Abstract
Second primary cancer (SPC) is a term used to describe a new primary cancer occurring in patients who had formerly been diagnosed with tumor. Even though SPCs appear to be related to primary cancers, they are actually entities that have arisen independently and not as a result of recurrence. This report is of the first case in literature of a patient hospitalized for the surgical treatment of 3 synchronous Head and Neck Cancers. A 66-year-old male was admitted to our hospital (Ospedale Degli Infermi—Biella, Italy) complaining about pharyngodynia. Three different lesions were identified through endoscopic examination and narrow band imaging: the first one on left tonsil, the second one on epiglottis, and the third one on right aryepiglottic fold. The case was subject to a multidisciplinary team analysis due to its complexity, then the surgery consisted in (1) CO2 laser left tonsillectomy, associated with (2) CO2 laser excision of the lesion on epiglottis free edge, and (3) CO2 laser excision of right aryepiglottic fold lesion. Synchronous tumors are among the most defiant challenges for surgeons since no international guideline specifies differentiated strategies to be adopted in patients affected by synchronous Head and Neck Cancers, therefore surgical planning must be tailored differently from patient to patient, and many unsolved questions still concern clinical treatments to be adopted.
Introduction
The term second primary cancer (SPC) refers to a new primary cancer occurring in patients who had previously been diagnosed with tumor. Nevertheless, SPCs are independent tumors that can affect any organ or tissue 1 at any time, showing no correlation to the primary tumor site. Second primary cancers are defined as synchronous if detected simultaneously or within 6 months after the primary tumor diagnosis, otherwise SPCs are defined as metachronous. 2
According to the Radiation Therapy Oncology Group, approximately as many as 23% of patients who have been diagnosed with Head and Neck Cancer (HNC) develop a SPC in treated regions. 3 At the same time, as stated by an international meta-analysis held in 1992, as many as 14.2% HNC survivors are diagnosed with SNC if followed up for more than 5 years. 4 However, it should be specified that residual cancer, a remaining portion of the original primary tumor, or disease recurrence could be misdiagnosed as SPCs, especially if occurring within 5 years after the primary HNC diagnosis.
The SPC’s etiology is multifactorial. Firstly, tobacco and alcohol consumption are among the main risk factors for SPCs to occur, owing to their synergistic effect combining mucosal inflammation determined by alcohol abuse with tobacco genotoxic effects. Secondly, human papilloma virus-16 infection, poor nutrition, bad dental hygiene, and exposure to carcinogenic substances may contribute to the onset of the disease.5-8 Thirdly, radiotherapy to treat the primary cancer as well as the prolonged consumption of alcohol and tobacco after the HNC diagnosis may rise the possibility for SPCs to occur. 5
In addition, a statistically relevant correlation between HNC and breast cancer has recently been discovered, suggesting a genetic predisposition to the disease in those patients who have a family history of breast cancer. According to several contemporary studies, “field cancerization,” which is the replacement of normal cell population by a cancer-primed cell population, is recognized to underlie the development of different types of cancer and it can explain the aerodigestive epithelium predisposition to develop multiple precancerous and cancerous lesions after the exposure to carcinogenic substances. 6
In addition, the frequent exposure to radiation of HNC survivors who are constantly checked-up and, therefore, most likely to be diagnosed with a second tumor, must also be considered as an influencing factor in the analysis of SPCs etiology. Finally, the detection of SPCs has lately been improved by the accuracy of sophisticated imaging modalities that can identify even the smallest lesions. 7
Patients who had previously been diagnosed with HNC and who present with synchronous SPCs account for 1% to 6% 8 ; head, neck, lungs, and esophagus are the most affected sites. These patients have traditionally been screened for synchronous SPCs through a comprehensive examination under anesthesia including endoscopic procedures, such as laryngo-pharyngoscopy, bronchoscopy, and esophagoscopy.
In addition, high sensitivity in identifying synchronous primary cancers can be offered by positron emission tomography and computed tomography (PET/CT) scans. Nevertheless, their exposure to false-positive results may lead to further investigations, meaning additional costs for the hospital and stress for the patient. For this reason, the universal use of PET/CT scans for the detection of SPCs is not the suggested procedure.9-11 On the other hand, promising results in SPCs prompt detection of both synchronous and metachronous tumors have been revealed by new endoscopic techniques such as narrow band imaging (NBI).11,12 This report is of the first case in literature of a patient hospitalized at Ear Nose and Throat Department of Biella Hospital for the surgical treatment of 3 synchronous HNCs.
Case Report
A 66-year-old male with a clinical history of chronic alcohol-related liver diseases and tobacco abuse was admitted to our hospital (Ospedale Degli Infermi—Biella, Italy) complaining about pharyngodynia; neither dyspnea nor dysphonia had been associated. Endoscopic examination and NBI highlighted 3 different lesions. The first one on left tonsil, ulcerated, hardened on palpation, 1.5 cm in diameter; the second one on epiglottis lingual surface (no involvement of epiglottis base was detected), vegetative, 0.8 cm in diameter; the third one on right aryepiglottic fold (piriform sinus apex free from disease), vegetative, 1.2 cm in diameter. No lymph node involvement was found on neck palpation.
Neither lymph node involvement nor distant metastases were detected through neck CT-scan with contrast and total body PET-scan. At the same time, no gastroesophageal lesions were detected through esophagoscopy. The diagnosis of squamous cell carcinoma was then confirmed through biopsy and histological examination.
The case was subject to a multidisciplinary team analysis, then the indication of surgical intervention was identified as the optimal approach. The surgery consisted in (1) CO2 laser left tonsillectomy, associated with (2) CO2 laser excision of the lesion on epiglottis free edge, and (3) CO2 laser excision of right aryepiglottic fold lesion. Bilateral neck dissection and a protective tracheostomy were then performed. The presence of carcinoma affecting the 3 sites was confirmed by postoperatory anatomopathological examination report, revealing pT1N0M0 staging grade (Table 1; Figure 1A and B).
List of the 3 Surgeries Performed Simultaneously for the Removal of the 3 Synchronous Tumors With Histological Outcome.
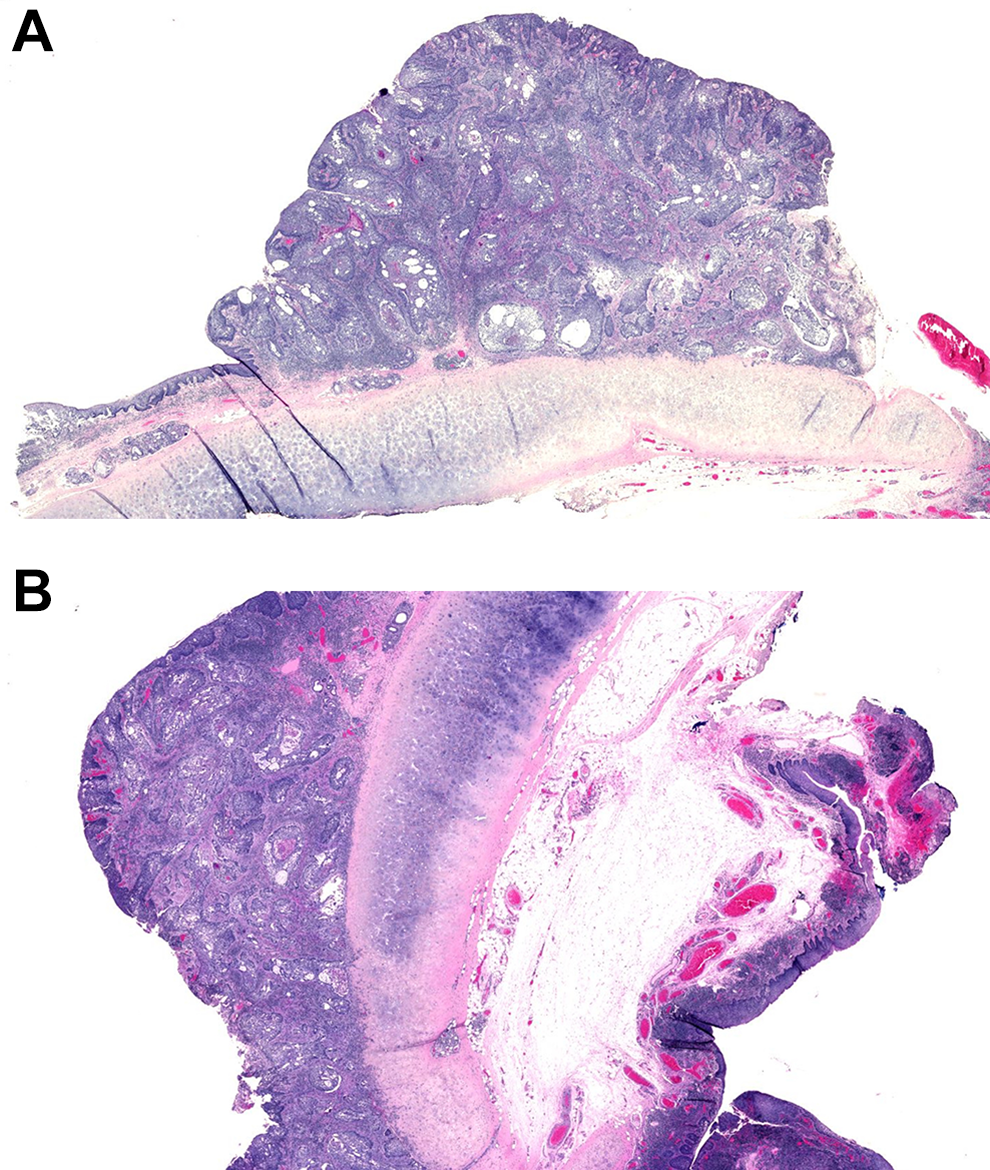
A and B, Histological images of the tumor on epiglottis lingual surface, showing the absence of invasion of the cartilage.
In order to prevent any disease recurrence and considering the close surgical margins found on left palatine tonsil, the patient was subject to adjuvant radiotherapy. Six months after the end of radiant therapy no endoscopic or radiologic signs of relapse were detected.
Discussion
Tobacco and alcohol abuse, exposure to carcinogens, genetic predisposition, and the biological process—known as “field cancerization”—in which normal cell population is replaced by a cancer-primed cell population are recognized among the main causes for the occurrence of SPCs. Because of well-documented HNC patients’ high predisposition to develop metachronous or synchronous tumors, careful check-ups through PET/CT scans and innovative endoscopic techniques such as NBI should be of primary importance after the recovery.
Careful and adequate follow-ups are essential not only to inspect the primary tumor location but also to check head and neck, as well as lungs and esophagus, which are more likely to be affected by SPCs. Unfortunately, no international guideline specifies, to our knowledge, differentiated and targeted strategies to be adopted in patients affected by synchronous HNCs. The heterogeneity of sites and staging grades of 2 or more distinct synchronous tumors may be among the main reasons for this lack of indication, therefore, therapeutic procedures are very difficult to standardize.
This lack of specific guidelines complicates the evaluation of compromised and fragile patients, often presenting with different concomitant pathologies, high risk of disease recurrence, and a long-standing history of alcohol or tobacco abuse. Synchronous tumors represent one of the most defiant challenges for surgeons since surgical planning must be tailored differently from patient to patient, owing to the simultaneous presence of multiple tumors affecting different sites. Therefore, the ability of the surgeon to handle the same intervention using different techniques and the communication among clinicians are essential.
In addition, the intervention should be as much conservative as possible in order to preserve patient’s chewing, swallowing, and speaking functions as well as an adequate quality of life. For this reason, laser or transoral robotic surgeries should be preferred in patients presenting with synchronous tumors, as these techniques minimize surgical excision, hence facilitating functional recovery and postoperative healing.
Our patient had all of 3 tumors removed in a single intervention through transoral CO2 laser surgery. Postoperative radiotherapy was still necessary owing to the presence of close surgical margins and to the high risk of disease recurrence. In conclusion, many unsolved questions still concern clinical treatments to be adopted in patients who are diagnosed with synchronous tumors. For example, whether adjuvant radiotherapy may have significant benefits or whether follow-ups should be planned closer in time are still open questions.
There is no doubt that international guidelines would represent a crucial support in planning specific treatments for patients presenting with synchronous tumors. Nevertheless, the heterogeneity of stages and sites of synchronous HNCs complicates the standardization process, hence communication between specialists taking part in multidisciplinary teams is essential to positively influence patients’ treatment planning. The setting is complex, and decisions must be made considering a lot of factors. Careful balancing of these factors allows individualized approaches to be tailored for any individual case.
Footnotes
Acknowledgments
The authors would like to thank Anna Lanza for her assistance in drafting and correcting the English language.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
