Abstract
A 58 year old male with a history of prostate adenocarcinoma presented with diplopia, severe headaches, and eye pain, consistent with sinusitis. Imaging was concerning for invasive fungal sinusitis (IFS) and an urgent ENT consultation was requested. Endoscopic sinus surgery was performed revealing metastatic prostate adenocarcinoma to the sinuses and anterior cranial fossa. The distinctive imaging features in this case are very useful when considering the divergent management options of IFS and metastatic sinus disease. These entities are likely to be encountered more frequently as immunomodulating therapies expand and prostate cancer continues to be a leading cause of death in males.
Introduction
Prostate cancer is the most commonly diagnosed male malignancy and is the fourth leading cause of cancer-related deaths in men around the world. 1 The mean age of diagnosis is 67 years. About 156 out of 100,000 men are diagnosed with prostate cancer each year. 2 The majority of prostate cancers are low grade and slow growing. 1 However, prostate cancer may occasionally metastasize to the bone (84%), distant lymph nodes (10.6%), liver (10.2%), thorax (9.1%), and skull (4%). 3 A total of 18% of patients have multiple metastases. 4 Prostate cancer metastatic to the sinuses is extremely rare.5,6
Invasive fungal sinusitis (IFS) is a critical infection that affects the sinuses when fungus is inhaled into the nasal passages and paranasal sinuses. 7 In susceptible patients, pathogenic fungi invade vascular spaces and tissues, leading to problems affecting the visual system and/or the central nervous system. 8 Most patients diagnosed with IFS have compromised immunity. 9 Individuals who have diabetes, use systemic steroids, take broad-spectrum antibiotics, or undergo chemotherapy are at higher risk of developing IFS. 10 IFS is an important clinical entity to recognize given its significant mortality rate (20%-80%). 7 We present an unusual case of an immunocompromized patient with a history of steroid use, broad-spectrum antibiotic use, and a known underlying malignancy appearing clinically to meet diagnostic criteria for IFS. Imaging and surgical management ultimately revealed that there was no invasive fungal process, but instead metastatic prostate cancer. The patient granted his verbal informed consent for the publication of this case report. Upon consultation, the local institutional review board and privacy office stated that verbal consent was sufficient.
Case Report
A 58 year old Caucasian male with prostate cancer and a recent history of severe, acute sinusitis presented to the emergency department (ED) with the chief complaints of intermittent diplopia, blurred vision, ocular pressure, and severe frontal headache. His advanced prostate cancer had been treated with castration, androgen deprivation therapy, and radiation therapy, without chemotherapy, 12 months prior to the current admission. Six weeks prior, the patient had been treated in a different ED where magnetic resonance imaging (MRI) showed orbital cellulitis and sinusitis. He was prescribed antibiotics, vancomycin, and meropenem via a peripherally inserted central catheter to treat the acute sinus infection.
Examination and Imaging
ENT was immediately consulted. Bedside endoscopy revealed mild edematous and inflamed mucosa with polypoid mucosal changes in the right sphenoethmoidal recess. No frankly purulent material or necrotic tissue was seen. Urgent imaging was then obtained. Computed tomography (CT) imaging demonstrated evidence of chronic pansinusitis disease with stranding of the right retroantral maxillary fat which raised the suspicion of IFS. MRI indicated bilateral frontal dural thickening with enhancement, as well as subdural enhancing lesions more prominent on the right, suggestive of intracranial extension of the sinus disease. Imaging also revealed a right frontal abscess measuring approximately 1.8 cm. CT and MRI showed complete obstruction of the maxillary sinuses with findings consistent with IFS, albeit with an unusual spiculated-type sunburst periosteal reaction (Figures 1 and 2).
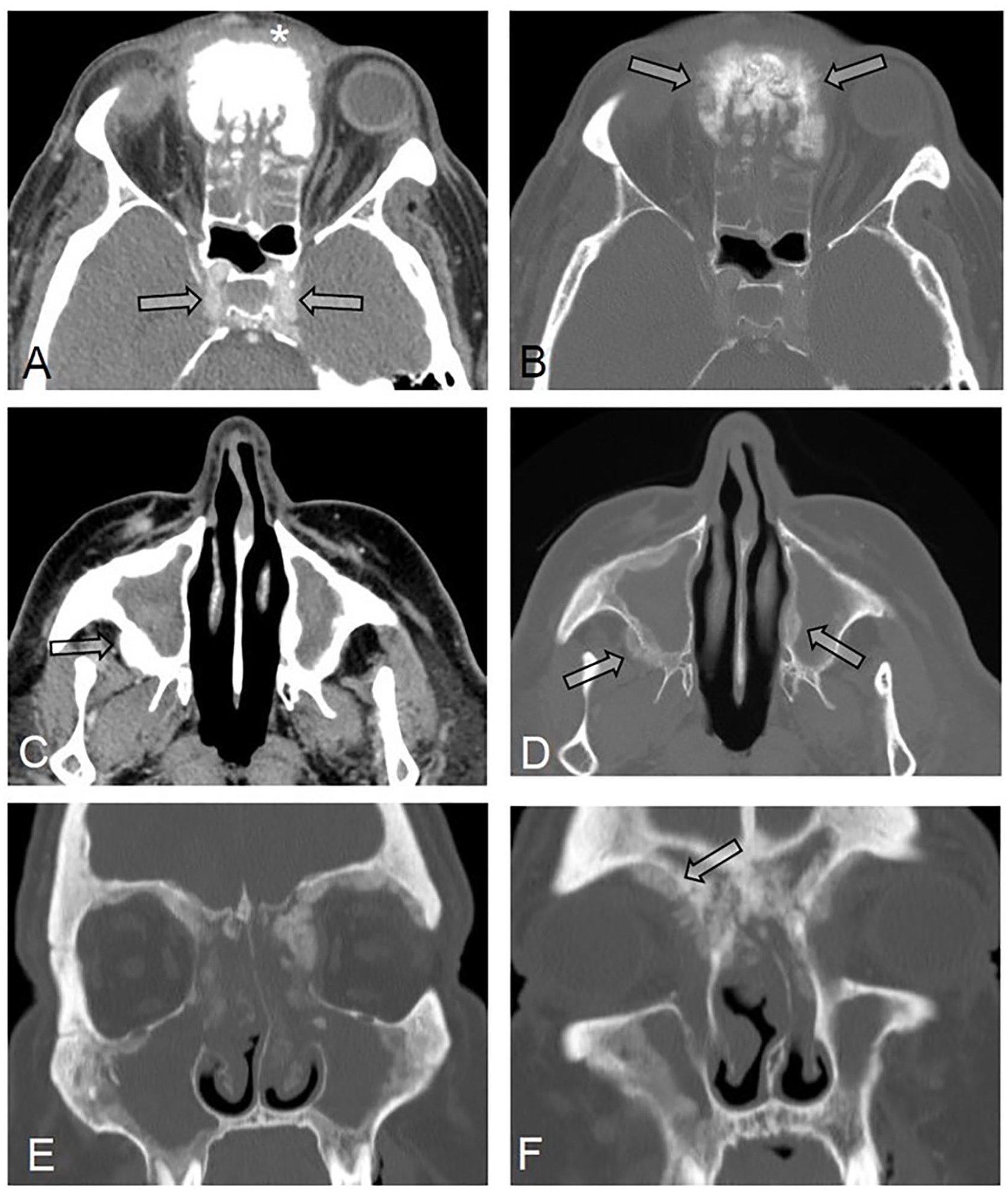
(A) Axial contrast-enhanced CT images of the paranasal sinuses showing complete obstruction of the ethmoid sinuses by isodense material associated with extensive predominantly anterior sinus wall bony thickening as well as swelling and stranding of the soft tissues anterior to the sinuses walls (Asterisk). Note the homogeneous enhancement of the cavernous sinuses and bilateral internal carotid arteries excluding cavernous sinus thrombosis and internal carotid artery (ICA) involvement by disease (Arrows). (B) Axial bone window at the same level showing extensive bone thickening with spiculated-type sunburst periosteal reaction (Arrows). (C) Axial contrast-enhanced CT images of the paranasal sinuses showing complete obstruction of the maxillary sinuses by centrally hyperdense and peripherally isodense material with diffuse sinus wall thickening in addition to soft tissue stranding in the right posterior periantral fat, a finding commonly seen with invasive fungal sinusitis (arrow). (D) Axial bone window at the same level showing extensive bone thickening and the same spiculated-type sunburst periosteal reaction (Arrows). (E, F) Coronal bone windows showing complete opacification of the ethmoid and maxillary sinus with extension to the nasal cavities and diffuse bone sinus wall thickening associated with intra-orbital extension along the medial wall and roof of the orbits. Again, note the unusual spiculated-type sunburst periosteal reaction (Arrow).
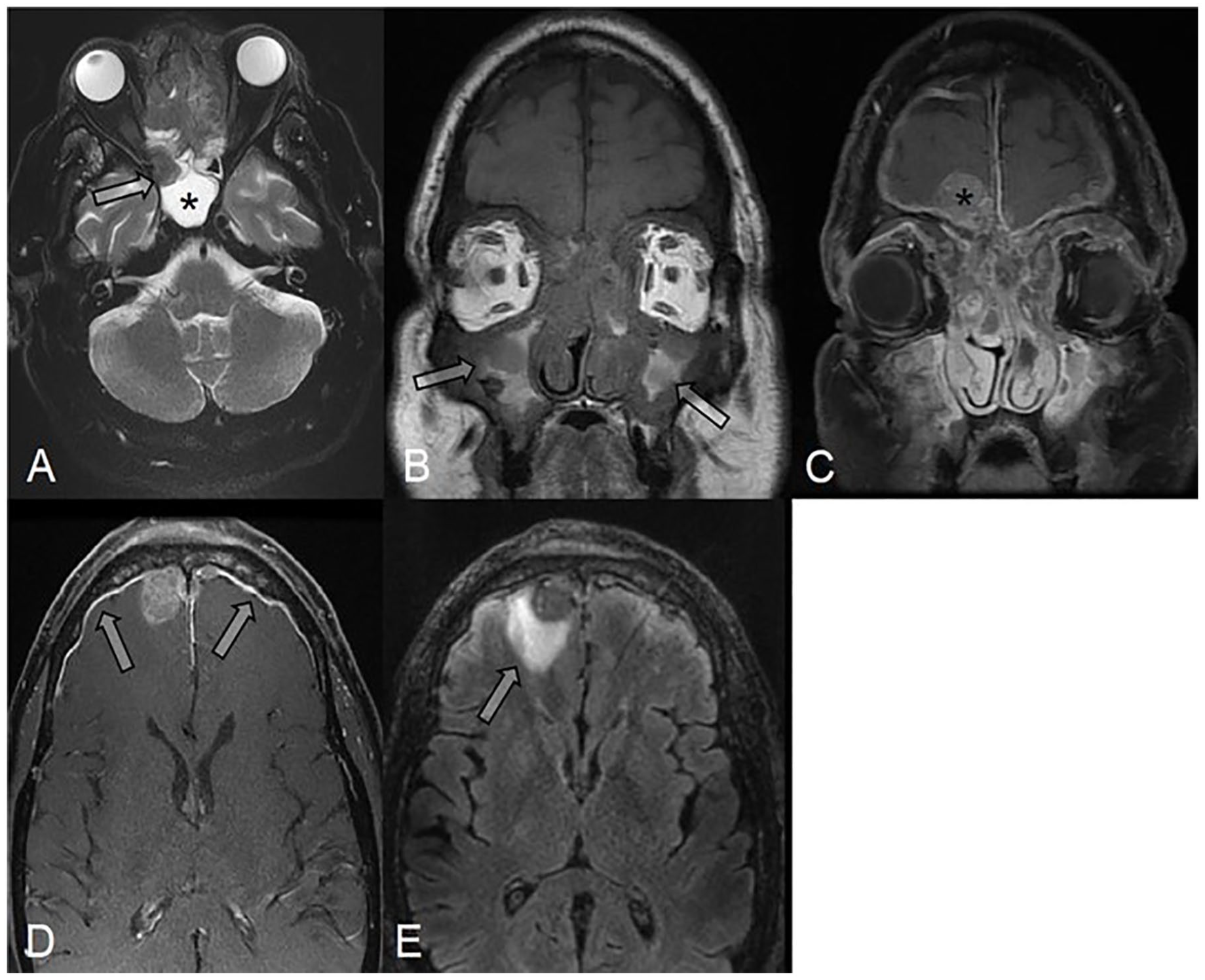
(A) Axial T2-weighted MRI of the brain showing the peculiar low T2 signal of the lesion (Arrow). T2 WI sequences are very helpful in differentiating the lesion from the T2 hyperintense post-obstructive fluid in the right sphenoid sinus (Asterisk), thus mapping the extent of the disease. (B) Coronal non-enhanced T1-weighted MRI showing the low T1 SI lesion invading the bones with trapped T1 hyperintense fluid centrally (Arrows). (C, D) Coronal and axial post-contrast T1 WI images of the sinuses better delineating the enhancing lesion and its intra-orbital extension and revealing an extensive dural-based intracranial extension (Arrow) with a dominant right frontal mass indenting the adjacent frontal lobe (Asterisk). (E) Axial T2-FLAIR images of the brain showing edema in the adjacent right frontal lobe (Arrows) which could be reactive or indicate parenchymal invasion.
Surgical Management
The patient was then transferred to a tertiary care center for rhinologic management of presumed IFS. Since no objective cranial nerve deficits were seen, neurology, ophthalmology, and infectious disease specialists were consulted. He was taken to the operating room for a biopsy and treatment. Endoscopy revealed normal nasal mucosa; however, the anatomic landmarks within each middle meatus were misshapen. An ethmoidectomy revealed contour irregularities of the skull base and multiple, mucosalized, 0.5 to 1.0 cm irregularly shaped lesions which were removed and sent for pathology. Submucosally, the lesions had the clinical appearance of cancellous bone. Frozen section analysis came back negative for fungal elements and suspicious for malignancy. At this point the procedure was terminated, awaiting the results of permanent pathology, which revealed high-grade prostate adenocarcinoma, leading to de-escalation of his antimicrobial therapy.
Discussion
This case was puzzling, as the patient’s overall clinical picture and lack of intranasal findings were inconsistent with the extent of his intracranial disease. However, IFS can have variable presentations 11 and the clinical presentation of diplopia and invasive sinus disease on imaging in the setting of malignancy were suggestive of IFS. Due to the high risk of mortality with IFS, vigilance in considering this diagnosis was warranted, and appropriate action was taken.
The rare and unique presentation of this case highlights the radiologic similarities of intracranial metastases and IFS. The clinical importance of distinguishing between these 2 entities is clear when one considers the divergent management approaches, both in terms of timing and extent of intervention. With IFS, emergent and aggressive surgical intervention is required. On the other hand, sinus metastases can be managed more conservatively, and surgical intervention is generally limited to diagnostic biopsy.
On imaging, the patient’s lesion resembled invasive fungal sinusitis especially with low T2 signal on MRI; however, the presence of spiculated-type sunburst periosteal reaction is unusual for IFS 12 and in retrospect would point more to metastatic prostate cancer.13,14
Both IFS and metastatic cancer can present with headaches, fever, and cranial neuropathies. Only IFS, however, is a surgical emergency. 7 Because divergent management protocols exist, correct diagnosis is essential. In this case report, the patient had multiple risks factors for IFS (immunocompromized with chronic steroid use), and symptoms and imaging were largely consistent with IFS.
This case highlights the unusual presentation of metastatic prostate cancer to the sinuses and the fact that it can mimic IFS. It also highlights some of the key differentiating factors for IFS, including limited symptoms and physical findings as well as the presence of spiculated-type sunburst periosteal reactions on MRI, both of which are unusual for IFS.
Footnotes
Acknowledgements
The authors thank Allison Foster, PhD, of Foster Medical Communications, for editing and administrative support. Dr. Foster was paid by the corresponding author.
Authorship Statement
The authors designed the study, collected the data, drafted the article, and approved the final version to be published.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with the World Medical Association’s Declaration of Helsinki.
Statement of Informed Consent
Verbal informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
