Abstract
Introduction
Glomangiopericytoma (GPC; sinonasal-type hemangiopericytoma) is a rare sinonasal tumor with a perivascular myoid phenotype. 1 GPC makes up less than 1% of sinonasal tumors, most of which start in the nasal cavity and spread to the paranasal sinuses; however, lone paranasal sinus involvement has been documented.2-4 People of all ages may be affected, with a peak prevalence in the seventh decade and a small female predominance.3,4 Nasal obstruction was the most prevalent symptom, followed by epistaxis, headache, facial pain or pressure, and anosmia. 5 GPC is a type of cellular spindle cell neoplasm with hemangiopericytoma-like vasculature and perivascular hyalinization of capillary-sized veins.4,6 Smooth muscle actin, muscle-specific actin, and vimentin are all positive in most cases.4,7,8 CD34 and S100 protein staining might be positive in a small percentage of GPC.4,6,8,9
GPCs can be mistaken for several spindle cell and vascular neoplasms that affect the sinonasal tract, the most frequent of which is a solitary fibrous tumor (SFT). 1 SFTs are rare neoplasms initially described in 1931 by Klemperer and Rabin. 10 Instead of mesothelial cells, SFTs are made up of mesenchymal cells. They typically originate in the serosa but they can also arise from other locations such as the urogenital system, mediastinal region, lungs, vulva, orbit, thyroid gland, nasopharyngeal area, larynx, and salivary glands. 11 SFT affects mostly adults (mean age: 52 years) with about equal sex predilection and can present clinically like any other obstructive nasal tumor with epistaxis and rhinorrhea 12 GPCs, like SFT, lobular capillary hemangioma (LCH), angiofibroma, and leiomyoma, should be distinguished from other heavily vascularized mesenchymal tumors. 13 However, challenges remain when differentiating GPCs from SFT.
Histopathologically, SFT exhibits areas of both hypocellularity and hypercellularity, similar to GPC. The vasculature in SFT includes staghorn-type and capillary-sized capillaries, resembling GPC. However, perivascular hyalinization is infrequent in SFT. Furthermore, cell distribution in SFT shows random overlap with uneven nuclear outlines, a contrast to GPC. Moreover, SFT displays strong staining for CD34, with less consistent actin staining. 12 Besides, Agaimy et al. observed that NAB2STAT6 translocation and nuclear STAT6 expression are highly specific and sensitive to SFT. 13 We report a case of sinonasal GPC with skull base involvement and unusually strong CD34 straining but negative STAT6 expression.
Case report
A 38-year-old male patient presented with a 3-year history of right-sided recurrent epistaxis. During this period, he also experienced occasional headaches and right-sided nasal obstruction. He denied hyposmia, drainage, visual changes, and facial numbness; and apart from smoking, he was otherwise healthy. On physical examination, he was in general good condition with unremarkable head and neck exam and intact cranial nerves II-XII. On nasal endoscopy, there was evidence of a right-sided reddish nasal mass originating medial to the middle turbinate and the right ostiomeatal complex was edematous; otherwise, the remaining endoscopy was unremarkable. A contrasted computed tomography (CT) of the paranasal sinuses was performed and it revealed an enhancing right ethmoid cavity lesion with bony erosion of the right fovea ethmoidalis, cribriform plate, perpendicular plate of the ethmoid, and superior septum, there was no intracranial or intraorbital extension (Figure 1). Magnetic resonance imaging (MRI) showed an enhancing, homogeneous mass on the contrasted T1-weighted image with homogeneous hyperintensity on the T2-weighted image (Figure 2). Given the vascularity of the lesion, a decision was made to directly proceed with surgical excision (Figure 3).
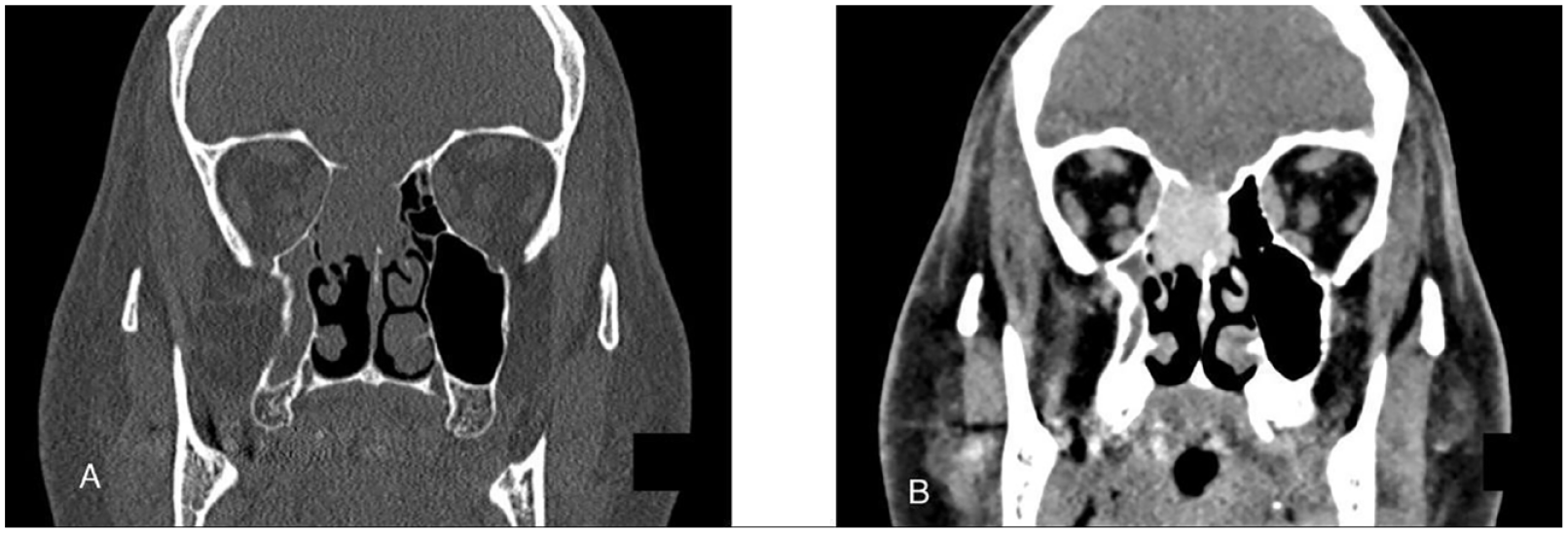
Preoperative computed tomography, coronal view, demonstrating right ethmoid cavity lesion with anterior skull base involvement (A) and contrast enhancement (B).
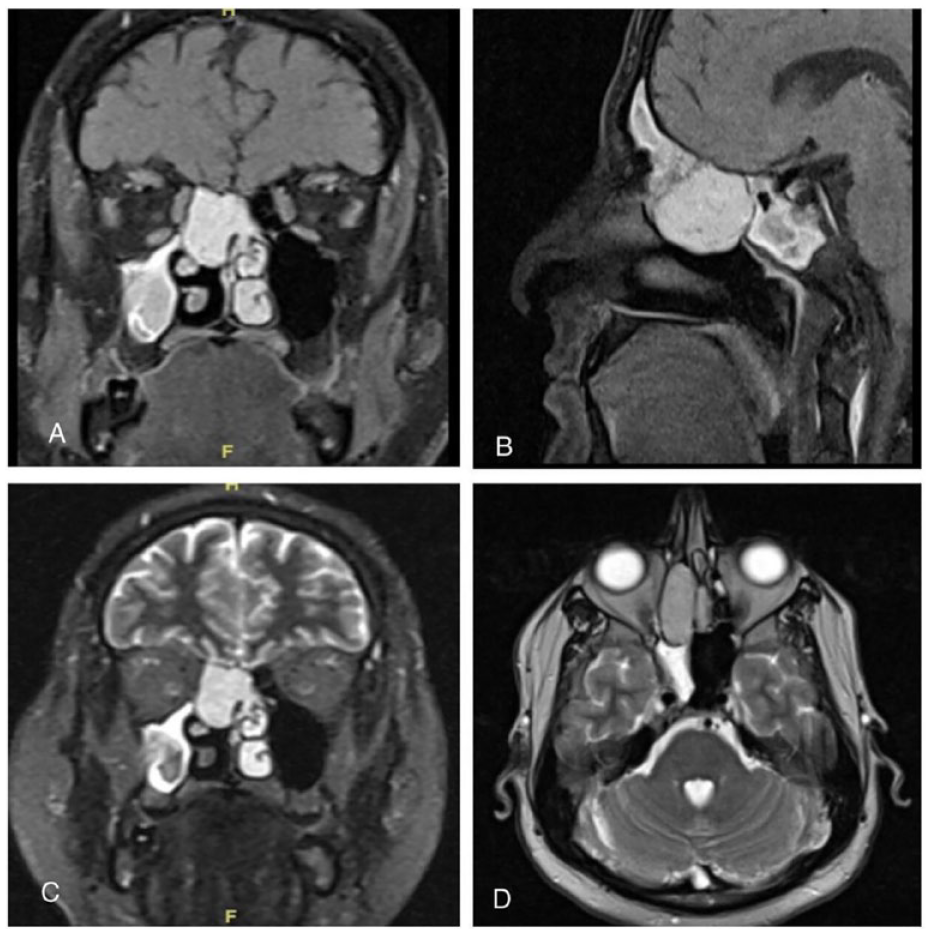
Preoperative MRI, coronal T1-weighted MRI with contrasts demonstrates right ethmoid, homogeneous, hyperintense mass abutting the anterior skull base (A) and similarly on sagittal view (B). Coronal T2-weighted MRI demonstrates the lesion within the ethmoid cavity (C), and the axial view demonstrates the relationship to the orbit (D).
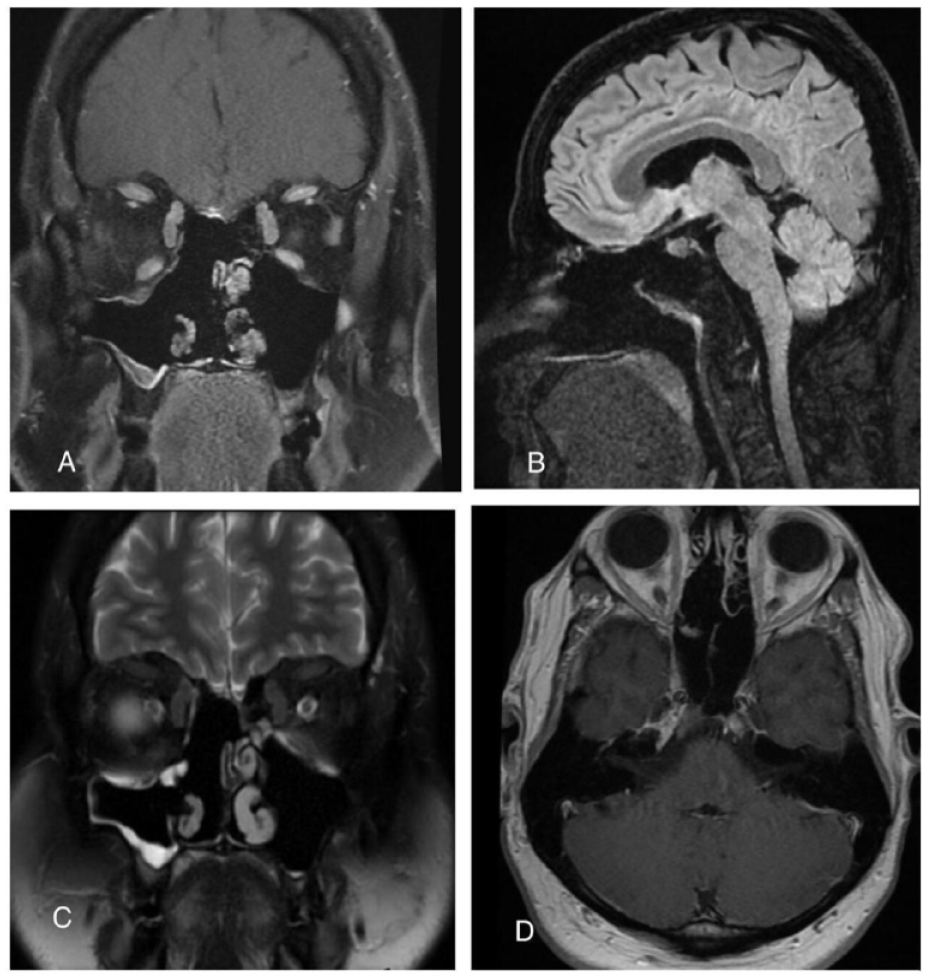
This is a 6-month postoperative MRI, showing a coronal T1-weighted image with contrast and no evidence of residual disease or recurrence (A). Similarly, on sagittal view (B). Coronal T2-weighted image with no evidence of disease. Axial T1-weighted image with similar findings (D).
An endoscopic approach was performed, and consent was made for a possible lynch approach if the need for anterior ethmoid artery ligation is required. Intraoperatively, the epicenter of the mas was the olfactory neuroepithelium part of the septum with extension into the right ethmoid cavity. A gross total resection was performed with preservation of the contralateral superior turbinate. Low-flow cerebrospinal fluid leak was encountered and reconstructed using a nasal floor mucosal graft. The patient did well postoperatively without complications and was discharged postoperative day two. Upon histopathological review, it showed spindle cell proliferation with hemangiopericytoma like vessels (Figure 4). CD34 was performed showing strong membranous and cytoplasmic staining (Figure 5). Given these results, our pathologists were suspecting an SFT; therefore, STAT6 was performed and showed negative nuclear staining. Accordingly, a diagnosis of sinonasal GPC was favored. The patient continued to do well upon 6-month follow-up, with preserved olfaction and no evidence of recurrence (Figure 6).
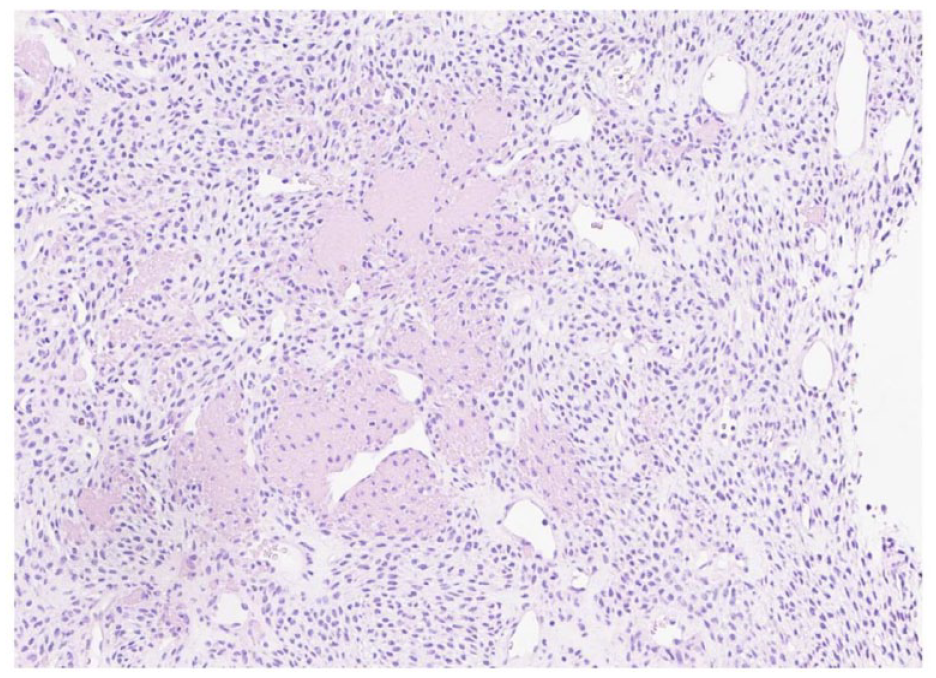
H&E section of the tumor shows spindle and oval-shaped nuclei with irregular and hemangiopericytoma like vessels. H/E stain ×200.
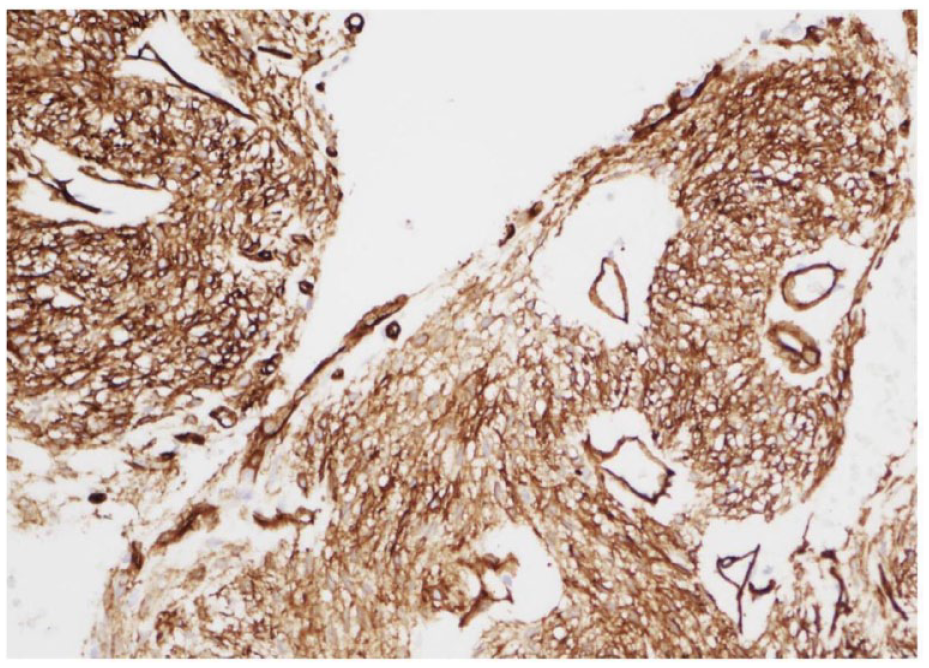
CD34 photomicrograph showing strong membranous and cytoplasmic staining with CD34 immunohistochemical stain ×200.
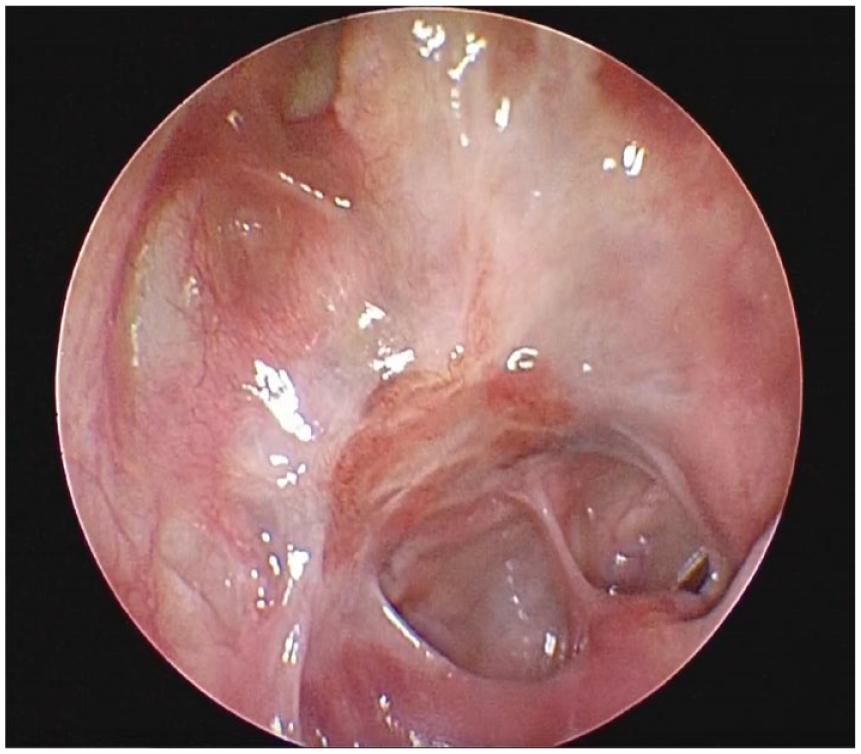
Postoperative nasal endoscopy at 6 months showed a healed skull base and without disease recurrence.
Discussion
GPC is an uncommon sinonasal perivascular tumor with a low risk of malignancy. 14 People of all ages are at risk, with a peak incidence in the seventh decade and a slight female predominance.3,4 The nasal cavity is the most prevalent site of GPC, with paranasal involvement and skull base extension being uncommon. 15
The most common symptom of GPCs was nasal obstruction, followed by epistaxis, headache, facial pain or pressure, and anosmia, in that order. 5 In our case, the patient exhibited recurrent epistaxis and nasal obstruction. GPCs are spindle cell and vascular tumors that affect the sinonasal tract, with SFT being the most common differential diagnosis. 1 SFT, LCH, angiofibroma, and leiomyoma are examples of vascular-rich mesenchymal tumors that should be distinguished from GPCs. 13
Differentiating these entities in the head and neck can be difficult due to overlapping imaging signals; hence, tissue collection (biopsy or resection) is the gold standard for diagnosis. Endoscopy, CT, and MRI may be utilized in the preoperative workup to evaluate the tumor’s extent, size, and characteristics, allowing for optimal preoperative planning. 15
In our case, MRI with contrast revealed a homogeneous, hyperintense mass in the right ethmoid abutting the front skull base (Figure 2A). Histological features such as epithelioid cells with myo-like cell differentiation and immunohistochemistry were utilized to reach the diagnosis. GPC is a cellular spindle cell tumor with hemangiopericytoma-like vasculature and perivascular hyalinization of capillary-sized vessels.4,6 In most instances, smooth muscle actin, muscle-specific actin, and vimentin are all positive.4,7,8 Although CD34 and S100 protein staining may be barely positive in a small percentage of GPC, they are nevertheless essential markers.4,6,8,9 SFT, like GPC, may include zones of hypocellularity and hypercellularity and a variable vasculature made up of staghorn-type and capillary-sized capillaries. However, perivascular hyalinization is uncommon in SFT. In contrast to GPC, the cells overlap and are dispersed more haphazardly, with irregular nuclear outlines. There are no noticeable coarse collagen bundles in a GPC. SFT, unlike GPC, the neoplastic cells stain strongly for CD34, with less consistent actin staining. 12
According to Agaimy et al., NAB2–STAT6 translocation and nuclear STAT6 expression are not present in GPC but are in SFTs that form in the sinus tract. 13 The histology slides of our patient revealed spindle and oval nuclei, along with a few irregular and hemangiopericytoma-like veins, raising suspicion of a diagnosis of a SFT (Figure 4). Interestingly, the STAT6 test for our patient returned negative, whereas the CD34 test returned positive. Therefore, the diagnosis was confirmed as GPC. Ultimately, optimal management for these tumors involves thorough surgical excision followed by extended surveillance. Given their elevated recurrence rate, which can reach up to 40%, and the majority of recurrences manifesting within 5 years post-excision, postoperative surveillance with regular nasal endoscopy is advisable. 16
Conclusion
Sinonasal GPC is a rare, non-malignant tumor that requires histopathological assessment for precise diagnosis, and it can be successfully removed using endoscopic resection after meticulous preoperative planning.
Footnotes
Acknowledgements
Not applicable.
Authors’ Note
The paper was presented at the International Federation of Otorhino Laryngological Societies (IFOUS) in Dubai on January 21, 2023.
Data Availability Statement
The data that support the findings of this study are available within the article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Consideration
All the data collected from this study were confidential, and no personal information was obtained from the participants to ensure their privacy.
Informed Consent Statement
Written informed consents were obtained from the patients for the publication of this case report.
