Abstract
Ameloblastic carcinoma (AC) is a rare and aggressive malignant epithelial odontogenic tumor making up less than 1% of malignant head and neck tumors. The majority of cases occur in the mandible with a minority occurring in the maxilla. Most occur de novo, while rare cases of AC have resulted from transformation from ameloblastoma. Here, we present a case in which a 30-year-old man presented with proptosis and a recurrent right temporal mass, which had been previously diagnosed as ameloblastoma on surgical pathology. CT findings demonstrated local invasion, and he was subsequently taken to the operating room for right craniotomy, infratemporal and middle cranial fossa tumor resection, and right modified radical neck dissection with reconstruction. Final pathology, which included areas of early focal necrosis, loss of peripheral palisading, and hyperchromatism, confirmed the diagnosis of ameloblastoma with transformation to AC. We further discuss radiologic and histopathological signs of this rare tumor, as well as recommended treatment modalities.
Introduction
Ameloblastic carcinoma (AC) is a rare malignant odontogenic tumor. However, amongst the odontogenic carcinomas, it is the most common malignancy. 1 The World Health Organization (WHO), in its 4th edition of the classification of head and neck tumors, classified odontogenic carcinomas as AC, primary intraosseous carcinoma, sclerosing odontogenic carcinoma, clear cell odontogenic carcinoma, ghost cell odontogenic carcinoma, as well as odontogenic carcinosarcoma and sarcoma. 2
A large age range is affected by AC, and it remains unclear whether the tumor has a predilection for males or females. 3 The mandible is the most common location, while a minority of cases occur in the maxilla. Most reported cases of AC occur de novo, while rarely they result from the transformation of benign ameloblastoma. Like ameloblastoma, AC can present aggressively. Painful swelling, rapid growth, paresthesia, facial palsy, and dysphonia are some characteristic features. 4 Due to a lack of large clinical series with long-term follow-up, there is no consensus on the optimal treatment of AC. Despite this, the therapy of choice is often surgical resection. 5
The present report discusses the clinical course and treatment of a 30-year-old man with AC of the mandible, occurring as a result of transformation from an initial ameloblastoma. We further discuss radiologic and histopathological findings helpful in diagnosing AC, as well as potential treatment modalities.
Case Report
A 30-year-old man was first diagnosed with ameloblastoma under his right third molar and underwent resection with mandibular hardware placement at an outside hospital. He had recurrence of his tumor 2 years later at the margin of his previous resection, which spanned across the midline of the mandible, requiring resection of the mandible bilaterally with hardware placement as well as soft tissue reconstruction with a fasciocutaneous radial forearm free flap (RFFF). He then had a third recurrence on the right mandible the following year, which was treated with tumor resection and partial hardware removal without reconstruction. A course of adjuvant radiotherapy was initiated at this time in Guatemala. The remaining hardware began to erode out of his right cheek, and a large mass became evident in the right temporal region. He was placed on dabrafenib 100 mg BID, but the mass grew despite therapy, and the dosage was thus increased to 225 mg QD. The patient self-discontinued the medication two years after initiation. At this time, he underwent presurgical evaluation for his right temporal region mass associated with sharp pain, headaches, and progressive right-sided vision loss, concerning for a fourth recurrence.
On physical exam, the patient had a firm, 5 × 6 cm right temporal mass, mild right proptosis, extrusion of his anterior mandibular hardware through the right cheek with no superimposed infection, and right jaw volume loss. He also displayed reduced light touch sensation over the mass, and an ipsilateral hemifacial palsy, likely secondary to tumor growth and multiple previous surgeries. Fine-needle aspiration (FNA) biopsy of the mass was positive for ameloblastoma, BRAF negative. Computed tomography (CT) scan of the face with contrast showed a locally aggressive mass in the right masticator space invading the right maxillary wall and anterior zygoma, malar eminence, sphenoid bone, as well as extradural extension into the middle cranial fossa (Figure 1A-D). (A) On CT of the face with contrast, there was a large heterogeneous enhancing mass lesion centered within the right masticator space with local osseous destruction and local invasion as seen in the sagittal view (yellow arrowhead). (B) The mass lesion is also seen on coronal view (yellow arrowhead). (C) and (D) Axial views in bone and soft tissue windows (yellow arrowhead). A linear soft tissue density along the superolateral margin of the fasciocutaneous flap was also noted in all views.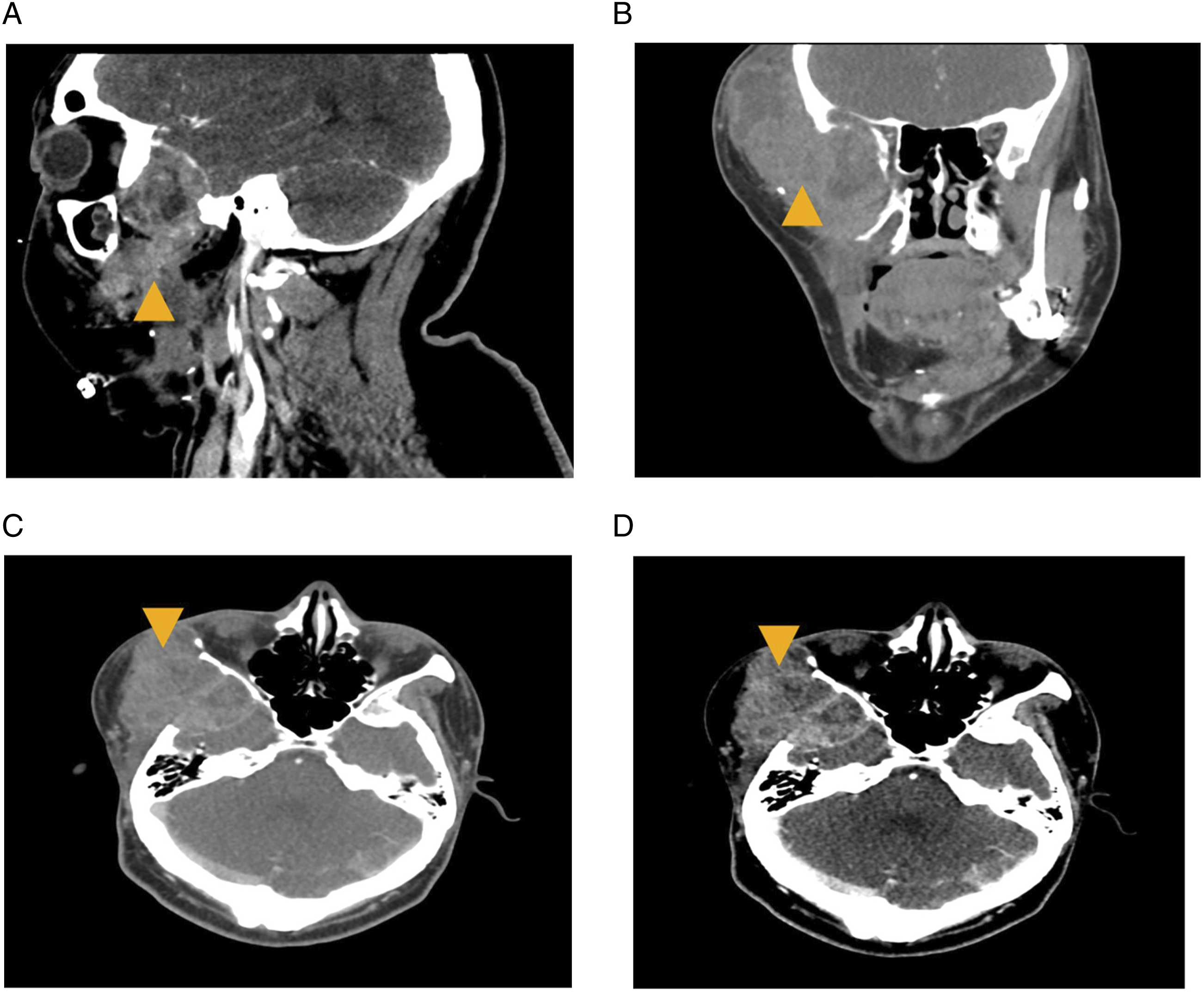
The patient was subsequently taken to the operating room for a right craniotomy, infratemporal and middle cranial fossa tumor resection, right modified radical neck dissection and right anterolateral thigh free flap reconstruction, and static right commissure suspension with a fascia lata sling. Final pathology of the specimen showed evidence suggestive of ameloblastoma with transformation to AC. Specifically, on high magnification there were foci of residual ameloblastoma that showed classic features such as columnar basal cells with palisading, polarization, and cytoplasmic vacuolization (Figure 2A). Alongside this were foci of early focal necrosis (Figure 2B), as well as loss of peripheral palisading, hyperchromatism, increased nuclear to cytoplasmic ratio, and nuclear pleomorphism (Figure 2C, D), indicating transformation to AC. (A) High magnification foci of ameloblastoma demonstrating columnar basal cells with palisading, polarization, and cytoplasmic vacuolization (black arrowhead). (B) Histologic examination of the surgical resection specimen showed ameloblastoma with foci suggestive of transformation to ameloblastic carcinoma. Early focal necrosis is seen (black arrowhead). (C) The stellate reticulum is somewhat retained in the tumor (black arrowhead). (C) and (D) Pathology of the specimen demonstrated loss of peripheral palisading and nuclear polarity (yellow arrowhead). The tumor also showed nuclear pleomorphism, hyperchromatism, increased nuclear to cytoplasmic ratio, and mitotic figures (black arrowheads).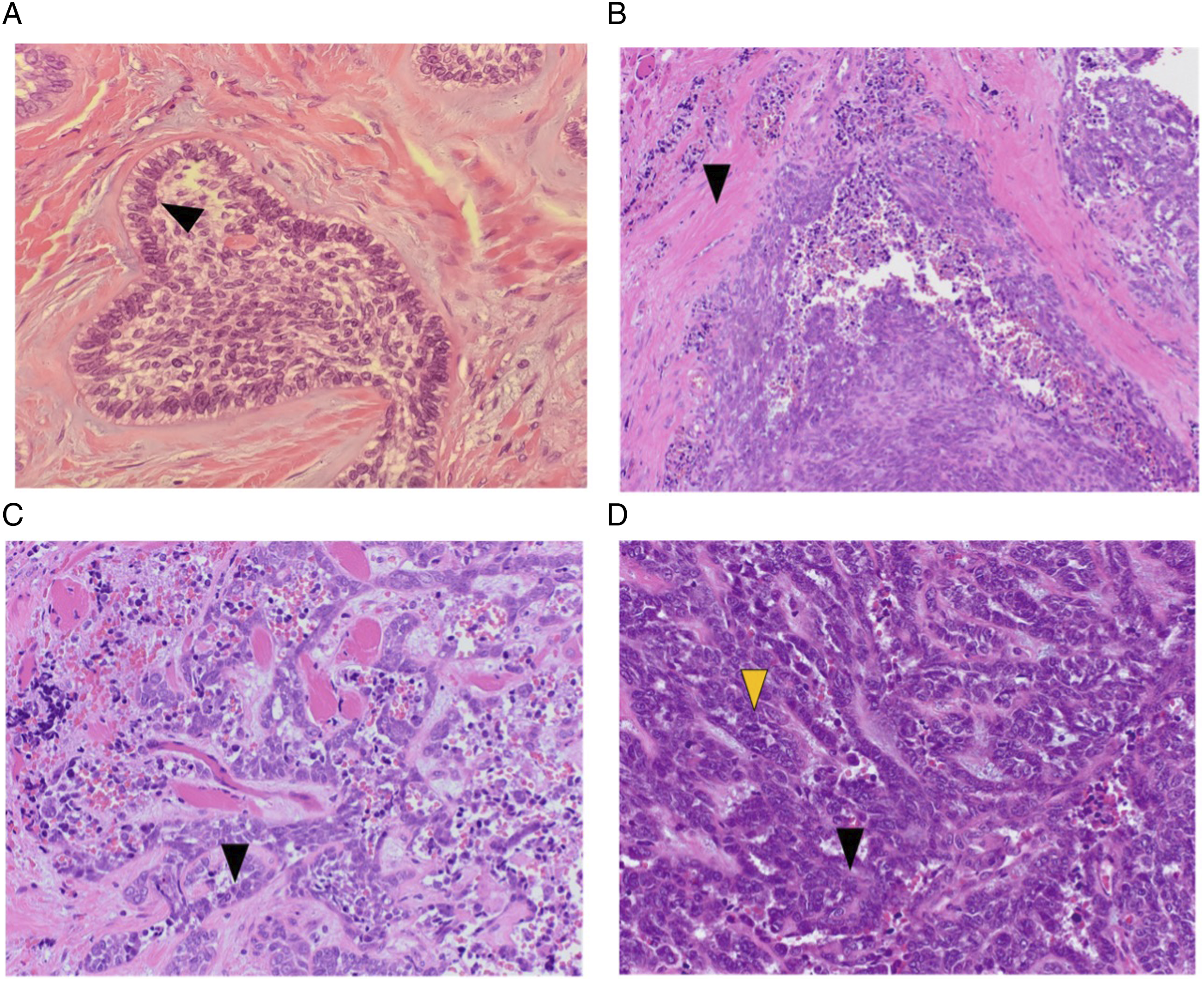
Discussion
AC is a rare and aggressive malignant epithelial odontogenic tumor, with only few cases reported in the literature. In fact, AC makes up fewer than 1% of malignant head and neck tumors. 1 Two-thirds of AC cases arise in the mandible, particularly in the posterior region, while the remaining one-third originate from the maxilla. 6 The majority of ACs seem to occur de novo, but there are some case reports which have described the transformation of ameloblastoma to AC. 4 In these instances, diagnosis is generally simpler, as there is a transition area on pathology between existing benign and malignant tumor.
There are two malignant variants of ameloblastoma, including metastasizing ameloblastoma, which microscopically appears benign, as well as AC, which retains some histopathologic features of benign ameloblastoma while also showing characteristics of malignancy. These include increased mitotic index, necrosis, atypical nuclei, and hyperchromatism. 7 Histopathology of AC may also resemble many other malignant odontogenic tumors classified by the WHO, including squamous odontogenic tumor, intraosseous squamous cell carcinoma, and clear cell odontogenic carcinoma. Histologic features more specific to AC are lack of a stellate reticulum, spindle-shaped epithelial cells with minimal differentiation, and islands of epithelium (Figure 2B-D).7,8
Similar to ameloblastoma, AC can present with an aggressive clinical course. Facial palsy, painful swelling, rapid growth, and paresthesia are among the most characteristic clinical features. 4 Moreover, AC may metastasize to regional lymph nodes, the lungs, or liver. When AC arises de novo, other tumors such as metastatic carcinoma of the jaw, primary intraosseous squamous cell carcinoma, central high-grade mucoepidermoid carcinoma, as well as carcinoma of nearby soft tissue invading bone, should be included on the list of differential diagnoses. 8
AC may demonstrate various radiographic features. It can present as a cystic lesion or large mass with resorption of bone. 3 At times, it can be difficult to differentiate between AC and ameloblastoma, as AC may have a well-defined or ill-defined border. 3 AC, however, often demonstrates bone erosion with focal radiopacities. 9 Furthermore, AC may include regions of necrosis with dystrophic calcification. 3 Thus, obtaining a CT scan of the face with contrast is useful, as findings are occasionally helpful in differentiating AC from ameloblastoma.
Considering the rarity of cases of AC, treatment remains controversial. However, in the case of AC arising from ameloblastoma, tumor resection with a surgical margin of 10–15 mm is the most common treatment modality. For primary AC, smaller margins have been reported. When there is clinically evident lymphadenopathy, modified radical neck dissection should be considered. 7 While some studies have suggested radiotherapy to be an effective treatment for AC, 10 others argue against its role in the treatment of this tumor. 8 Adjuvant radiotherapy may be considered in cases with significant soft-tissue involvement or perineural invasion, or positive surgical margins. 10 With regards to chemotherapy, the evidence in the literature is limited. The few studies which have reported outcomes have demonstrated that chemotherapy has limited value for AC. 7
Ultimately, the prognosis of AC is largely driven by risk of local recurrence, which has been reported to occur between 6 months and 11 years after treatment. Reports on the rate of recurrence range from 50% to 90%, whereas rate of metastasis is reported around 30%. The lungs are the most common site of metastasis, while there have also been cases of bone, brain, and liver metastases. Distant metastasis has been reported to occur between 4 months and 12 years post-surgery. 7 Therefore, long-term follow-up for recurrence is important for patients diagnosed with AC.
We presented a case of ameloblastoma in the mandible with transformation to AC. The tumor was locally invasive and represented the patient’s fourth recurrence of the mass. The patient underwent right craniotomy, infratemporal and middle cranial fossa tumor resection, and right modified radical neck dissection with reconstruction. Histologic sectioning showed areas of ameloblastoma with regions of transformation to AC, confirming the diagnosis. Due to the rarity of this malignancy, however, the most effective treatment modalities continue to remain unclear.
Footnotes
Acknowledgments
We would like to thank Wesley Naritoku, MD, Ph.D. (Associate Professor, Program Director) and Philip Petersen, MD (Resident Physician) from the Department of Pathology and Laboratory Medicine of USC for the provision and interpretation of the histopathological images. We would also like to thank Saif Azam, MD (Resident Physician) from the Department of Radiology of USC for provision of the radiologic images.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
