Abstract
Introduction
Obstructive sleep apnea syndrome (OSAS) is defined as apnea and hypoventilation caused by collapse or obstruction of the upper airways during sleep, which may lead to damage to multiple organs and systems due to frequent nocturnal decreases in blood oxygen saturation. 1 Hypoxic stimulation can cause oxidative stress, activation of inflammatory pathways in the body, and an increase in the number of inflammatory mediators and inflammatory cells. 2 Currently, the gold standard for diagnosing OSAS remains polysomnography (PSG). However, the PSG examination is relatively cumbersome, time-consuming, and labor-intensive. Therefore, in recent years, attempts to find hematologic biomarkers to assist in the diagnosis of OSAS have become the direction of research. Some meta-analysis reviewed that C-reactive protein (CRP), IL-6, TNF-alpha, IL-8, neutrophil-to-lymphocyte ratio (NLR), homocysteine, vascular cell adhesion molecule-1, and vascular endothelial growth factor were higher in patients with OSAS and could be OSAS-associated potential biomarkers.3 -6 The neutrophil count and NLR increased in patients with OSAS, and were positively correlated with the apnea hyponea index (AHI) and negatively correlated with the minimum and mean oxygen saturation.7 -9 In addition to routine blood examination, CRP, procalcitonin (PCT), erythrocyte sedimentation rate (ESR), and heparin-binding protein (HBP) are also widely used biomarkers reflecting systemic inflammation in clinical practice. 10 Among these, numerous studies have confirmed that CRP has been confirmed to have increased levels in the peripheral blood of OSAS patients,11 -13 the PCT did not show statistically significant alterations in the OSAS group, 14 and the ESR was significantly higher in patients with OSAS and closely correlated with PSG parameters. 15 However, we did not retrieve HBP studies in patients with OSAS.
From the above studies, we know that patients with OSAS may have an altered systemic inflammatory state. However, related studies are not sufficient, and the value remains to be explored further. Finding markers with high sensitivity and specificity that can predict OSAS from among many inflammatory markers will benefit clinical diagnosis and treatment.
HBP, a neutrophil-secreted protein with the function of binding heparin, was officially named in 1991 and has been used in the diagnosis of infectious diseases since 2010 as a novel clinical marker of inflammation. 10 Compared to the conventional blood routine examination, CRP, ESR, and PCT, HBP has a higher sensitivity and specificity for bacterial infections.10,16,17 In recent years, our institution has included HBP in the routine examination of hospitalized surgical patients, and we have unexpectedly found that the vast majority of OSAS patients appear to have increased HBP values. However, it has not been reported whether HBP increases in patients with OSAS, whether it is an independent risk factor for OSAS, and whether it can be used as a marker to predict OSAS. In view of this, we collated and analyzed the available clinical data to investigate the alteration of systemic inflammatory status in OSAS patients and the predictive value of HBP for OSAS.
Methods
Participants
A retrospective study of patients with OSAS who were hospitalized in the affiliated Jiangning hospital of Nanjing medical university from January 1, 2020, to December 31, 2022, was conducted. Inclusion criteria for patients in the OSAS group: ① age ≥16 years old and ② symptoms and PSG results according to the OSAS diagnosis and treatment guidelines. 1 Exclusion criteria: ① acute infection of any organ or tissue or acute onset of chronic infection within the last 1 month and ② patients with malignancy. Patients with non-acute infectious diseases who were hospitalized for surgery during the same period were selected for the control group. Inclusion criteria for the control group: ① age ≥16 years old; ② patients admitted for vocal cord polyps and epidermoid cysts of the ear; and ③ Epworth Sleepiness Scale scores all less than 6 points. Exclusion criteria for the control group: ① acute infection of any organ or tissue or acute onset of chronic infection within the last 1 month; ② patients with malignancy; and ③ any OSAS symptoms such as nasal congestion, sleep snoring, open mouth breathing, and apnea within the last 3 months.
Method
The gender, age, height, weight, AHI, neutrophil count, lymphocyte count, PCT, and HBP of the patients were recorded and body mass index (BMI) and NLR were calculated. The OSAS group was further divided into a “severe OSAS group (sOSAS)” and “non-severe OSAS group (non-sOSAS)” according to whether the AHI was greater than 30 times/h.
Statistical Analysis
Statistical software IBM SPSS 26 and Graphpad Prism 6 were used for statistical analysis of the data. Two independent samples’ t-test or nonparametric test was used to compare the differences between the OSAS group and the control group for each index according to the results of the normal distribution test and the homogeneity test of variances. The sensitivity and specificity of the HBP cutoff point as a basis for predicting OSAS were calculated, and the sensitivity+specificity-1 was the Youden index, and the value corresponding to the maximum Youden index was the best cutoff value. The area under the curve (AUC) was obtained by plotting the receiver operating characteristic curve (ROC) with sensitivity as the vertical coordinate and 1-specificity as the horizontal coordinate, and the AUC was used to judge the predictive value of OSAS. Logistic regression analysis was performed to determine whether the indicators were independent risk factors for OSAS. OR > 1 was considered a risk factor and OR < 1 was considered a protective factor. The difference was considered statistically significant by the test level P < .05.
Result
A total of 79 patients were included in the OSAS group, including 66 males and 13 females with a mean age of 34.08 years. A total of 28 controls (20 males and 8 females) were included after screening according to age and sex matching and inclusion and exclusion criteria. There were no statistical differences in age (P > .05) and gender ratio (χ2 = 3.07, P > .05) between the 2 groups, which were comparable (Table 1). The BMI was higher in the OSAS group compared to the control group, and there was a statistical difference between the 2 groups (P < .0001, Table 1). To exclude the bias produced by BMI on inflammatory markers, each inflammatory marker was analyzed separately using a statistical approach (analysis of covariance, ANCOVA), and the corrected P value was used.
Comparison of each index between OSAS group and control group.
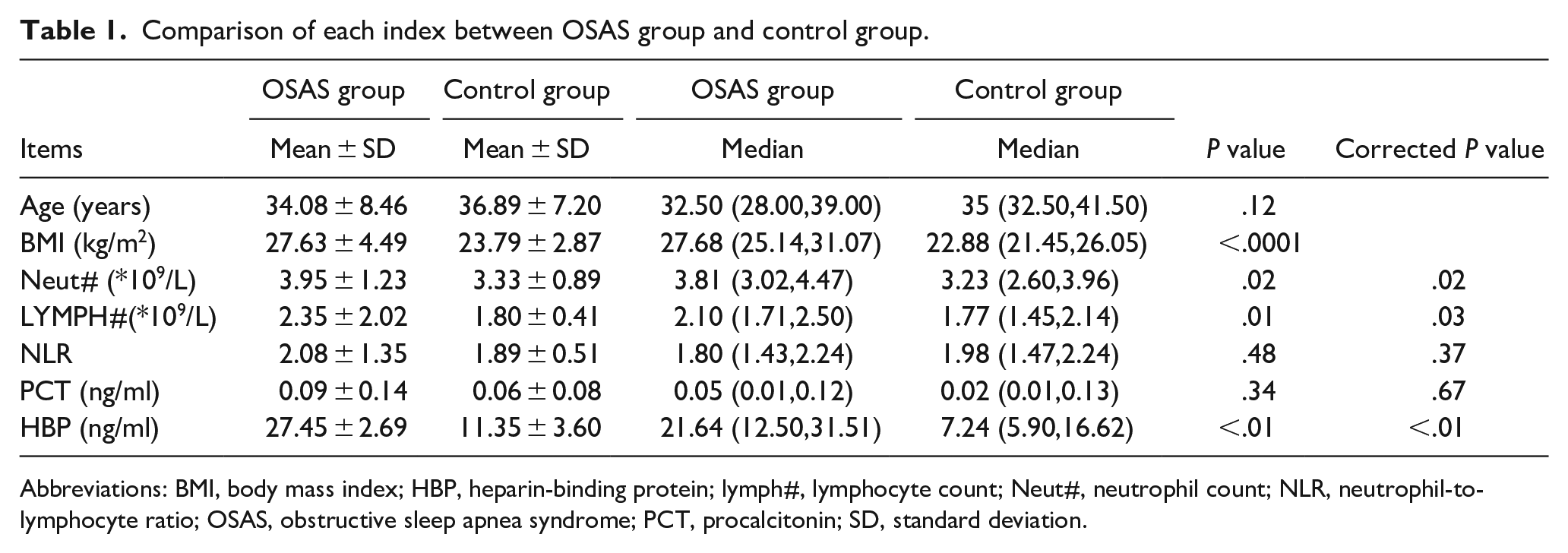
Abbreviations: BMI, body mass index; HBP, heparin-binding protein; lymph#, lymphocyte count; Neut#, neutrophil count; NLR, neutrophil-to-lymphocyte ratio; OSAS, obstructive sleep apnea syndrome; PCT, procalcitonin; SD, standard deviation.
Differences of Each Inflammatory Marker Between the 2 Groups (Table 1)
Differences in Neutrophil Count Between the 2 Groups
The neutrophil count was higher in the OSAS group than in the control group by the t-test and the difference was statistically significant (3.95 ± 1.23 vs 3.33 ± 0.89, P = .02) (Table 1, Figure 1A).
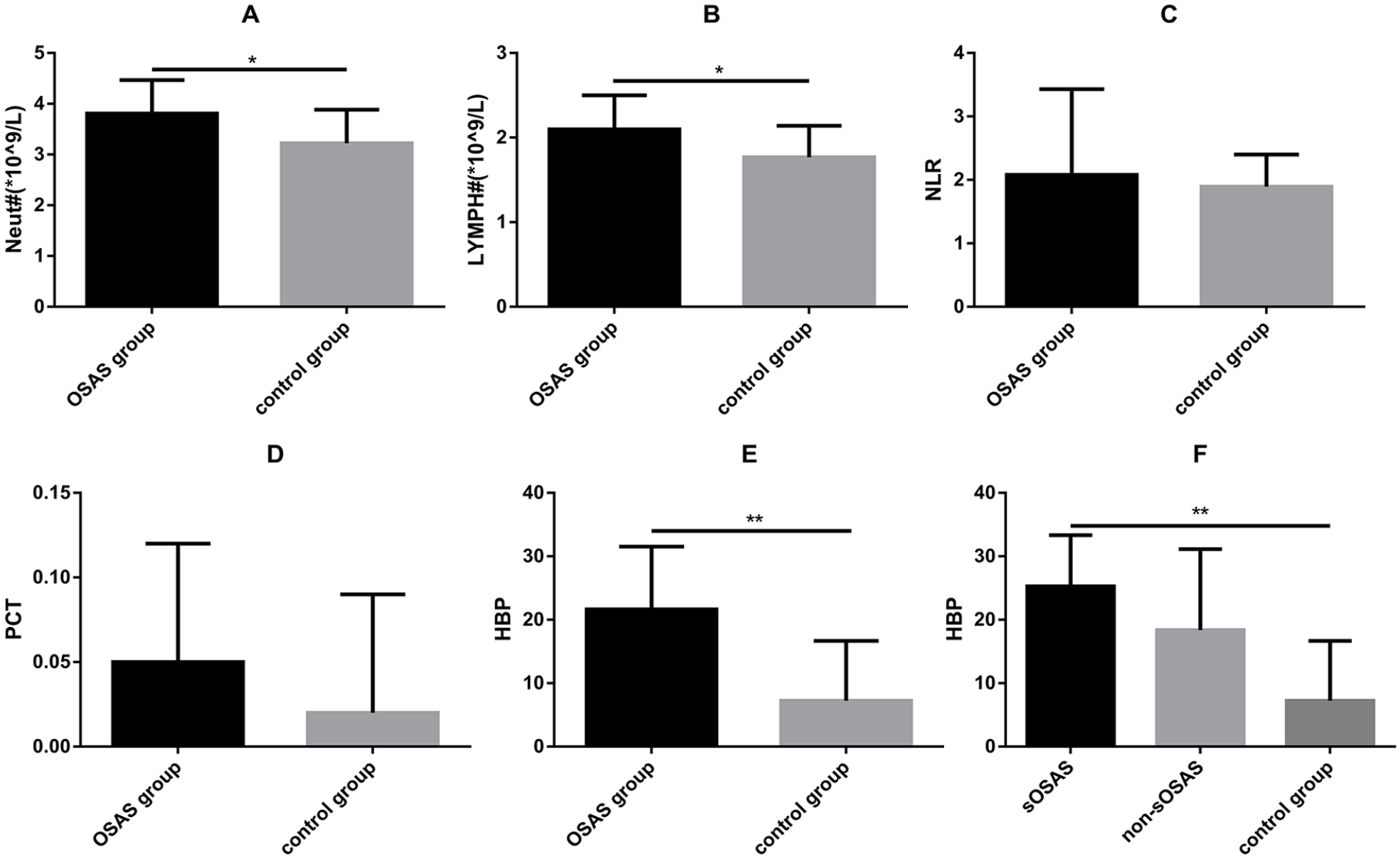
Differences in each inflammatory biomarker between the groups. A: Neut#(*109/L), neutrophil count (*109/L); B: LYMPH#(*109/L), lymphocyte count (*109/L); C: NLR, neutrophil-to-lymphocyte ratio; D: PCT, procalcitonin (ng/ml); E: HBP, heparin-binding protein (ng/ml); F: HBP in subgroups; sOSAS: severe OSAS; non-sOSAS: non severe OSAS. *P < 0.05. **P < 0.01.
Differences in Lymphocyte Count Between the 2 Groups
The lymphocyte count was higher in the OSAS group than in the control group by the t-test and the difference was statistically significant (2.35 ± 2.02 vs 1.80 ± 0.41, P = .03) (Table 1, Figure 1B).
Differences in NLR Between the 2 Groups
The NLR in the OSAS group was higher than that of the control group by the t-test, but the difference was not statistically significant (2.08 ± 1.35 vs 1.89 ± 0.51, P = .37) (Table 1, Figure 1C).
Differences in PCT Between the 2 Groups
The PCT was higher in the OSAS group than in the control group by the t-test, but the difference was not statistically significant (0.09 ± 0.14 vs 0.06 ± 0.08, P = .67) (Table 1, Figure 1D).
Differences in HBP Between Groups
The HBP in the OSAS group was higher than that of the control group by the t-test and the difference was statistically significant (27.45 ± 2.69 vs 11.35 ± 3.60, P < 0.01) (Table 1, Figure 1E and 1F). The OSAS group was divided into the sOSAS group and the non-sOSAS group for comparison with the control group, and the results showed that the patients in the sOSAS group had significantly higher HBP than the control group (32.32 ± 5.89 vs 11.35 ± 3.60, P < 0.01), while the other 2 comparisons were not statistically different (P all > .05).
Predictive Value of HBP for OSAS
The analysis of HBP’s ROC curve for predicting OSAS revealed that the AUC of HBP was 0.79 (95% CI: 0.68–0.90), indicating that HBP has a predictive value for OSAS with moderate precision. The Jorden index was maximum when HBP >10.73 ng/ml, which was the best predictive cutoff value, with a sensitivity of 80.49% and a specificity of 70.83% (P < .0001, Figure 2. Red line).
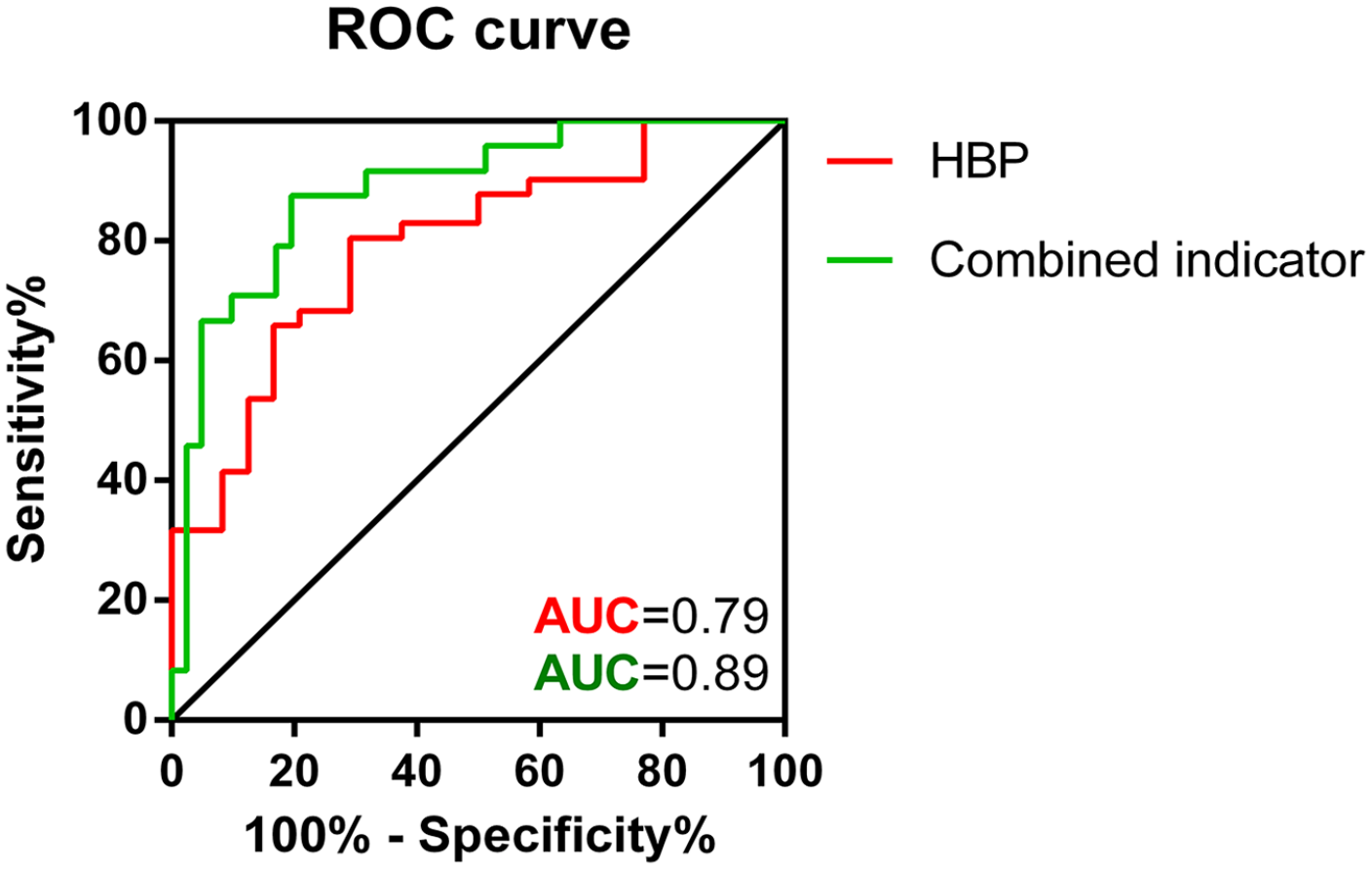
Predictive value of HBP (Red line) and Combined indicator (Green line) for OSAS. AUC, area under the curve; HBP, heparin-binding protein; OSAS, obstructive sleep apnea syndrome.
Predictive Value of the Combined Indicator for OSAS
Binary logistic regression was performed on the 4 statistically different indicators (BMI, neutrophil count, lymphocyte count, and HBP) to obtain the predicted probability values, and ROC curve analysis was performed using the predicted probability values to obtain the combined diagnostic value for OSAS. The results showed that the AUC of the combined indicator was 0.89 (95% CI: 0.80–0.97), indicating that the combined diagnosis had a predictive value for OSAS and improved the diagnostic value compared to HBP. The Jorden index was highest when the predictive probability value was >.66, which was the best predictive cutoff value, with a sensitivity of 80.49% and a specificity of 87.50% (P < .0001, Figure 2. Green line).
Analysis of Independent Risk Factors for OSAS
The results of this study showed a statistically significant increase in BMI, neutrophil count, lymphocyte count, and HBP in the OSAS group, and logistic regression was performed on these 4 indicators to analyze whether they were independent risk factors for OSAS (Figure 3). The results suggested that the regression equation was well fitted (Negoelco R2 = 0.60, Hosmer-Lemay P = .55) and that BMI and HBP were independent risk factors for OSAS (PBMI < 0.001, PHBP <0.01). Each unit increase in BMI increased the risk by 48.7%, and each unit increase in HBP increased the risk by 10.3%. However, neutrophil and lymphocyte count were not independent risk factors for OSAS (P = .179, P = .81, respectively).
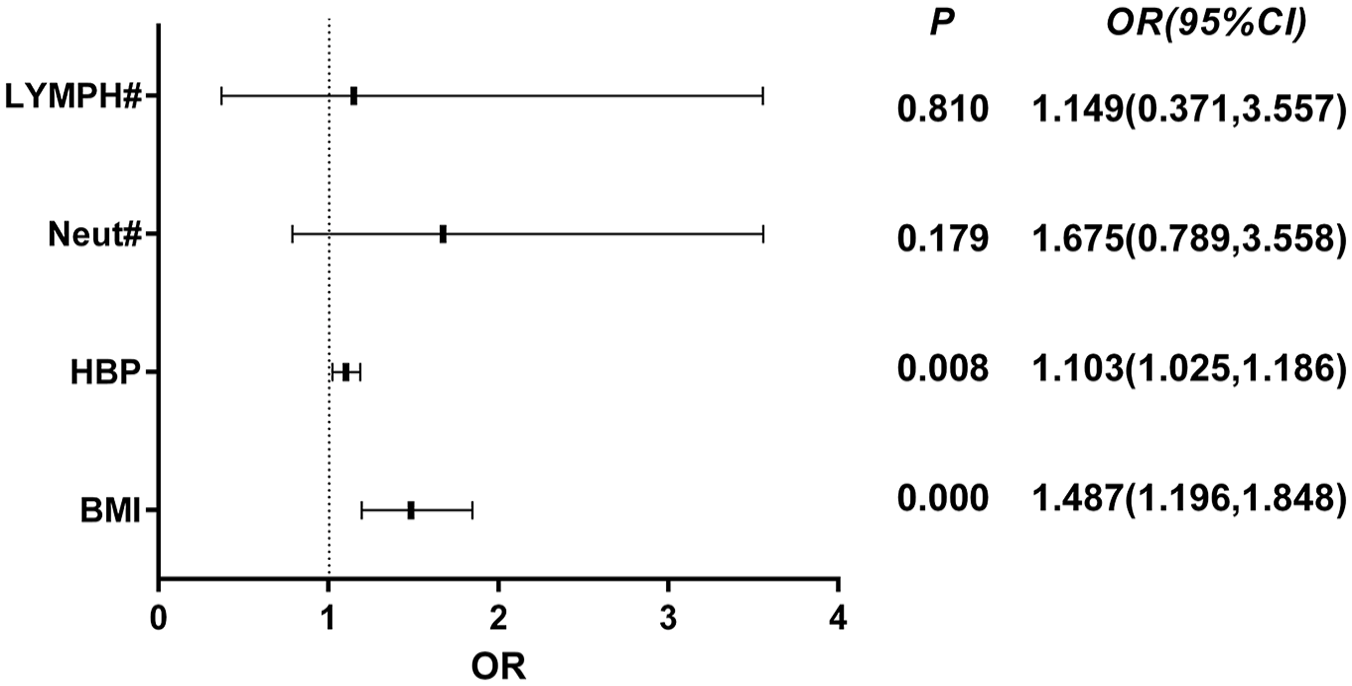
Forest diagram of OSAS risk factors. BMI, body mass index; HBP, heparin-binding protein; Neut#, neutrophil count; lymph#, lymphocyte count; OR, odds ratio; CI, confidence interval.
Discussion
Epidemiological studies have shown that the prevalence of OSAS in adults ranges from 23.4% to 49.7%, with males more affected than females. 18 Furthermore, the prevalence of OSAS in children is nearly 10.47%. 19 The large population of patients has caused great pressure on human health and socioeconomic development, but has not attracted sufficient attention from the whole society. 20 The harm of OSAS is not only snoring, and the goal of treatment is not just to widen the airway to solve the snoring problem; the deeper meaning of treatment is to prevent or reverse a series of complications caused by OSAS and changes in systemic immune status.21,22 Patients with OSAS are accompanied by frequent nocturnal decreases in oxygen saturation, while prolonged intermittent hypoxia and reoxygenation trigger an oxidative stress response that increases the expression of numerous inflammatory mediators and inflammatory cells.23,24 In recent years, NLR has been widely used in immune diseases and inflammatory responses, such as cardiovascular diseases, diabetes, and tumors.25,26 Previous studies have shown that peripheral blood NLR increases in patients with OSAS compared to the population without OSAS and increases with higher BMI.12,27 Our results showed a significant increase in neutrophil and lymphocyte count in OSAS patients compared to controls; however, the increase in NLR was not statistically significant. Neutrophil counts suggest statistically significant elevations in all existing studies, and the main reason for the difference in NLR is the difference in lymphocytes. Bozkuş et al 12 found that the number of lymphocytes in normal controls was intermediate between overweight OSAS and obese OSAS patients. We speculate that BMI may be an important factor affecting the results. In our study, the BMI was higher in the OSAS group than in the control group, but due to the small sample size, it was not possible to stratify the analysis by BMI, so the ANCOVA analysis was used to correct for the bias caused by BMI, and the results showed that the lymphocyte count was still higher in the OSAS group than in the control group. Although NLR was not statistically significant between the 2 groups in our study, it was higher in the OSAS group, indicating that the lymphocyte count did not increase as much as the neutrophil count. It is inferred that OSAS patients, who were under long-term hypoxia and oxidative stress, have an altered systematic inflammatory state, especially neutrophil inflammation.
PCT has been considered one of the biomarkers that reflect neutrophil inflammation in recent years. Sozer et al 14 have compared PCT levels in patients with different degrees of OSAS and healthy controls, but none of the results found a statistical difference. In our study, PCT was slightly higher in OSAS patients than in controls, but there was also no statistical difference. It has been suggested that PCT is usually elevated after the release of toxins by bacteria leading to infection and is more sensitive to reflect the activity of the inflammatory response, while it is generally at a lower level in the early stages of infection.10,28 None of the patients with OSAS in our group had an active infection, which could explain why PCT was not significantly elevated in patients with OSAS. But PCT was slightly higher in the OSAS group, still suggesting an altered systemic inflammatory state and an increased potential risk of infection in OSAS patients. However, more studies are needed to confirm whether the increase in PCT is more significant in OSAS patients than in non-OSAS patients in the presence of acute infection.
Current studies suggest that HBP is a neutrophil-secreted granule protein, and although the exact mechanism of secretion is unclear, HBP is more likely to be significantly elevated in the early stages of infection and has shown great value in early warning of bacterial infection and in determining the severity and prognosis of infection.16,28 Neutrophilia in OSAS patients has been confirmed by several studies, and therefore we hypothesized that HBP may have potential research value in altering systemic inflammatory status in OSAS patients. Given that there is no literature on the level of HBP expression of patients with OSAS yet, we explored it, and the results showed a significant increase in HBP in the OSAS group compared to the control group, especially in the severe OSAS group, and the difference was more significant than that of neutrophils. Furthermore, the analysis of the ROC curve also showed that HBP had a moderate precision predictive value with high sensitivity and specificity for OSAS (AUC = 0.79). Our study also performed ROC curve analysis of the value of 4 indicators (BMI, neutrophil count, lymphocyte count, and HBP) that were statistically different between the 2 groups for the combined diagnosis of OSAS, and the results suggested that the value of the combined diagnosis improved further compared to HBP (AUC = 0.89). Although BMI, neutrophil count, lymphocyte count, and HBP were all statistically significantly increased in the OSAS group, it is ambiguous what the independent risk factors for OSAS are. Therefore, logistic regression was used in this study to analyze the results, which showed that both BMI and HBP were independent risk factors for OSAS. 48.7% of the risk increased for each unit increase in BMI, and 10.3% of the risk increased for each unit increase in HBP. However, neither neutrophil nor lymphocyte count was an independent risk factor for OSAS. It is evident that HBP has an important research value in the pathophysiological mechanisms of OSAS. HBP has been suggested to be involved in cardiovascular endothelial cell injury and cancers,29 -31 and patients with OSAS have an increased risk of complicating with cardiovascular disease and cancers.32,33 Therefore, we speculate that HBP could play an important role in the development of complications in OSAS patients, but the exact mechanism needs to be confirmed by further studies.
There are some shortcomings in this retrospective study. First, the sample size is not large enough, especially for patients with mild and moderate OSAS, which prevented the stratification analysis of mild, moderate, and severe OSAS by AHI. Additionally, medical record writing often neglected documentation of oxygen saturation values, so the correlation between each inflammatory biomarker and oxygen saturation could not be analyzed. Despite the above shortcomings, this study reveals for the first time the research value of HBP in the altered systemic inflammatory status of patients with OSAS and suggests that HBP can be used as a novel biomarker to predict OSAS. When PSG examination is inconvenient, OSAS can be diagnosed preliminary according to the combination of BMI, peripheral blood routine, and HBP. Furthermore, it is hypothesized that patients with OSAS may have weaker resistance to bacteria and are more prone to more serious infections when encountering bacterial invasion, requiring more attention. Additionally, HBP has potential research value on the mechanism of OSAS secondary to systemic complications.
Conclusion
This is the first study of its kind to objectively examine HBP in patients with OSAS, and HBP is an independent risk factor that can serve as a diagnostic biomarker in OSAS. Patients with OSAS have an altered systemic inflammatory state and may be more prone to severe bacterial infections.
Footnotes
Acknowledgements
We thank Nanjing Medical Science and Technology Development Fund for providing financial support to this study.
Authors’ Contributions
Yang and Xue contributed equally to this study, they conducted statistical analysis, and drafted the article. Tao and Shi mainly participated in article retrieval and data acquisition. Li designed this study and reviewed and revised the article. All authors read and approved the final article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Project Supported by Nanjing Medical Science and Technology Development Fund (No. YKK22217).
Ethical Statement
This study was approved by the ethics committee of The Affiliated Jiangning Hospital of Nanjing Medical University (Nos.2022-03-007-K01). This study was a retrospective observation of whether some of the biomarkers examined in the preoperative peripheral blood of OSAS patients differed from those of non-OSAS patients, the study did not involve treatment, did not increase the risk of patient treatment, did not increase the financial burden on patients, and did not expose patient privacy, therefore, the ethics committee waived informed consent from patients.
Consent for Publication
As there is no patient identifying data, consent for publication was not required.
Data Sharing Statement
Data will be made available by the corresponding author upon reasonable request.
