Abstract
Introduction
Sinonasal mucosal melanoma (SNMM) is a rarely seen malignancy. It is more likely to develop in the nasal cavity (approximately 80%) than in the paranasal sinuses (approximately 20%).1 -3 Patients with an SNMM mostly complain of symptoms such as nasal congestion and rhinorrhea. 4 Therefore, an advanced stage of SNMM is found in most patients, and a poor prognosis is difficult to avoid. Previous studies have shown the recurrence rate of SNMM to be approximately 50% after initial treatment. The rate of distant metastasis rate is 58.5%, and the 5-year patient survival rate ranges from 10% to 30%.5 -7
No previous report has described an SNMM that develops in conjunction with another type of tumor occurring in the contralateral nasal cavity. When bilateral sinonasal tumors develop, the occurrence of SNMM can be easily overlooked. Here, we report a case of SNMM with a sinonasal inverted papilloma (SIP) developing in the contralateral nasal cavity, with the aim of providing some suggestive information for surgeons.
Case Report
A 74-year-old male was admitted to the hospital complaining of persistent bilateral nasal congestion for 1 month. He also reported coughing up blood and pain in the left forehead. He denied any family history of malignancy. Nasal endoscopy revealed a dark gray neoplasm that completely occupied the left nasal cavity, and also a pink neoplasm located in the right olfactory cleft (Figure 1A and B). Sinus computed tomography (CT) showed opacification in the left nasal cavity and right olfactory cleft (Figure 1C). The signal intensity of magnetic resonance imaging (MRI) in both nasal cavities was high (Figure 1D), and contract-enhanced imaging showed inhomogeneous enhancement in both nasal cavities, and linear dark signal intensity (Figure 1E). The signal intensity of fat-suppressed T2-weighted imaging was high (Figure 1F).
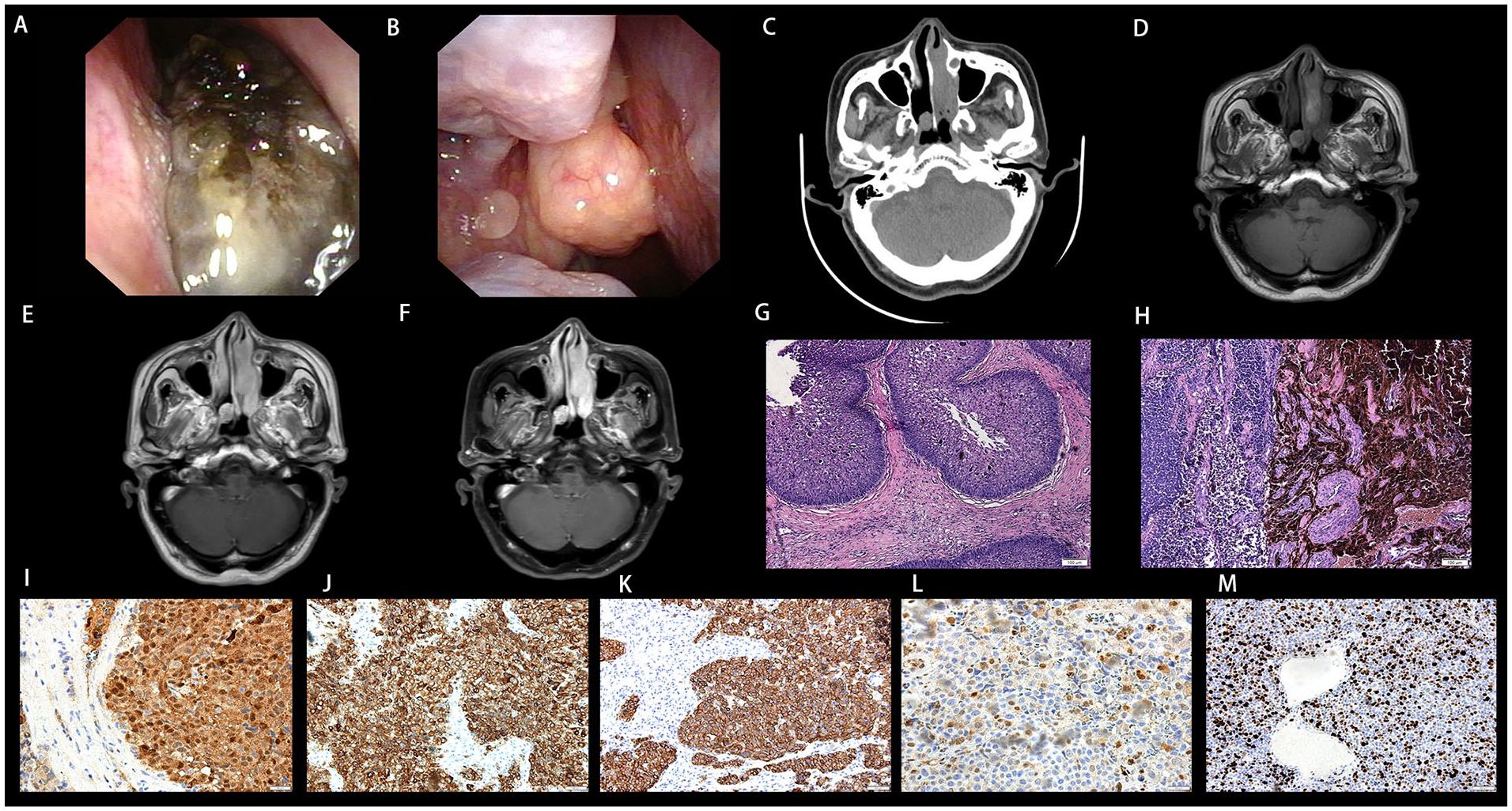
Nasal endoscopy (A; right) and (B; left) showed the morphology of tumors. Axial computed tomography (C) revealed the extent of each tumor. (D) Axial T2WI image of the tumors. (E) Contrast-enhanced MR images of the tumors. (F) Fat-suppressed T1wC image of the tumors. The histologic findings of SIP (G) and SNMM (H). The histopathological features of SNMM revealed large amounts of melanin pigmentation in the tumor cells. Positive staining for S-100 (I), MelanA (J), HMB45(K), and CyclinD1 (L). (M) 70% for Ki67. (Hematoxylin and eosin staining, magnification, G, H, ×100; Immunostaining, magnification, J, K, and M, ×200; I and L, ×400). T2WI, T2-weighted imaging; T1wC, T1-weighted contrast-enhanced; SNMM, sinonasal mucosal melanoma; SIP, sinonasal inverted papilloma.
Nasal surgery was performed, and an intraoperative histopathological examination showed the left tumor to be SNMM and the right tumor to be an SIP (Figure 1G and H). During surgery, the SNMM was found to originate from the bottom of the nasal cavity. To minimize the risk for recurrence of both tumors, both bilateral nasal tumors were resected, and the mucus and risky bone areas were completely removed.
The right tumor had a pink tip, the left nasal tumor was black, soft, and could easily bleed. A postoperative histopathological examination of the SNMM showed the tumor to be S-100 (+), MelanA (+), HMB45 (+), CyclinD1 (+), CK (−), Syn C (−), HNF1β (−), and have a 70% positivity for Ki67 expression (Figure 1I to M).
Based on the malignancy of the SNMM, further radiotherapy and chemotherapy were recommended; however, the patient refused those treatments. Seven months later, the patient presented again, with complaints of left nasal congestion, rhinorrhea, and headache for a period of 1 month. He reported no discomfort in the right side. A physical examination showed that the left nasal cavity was completely obstructed. Nasal endoscopy, CT, and enhanced MRI showed that the left nasal cavity was completely occupied by a neoplasm that extended to the nasopharynx (Figure 2A to D). No neoplasm was found in the right nasal cavity. A further positron emission tomography-computed tomography examination suggested the recurrence of SNMM in the left nasal cavity and indicated extensive distant metastases in the patient’s bones, organs, and lymph nodes (Figure 2E). However, the patient refused all types of further therapy and chemotherapy treatment and died after 4 months of conservative management.
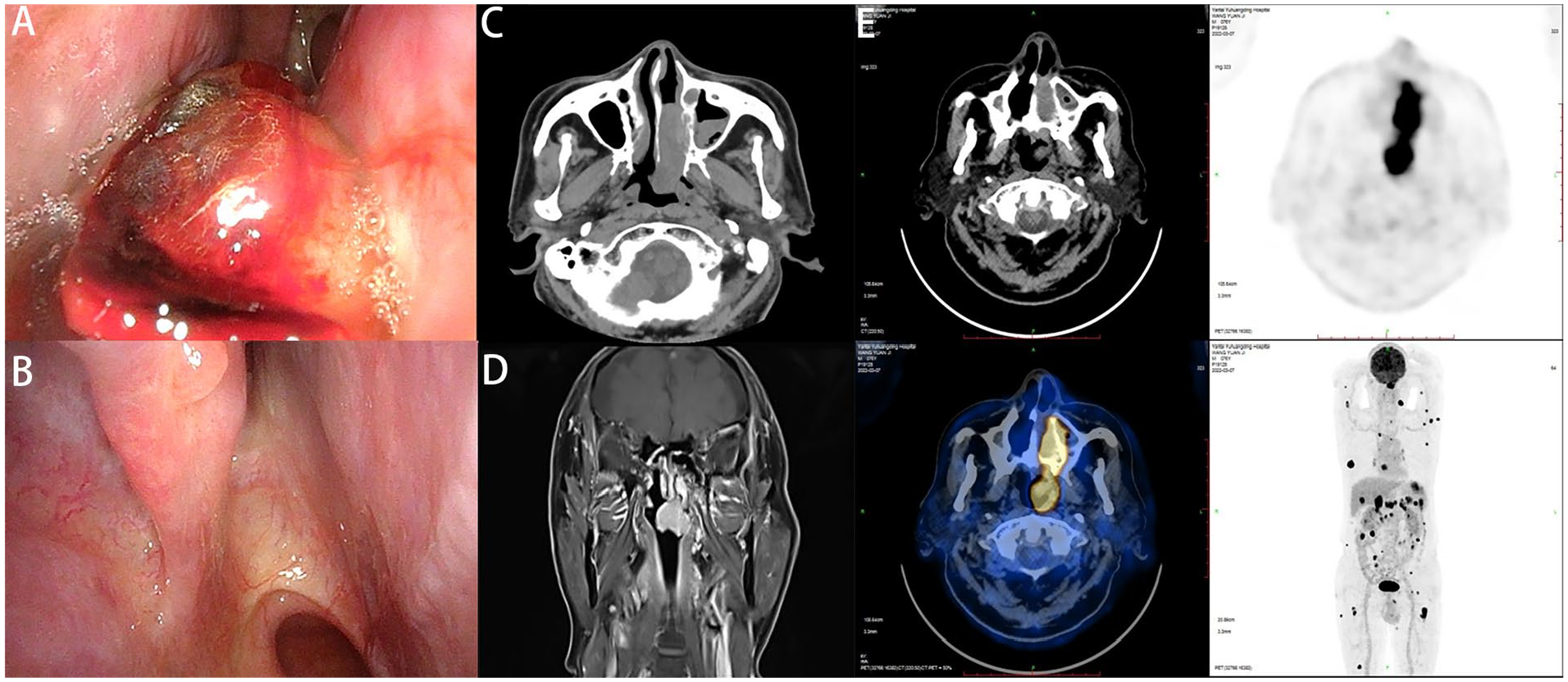
Nasal endoscopy (A; left) and (B; right) showed the recurrence of left nasal swelling, while the right nasal cavity was clean. Computed tomography (C) revealed adjacent bone destruction caused by swelling. Magnetic resonance imaging (D) revealed irregular signals in the left nasal cavity as well as in the maxillary sinus. PET-CT (E) revealed extensive distant metastases. PET-CT, positron emission tomography-computed tomography.
Discussion
Melanoma is an uncommon malignancy. When present, it mainly originates from neural crest cells and is regarded as a highly malignant tumor of cutaneous origin with a rapid course of development. Malignant melanoma in the upper respiratory region accounts for only 0.3% to 2% of all systemic malignant melanomas,8 -11 while SNMM accounts for >72.0% of all malignant melanomas in the head and neck region. 12 In patients with SNMM, the rich vasculature and lymphatic vessels in the nasal cavity contribute to the tumor’s early spread and metastasis. 13 As a result, patients with SNMM tend to have a poor prognosis and experience tumor recurrence or metastasis. When compared to the rarity of SNMM, the presence of an SNMM accompanied by another type of tumor in the contralateral nasal cavity is much less common. Here, we describe a case of SNMM with an SIP in the contralateral nasal cavity, for the purpose of providing suggestive information for surgeons.
SNMM is one type of unilateral malignant nasal tumor. No bilateral SNMMs have been reported. Similar to SNMM, an SIP usually affects only unilateral sinonasal structures and may be neglected in patients with bilateral nasal tumors. SIP is a benign epithelial-derived tumor that accounts for 0.5% to 4.0% of all nasal tumors. 14 It has a predilection to reoccur, and 5% to 15% of SIPs develop into squamous cell carcinoma. 14 Therefore, it is very important to correctly diagnose an SIP as early as possible to prevent its malignant transformation.
CT can show the location and extent of a nasal tumor and soft tissue opacification with or without the erosion of surrounding bone. MRI is more specific than CT. The typical signal intensity of SNMM is defined as a high signal on T1WI and a low signal on T2WI because the pigment in the tumor is paramagnetic and bleeding often occurs. 12 However, the MRI features of SNMM can vary due to differing amounts of melanin, and up to 33% of cases are amelanotic; only 41.6% of patients have typical MRI findings. 15 A mass with a convoluted cerebriform pattern (CCP) on T2- or enhanced T1-weighted images suggests SIP15,16; however, the MRI features of the current case were untypical. It can be challenging to properly analyze the morphological features on MRI images and use them to distinguish SNMM and SIP because the features are nonspecific and overlapping. A diagnosis based on imaging can be misleading, and a bilateral tumor must undergo a pathological examination to aid in its diagnosis. Imaging examinations performed during the patient’s first visit showed no invasion into adjacent tissue or bone; as a result, there was a high risk for preoperative misdiagnosis.
In the present patient, a nasal endoscopy examination showed that the tumor in the right nasal cavity was polyp-like with a pink surface, while the tumor in the left nasal cavity had a gray-black surface; however, nasal melanomas can exhibit polyp-like, nodular, or cauliflower-like morphologies. Furthermore, based on the amount of melanin present, the tumor’s surface can exhibit different colors.4,6,17 Moreover, some bilateral nasal polyps can appear as pink or pale gray growths and have different surface morphologies or colors. 18 Therefore, the presence of bilateral nasal neoplasms reduces the possibility of uncommon tumors, but makes it difficult to distinguish among a nasal melanoma, SIP, and nasal polyp. Pathological examination is the gold standard for diagnosing a tumor, whether benign or malignant, and can effectively guide future treatment. While nasal malignant melanoma is characterized by the presence of typical melanosomes in tumor cells, the number of melanosomes varies dramatically during different stages of the tumor’s development. Furthermore, because melanosomes are also found in other tumors including rhabdomyosarcoma, olfactory neuroblastoma, sinonasal undifferentiated carcinoma, and lymphoma,19,20 a subsequent definitive examination by immunohistochemistry is required. While S-100 protein is the standard marker for making an immunohistochemical diagnosis of malignant melanoma, Melan A has a sensitivity similar to that of S-100 protein for detecting malignant melanoma. In contrast, HMB45 has better specificity but lower sensitivity than S-100 protein for identifying malignant melanoma. Furthermore, Ki67 antigen is a diagnostic and prognostic marker for melanoma. Patients with <35% Ki67 staining have a better survival rate than patients with >35% staining21,22; however, Ki-67 overexpression is associated with SIP recurrence and malignant transformation.22 -24 In summary, the combined use of staining for S-100, Melan A, HMB 45, and Ki-67 can effectively assist in diagnosing SNMM.1,6 The commonly seen benign nasal tumor SIP is much easier to diagnose, as it is characterized by inverted growth of the hyperplastic epithelium into the underlying stroma. 25
Surgical excision is empirically recommended as first-line treatment for uncommon malignant carcinomas such as SNMM, and especially for tumors that tend to be locally aggressive, recurrent, and potentially metastatic. 26 In this case, radical surgical excision was necessary, with the negative surgical margins being confirmed during surgery to improve the patient’s quality of life. 27 Although SIP has a tendency to reoccur after surgery, its complete excision is a relatively easy procedure for an experienced surgeon. In contrast, treatment for SNMM is rather difficult, as the site of the primary tumor is often difficult to determine due to extension of the lesion at the time of diagnosis. Furthermore, clean margins may be difficult to achieve due to anatomic constraints and functional considerations. 1 The early diagnosis and treatment of SNMM are pivotal factors affecting its prognosis.5 -7 Given those circumstances, increasing evidence supports the use of postoperative radiation therapy for SNMM; however, the optimal radiation dose and fractionation regimen remain undefined.28 -31 Although there is no good evidence indicating that radiotherapy can prolong or improve patient survival, it does reduce local recurrence.28 -31 Conventional chemotherapeutic agents do not seem to have a good effect on regional and distant metastases. 31 The patient described in this report refused recommended radiotherapy and chemotherapy after undergoing surgeries, and that might be one reason for the rapid progression of SNMM in the patient. While new therapeutic techniques such as biological drugs, immunological treatments, and cellular agents are emerging, no clinical evidence gathered during their early development stage indicates that those treatments prolong life expectancy.32,33
This patient suffered a rapid progression of SNMM. Huang et al 34 reported that the mean time from surgery for SNMM to its local recurrence was 5 months. Our patient had recurrent SNMM that extensively invaded the nasopharynx at 7 months after surgery. It has been reported that melanomas occurring in the sinuses have the worst prognosis, while those occurring in the nasal cavity have a better prognosis.5,35,36 The patient described here had multiple metastases in multiple tissues and organs at the time of his second consultation. As a result, he had a poor prognosis, even after treatment with radical surgery and postoperative radiotherapy. While early diagnosis and treatment are necessary, the coexisting SIP in the contralateral nasal cavity of this patient made the diagnosis of SNMM much more difficult, because this type of uncommon coexisting tumors is rarely seen, even by experienced surgeons.
In summary, physicians should be alert for different types of bilateral nasal tumors, and especially when a patient’s morphological and imaging features are atypical. In such cases, a pathological examination should be used to make a final diagnosis. Although the prognosis for SNMM is extremely poor, patients should receive comprehensive multidisciplinary postoperative treatment and timely follow-up based on changes in their condition.
Footnotes
Correction (September 2023):
Affiliation 1 and corresponding author address have been updated in the article.
Authors’ Note
Further information and requests for resources should be directed to Xicheng Song (
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Key Research and Development Program of Shandong Province (2022CXPT023) and The Taishan Scholars Project, Grant/Award Number: ts20190991.
