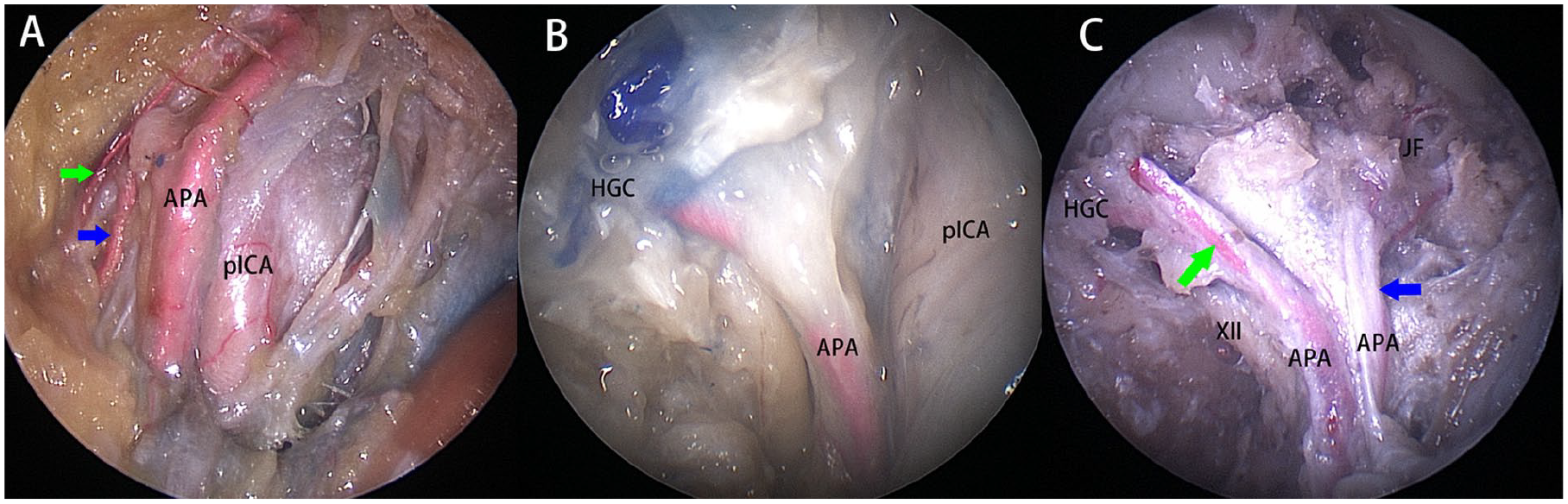
Left side of the specimen, viewed by 0° scope. (A) The branches of APA (blue and green arrows); (B) when coursed upward close to the HGC, the APA located at the posteromedial aspect of the pICA; (C) the branches of APA nurturing the HGC (green arrow) and JF (blue arrow). APA, ascending pharyngeal artery; HGC, hypoglossal canal; JF, jugular foramen; pICA, parapharyngeal internal carotid artery.
Intraoperative Validation
The technical details of the endoscopic transoral approach for extirpation of tumors arising from the PPS has been previously reported.8,20 Tumors arising in the pre-styloid compartment in this cohort included pleomorphic adenoma (n = 7), basal cell adenoma (n = 1), and cavernous hemangioma (n = 2). The APA was not encountered in any of the patients with pleomorphic adenoma or cavernous hemangioma (n = 9, 90%). In the patient with a basal cell adenoma (pre-styloid compartment; Figure 4A), however, both the APA and the pICA had been displaced to the medial aspect of the tumor. The tumor was subsequently removed en bloc with preservation of both vessels (Figure 4B).
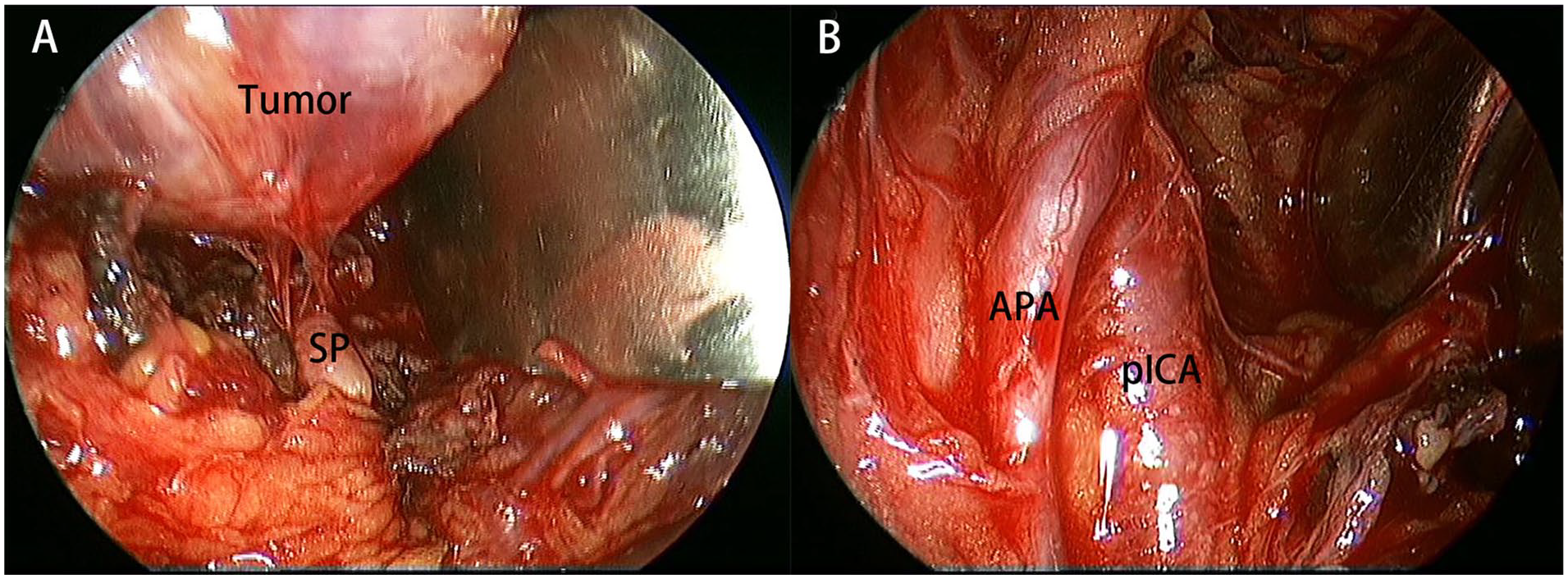
Left PPS, viewed by 0° scope. (A) The tumor located anterior to the SP; (B) intraoperative identification of the APA and the pICA. APA, ascending pharyngeal artery; pICA, parapharyngeal internal carotid artery; SP, styloid process.
All retro-styloid compartment tumors included in this cohort were schwannomas (n = 10). In all these 10 patients, the pICA was displaced into the anteromedial aspect of the tumor (Figure 5A). While the APA was identified in 8 patients (80%) on the preoperative magnetic resonance imaging (MRI) at the anteromedial aspect of the pICA (Figure 5B).
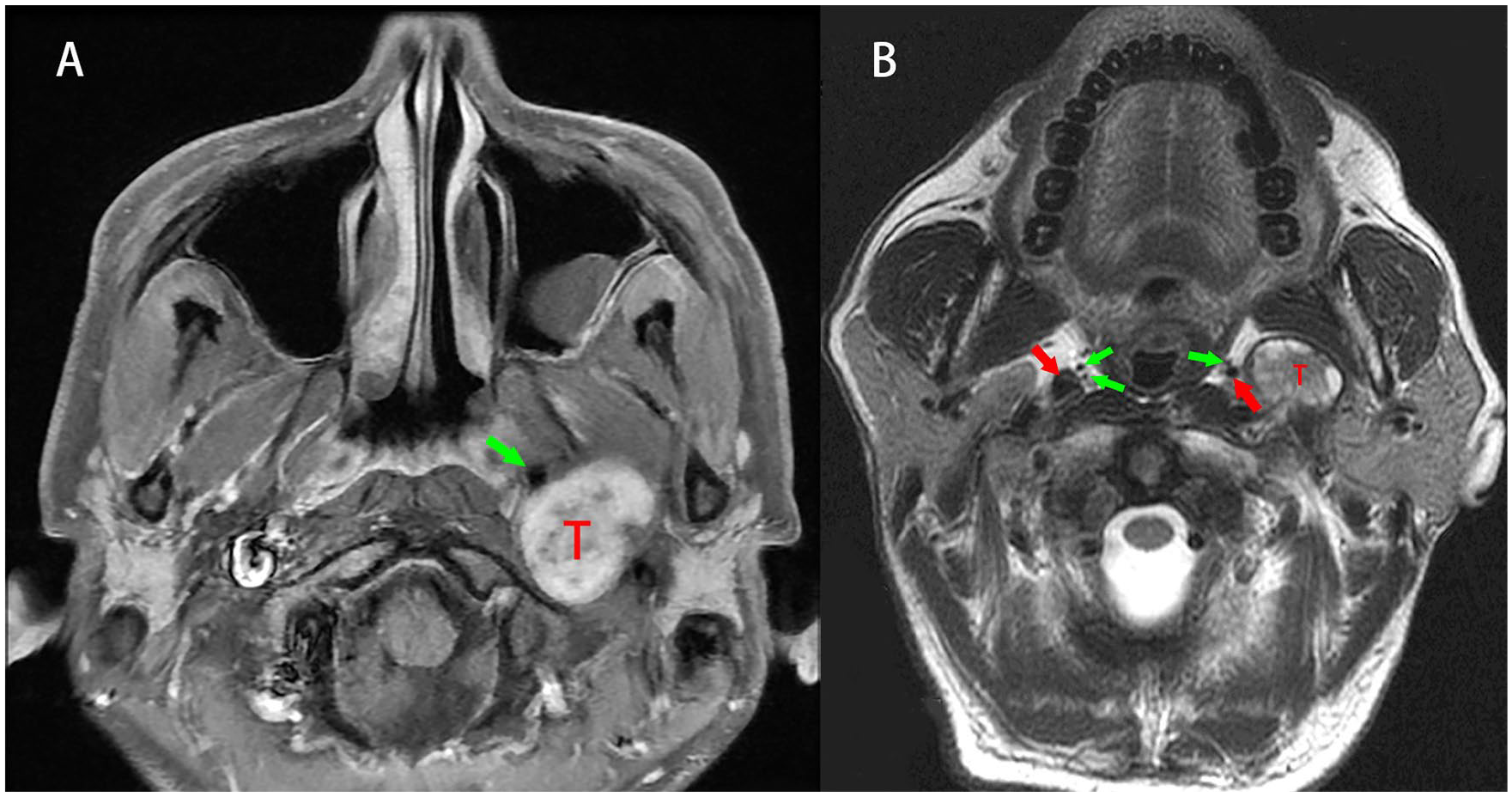
(A) The pICA was displaced into the anteromedial aspect of the tumor; (B) the branches of APA (green arrows) and pICA (red arrows) on preoperative MRI. APA, ascending pharyngeal artery; MRI, magnetic resonance imaging; pICA, parapharyngeal internal carotid artery.
During the transoral resection of tumors arising in the retro-styloid compartment, the APA served as a sentinel landmark for the pICA (Figure 6A). The pICA was identified at the posterior aspect of the APA in 8 patients (80%, Figure 6B), which was in accordance with the presence of APA in MRI scan. All tumors were removed without injury of these arteries. Branches of the APA were observed coursing vertically in 3 patients (37.5%, Figure 6C). In the remaining 2 patients with retro-styloid tumors, the APA was not encountered, and the medial border of the tumor was dissected following identification and preservation of the pICA.
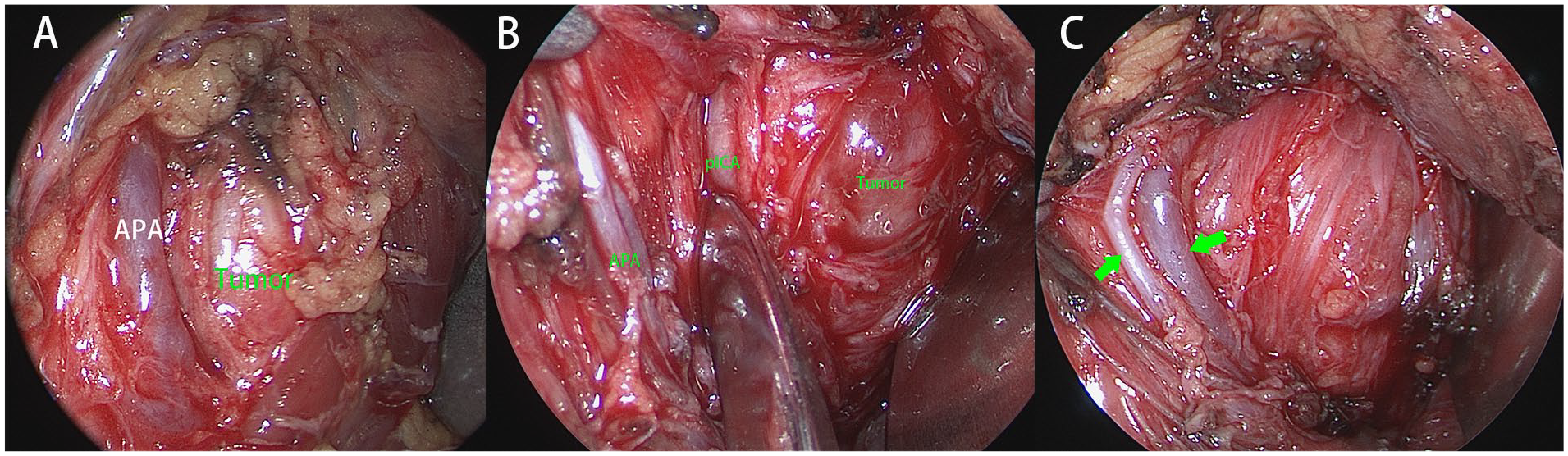
Left PPS, viewed by 0° scope. (A) The APA locates at the anteromedial aspect of the tumor; (B) the pICA locates at the posterior aspect of the APA; (C) branches of APA (green arrows). APA, ascending pharyngeal artery; pICA, parapharyngeal internal carotid artery; PPS, parapharyngeal space.
Discussion
The complex neurovascular relationships within the PPS, pose significant challenges for extirpation of lesions originally arising from the PPS. 21 Therefore, additional surgical landmarks like the APA may help complete a safe surgical procedure in the PPS. 15 Anatomical variations of the APA and their implications were explored using cadaveric dissections and confirmed in live surgeries; thus, adding to our knowledge of the PPS surgical anatomy.
The APA routinely branched from the external carotid artery and coursed vertically upward toward the skull base.3,22 The APA has several branches, including the superior pharyngeal branch and the meningeal branch.17,23 In this cadaveric study, the APA was detected to constantly course surrounding the pICA (at the medial and posteromedial aspects of the pICA in 60% and 20% of specimens respectively) at the retro-styloid compartment; and as such, the APA may serve as an additional landmark to localize the pICA intraoperatively, especially when employing an endoscopic approach.
Others described the external carotid artery traveling vertically or diagonally in the retro-styloid compartment, whereas the branches of the APA may course in both the pre- and retro-styloid compartments.15,19 This cadaveric study demonstrated that the APA was identified in the retro-styloid compartment in all 20 sides. In this cohort of 10 patients with pre-styloid tumors; however, the APA was intraoperatively encountered in one patient only (10%). We analyzed this phenomenon in our previous publication, considering that the basal cell adenoma was arising from the small salivary gland around the styloid process, masquerading as a retro-styloid PPS tumor to displace the carotid artery and APA medially. 20 In the clinical setting, we also observed that the main trunk of the external carotid artery was occasionally encountered during the procedures in the pre-styloid space, whereas the APA was rarely exposed during pre-styloid PPS tumor removal. Therefore, we may consider that the role of APA for procedures within the pre-styloid compartment is relatively limited.
External approaches including the transcervical, transcervical-parotid with or without mandibulotomy have been routinely performed for the extirpation of tumors in the retro-styloid compartment.24,25 The pICA is often displaced to the medial or anteromedial aspect of these tumors. 8 Although the potential risk of injury to the pICA exists, endoscopic approaches have been successfully applied for resection of retro-styloid tumors.8,26 In this cohort of retro-styloid tumors, the pICA was displaced into the anteromedial aspect of the tumor, and the APA was identified to at the anterior aspect of the pICA in 80% of preoperative MRIs and validated in surgical dissections. In these 8 patients, the APA could be adequately revealed and protected, and it is not necessary to sacrifice the artery. Therefore, suggesting that the APA serve as a valuable landmark for endoscopic procedures within the retro-styloid compartment. However, the APA was not constantly presented for all patients who harbored the schwannoma (20% absence). Compressing the artery by the tumor might constitute the contributing factor; and as such, presence of the APA on preoperative MRI may be a valuable reference factor to facilitate its intraoperative usage. Moreover, the paraganglioma often arises from the vagus or the sympathetic trunk in the retro-styloid space, and the open approaches are traditionally adopted for management of these paragangliomas. It is not difficult to control the APA from an open incision; and as such, the role of APA for paraganglioma management is limited via the open approach.
Given that the APA can branch to supply the lower cranial nerves (CN IX to XII) and has collateral communication with the vertebrobasilar artery system, the intraoperative identification and protection of the APA may be of critical importance. 17 A preoperative high-resolution MRI screening can help in its identification and to define its relationship with the tumor and the pICA. Moreover, the APA may supply tumors arising in the PPS, especially for paragangliomas in the PPS. A preoperative digital subtraction angiography can help determine if the APA is the dominant nurturing vessel. 18 In this regard, it may advantageous to embolize the APA preoperatively especially for vascular tumors such as paragangliomas. 27 Based on our experience, however, the APA was rarely detected to supply the schwannoma. Therefore, for patients with schwannoma selected in this cohort, the nutrition function by the APA to the schwannoma was limited.
Based on our observation, the tumor size does not impact the relationship between the APA and the tumor in PPS. Instead, the tumor origin from the pre- or retro-styloid compartment may determine the relative relationship between the tumor and the artery. To extirpate the tumor in the PPS en bloc via the transoral approach, we have previously reported that the maximal diameter of the tumor was 5 cm.8,20 For schwannomas arising from the retro-styloid space, however, the diameter is not the determining factor due to the fact that the tumor can be removed in an intracapsular and piece-by-piece fashion.
In a previous study, the tensor veli palatini muscle was used as the landmark for intraoperative identification of the pICA via an endonasal corridor. 28 Based on this cadaveric dissection, however, the APA was identified lying between the tensor veli palatini muscle and the pICA in 60% of specimens. Therefore, following identification of the tensor veli palatini muscle, the APA may serve as a complementary landmark for identification of the pICA through an endonasal or transoral corridor. Furthermore, particular attention is desirable when elevating and dividing the tensor veli palatini muscle, in case to avoid inadvertent injury of the APA.
The authors acknowledge the significant limitations included in the present study. All cadaveric dissection involved in the PPS was performed through an endonasal corridor, whereas all tumors in the clinical series were resected an endoscopic transoral corridor. Although a significant bias may exist, the anatomical principles and the neurovascular relationships within the PPS are adequately described. Moreover, while the value of using the APA as a landmark for procedures within the PPS has been validated for the endoscopic transoral corridor, its implication for endonasal resection remains undefined. In addition, one should note that the transoral corridor for resection of retro-styloid tumors is associated with significant technical difficulty and risk; thus, should only be performed by surgical teams with adequate experience in skull base and infratemporal fossa.
Conclusion
With identification of the APA on preoperative MRI, it may serve as an additional landmark for extirpation of tumors arising in the retro-styloid PPS via the endoscopic approaches.
Footnotes
Authors’ Note
Abstract was accepted as poster presentation at the Chinese Annual Meeting of Otolaryngology–Head and Neck Surgery, Suzhou, November 2021.
Data Availability Statement
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: N. London holds stock in Navigen Pharmaceuticals and was a consultant for Cooltech Inc. The other authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This article was supported by the Beijing Municipal Administration of Hospitals Incubating Program (PX2024008).
