Abstract
Introduction
Glomangiopericytoma (GPC) is a rare neoplasm, unique to the sinonasal tract, representing less than 0.5% of all sinonasal tumors. 1,2 Initially described as hemangiopericytoma in 1942 by Stout and Murray and redefined as glomangiopericytoma by Granter in 1998, GPC is a pericytic neoplasm with features similar to hemangiopericytoma/solitary fibrous tumor and glomus tumor and which was ultimately reclassified as its own unique entity in 2005. 3,4 Sinonasal GPCs differentiate from capillary pericytes, the smooth muscle cells surrounding small vasculature. 3 -5 These tumors have low malignant potential, and act less aggressively than their hemangiopericytoma/solitary fibrous tumor counterparts. 6,7 Etiology remains unknown, but possible increased vascularity from trauma, pregnancy, hypertension, or corticosteroid use has been postulated. 1,2
Presenting symptoms typically include unilateral epistaxis, nasal obstruction, and headache. 1 Initial evaluation includes computerized tomography (CT) or magnetic resonance imaging (MRI) and nasal endoscopy. Definitive diagnosis is made upon histologic evaluation of resected tissue. 1,6 The gold standard for treatment is complete endoscopic surgical resection. 1 -4,6 -8 Recurrence rates are low, and usually occur within 5 years of definitive management. 1,9 Recurrence is thought to result from incomplete surgical resection. 1
Accounts of sinonasal GPC in the literature remain sparse, and further reports help to strengthen the knowledge base of this rare tumor. This review of 6 patients, to the best of our knowledge, represents the largest single institution series to date.
Methods
Retrospective chart review of patients with an ICD (International Classification of Diseases) diagnosis code for sinonasal GPC was performed. Demographic data including gender, race/ethnicity, age, past medical history, and presenting symptoms were extracted from the electronic medical records. Additionally, pre-operative and any post-operative imaging, office endoscopy notes and operative reports as well as pathology and immunohistochemistry reports were reviewed when available. All ancillary testing was performed as part of routine pathologic evaluation.
Results
Six patients with sinonasal GPC were identified from 2009 through 2021. All patients had imaging reports and operative data available. There was an additional (7th) patient who only underwent pathology review through our institution and, therefore, was not included in this series.
Demographics
The mean and median age of diagnosis was 58.7 and 58.5 years respectfully. There was a male predominance with 83% male (n = 5) and 17% female (n = 1). All patients reported Caucasian race and non-Hispanic ethnicity. The most common comorbidities included type 2 diabetes mellitus and hypertension (n = 4/6, 67%). Other specific comorbidities are reported in Table 1, which provides a summary of our cohort.
Demographic Data of Patients With Sinonasal Glomangiopericytomas.
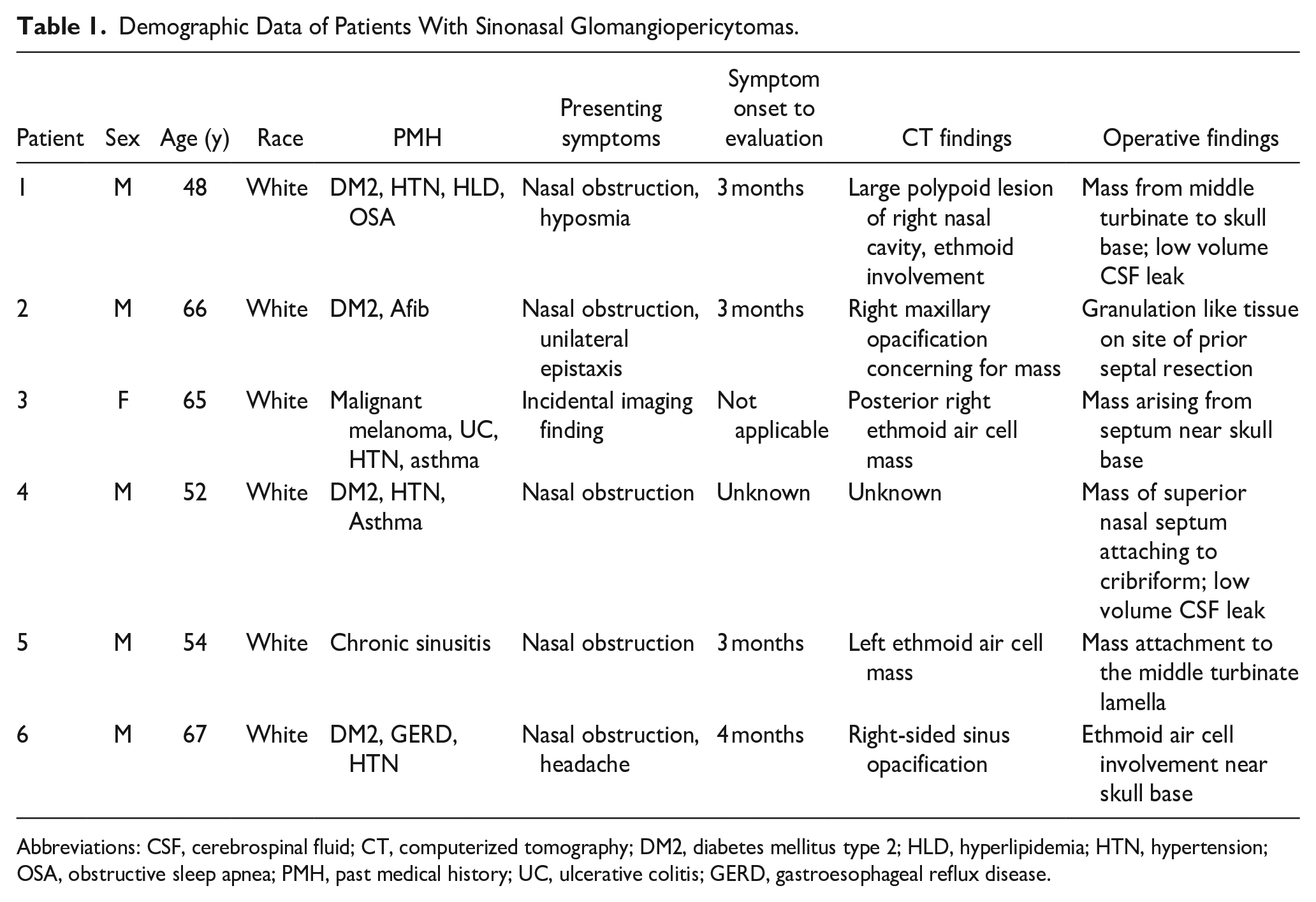
Abbreviations: CSF, cerebrospinal fluid; CT, computerized tomography; DM2, diabetes mellitus type 2; HLD, hyperlipidemia; HTN, hypertension; OSA, obstructive sleep apnea; PMH, past medical history; UC, ulcerative colitis; GERD, gastroesophageal reflux disease.
Initial Presentation
Presenting symptoms and initial work up were available for all patients. Eighty-three percent (n = 5/6) of patients reported unilateral nasal obstruction as the reason for evaluation. The remaining patient’s lesion was identified on imaging obtained as part of a clinical trial protocol for malignant melanoma. Other reported symptoms included epistaxis and headache (Table 1). Symptoms were present for an average of 3 months before evaluation. Eighty-three percent (n = 5/6) of patients had preoperative CT imaging available in our system, and 50% (n = 3/6) underwent subsequent MRI prior to treatment. All tumors were found to be soft tissue density on CT, and hyperintense on T1 weighted post contrast imaging (Figure 1).
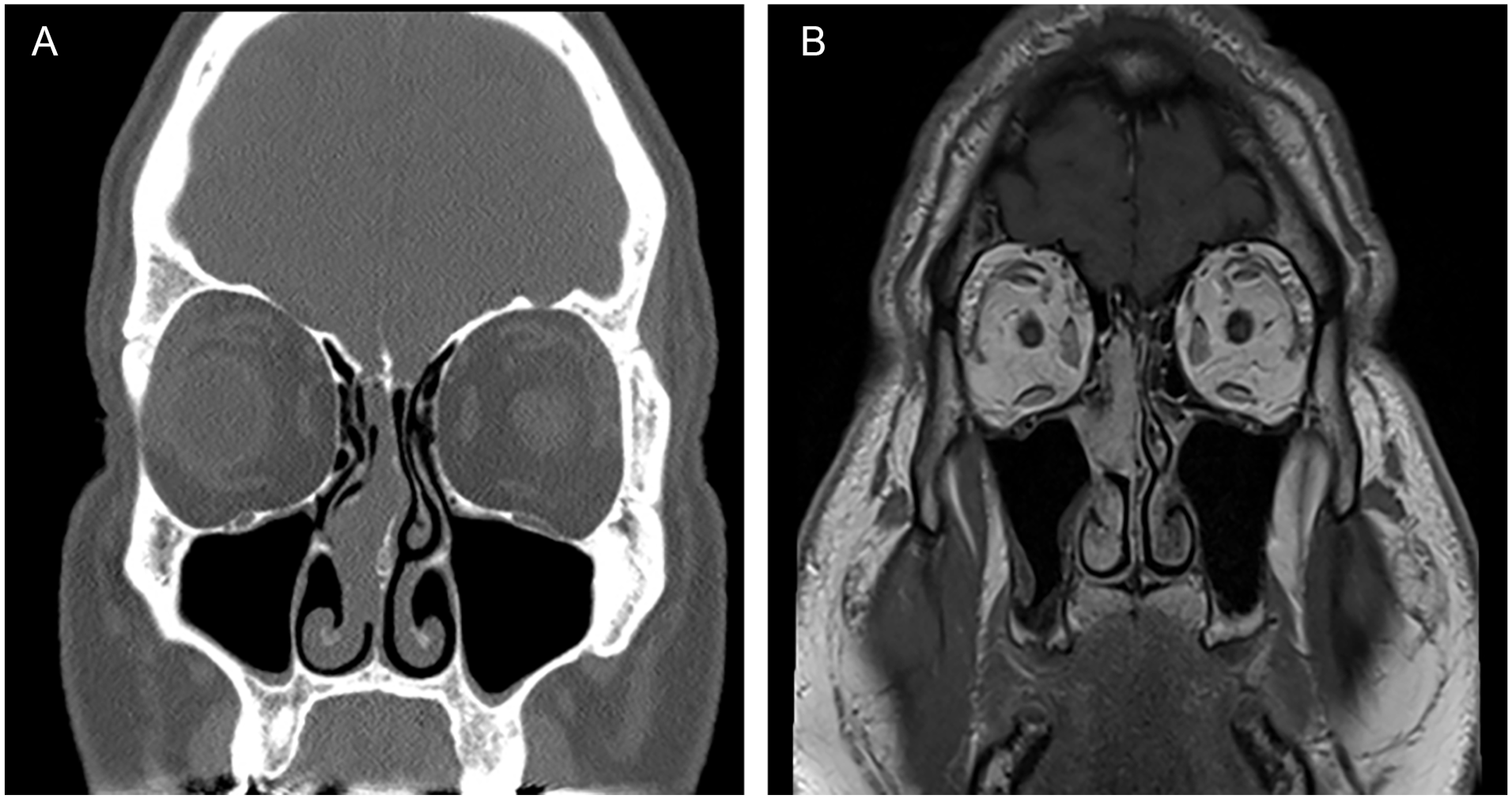
Representative pre-operative imaging. (A) CT with central soft tissue density mass. (B) T1 post-contrast MRI with central hyperintense mass. CT, computerized tomography; MRI, magnetic resonance imaging.
Management
All 6 of the patients treated at our institution underwent endoscopic surgical excision of the mass. Half (n = 3/6) of patients had undergone initial biopsy or endoscopic excision of the lesion and were referred for concern for residual tumor or inadequate resection. Operative reports and in-office endoscopy notes described a polypoid erythematous or violaceous mass in the nasal cavity clearly different from typical nasal polyposis (Figure 2). All tumors were noted to extend/have attachments to the skull base near the cribriform plate. One operative report noted clear elevation of the tumor off the underlying bone. No other reports commented on sub-mucosal extension, but bone was included in surgical margins. One third (n = 2/6) of patients underwent nasoseptal flap creation for preemptive cerebrospinal fluid (CSF) leak repair intra-operatively. However, no patient had post-operative CSF leak concerns. All tissue appearing to be involved with the tumor was able to be resected in all patients.
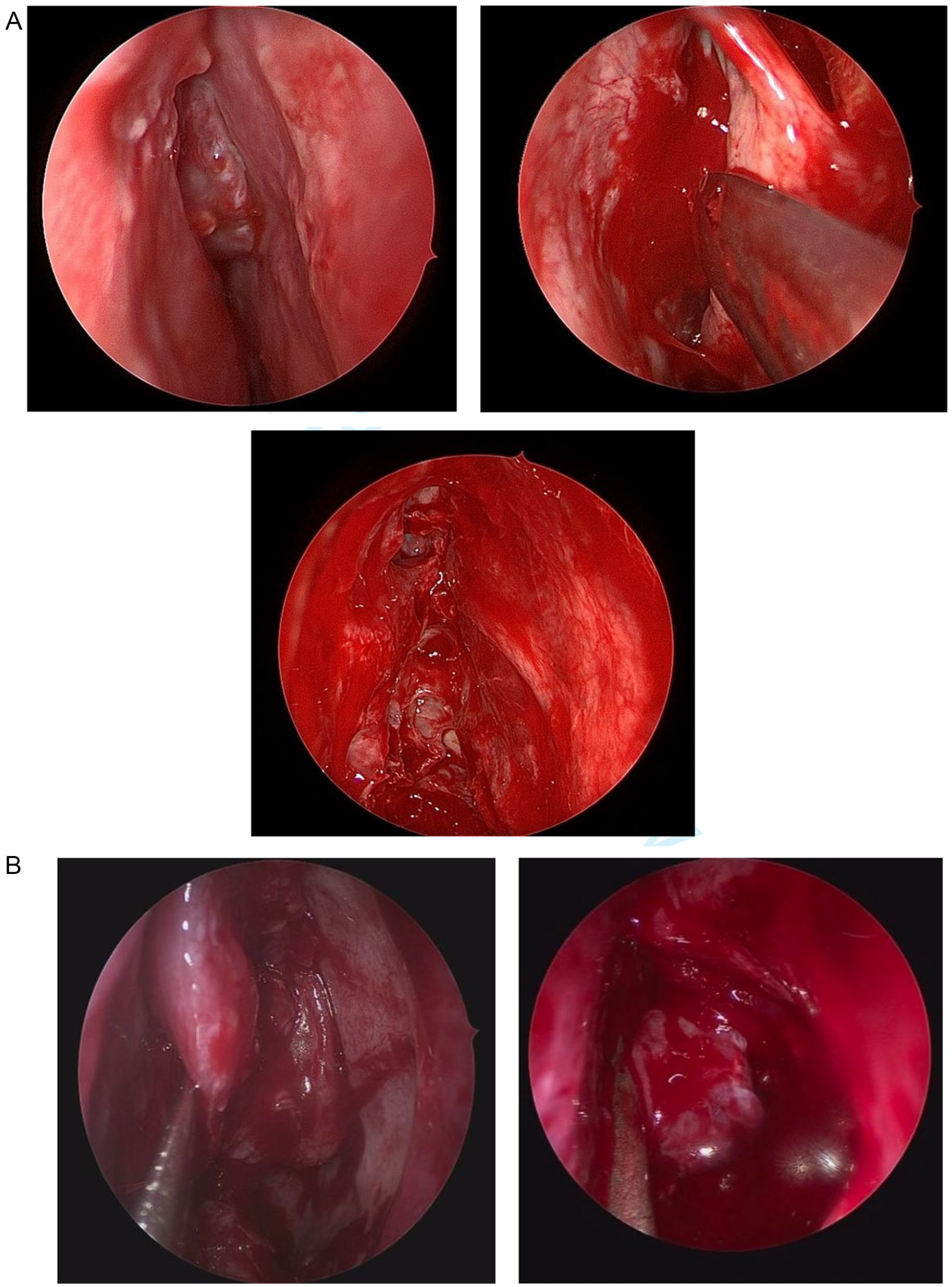
Representative intraoperative endoscopic images. Polyploid masses abutting the nasal septum (A) and superior turbinate (B).
Diagnosis
All resected masses exhibited similar histology, with cellular proliferations of neoplastic oval to spindled cells arranged vague fascicles. Vessels exhibited frequent staghorn forms and extravasated blood and perivascular hyalinization were frequently seen. Cytologic atypia was minimal, and mitoses were very rare. Immunohistochemical (IHC) testing for smooth muscle actin was documented on 5 of the 6 specimens and was positive in all. The remaining pathologic report listed diagnosis as “Sino-nasal GPC” without describing specific tests conducted. Beta-catenin was performed on 2 specimens, exhibiting nuclear positivity. Additional IHC was performed on several specimens and all were negative for cytokeratins, desmin, SOX10, STAT6, and S100. These histopathologic and immunophenotypic findings confirmed the diagnosis of sinonasal GPC for each patient (Figure 3).
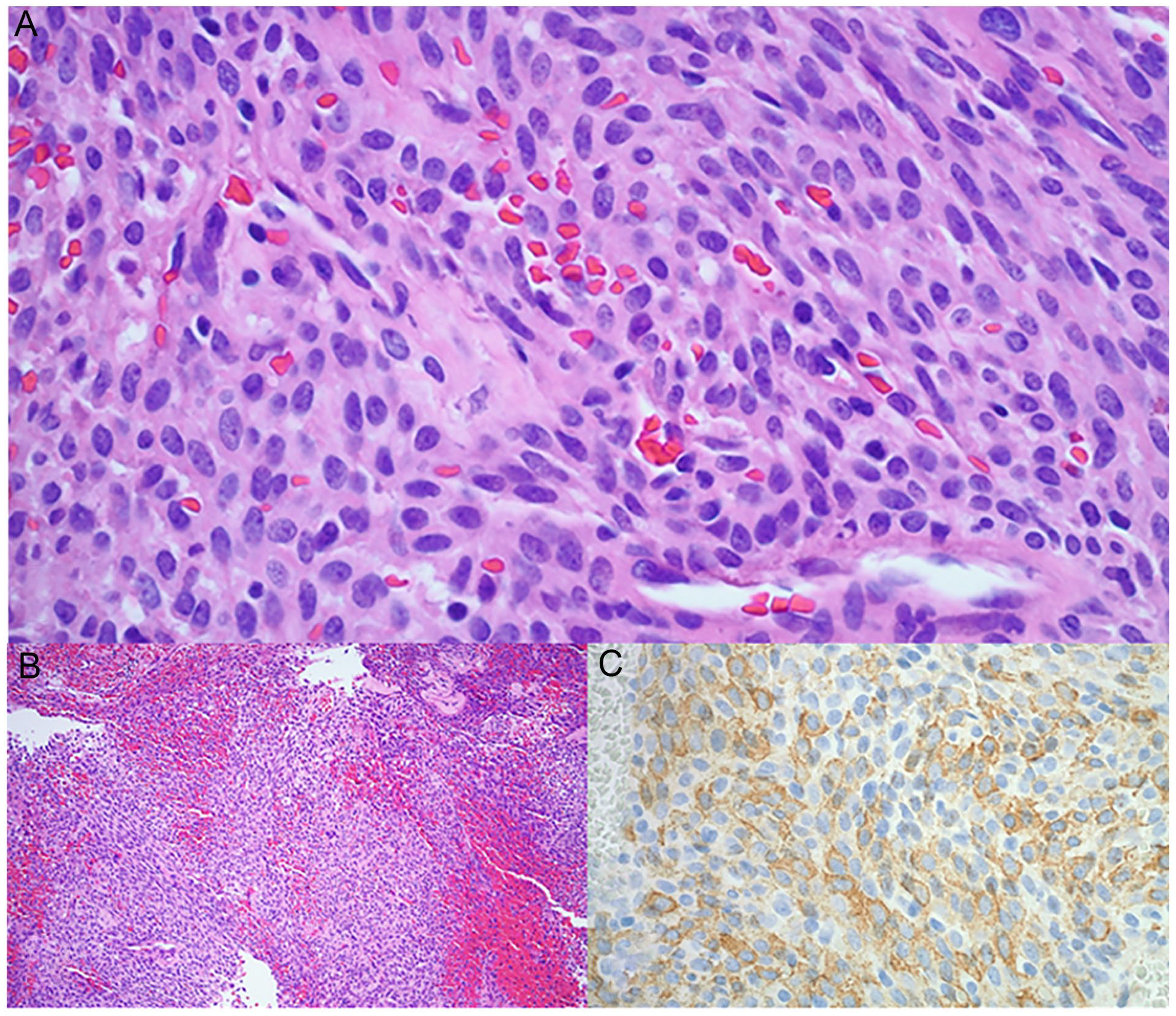
A high-power H&E photomicrograph (A, 40×) shows spindled cells in short fascicles surrounding vessels. A low-power H&E view (B, 10×) highlights the vascular pattern of the neoplasm. SMA is diffusely positive (C). H&E, hematoxylin and eosin; SMA, smooth muscle actin.
Follow-Up
Eighty-three percent (n = 5/6) of patients who underwent resection within our institution were seen back in clinic for follow-up. In-office nasal endoscopy was performed on all patients without obvious disease recurrence. Only 33% (n = 2/6) of the patients had subsequent imaging (MRI in both) which demonstrated no disease recurrence. Patients were followed up for variable periods of time, ranging from 11-month post-operative to 10 years with annual visits. There has been no evidence of disease recurrence to date in any of the study patients.
Discussion
Our series details the largest single institution review of sinonasal GPCs reported in the literature. We detail the demographics, work up, management, and diagnosis of this tumor over the past decade without significant changes within our cohort over this period.
The average age of diagnosis at 58.6 years agrees with prior reports in the literature of usual presentation in 6th or 7th decade of life. 1,5,9 While sinonasal GPCs have been reported with slight female predominance, our series was predominantly male. 9 However, most of our patients did have hypertension, a commonly cited comorbidity thought to promote vascularity. 1 The primary presenting symptom of nasal obstruction in our patients aligns with Thompson’s 2003 review and other reports in the literature with most common symptomology being obstruction and epistaxis. 9
All patients treated within our system had preoperative CT imaging which consistently demonstrated non-specific findings of mass-like lesion of the para-nasal sinuses extending up toward the skull base. Operative reports, and in-office endoscopy notes that described the mass documented erythematous or violaceous polypoid tissue. These descriptive features in our series have been consistently documented in the literature as classic GPC presentation. 10
The gold standard for treatment of sinonasal GPC is complete surgical excision. Our series reflects this, with endoscopic resection of all involved tissue and without evidence of disease upon available follow-up examinations and imaging. Additionally, we demonstrate re-resection of residual tumor as appropriate management for tumors with inadequate original resection. Two of our patients underwent re-resection with adequate margins, and had no disease recurrence upon available follow-up.
The follow-up for our series was varied. All patients except 1 were evaluated for at least 1 post-operative appointment. Our longest follow-up was 10 years, with yearly visits. There is no standardization of follow-up for GPC disease recurrence to our knowledge. Enzinger et al report a 17% risk of recurrence after excision, with greatest risk in the first 5 years post diagnosis. 11 Park et al report a similar recurrence risk in their recent systematic review, of 16% post-initial treatment. 12 They also describe certain IHC findings perhaps indicating more aggressive tumors, but no effect on disease-free survival. 12 While we cannot make recommendations based upon our institution’s experience, it is reasonable to consider scheduled follow-up for several years for patients even with adequate resection intra-operatively.
Conclusions
We present the largest known individual institution review of sinonasal GPC. Our findings reflect those of other cases in the literature. Additionally, the treatment for sinonasal GPC has not changed within our institution, or as reported in the literature, over the past decade. This low-malignant-potential tumor should be considered when imaging and endoscopy demonstrates a soft tissue polypoid vascular nasal mass, as treatment for cure is complete endoscopic resection. Future studies and reports may standardize post-operative follow-up and identify at-risk groups.
Footnotes
Authors’ Note
Abstract was accepted as poster presentation at the American Rhinologic Society Meeting, Philadelphia, PA, September 2022.
Author Contributions
Dr. Schauwecker was responsible for chart review, figure compilation, and manuscript writing. Dr. Perez provided pathology image and description. Dr. Chandra provided guidance for project ideas and importance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. For this type of study formal consent is not required.
