Abstract
Objectives
Schwannoma arising from cervical sympathetic chain (CSC) is a rare clinical entity. CSC is hard to preserve in CSC schwannoma (CSCS) surgeries, resulting in Horner’s syndrome (HS) and first bite syndrome (FBS). This article aims to explore our experience in successful preservation of CSC in CSCS surgeries.
Methods
This is a retrospective review of CSCS cases treated at our tertiary medical institution between Apr 2018 and March 2022. Only cases with successful intraoperative preservation of CSC were included.
Results
In total, 3 cases were included. There were 2 male patients and 1 female patient. Their age was between 38 years old and 66 years old (average and median age was 52 and 51 years old, respectively). Presenting symptom was neck mass for all them, 2 of which were on the left side, while 1 was on the right. Intracapsular enucleation of the CSCS was attempted and achieved in all 3 cases. Hemorrhage from the inner surface of capsules was diffuse and heavy. Constant saline irrigation, suction, and bipolar coagulation were needed to achieve hemostasis. Postoperative HS presented between 4 hours and 14 hours after surgery for all 3 patients, which took 1 month to 3 months to recover, whereas FBS did not occur in any patient. Median duration of follow-up was 6 months. No recurrence was observed.
Conclusions
Intracapsular enucleation should be attempted in CSCS surgeries, although hemorrhage might be diffuse and hard to control. When intracapsular enucleation was achieved, postoperative FBS can be avoided, while postoperative HS could occur, but its recovery was satisfactory.
Keywords
Introduction
Schwannomas are neoplasms originating from the myelin-producing Schwann cells of the nerve sheath, 1 which are typically benign, slow-growing, and solitary. 2 Schwannomas arising from the cervical sympathetic chain (CSC) are extremely rare, and the largest number of CSC schwannoma (CSCS) cases from a single institution ever reported in literature was 12 cases. 3
CSCS is often asymptomatic, and referral is usually initiated by neck mass, although vague symptoms like dysphagia and sore throat may be present, 3 and a unique case of secretory CSCS causing hypertension associated with raised urinary catecholamine degradation by-products has been reported. 4 Because the CSC runs in a relatively loose fascial compartment, compression by CSCS, as seen with acoustic neuromas or facial nerve schwannomas, is exceedingly rare. 5 Hence, preoperative Horner’s syndrome (HS) is uncommon for CSCS, although there have been several such cases reported in literature. 5 -8 Schwannomas are radioresistant, and malignant transformation has been reported; therefore, surgical excision via an external approach is the treatment of choice. 2 However, head and neck surgeons have found it hard to separate the mass from the CSC in operations. Hence, not uncommonly, the CSCS is excised with part of the nerve, although the dissection is meticulously performed. 5,7,9 -12 And postoperative HS is commonly encountered in up to 91.1% of cases. 13
First bite syndrome (FBS) is another potential complication after surgery of CSCS, which is very disturbing. Pain in the parotid area can be quite severe on the first bite of food. As more food is ingested, the pain associated with chewing and biting decreases, 5 but returns at the first bite of each subsequent meal. 1 Intensive ear pain is increased with strong sialogogues, 5 such as tart or bitter foods. 10 Since CSC is frequently sacrificed in CSCS surgeries, FBS is not uncommonly encountered, 1,10 and could occur in up to 21.1% of cases. 13
Yet, when lucky enough, CSCS can be excised without sacrificing CSC, and both HS and FBS can be avoided. 2,14 Here, we report our experience with 3 cases of successful intracapsular enucleation of CSCS.
Patients and Methods
A retrospective review of CSCS cases treated at our tertiary medical institution between Apr 2018 and March 2022 was conducted. Only cases with successful intraoperative intracapsular enucleation of CSCS and preservation of CSC were included. The clinical and demographic data was retrieved from case notes. The protocol of the research project was approved by the Bioethics Committee of Beijing Friendship Hospital, and it conformed to the provisions of the Declaration of Helsinki. A written consent form was obtained from the patients.
Diagnosis
All 3 patients complained of a growing neck mass, with no other symptoms. Preoperative cervical CT and/or MRI was routinely performed to evaluate the neck mass, especially for its relationship with vital structures like internal jugular vein (IJV), and carotid arteries, including internal carotid artery (ICA), external carotid artery (ECA), and common carotid artery. When the mass was found to be posterior to the carotid sheath, and likely to be a schwannoma, Furukawa criteria
15
was useful in predicting the mass to be originating from CSC, because there was no separation between IJV and ICA (see Figure 1A). Preoperative, intraoperative, and postoperative images of Case 3. A, Preoperative MRI T1 image demonstrated a homogenous mass (blue arrow), pushing the carotid arteries and internal jugular vein (IJV) anteriorly, without separating them. B, Intraoperative image showed vagus nerve at the tip of the monitoring probe, while the common carotid artery (yellow star) lied anteromedially, and the cervical sympathetic chain schwannoma (CSCS) lied posteriorly (blue arrow). C, Intracapsular enucleation was achieved, and the complete mass with a diameter of about 3 cm was demonstrated. D, Partial ptosis of the left upper eyelid presented at 4 hours after surgery.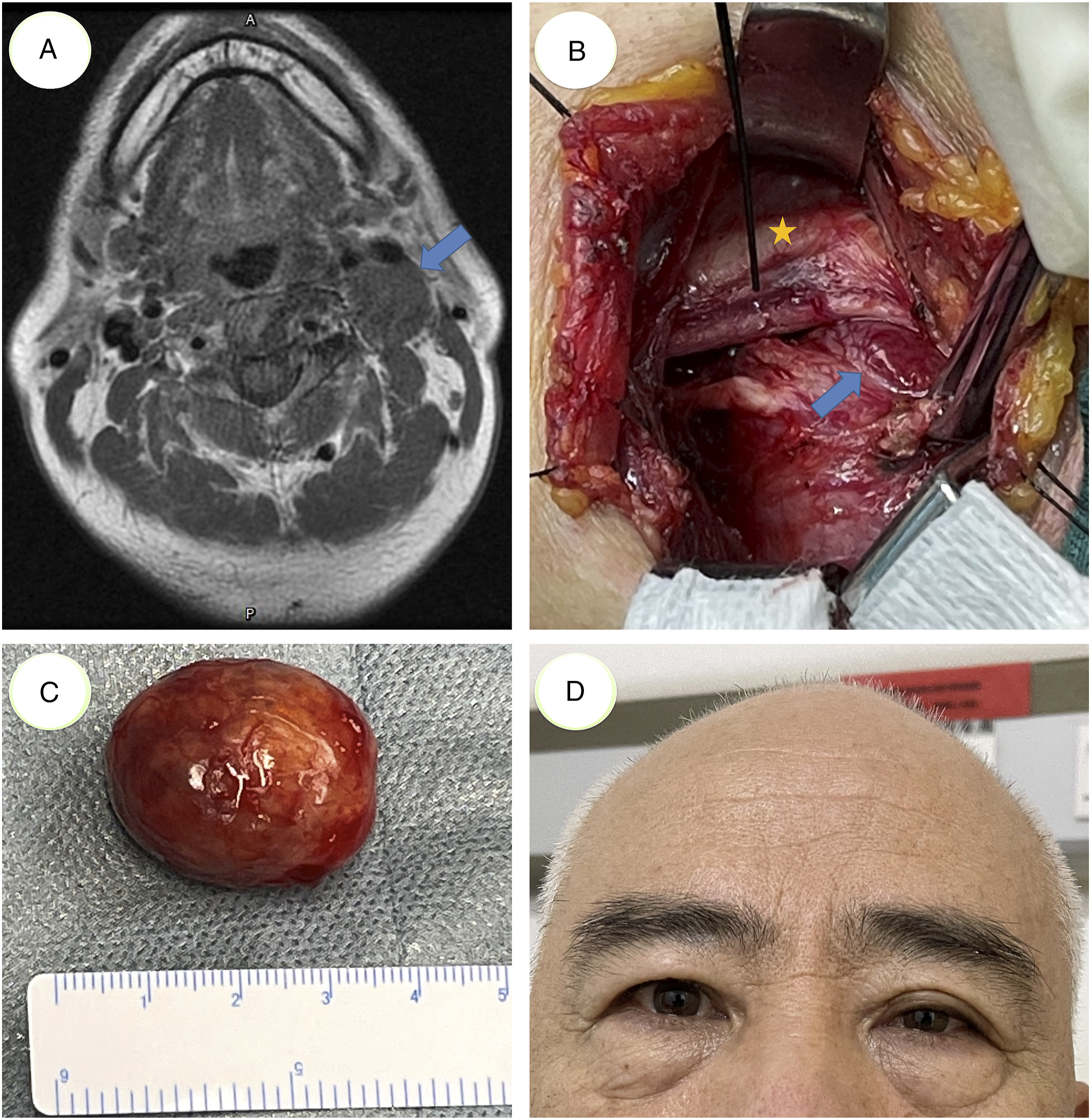
Surgical Procedure
A transverse skin incision was made along the nearest skin crease over the mass. Subplatysmal flaps were elevated, and the anterior border of the sternocleidomastoid muscle (SCM) was exposed. Then SCM was mobilized and retracted laterally to expose the carotid sheath. A mass posterior to the carotid sheath was identified, and IJV was mobilized and retracted medially. Recurrent laryngeal nerve monitoring was routinely utilized in our procedures, to confirm the vagus nerve (see Figure 1B), which also helped identifying the CSC, as recommended by literature. 11 Now the nerve fiber of CSC could be distinguished from the eccentric schwannoma under loupe magnification, and a longitudinal incision on the surface of CSCS capsule was made along the long axis of the schwannoma and away from CSC nerve fiber. Then, the schwannoma was meticulously dissected from the surrounding capsule, and en bloc excision was achieved (see Figure 1C). After careful hemostasis, the surgical field was irrigated with peroxide, saline, diluted iodophor, and saline, sequentially. Negative-pressure suction device was placed, and incision was closed.
Follow-up
Patients were routinely followed up every month after surgery in the first 6 months, to observe postoperative complications, especially the dynamic changes of HS. Then, they were followed up every 3 months.
Results
Clinical Data of This Series of Cervical Sympathetic Chain Schwannoma Patients.
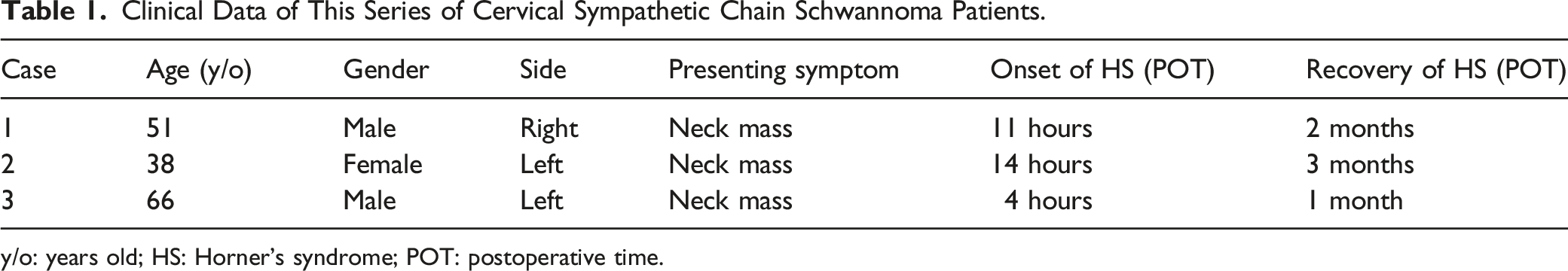
y/o: years old; HS: Horner’s syndrome; POT: postoperative time.
Intracapsular enucleation of the CSCS was attempted and achieved in all 3 cases. Hemorrhage from the inner surface of capsules, especially at the superior pole, was diffuse and heavy for all 3 cases. Constant saline irrigation, suction, and bipolar coagulation were needed to achieve hemostasis, which is quite demanding and time consuming. The hemorrhage in Case 2 was especially heavy, and her preoperative MRI showed significant contrast enhancement (figure not shown).
Postoperative HS presented at 11 hours, 14 hours, and 4 hours after surgery, which took 2 months, 3 months, and 1 month to recover, for the 3 cases, respectively (shown in Table 1). It almost seemed that the sooner HS presented postoperatively, the quicker it would recover. But this case series is too small to draw such a conclusion.
For Cases 1 and 3, HS only presented as partial ptosis (see Figure 1D), while for Case 2, both partial ptosis and slight miosis were observed. For all 3 cases, neither hemifacial anhidrosis nor enophthalmos was present. Fortunately, FBS did not occur in any patient. Recovery of all 3 patients was otherwise uneventful.
Median and average duration of follow-up were 6 months and 19 months, respectively. No recurrence was observed.
Discussion
HS is characterized by the classic triad of miosis, partial ptosis, and hemifacial anhidrosis, because the release of norepinephrine from postganglionic oculo-sympathetic fibers, which innervate the muscle of Müller within the eyelid, the dilator of the iris, and the sweat glands of the face, is inhibited. 16 Although most authors reported postoperative HS to be very asymptomatic with no patient requiring intervention, 5,9 quality of life can be affected by distortion of the eyelid and pupils affecting facial aesthetics, visual impairment, and pain. 13
For a case that could not preserve CSC, miosis and ptosis were reported to occur on postoperative day 2, and additional clinical findings of HS, including enophthalmos, anhidrosis, and vasodilatation of the ipsilateral face would present during follow-up of 20 months after surgery. 9 And for another CSCS case at our institution without CSC preservation, miosis and ptosis presented on postoperative day 3 (data not reported in this series). However, in this series with preserved CSC, ptosis and/or miosis would present at 4–14 hours after surgery. It is quite awkward that we could not interpret this phenomenon, even after consultation with Neurology Department. It is our assumption that, in our attempt to achieve hemostasis, heat injury might be inflicted upon the adjacent CSC nerve fibers, even with constant saline irrigation; and this injury would somehow accelerate the appearance of HS. But even this reasoning could not be applied to all scenarios: hemostasis in Case 2 was the most difficult and hence potential heat injury and stimulation to CSC should be the worst; yet the presence of postoperative HS was the latest, although its recovery was indeed the slowest.
On the other hand, it is assumed that FBS may be caused by loss of sympathetic input to the parotid gland after severing of the CSC. This results in a denervation hypersensitivity of the sympathetic receptors that control myoepithelial cells in the parotid gland. 5 This pain can be quite disturbing and may deter the patient from eating. Symptoms resolved slowly over time and have been reported to continue for more than 6 months before beginning to subside. 5,10 Treatment includes restriction to bland foods, anti-inflammatory medication, and oral carbamazepine (100–200 mg twice daily) with severe pain; 1,10,17 and for patients who could not benefit, pregabalin 75 mg twice daily (for 2 weeks) might be needed. 1 Besides, intraoperative sacrifice of CSC could result in severe facial pain for as long as 2 years even with medical management, which would make the patient unable to work. 18 Hence, maybe it is best to preserve the CSC during operations, whenever possible, to prevent the occurrence of FBS.
Dysfunction of the nerve of origin may not only result from axons being stretched over the tumor capsule but also adjacent nerves may be affected by direct pressure; therefore, importantly, specific nerve dysfunction does not indicate with certainty the exact site of origin of the tumor. 16 Indeed, it is the opinion of some authors that determination of the nerve of origin often remains elusive and diagnosis is not often made until the time of surgery. 10,11 Luckily, there is an anatomic pattern that may be helpful. The CSC is located posterior to the carotid sheath structures. Consequently, lesions of the CSC, as they grow and expand, will tend to push the common carotid artery, or ICA and ECA, anteriorly. The IJV will also be pushed anterolaterally. 5 And hence, no separation can be observed between the IJV and carotid arteries. In contrast, for masses arising from the vagus nerve which grow between the carotid arteries and the IJV, there will be an increase in the distance between the artery and vein (separation). 15 This paradigm was first proposed by Furukawa et al, 15 although a caveat must be added, because a CSCS that splayed the IJV and ICA had been reported. 3 And a recent review has demonstrated that ICA and IJV splaying with medial ICA displacement carries a 75% probability of vagal nerve schwannoma; while the absence of such splaying with lateral ICA displacement carries an 87% probability of CSCS. 19
It is also worth noticing that CSCS could develop at the level of carotid bifurcation and cause splaying of the carotid bifurcation (the lyre sign), which should be differentiated from carotid body tumor, based on the vascularity of the mass, 12,16,20 and the observation that CSCS neither fills the crotch of the carotid bifurcation nor encases the arteries. 15 Yet, it is confusing that CSCS can show avid enhancement on MRI, 1 which might be associated with histological findings such as intra-tumoral hemorrhage and vasodilation, 12 and it could further be supported by our intraoperative findings of diffuse hemorrhage from inner surfaces of capsules, which indicated abundant blood supply to the schwannoma. Therefore, extreme caution must be taken when managing these conditions, and intraoperative hemostasis could be troublesome. Besides, if MR images are acquired beyond the first 60 seconds after the contrast injection, they can show marked enhancement due to pooling of contrast from poor venous drainage, simulating a hypervascular lesion. 3
Conclusion
Intracapsular enucleation should be attempted in CSCS surgeries, although hemorrhage might be diffuse and hard to control, especially for those that demonstrate significant enhancement on MRI with contrast. When CSC was preserved, postoperative FBS can be avoided, while postoperative HS could occur, but its recovery was satisfactory.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Beijing Hospitals Authority’s Ascent Plan [grant number DFL20220102].
