Abstract
Introduction
Antibiotics are important in the treatment of acute exacerbations of chronic rhinosinusitis (AECRS).1,2 AECRS is defined as a sudden worsening of chronic rhinosinusitis (CRS) symptoms with a return to baseline after treatment. 1 Patients with CRS often receive multiple courses of oral antibiotics throughout their lifetime and are more likely to harbor antibiotic-resistant pathogens compared to patients with acute rhinosinustis.3,4
A few studies have reported effectiveness of topical antibiotics in CRS; however, higher level evidence has failed to consistently demonstrate its benefits.5,6 Although the International Consensus Statement on Allergy and Rhinology recommended against the routine use of topical antibiotics in CRS, it did state that this therapy may be beneficial in specific situations as an alternative to intravenous antibiotics or for organisms resistant to oral antibiotics. 1
Adverse effects with oral antibiotics range from mild to life threatening and can further limit treatment options for patients with AECRS. In these patients, topical antibiotics may be a reasonable alternative. To our knowledge, the administration of antibiotic irrigations in patients unable to take the oral form has not been previously investigated. The objectives of this pilot study were to determine the safety and efficacy of antibiotic irrigations for AECRS in patients with allergic reactions or adverse effects with the oral form of the medication.
Patients and Methods
Institutional Review Board’s exemption was obtained (IRB# 21-001756). This retrospective review included adult patients treated at a tertiary care center from January 2017 to January 2023 by a single rhinologist (JDS). Inclusion criteria included diagnosis of AECRS, treatment with antibiotic irrigations, and a documented allergy or adverse event to the systemic form of the antibiotic. Exclusion criteria included nonadherence to the full treatment course, inadequate follow-up, <18 years of age, and concurrent treatment with an oral antibiotic.
Patients were diagnosed with AECRS based on symptoms and the presence of mucopurulence on nasal endoscopy. In all cases, endoscopically guided nasal cultures were obtained from the purulent drainage. Patients were prescribed antibiotic irrigations as the sole treatment based on cultures and sensitivities. The decision to prescribe antibiotic irrigations was made with careful consideration of the patient’s disease course. Often, patients experienced multiple exacerbations of CRS despite oral antibiotics. In addition, previous adverse reactions to oral antibiotics precluded their administration, while limiting alternative treatment options. Whenever possible, an antibiotic that had not previously caused anaphylaxis was prescribed. Furthermore, the antibiotic that caused milder adverse effects was preferred over those associated with more severe effects in each patient. Because there is minimal research evaluating the potential risks of bacterial resistance with administration of topical antibiotics, 7 prescription of certain antibiotics was avoided in patients with high chance of disease recurrence. For instance, levofloxacin/ciprofloxacin was avoided in patients with culture positive P. aeruginosa to avoid the potential risk of developing resistance to its oral form. A detailed informed consent was obtained from all patients prior to initiating treatment.
Antibiotic irrigations were prescribed twice daily for 1 month. Patients were reevaluated after treatment completion to determine response. Antibiotics included trimethoprim/sulfamethoxazole (TMP/SMZ) (80 mg/50 mg capsule/240 mL saline), levofloxacin (100 mg capsule/240 mL saline), and ciprofloxacin (500 mg capsule/240 mL saline), which were obtained from Cascade Specialty Pharmacy (Poulsbo, Washington). Demographic information and outcomes data were collected.
Results
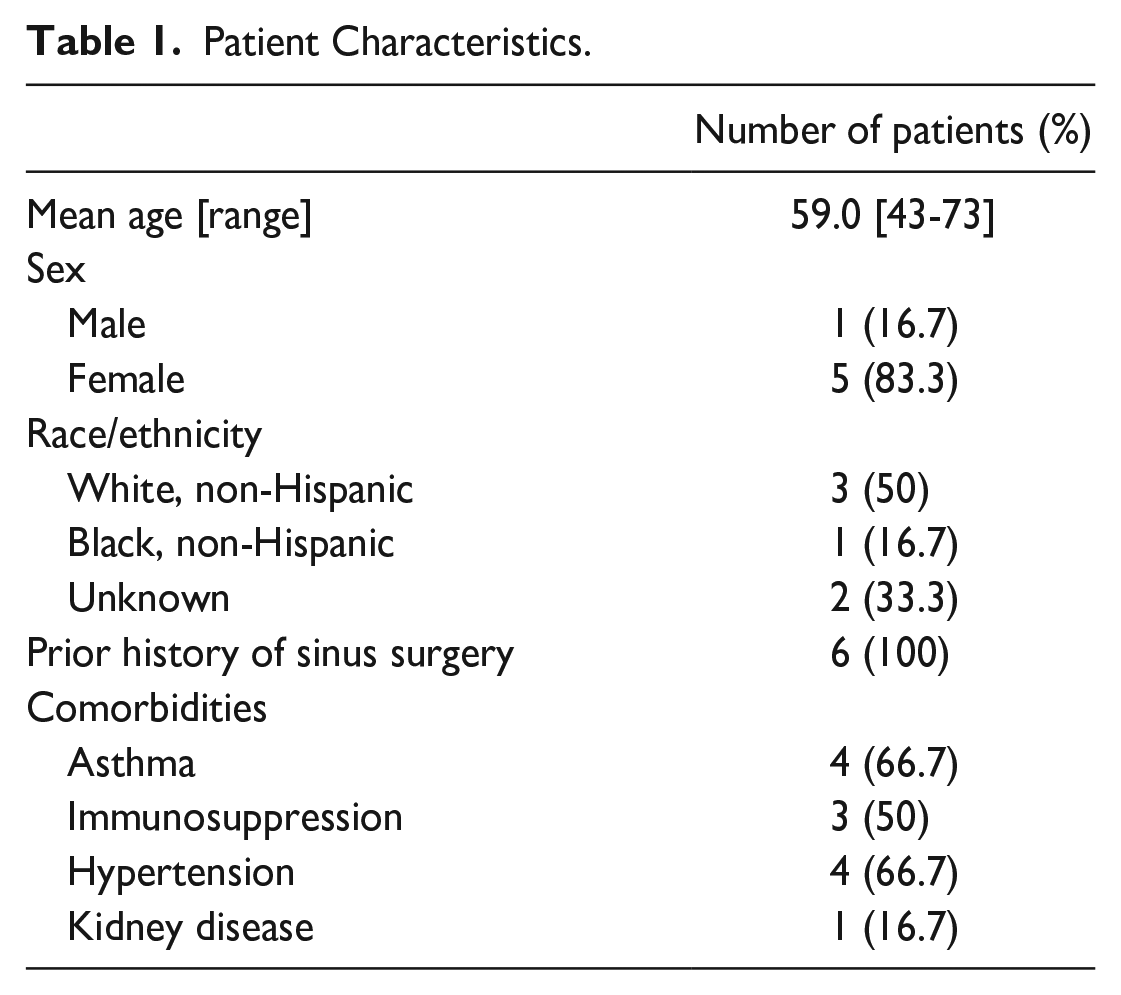
Six patients met the inclusion criteria, resulting in 7 treated cases of AECRS. There were 5 females and 1 male, with a mean age of 59.0 years (range: 43-73; Table 1). All patients had a history of endoscopic sinus surgery (ESS). Five patients with adverse effects to oral TMP/SMZ received TMP/SMZ irrigations. One patient also had an adverse reaction to oral levofloxacin and was treated with levofloxacin irrigations during a separate AECRS episode. One patient with an adverse reaction to oral ciprofloxacin was treated with ciprofloxacin irrigations. Adverse reactions to systemic TMP/SMZ included anaphylaxis, rash, fever, diarrhea, nausea, and vomiting; the reaction to systemic levofloxacin and ciprofloxacin was Achilles tendinopathy and rash, respectively.
Patient Characteristics.
Pretreatment culture results are shown in Table 2. There were no allergic or adverse reactions to the antibiotic irrigations in this patient cohort. Following treatment, 1 case (14.3%) resulted in complete resolution of infection, 1 (14.3%) had partial improvement, and 5 (71.4%) had minimal to no endoscopic improvement at the subsequent clinic visit. Among cases with partial or no response on endoscopy, posttreatment cultures revealed that 1 case was culture negative despite clinical evidence of persistent infection, whereas 5 cases had no treatment effect and were still culture positive with either the same or new bacteria.
Outcomes of Antibiotic Irrigations in Patients with Concomitant Adverse Reactions.
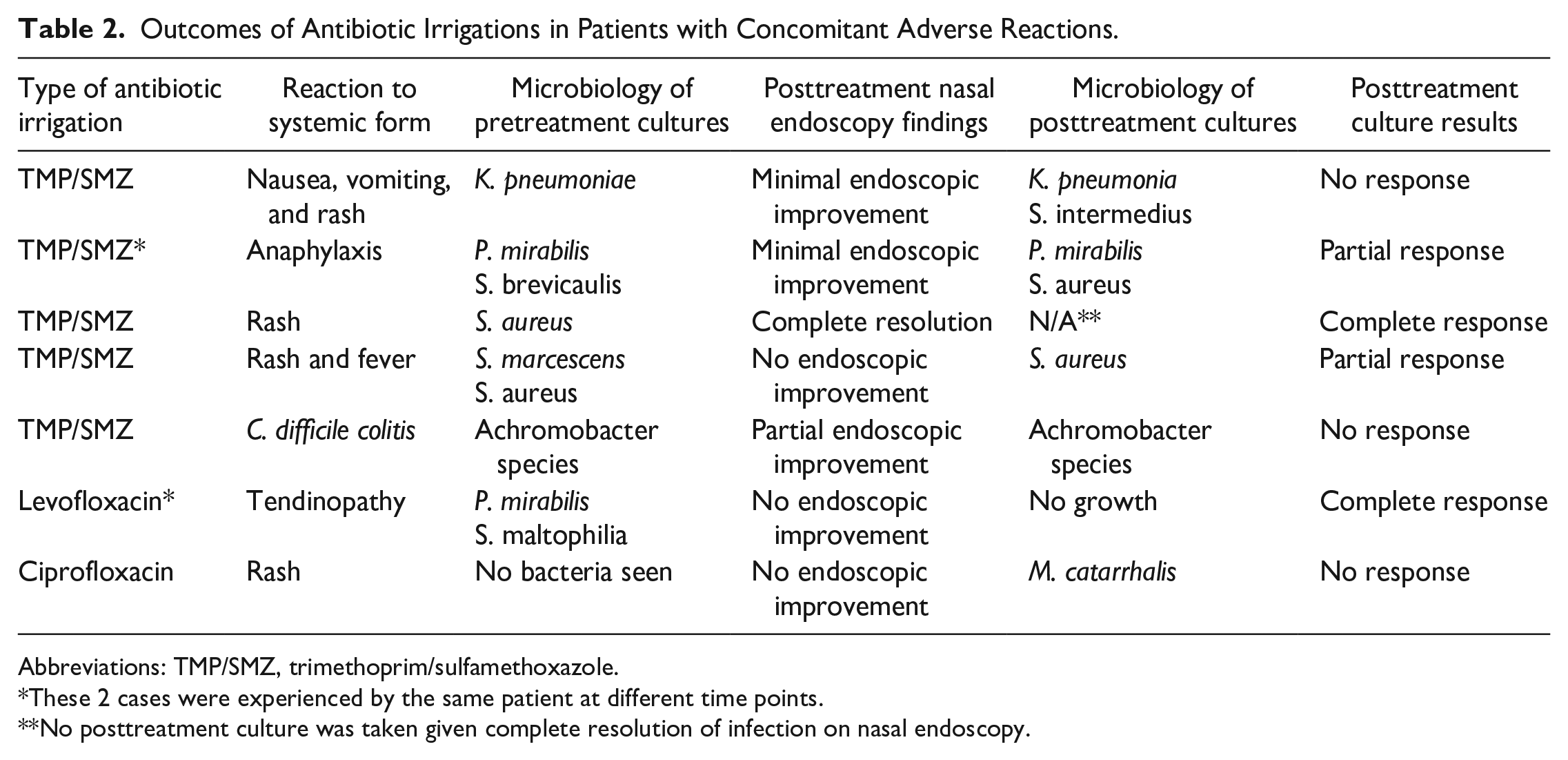
Abbreviations: TMP/SMZ, trimethoprim/sulfamethoxazole.
These 2 cases were experienced by the same patient at different time points.
No posttreatment culture was taken given complete resolution of infection on nasal endoscopy.
Discussion
In the current study, topical administration of TMP/SMX, levofloxacin, or ciprofloxacin in patients with an adverse reaction to its systemic form did not cause any side effects or allergic reactions. These findings suggest that topical administration of antibiotics may be a safe alternative for patients with adverse effects to the systemic form. Currently, no prior study has examined whether adverse reactions to a systemic antibiotic also occur when the medication is delivered topically via sinonasal irrigations.
An important consideration in administering topical antibiotics to which the patient has reported a previous allergy is the potential systemic absorption of the irrigation. Two studies have investigated the systemic absorption of intranasal administration of topical gentamicin with conflicting results. One study found that 10 of the 12 post-ESS patients with CRS (83%) who were treated with an average of 7 weeks of gentamicin irrigations had detectable serum gentamicin levels after treatment. 8 Conversely, a prospective study of 20 patients who received gentamicin irrigations intraoperatively during ESS found only 3 patients (15%) with detectable serum levels of gentamicin post-irrigation. 9 The conflicting results between these studies is likely due to the fact that the former used antibiotic irrigations for an average of 7 weeks, while the latter assessed one-time dosing. Finally, a prospective study investigating systemic absorption of sinonasal tobramycin and colistin sulfomethate sodium irrigations found 2 of 10 patients with detectable tobramycin levels well below toxic levels and no patient with detectable colistin A or B levels. 10 Additional studies are needed to determine systemic absorption of antibiotics such as TMP/SMX, levofloxacin, and ciprofloxacin via the nasal mucosa, with a focus on safety in patients with antibiotic allergies. Because absorption rates for these antibiotics are currently unknown, caution is advised when prescribing them topically to patients who have experienced previous adverse reactions.
Finally, this study found that only 1 case (14.3%) resulted in complete resolution of infection following antibiotic irrigations. Notably, a resolution response was seen in the patient with pretreatment culture results of S. aureus, a known biofilm-producing microorganism. Although another purported benefit of topical antibiotic administration is biofilm penetration, efficacy of topical antibiotic use on S. aureus-associated AECRS is lacking. However, animal and in vitro studies examining the effect of topical antibiotics on S. aureus biofilms found significant reduction in biofilm surface area, suggesting potential benefits of topical antibiotic delivery.11,12 Overall, the use of topical antibiotics in the treatment of CRS and AECRS remains controversial; however, in some patients it represents a viable option prior to initiation of parenteral antibiotics when there are no oral antibiotic alternatives.
There are several study limitations. This was a retrospective, single institution study with a small patient cohort, which introduces bias and potentially limits its broader applicability. Larger, prospective studies are needed to investigate the safety and efficacy of antibiotic irrigations in patients with adverse reactions to the systemic form of the antibiotic.
Footnotes
Author’s Note
This article was presented as a poster at the American Rhinologic Society’s Spring Meeting in Dallas, TX on April 28-29, 2022.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
