Abstract
Objective:
Brachial plexus schwannoma (BPS) is a rare clinical entity that poses a significant challenge for head and neck surgeons due to its neuroanatomical complexity and potential severe complications, such as major motor or sensory neurological deficits or pain of the corresponding upper extremity. This article summarizes our experience in its diagnosis and intracapsular enucleation with intraoperative neuromonitoring (INM).
Methods:
A retrospective review of BPS cases treated at our tertiary medical institution was conducted between April 2020 and May 2023. The clinical and demographic data were retrieved from case notes.
Results:
Totally, 3 cases were included. All 3 patients were male, aged 43 to 54 years (median age = 52). The presenting symptom was a palpable supraclavicular mass in all these cases (2 on the left side and 1 on the right side). Neuromonitoring was performed with a 4-channel nerve integrity monitor, with the electrodes placed in the upper arm and forearm muscles, as demonstrated in the literature. After exposing the mass and identifying its origin from the brachial plexus, a unipolar stimulating probe was used to stimulate the tumor surface or the nerves with a 1.0-mA current, and a longitudinal incision into the tumor capsule was made along a carefully mapped line with no INM response. Then the mass was carefully exposed and meticulously dissected from its capsule to achieve an intact enucleation. Immediate postoperative neurological deficit was documented in Case 1 as a mild grasping weakness. The recovery of the other 2 patients was uneventful. The follow-up duration was 7 to 38 months (median = 8 months). The minor motor deficit of Case 1 recovered completely 1 month after surgery. No recurrence of BPS was observed.
Conclusions:
Intracapsular enucleation with INM could result in complete removal of BPS and minimal postoperative neurological deficit, whose recovery was quick and satisfactory.
Keywords
Introduction
Schwannomas are rare, slow-growing tumors that arise from Schwann cells that form the myelin sheath of nerve fibers. 1 Clinical presentation varies depending on its anatomic site. A slow-growing swelling and lesion without pain and neurological features are the common presenting symptoms in schwannoma. 2 Concerning brachial plexus schwannoma (BPS), a palpable mass is the most common clinical presentation, followed by paresthesia or numbness, pain or tenderness, and motor deficits of extremities. 3
Since a schwannoma is well encapsulated and eccentric and eventually displaces the fascicles of the nerve to the periphery of the mass, 4 it is generally believed that a schwannoma can be enucleated from the nerve without producing a neurological deficit.5,6 However, this may not always be the case. In a series of 56 patients with upper limb schwannomas, immediately after meticulous enucleation procedure without intraoperative neuromonitoring (INM), 41 (73.2%) patients developed a new neurological deficit, 10 (17.8%) of whom had a major deficit such as severe motor or sensory loss, or intolerable neuropathic pain. After a mean of 25.4 months, 17 (30%) still had hypoesthesia, paresthesia, or mild motor weakness. 5 In another similar series of 19 patients with BPS, 5 patients presented with immediate postoperative neurological deficits, 2 of whom had motor deficits that recovered after a follow-up of 2 and 5 years, respectively. 7 Hence, although BPS is rare,7 -9 it poses a significant challenge for head and neck surgeons due to the neuroanatomical complexity of this region and the potential of jeopardizing the patient’s quality of life, productivity, and economic stability.
Larger BPS cases were associated with a greater risk of neurologic deficit after surgery, 7 and the mean tumor size was significantly larger in patients with a major neurological deficit than in those with a minor or no deficit (P = 0.01). 5 Hence, observation might not be the best recommendation for patients with BPS, and a carefully planned early enucleation procedure with INM should be scheduled. 9 Here, we report our experience with 3 cases of successful intracapsular enucleation of BPS under INM, with only one case of minor postoperative motor deficit, which quickly recovered.
Patients and Methods
A retrospective review of BPS cases treated at our tertiary medical institution was conducted between April 2020 and May 2023. The clinical and demographic data were retrieved from case notes. The protocol of the research project was approved by the institutional review board, and it conformed to the provisions of the Declaration of Helsinki. A written consent form was obtained from the patients.
Diagnosis
The presenting symptom was a palpable cervical mass in the supraclavicular region in all 3 cases. Tinel’s sign was positive in all the cases. After ultrasonography revealed the masses to be possibly of nerve origin, MRI with contrast was scheduled for further evaluation of the masses, which showed well-defined, T1 isointense, and T2 hyperintense masses with homogeneous enhancement (see Figure 1A). The masses were contiguous with certain components of the brachial plexus. Therefore, a tentative diagnosis of BPS was proposed.
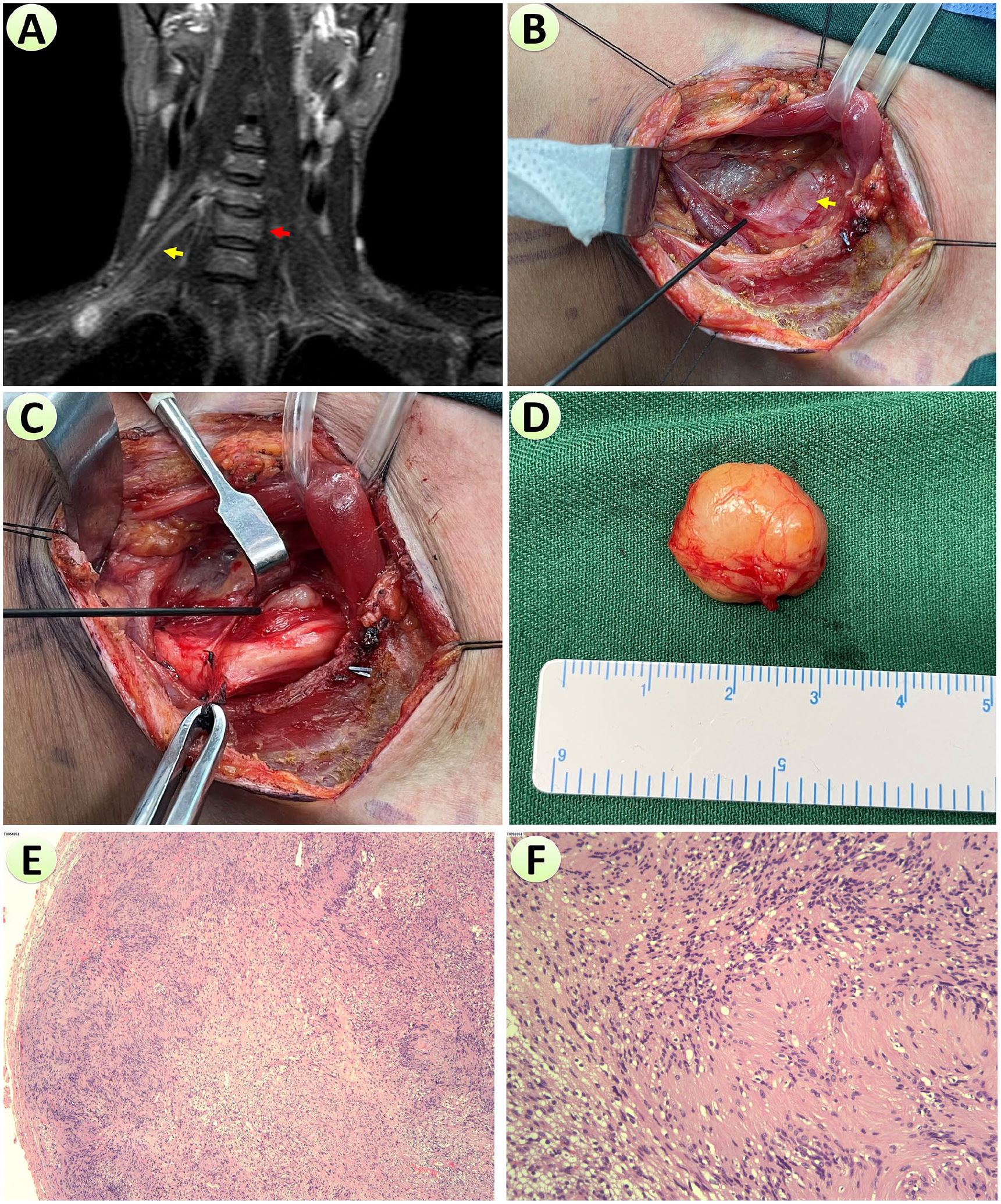
Preoperative evaluation, intraoperative findings, and pathology figures of Case 3 with intraoperative neuromonitoring (INM). (A) Preoperative T2 MRI image showing a well-defined, hyperintense mass in the right supraclavicular neck. On the superior margin of the sixth cervical vertebra (right arrow), the ventral rami of the C6 (yellow arrow) are contiguous with the mass (image with maximal mass diameter not shown here, for a better demonstration of the relationship with C6). (B) Intraoperative image showing the exposure of the mass (yellow arrow). Nerve fascicles of the upper trunk of the brachial plexus (at the tip of the monitoring probe) could be seen on the surface of the mass. (C) Intraoperative image showing the incision on the capsule of the mass at the tip of the monitoring probe. The surface of the capsule of the mass was mapped for neural response with INM at 1.0 mA, and the incision was made where no response was elicited behind the posterior division of the upper trunk. (D) Intactly enucleated brachial plexus mass from the posteromedial surface of the posterior division of the upper trunk. (E) On the 40× image, the tumor was well defined with an intact capsule, and alternating Antoni A zones with dense cellular arrangement and Antoni B zones with sparse cellular arrangement were seen. (F) On the 200× image, Antoni A area consisted of palisading spindle-shaped Schwann cells with abundant eosinophilic cytoplasm forming varocay vesicles, while Antoni B area consisted of reticulated stellate Schwann cells, with ovoid cell shape and microcystic formation.
Surgical Procedures
All surgical procedures were performed under 2.5× loupe magnification using microsurgical techniques. Neuro-monitoring was performed with a four-channel nerve integrity monitor (NIM3; Medtronic, Memphis, TN, USA). Electrodes were placed in the muscles of the upper arm and forearm, including the deltoid, biceps brachii, triceps brachii, and brachioradialis, as demonstrated by Lee et al. 9 After exposing the mass and identifying its origin from the brachial plexus (see Figure 1B), a unipolar stimulating probe was used to stimulate tumor surface or the nerves by applying a 1.0-mA current. A longitudinal incision into the tumor capsule was made along a carefully mapped line with no INM response (see Figure 1C). Then the mass was carefully exposed and meticulously dissected from its capsule to achieve an intact removal. All brachial plexus components were carefully preserved in this process, and efforts were made to minimize the tension imposed on them. Then the surgical field was irrigated with peroxide and normal saline. Final confirmation of all exposed brachial plexus components showed good INM response. An absorbable gelatin sponge soaked with dexamethasone was placed over the brachial plexus for protection. A negative-pressure drain was placed, and the incision was closed.
Follow-up
The patients were followed up every month after surgery to observe for postoperative neurological deficits. After recovery of neurological deficits, the patients were followed up every 6 months with MRI for tumor recurrence.
Results
The 3 cases included were all male, with an age range of 43 to 54 years (average and median ages were 49.7 and 52 years, respectively). The presenting symptom was a palpable supraclavicular mass in all 3 cases (see Table 1), with 2 on the left and 1 on the right side. No preoperative motor or sensory neurological deficit, or pain of the upper extremity was noted.
Patient Characteristics of this Series and Literature.
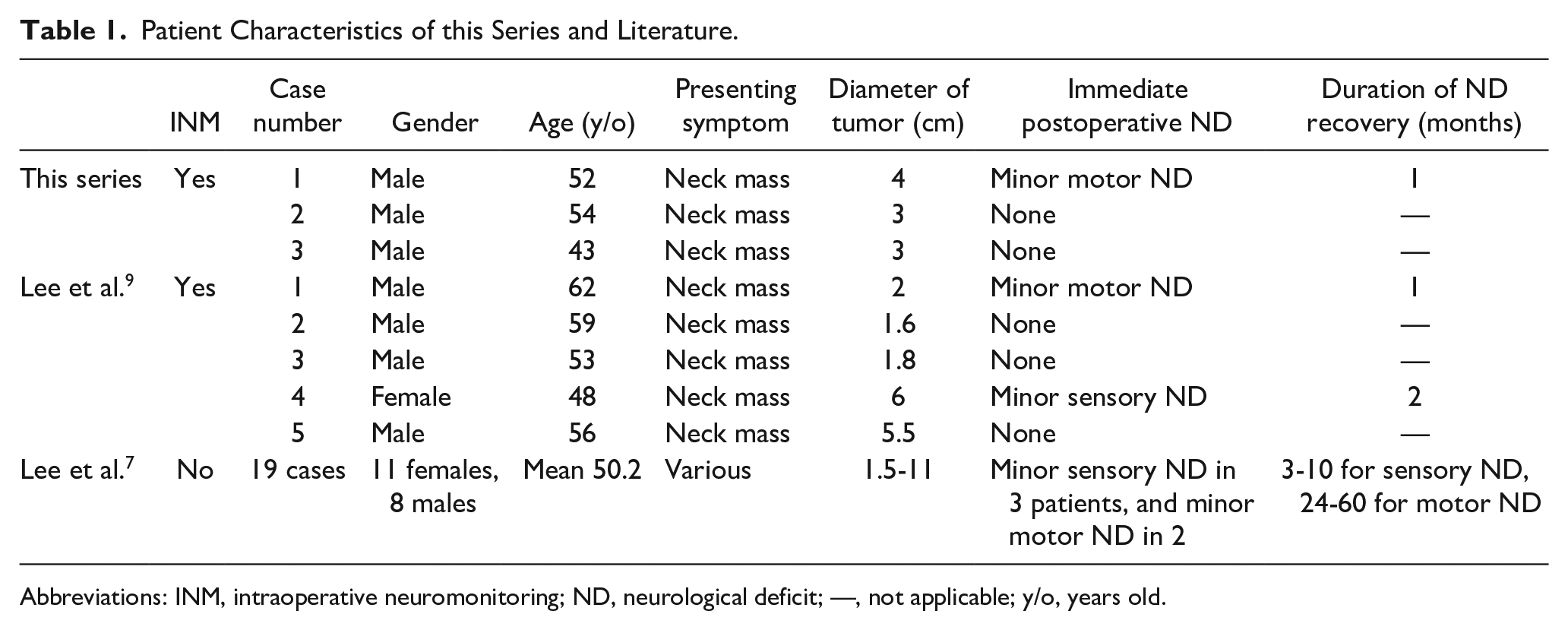
Abbreviations: INM, intraoperative neuromonitoring; ND, neurological deficit; —, not applicable; y/o, years old.
The surgical procedures of all 3 patients were uneventful. After dividing the sternocleidomastoid muscle overlying the palpable mass, and inferior retraction of the omohyoid muscle, the brachial plexus and mass could be exposed. Then INM stimulation of individual nerves using a 1.0-mA current could identify particular trunks and divisions based on the response of upper extremity muscles; and the origin of BPS could be identified: two were from the upper trunk and one was from the posterior division of the upper trunk. The incision into the tumor capsule should be carefully mapped with INM at 1.0 mA and made where no response was elicited. As in Case 3, after elevating the fibrous connective tissue overlying the anterior and posterior division of the upper trunk, the region between the anterior and posterior divisions (anterolateral surface) showed immense neural response. Only a thin line could be drawn on the posteromedial surface behind the posterior division without INM response. An incision into the capsule was then made longitudinally along this line to reveal the mass (at the tip of the monitoring probe, see Figure 1C). An Allis clamp was used to hold the elevated fibrous tissue to slightly rotate the posterior division anterolaterally, for better exposure of the mass, without retracting the posterior division with a surgical instrument. Then the mass was carefully dissected from the capsule, and intact enucleation was achieved (see Figure 1D) for all 3 cases.
Immediate postoperative neurological deficit was documented in Case 1 as a mild grasping weakness. The recovery of the other 2 patients was uneventful. The drain was removed when daily drainage was less than 10 mL, and the patient was discharged the next day. Pathology reports confirmed the diagnosis of schwannoma in all 3 cases.
The duration of follow-up was 7 to 38 months (average and median durations were 17.7 and 8 months, respectively). The minor motor deficit of Case 1 recovered completely 1 month after surgery. No recurrence of BPS was observed.
Discussion
First reported by Virchow in 1908, 10 schwannoma could arise along the course of a nerve. It might affect the third to twelfth cranial and peripheral and autonomic nerves. 11 Most schwannomas are benign; however, malignant tumors are rarely encountered.7,12 The preoperative diagnosis of schwannoma is challenging, as CT, MRI, and ultrasound-guided fine needle aspiration (FNA) all have relatively low accuracy.6,9,13 -15 Consequently, histology is still the gold standard for its diagnosis because of its atypical clinical symptoms and lack of specific images. 1 Schwannoma has a macroscopic appearance of a well-limited encapsulated mass; an eccentric fashion can be macroscopically seen through its sectioning. 16 Microscopically, areas of dense spindle cells (Antoni A) and loosely arranged cells (Antoni B) may be seen 6 ; wavy nuclear contours, S-100 protein expression, and a well-formed collagenous capsule are consistent findings. 12
It has been reported that preservation of neurological function is more likely in patients presenting intact and those not having undergone a previous biopsy procedure or resection than in those with such manipulation. 17 The incidence of neurological symptoms was reported to increase two-fold after exploration. 18 Hence, open biopsy and FNA are not recommended due to the risk of iatrogenic nerve injury, sometimes resulting in a permanent neurological deficit, or leaving a scar that might interfere with the meticulous dissection required for the enucleation of BPS.5,7,19
The brachial plexus is formed from the ventral nerve roots of C5 to T1: C5 and C6 give rise to the upper trunk, C7 to the middle trunk, and C8 and T1 to the lower trunk. 13 Anterior cervical supra-, infra-, or even trans-clavicular approaches and posterior subscapular approaches could be utilized for the surgical resection of BPS. The anterior cervical approach is quick and safe, allowing much better control of the important vascular and neural structures.11,20 However, the posterior approach is also invaluable, whose indications include the following: (1) If the lower plexus roots (C8 and T1) or trunk are affected by the tumor; (2) If the patient has a history of radiation therapy or resection, severely scarring the anterior plexus; and (3) If the proximal spinal nerves, especially those at an intra-foraminal level, are affected by the tumor. 17
It is our experience that INM is invaluable for enucleating BPS. In the series of Park et al., 5 the dissection and enucleation of BPS were meticulous, except for the use of INM. Yet, unfortunately, as many as 73.2% of patients presented new neurological deficits, with 17.8% deemed as major deficits. However, in our series and Lee et al., 9 with the help of INM, only minor deficits (temporary paresthesia or mild loss of grip strength) developed, with significantly reduced duration of recovery (from a mean of 25.4 to 1 month).5,9 We assume that the difference in neurological outcome resided partly in the choice of position of longitudinal incision on the surface of BPS. In our series, we found that only a thin line of no INM response among nerve fascicles could be drawn on tumor surfaces, which would be the line of our incision and might not be easily accessed as it might be on the posteromedial surface (see Figure 1C). Although this option might increase the difficulty of dissection, it might help avoid injuring the communication among nerve fascicles, resulting in better neurological outcomes. This approach was consistent with Lee et al., 9 who would make this incision with fewer neural fascicles with INM detection. In addition, we were so lucky that we did not need to divide any nerve fascicles entering or leaving the tumor substance. Although some authors believe that such a fascicle should be sacrificed to remove the mass en bloc,7,21 others have reported that small pieces of tumor tightly adhering to a functional fascicle could be left unresected. 9 We endorse the latter notion, because we believe that a functional nerve fascicle of the brachial plexus might be too valuable to sacrifice for a mostly benign lesion. However, if a nerve fascicle is entrapped in the mass, cannot be dissected, and is proven nonfunctional by INM, it can be resected with the mass. 3
Another technique worth mentioning is the careful handling of brachial plexus fascicles. After incising BPS capsules, we would carefully expose the tumor surface. Then the tumor was gently lifted with a small hook to create tension between the capsule and the tumor, which were separated with a thin dissector. After circumferential separation of the tumor from the capsule, the tumor could be lifted out; and its base was then carefully separated from the capsule for complete enucleation. It was imperative not to exert tension on brachial plexus fascicles, and we tried our best to avoid retracting brachial plexus elements with surgical instruments to expose the tumor. Sometimes, we would use elevated fibrous tissue overlying the brachial plexus to slightly rotate nerve fascicles (without retracting them) to expose the tumor better. This technique could be another reason for our favorable neurological outcomes.
Although the small number of included patients significantly limited our study, it could expand the knowledge of enucleation procedures of BPS with INM, regarding its feasibility, safety, and effectiveness. Hence, our work might contribute to literature, because such endeavor has been so few that its advantages needed the validation of larger cohorts.
Conclusion
Although rare, BPS is very challenging for head and neck surgeons. Once discovered, intracapsular enucleation procedures with INM should be scheduled to achieve complete removal and minimize postoperative neurological deficits. After such surgical treatment, minor motor or sensory, neurological deficits might develop, but quick and complete recovery could be expected.
Footnotes
Acknowledgements
Not applicable.
Author Contributions
Juan Shi and Kai-Xuan He reviewed medical records, analyzed data, and participated in patient follow-up. Yu-He Liu made contributions to revising articles. Yan-Bo Dong participated in treatment of patients. Cheng Lu participated in treatment of patients and made major contributions to the revising of multiple versions of the articles. Wan-Xin Li participated in treatment of patients, reviewed literature, and wrote multiple versions of the articles.
Availability of Data and Materials
All data, models, and code generated or used during the study appear in the submitted article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Beijing Hospitals Authority’s Ascent Plan (grant number DFL20220102) and Beijing Friendship Hospital Seed Project, Capital Medical University (grant number YYZZ202125). National Natural Science Foundation of China for Young Scholars (grant number 82303642), and the Foundation for Scientific Research and Cultivation, Capital Medical University (grant number PYZ22076).
Ethics Approval
The protocol for the research project has been approved by Bioethics Committee of Beijing Friendship Hospital, Capital Medical University.
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with the institutional review board’s approved protocols (2023-P2-015-01).
Statement of Informed Consent
Written informed consent was obtained from the patients for their anonymized information to be published in this article.
