Abstract
Introduction
Inverted papillomas of the middle ear are extremely rare tumors that carry an increased risk of recurrence and malignant transformation. There are currently 59 cases of middle ear inverted papillomas reported in the literature. The objective in this study was to systematically evaluate outcomes regarding middle ear inverted papillomas with respect to demographics, anatomical tumor sites, malignant transformation status, recurrence rate and HPV status.
Study Design
Retrospective case series and systematic review.
Methods
A systematic review was completed on June 25, 2020 with a search strategy including PubMed, Embase, Scopus and Google Scholar. This revealed 181 articles. Full-text review was completed, and 66 articles were included. 115 articles were eliminated due to duplication of articles from databases, article titles not applicable to the aims of the systematic review and articles describing inverted papilloma of body sites other than middle ear.
Discussion
Thirty-one cases of primary inverted papillomas of the middle ear were found in the literature with an additional 26 cases of secondary tumors. Four case reports did not specify primary versus secondary. The malignant transformation rate was 34.4% with a 53.6% recurrence rate. Treatment of middle ear inverted papillomas is primarily surgical with adjuvant radiation therapy considered for patients with recurrence or malignant transformation. Frequent clinical follow up of these patients is critical due to the increased rate of recurrence and malignant transformation.
Conclusion
Inverted papillomas of the middle ear are rare tumors that carry a high risk of recurrence and malignant transformation necessitating complete resection and frequent clinical follow up.
Introduction
Primary and secondary inverted papillomas (IPs) of the middle ear are extremely rare tumors, with only 56 cases in the current literature. Previous literature has recommended a multi-modality treatment for middle ear IPs to include surgical excision with adjuvant radiotherapy for recurrent disease or malignant transformation. However, due to the rarity of these lesions, universal recommendations are lacking. In addition, multifocal disease can be difficult to completely eradicate surgically and may require imaging, in addition to endoscopy, for long-term surveillance. The sparse information on middle ear IPs prompted the authors to document all known cases in the literature in an effort to better understand and treat this rare disease.
The objective in this study was to systematically evaluate outcomes regarding middle ear inverted papillomas with respect to demographics, anatomical tumor sites, malignant transformation status, recurrence rate and HPV status. The information obtained in this study will help us further understand this rare disease and help clinicians to accurately diagnose, treat and monitor these patients for recurrence and malignant transformation. We also report on two new cases of IPs involving the middle ear (representing case #60 and #61).
Methods
A systematic review was completed on June 25, 2020 with a search strategy including the following terms (MeSH strategy Supplemental 1). Articles were screened based upon abstract to ensure correct allocation. This revealed 181 articles. Full-text review was completed and ultimately 66 articles were included. Overall, 115 articles were eliminated due to duplication of articles from databases, article titles not applicable to aim of systematic review and articles describing inverted papillomas of body sites other than middle ear (see PRISMA flowchart). Reported data included first author, year, age, sex, sites involved, primary versus secondary tumor, laterality, malignant transformation, recurrence, and HPV status were collected. Time to recurrence and time disease-free were also reported for recurrent tumors and collected. All case reports were published in English or translated into English. Statistical data analysis of all known cases of IP of the middle ear in the current literature was analyzed with SPSS (v.25, Armonk, NY). Categorical variables were assessed with chi-square. Continuous data was evaluated with t-tests. Continuous data was represented as median (25%, 75%) when the data was not normally distributed and was reported as mean ± std dev when the data was normally distributed.
Consent was obtained from both patients to publish their clinical data.
Results
Case #1
A 49-year-old Caucasian male with a history of a prior sinonasal IP of the left maxillary sinus presented for evaluation in clinic. He had undergone prior resection of his IP 6 years ago at an outside hospital with no evidence of dysplasia or malignant transformation on pathology. Since that time, he began to have otalgia, aural fullness, and hearing loss in the right ear. Audiometry showed SNHL of the right ear decreased to 5–15 dB (Figure 1) and unchanged mixed hearing loss in the left ear. Otoscopic examination showed an intact right tympanic membrane with a mass medial to it. The external ear canal was normal, without inflammation or masses. CT scan showed a stable soft tissue density in the right middle ear cavity approximating the tympanic membrane and mesotympanum. The right mastoid was well aerated, and no obvious bony erosion was noted. He underwent a right canal wall up tympanomastoidectomy in February 2020. Intraoperatively, a mass was found posterior to the right tympanic membrane occupying the mesotympanum along with polypoid material densely filling the middle ear. Dense polypoid material was removed from the protympanum, mesotympanum, epitympanum and medial tympanic membrane. Pathology was consistent with an inflamed inverted papilloma with mild to moderate dysplasia. The patient had an unremarkable post-operative course and was discharged on ciprofloxacin ear drops. Three months post-operatively the patient returned to clinic with complaints of otorrhea and otalgia in his right ear. Physical examination revealed recurrence of a small papilloma along his right tympanic membrane. The patient has not returned for further follow-up since that time.
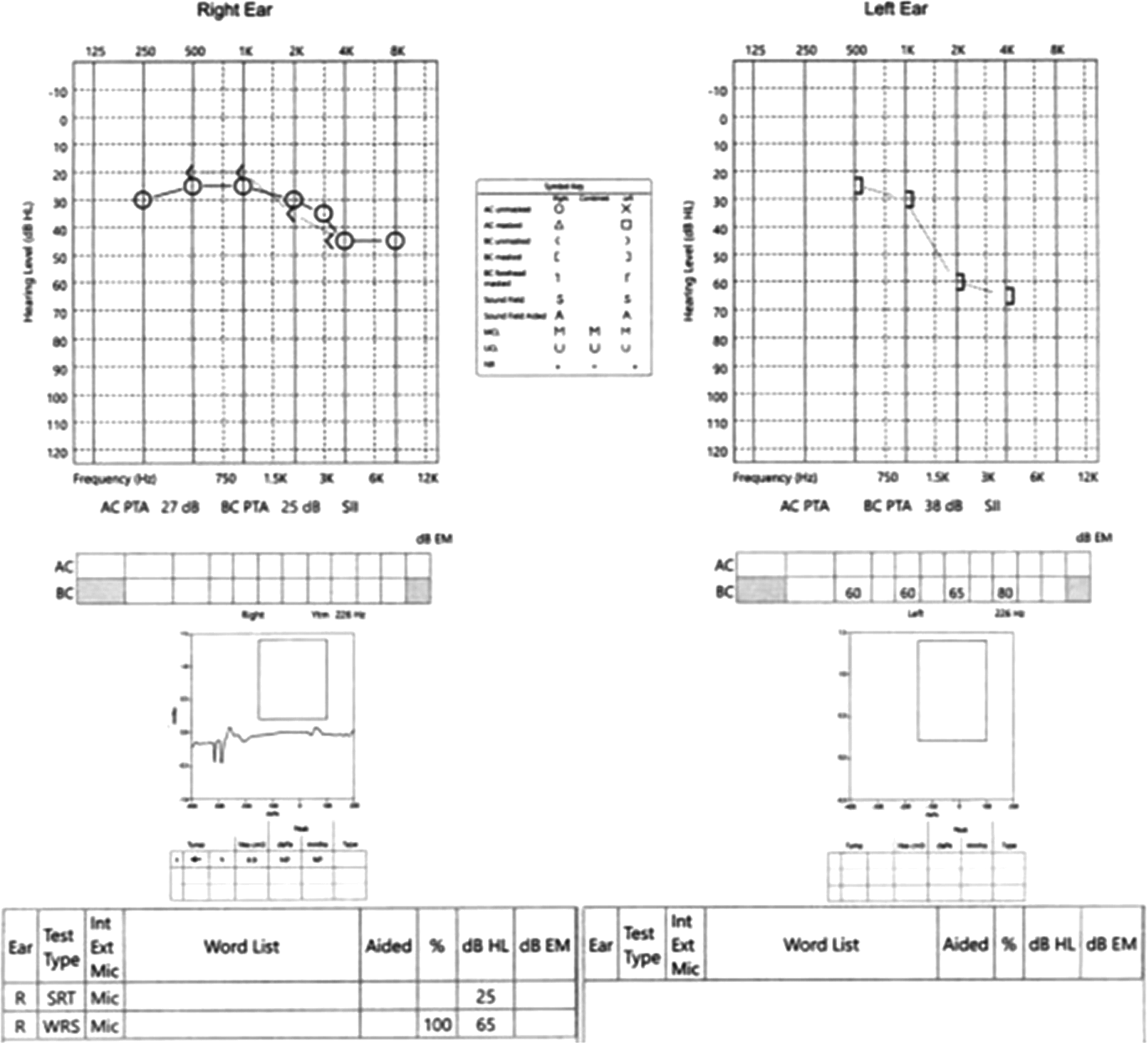
Audiogram showing sensorineural hearing loss of the right ear decreased to 5-15 dB.
Case #2
A 70-year-old African American male with a history of nasal polyposis, multiple dysplastic sinonasal IPs, field cancerization of the nasal cavity and prior renal transplant due to ESRD presented with new onset right sided aural fullness, otorrhea, and headaches. One year prior to presentation, the patient underwent surgical resection of a right maxillary sinus IP that filled the nasal cavity and extended to the nasopharynx. At that time, pathology showed an IP with high-grade dysplasia. He then underwent adjuvant radiotherapy (60 Gy in 30 fractions). On audiometry, the patient had bilateral mixed hearing loss that was unchanged from prior exams (Figure 2). He denied otalgia or nasal congestion. Nasal endoscopy showed a papilloma in the right nasopharynx and fossa of Rosenmuller obstructing the eustachian tube. There was no evidence of tumor recurrence in the right maxillary sinus or orbit wall. The patient then underwent resection of the nasopharyngeal tumor. Pathology again demonstrated squamous papilloma with high-grade dysplasia. Two months post-operatively, the patient returned to clinic complaining of increased otalgia and conductive hearing loss in his right ear with a new facial nerve weakness (House-Brackman 2). CT scan showed soft tissue densities in the right middle ear and mastoid cavity with complete occlusion of the right external ear canal (EAC) but without bony erosion (Figure 3). The patient underwent right canal wall down mastoidectomy with resection of tumor and blind sac closure of EAC. Pathology demonstrated moderately differentiated squamous cell carcinoma (SCC) with papillary features. The tumor histology showed similarities to the IP resected 2 months prior, suggesting the tumor was a direct extension of the nasopharyngeal IP. Four months following XRT completion, the patient was found to have recurrence of the tumor in his left middle ear extending to his EAC. He then underwent left subtotal temporal bone resection with closure of the left EAC. Pathology again showed an IP with high-grade dysplasia with resemblance to the prior inverted papilloma. Patient has since had recurrence of IP in left maxillary sinus treated by modified medial maxillectomy. He was noted in late 2020 to have a new nasopharyngeal lesion as well as concern for an infraglottic lesion. Biopsy confirmed recurrence of SCC in the nasopharynx and infraglottis. He underwent SBRT (35Gy in 5 fractions) to the infraglottis with a good response to therapy. However, his PET/CT showed evidence of metastasis to the lungs with continued progression and a new tracheal mass. The tracheal mass was biopsied 8/2021 and was noted to be SCC. He is now under palliative care. There has been no recurrence of SCC within the ear.

Audiogram showing bilateral mixed hearing loss.
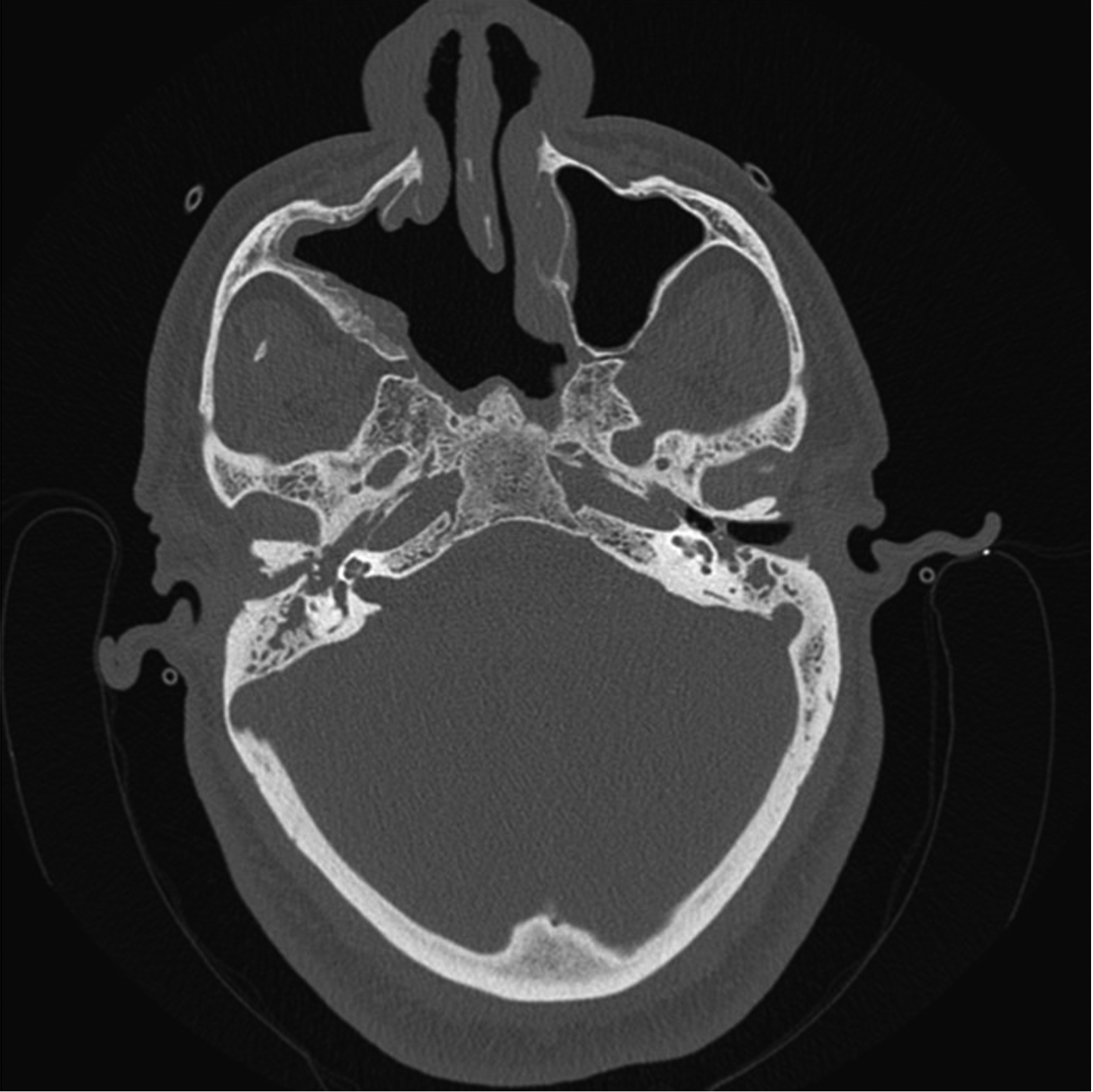
Non-contrasted computed tomography showing soft tissue densities in the right middle ear and mastoid cavity with complete occlusion of the right external ear canal.
Known Cases of Middle Ear IP in the Literature
Thirty-one cases of primary inverted papillomas of the middle ear have been reported in the last 35 years (Table 1). An additional twenty-six cases of secondary tumors have also been reported. Four case reports did not specify primary versus secondary. The mean age of all patients with IPs involving middle ear (primary and secondary) was 52.41 years ± 16.67. Right-sided disease was found in 37.7% (23/61) cases of middle ear IPs with left-sided disease representing 32.8% (20/61) (Table 2). Three cases were bilateral, including Case #2 of this study, while the remaining 14 cases did not report laterality (Table 3). Our study found that the recurrence rate was 53.6% (30/56) with a malignant transformation rate of 34.4% (21/61), which is in line with previous reported numbers (Table 4 & 5). Of the 61 cases of middle ear IPs (primary and secondary), only 20 reported HPV status (Table 1). Of the 20 cases, only 35% (n = 7) indicated that the patient was positive for HPV infection. Table 1 below summarizes the data from all known cases of inverted papillomas of the middle ear.
Cases of Inverted Papilloma of the Middle Ear.

ME = middle ear; MA = mastoid cavity; EAC = external ear canal; TF = temporal fossa; ET = Eustachian Tube; NS = not stated.
**Non-primary tumors included sinonasal tumors recurring in ME or sinonasal tumors extending to ME.
Patient Demographics.

Disease characteristics of known cases.

Recurrence rates of known cases.

Malignant transformation rates of known cases.

Recurrence rates of known primary tumors.

Discussion
Inverted papillomas are benign tumors with malignant potential that are most commonly found within the sinonasal cavity. Malignant transformation rates of sinonasal IPs are reported as ∼10%, with an approximately 15% recurrence rate [1–5]. The sinonasal cavities are lined with ciliated columnar epithelium with goblet cells known as Schneiderian epithelium. Therefore, tumors arising from this epithelium are referred to as Schneiderian papillomas. There are 3 histological types of Schneiderian papillomas: oncocytic, endophytic and inverted papillomas. IPs are the most common type of Schneiderian papilloma, composed of a fibrovascular core surrounded by squamous, transitional or respiratory epithelium [5].
While the sinonasal cavity is composed of Schneiderian epithelium, the middle ear is not. The etiology of middle ear IPs is not well understood, but secondary spread to the middle ear is thought to occur via the eustachian tube connection to the nasopharynx [6]. Thus, while the origin of primary IPs of the middle ear is unclear, three hypotheses have been proposed. The first hypothesis suggests sinonasal IP cells migrate to the middle ear via the eustachian tube. This is supported by the significant percentage of patients who had IPs of the ipsilateral sinonasal cavities (44%) [7]. The second suggests abnormal migration of embryonic Schneiderian epithelial cells into the mucosa of the middle ear with subsequent proliferation. The third hypothesis theorizes that chronic otitis media may stimulate the development of Schneiderian mucosa in the middle ear. The third hypothesis is supported by the increased incidence of chronic otitis media in patients with IPs of the middle ear [8]. However, chronic otitis media may also be due to local inflammation secondary to the IP or obstruction of the eustachian tube. More detailed case reports of patients with middle ear IPs will help shed light on the exact etiology of this disease process.
HPV status
Unlike sinonasal IPs, the association of HPV with IPs of the middle ear is unclear. The rate of HPV co-infection in patients with middle ear IPs is 14–35% [7, 9]. Coca-Pelaz et al, postulates that the HPV infection rate can vary even more, from 0–85% [6]. Many studies do not report the HPV status of patients. Of the 58 cases of middle ear IPs (primary and secondary), only 20 reported HPV status (Table 1). Of the 20 cases, only 35% (n = 7) indicated that the patient was positive for HPV infection. Alghamdi et al suggest that HPV infection should play a role in the development of middle ear IPs due to the relationship of HPV to sinonasal IPs [9]. Thus, vaccination against HPV may be protective in patients prone to developing recurrent sinonasal or middle ear IPs.
Presenting Symptoms
The most common presentation of IPs of the middle ear is hearing loss (78%), but other symptoms include otorrhea (39%), otalgia (30%), aural fullness (17%), facial nerve palsy (4%) and tinnitus (4%) [5, 10]. In the case of our patients, Case #1 presented with otalgia, aural fullness and SNHL. Case #2 presented to clinic multiple times with symptoms including hearing loss, otalgia, aural fullness, otorrhea, headache and facial nerve palsy. CT scans, along with otoscopic examination, are commonly used to diagnose IPs of the middle ear due to their ability to accurately visualize the middle ear [8]. MRI scans are frequently used to confirm the diagnosis.
Malignant Transformation and Recurrence
Despite the unclear origin of middle ear IPs, they are known to have higher rates of recurrence with increased risk of malignant transformation. IPs of the middle ear have been reported to have a 57–67% recurrence rate and a 35–41% rate of malignant transformation [5–7]. recurrence rate was 53.6% (30/56) (Table 6) with a malignant transformation rate of 34.4% (21/61) which is in line with previous reported numbers. This high rate of recurrence is hypothesized to be due, at least in part, to the intricate anatomy of the middle ear limiting complete resection of the tumor. Coca Pelaz et al postulates the recurrence rate can be decreased from 67% to around 10–15% with wider and more aggressive surgical resection[6]. Coca-Pelaz et al, Dingle et al, and Penaranda et al, report rare cases of middle ear IPs with intracranial invasion. The high rate of recurrence and malignant transformation coupled with difficult to treat locations highlight the need for aggressive treatment and long-term follow up to monitor for recurrence. Recurrent or residual disease pose serious risks for malignant transformation.
Management
Although no treatment algorithm exists, primary treatment of middle ear IPs is surgical resection [10]. Wide excision is necessary to prevent recurrence and the literature has reported tympanomastoidectomy to temporal bone resections and petrosectomy for management of these tumors [10, 11]. Recurrence rates can be as low as 10–15% with aggressive, wide surgical excision [10, 11]. However, complete resection is often difficult, even with experienced surgeons. Radiation therapy may be indicated for tumors that recur, have malignant transformation, or if unable to be completely resected. Routine long-term follow-up is necessary given the risk of recurrence and malignant transformation. Imaging modalties, like CT and MRI, are utilized in surveillance due to anatomic limitations that limit clinical exam [8, 12].
Conclusion
Primary and secondary inverted papillomas of the middle ear are uncommon, but have a higher rate of recurrence and malignant transformation when compared to their sinonasal counterparts. Management of these benign tumors is with aggressive, initial surgical intervention, while radiation therapy is reserved for those with unresectable, recurrent, and/or malignant disease. Patient education regarding close clinical follow up is integral to surveillance of these tumors.
MeSH search strategy:
PubMed = 39 results
("Papilloma, Inverted"[Mesh] OR Inverted Papilloma*[tiab] OR Schneiderian[tiab]) AND (“Ear, Middle”[Mesh] OR Middle ear*[tiab] OR Tympanic Cavit*[tiab] OR Typanum*[tiab])
All 39 results used in systematic review
Embase = 97 results, 30 of which are not found in PubMed. After running search, see “Sources” in the menus to the left of results. Click on the Embase portion of the diagram to limit to only those results.
12 of 30 results used. Eighteen results excluded due to article titles not applicable to aim of systematic review and articles describing inverted papillomas of body sites other than middle ear.
Scopus = 105 results
((((middle AND ear ) AND ( inverted OR schneiderian ) AND papilloma ) ) )
8 of 105 results used. 97 results excluded due to repetitive articles from PubMed and Embase search and article titles not applicable to the aim of the systematic review.
Google Scholar = 7 results
Supplemental Material
sj-docx-1-aar-10.1177_21526575221130711 - Supplemental material for Inverted Papilloma of the Middle Ear: Two New Cases and Systematic Review
Supplemental material, sj-docx-1-aar-10.1177_21526575221130711 for Inverted Papilloma of the Middle Ear: Two New Cases and Systematic Review by Peter L. Miller, Erika Walsh, Do-Yeon Cho, Bradford A. Woodworth and Jessica W. Grayson in Allergy & Rhinology
Footnotes
Ethics approval and consent to participate
Both patients consented to participation and IRB approval was obtained.
Consent for Publication
Both patients consented to publication.
Author Contribution(s)
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest Statement
Brad A. Woodworth is a consultant for Medtronic, Cook Medical, and Smith and Nephew.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
