Abstract
Objective
To investigate the possible causes and treatment methods of laryngeal stenosis after radiotherapy following supracricoid partial laryngectomy with cricohyoidoepiglottopexy (SCPL-CHEP).
Methods
The data of seven patients with laryngeal stenosis after radiotherapy following SCPL-CHEP were analysed retrospectively. All patients were diagnosed with mid-stage or advanced laryngeal carcinoma before surgery, and the pathological type was squamous cell carcinoma. All patients met the requirements for SCPL-CHEP surgery. When laryngeal stenosis was found during the post-surgical follow-up period, patients were immediately given the appropriate treatment according to their conditions.
Results
All seven patients had laryngeal stenosis. One patient underwent granulation resection using a laryngoscope, four patients underwent granulation removal + low-temperature plasma ablation using a laryngoscope, and two patients underwent laryngeal dehiscence surgery + laryngotracheal T-tube placement. All patients recovered well after surgery, with patent airways.
Conclusion
Laryngeal stenosis in patients with mid- and late-stage laryngeal carcinoma is one of the rare complications of SCPL-CHEP. Second-stage laryngeal dilatation can be selected according to the patient’s laryngeal stenosis. Most patients with laryngeal stenosis can be extubated completely.
Introduction
Supracricoid partial laryngectomy with cricohyoidoepiglottopexy (SCPL-CHEP) was first proposed in the 1950s. After several improvements, it has gradually become the standard procedure for SCPL-CHEP, and it was widely used in North America and Western Europe in the 1990s.1-4 Supracricoid laryngectomy is a well-tolerated procedure that results in excellent voice and swallowing function after long-term follow-ups. However, patient selection is crucial to achieving an acceptable result in terms of pathological and functional control. 5
The stage of laryngeal carcinoma is determined by the pathological examination of local laryngeal tissue. Patients with middle and partially advanced laryngeal cancer will experience symptoms of partial aphonia, dyspnea and dysphagia. 6 The SCPL-CHEP procedure is effective for middle and partially advanced laryngectomies,6-9 particularly when the tumour invades both glottic and supraglottic regions, since it avoids total resection while retaining function.10,11 Nonetheless, some patients who undergo surgery and radiotherapy experience postoperative laryngeal stenosis and intubation difficulties – a relatively rare complication after SCPL-CHEP. Luc Bron 7 and Roberto A. Lima et al. 12 reported the proportion of postoperative laryngeal stenosis to be relatively low, and thus, laryngeal stenosis has not been studied in depth. Once laryngeal cavity stenosis occurs, it often leads to decannulation failure. At present, the common methods for the treatment of laryngeal stenosis include routine throat shearing and narrow scar resection, cryogenic plasma ablation and T-tube dilation. The cause of laryngeal stenosis is currently unknown.
This study analysed the data of 7 patients who underwent radiotherapy after SCPL-CHEP in our hospital and explored the possible causes and treatments of laryngeal stenosis after SCPL-CHEP in patients with mid-stage and advanced laryngeal cancer.
Methodology
Patient Population and Features
Seven patients underwent SCPL-CHEP in our hospital from January 2013 to April 2021. All were males, aged 47-70 years, with an average age of 60 ± 8.44 years. The inclusion criterion was laryngeal stenosis after SCPL-CHEP surgery in patients with advanced laryngeal cancer, and the possibility of recurrence was excluded. All surgeries were performed by a senior head and neck surgeon with almost 20 years of surgical experience in CHEP in our hospital.
All 7 patients with mid-stage and advanced laryngeal cancer who underwent SCPL-CHEP had decannulation failure due to lumen laryngeal stenosis following surgical radiotherapy. Two cases were unable to extubate 6 months after surgery, 1 case was unable to extubate 10 months after surgery, 3 cases were unable to extubate 1 year after surgery and 1 case was unable to extubate 2 years after surgery.
Type and Stenosis of Laryngeal Cancer
According to the medical records of the 7 patients with laryngeal stenosis after SCPL-CHEP, they had highly differentiated squamous cell carcinoma in the middle and advanced stages. They underwent preoperative electronic fibreoptic laryngoscopy to determine the Myer–Cotton grading. 13
Treatment Method
The patients were asked about the history of their postoperative laryngeal stenosis, which comprised mostly mid-stage and advanced laryngeal cancer. The patients’ previous medical records and surgical histories of SCPL-CHEP were checked to determine specific information. Electronic fibreoptic laryngoscopy and computed tomography examinations of the throat were performed to determine the new biological size of the throat, which was the cause of decannulation failure.
The following surgical options were used for patients with laryngeal stenosis after radiotherapy and SCPL-CHEP. First, routine throat shearing and narrow scar resection were performed under total anaesthesia using laryngoscopy. Second, cryogenic plasma ablation using laryngoscopy was used. Third, when the supported laryngoscopy approach was invalid, T-tube dilation was performed. The patients received anti-infection and rehabilitation treatment after surgery.
Concurrent platinum-based chemoradiotherapy or surgery followed by postoperative RT is recommended for node-positive supraglottic carcinoma (T1–T2 N1+, stage III–IV) in patients whose performance status is sufficient to tolerate the above treatment. 14
Results
General information of patients (n = 7).

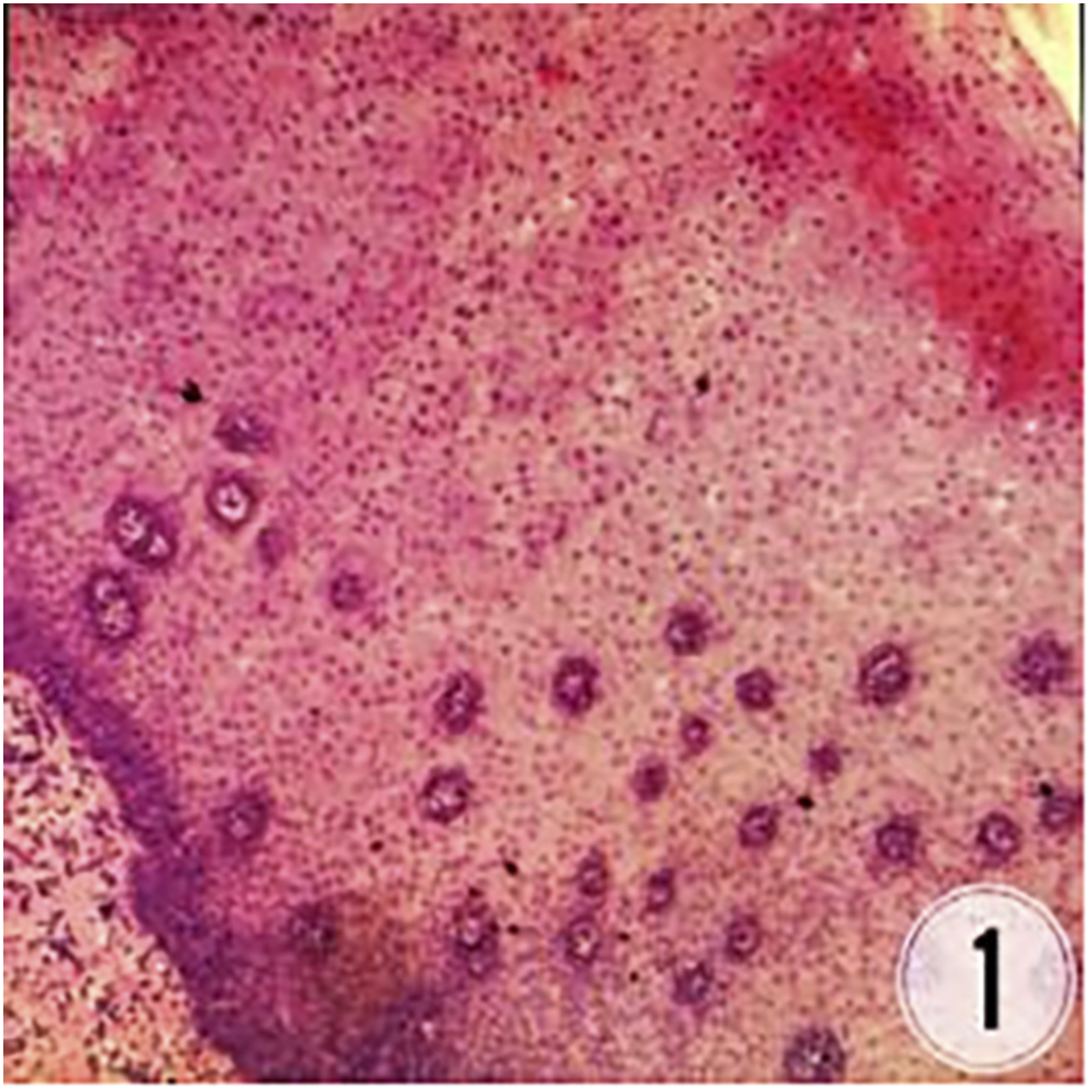
New biopathologic section of patients (laryngeal) polyp, squamous epithelial light-moderate atypical hyperplasia.
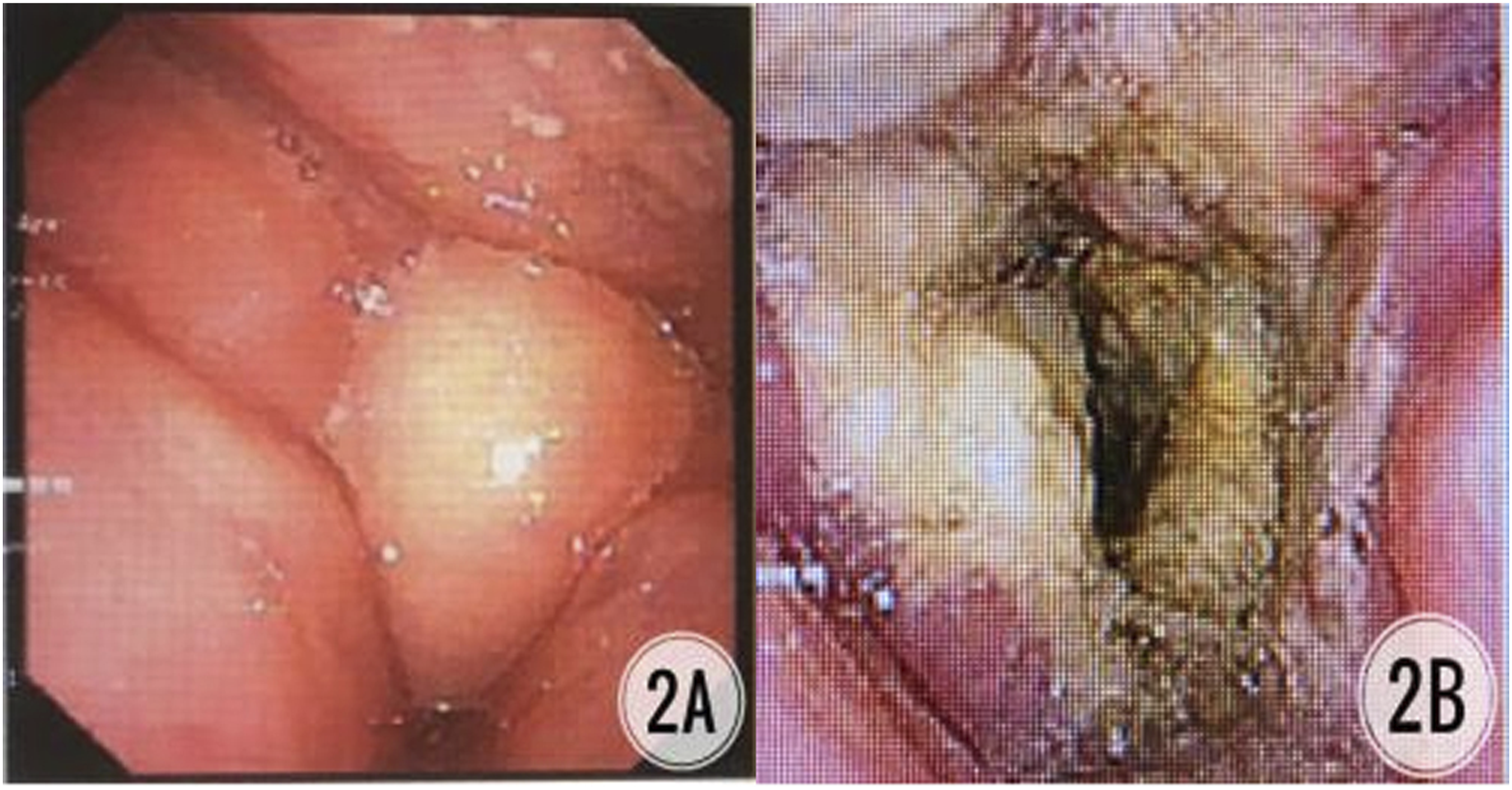
Patients with middle and advanced squamous cell carcinoma, underwent granulation removal + low-temperature plasma ablation using a laryngoscope. Notes: A. Preoperative larynthroscopy showed nodular tissue stenosis in the larynx cavity; B. Postoperative laryngoscope showed the airway reopening.
Two patients with mid-stage and advanced squamous cell carcinoma had laryngeal stenosis and extraction difficulties after SCPL-CHEP. One patient had laryngeal stenosis 1 year after surgery. We performed a routine granulation resection, and the tube extraction was successful. Another patient had a difficult stenosis extraction 2 years after surgery, but there was no follow-up; the stenosis occurred again 4 years later. In this patient, we performed low-temperature plasma ablation of the laryngeal hyperplasia tissue and achieved good postoperative airway opening (Figure 3). Patients with middle and advanced squamous cell carcinoma, underwent granulation removal using a laryngoscope. Notes: A. Preoperative larynthroscopy showed nodular tissue stenosis in the larynx cavity; B. Postoperative throat CT showed local obstruction in the throat cavity and airway reopening.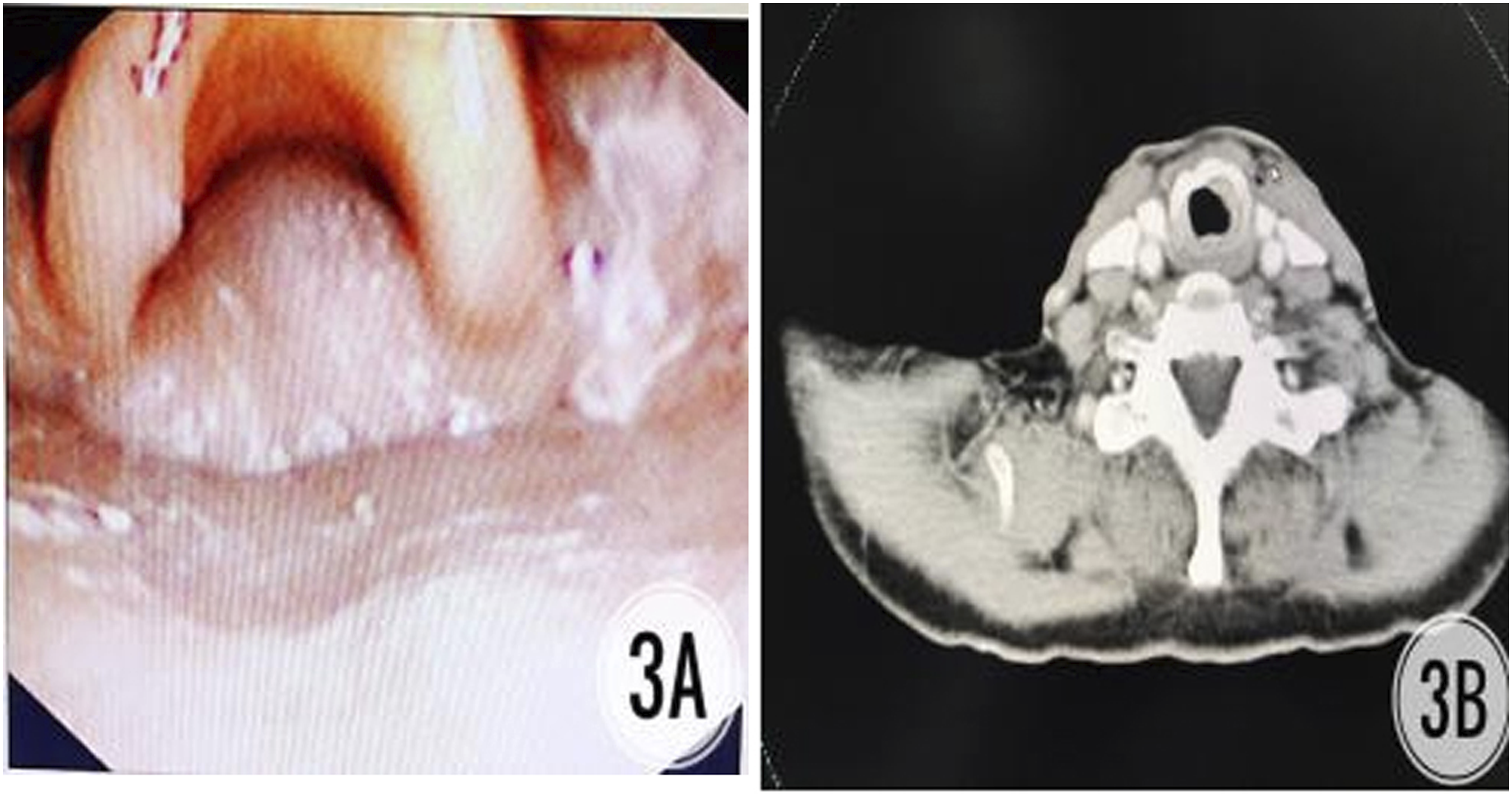
Two patients with mid-stage and advanced squamous cell carcinoma had laryngeal stenosis and difficulty in decannulation 1 year after SCPL-CHEP. We performed supported laryngoscope granulation resection + low-temperature plasma ablation of the hyperplasial tissue of the laryngeal cavity, expanded the residual throat cavity and successfully removed the tube. However, the patients had scarring and progressive breathing difficulties 2 months after surgery. Consequently, we performed a laryngoscopy on these patients and observed small residual sound-valve cracks along with hyperplasia and stenosis of the surrounding tissue. We considered this to be scar hyperplasia. In a rebiopsy, CK (−), EMA (+), P63 (−), CK5/6 (−), LCA (+) and fibre tissue hyperplasia were used to identify recurrence. We performed a laryngotomy and inserted a T-tube via the throat scar. This tube was removed after 1 year. No new tumours were found during the follow-up, and the airway opening was good (Figure 4). Patients with middle and advanced squamous cell carcinoma, underwent granulation removal + low-temperature plasma ablation using a laryngoscope, then performed the larynotomy and inserted T-tube by throat scar. Notes: A. CT showed narrowing of throat stenosis before underwent granulation removal + low-temperature plasma ablation using a laryngoscope; B. 2 months after surgery, laryngeal CT showed small residual sound-valve crack, peripheral tissue hyperplasia and stenosis, and scar hyperplasia was considered; C. Biopsy indicated fibre tissue hyperplasia to eliminate recurrence; D. Scar removal, CT image after T-tube inserted; E. The airway was reopening in the second year of follow-up after extraction.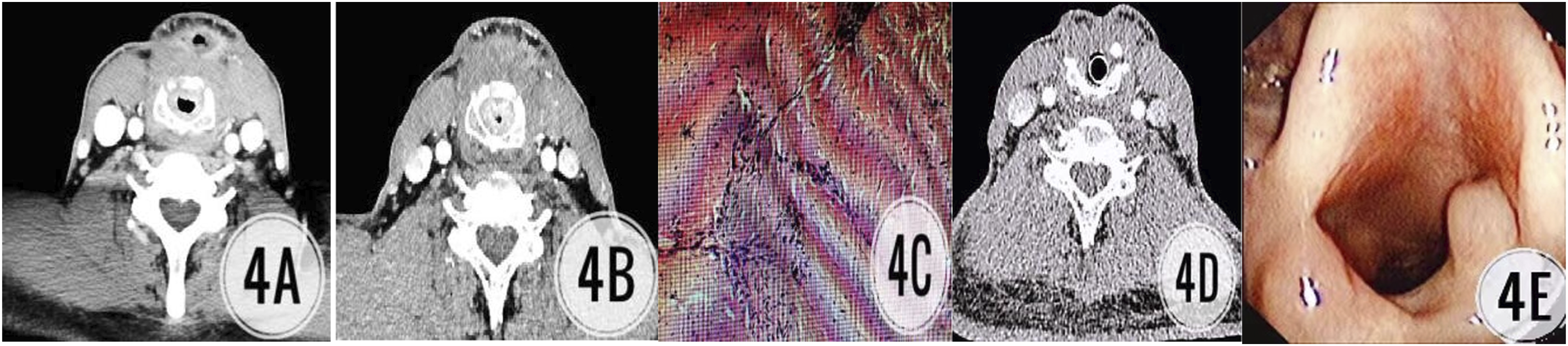
The above results indicate that laryngeal stenosis caused by scar hyperplasia could be treated by laryngotomy and the insertion of a T-tube via the throat scar.
Discussion
Previous reports have shown that SCPL-CHEP has a 5-year local control rate of around 85%–95% in patients with stages T2 and T3, and the decannulation rate is nearly 100%.1,15 However, such patients have no postoperative supplementary radiotherapy treatment, and only individual patients with postoperative lymphatic metastases receive radiotherapy.16-20 In the 43 patients with CHEP or CHP reported by Roberto A. Lima, 12 the proportion of postoperative laryngeal stenosis was 9.3% (4 cases). The tube was successfully removed after laser treatment in 1 patient with throat stenosis, and the remaining 3 patients needed a tracheotomy to breathe. All these cases occurred soon after surgery. In Luc Bron’s report, 7 the proportion of postoperative laryngeal stenosis was 1.4% (1 case); this also happened before extraction, and the tube was successfully removed with laser treatment. Eduardo M. Diaz 21 conducted long-term studies of the probability of postoperative non-neoplastic tracheal stenosis in patients who underwent SCPL. He followed 376 cases from 1975 to 1995 and categorised postoperative non-tumour symptomatic stenosis into two groups. Group 1 consisted of patients who could not remove the tube until 60 days after surgery, and Group 2 consisted of patients with breathing difficulties after successful extraction. In total, 14 cases (3.7%) of non-tumour symptomatic laryngeal stenosis occurred among the 376 cases of laryngeal squamous cell carcinoma. There were 7 cases in Group 1 (1.85%) and 7 cases in Group 2 (1.85%). Although comprehensively visible, laryngeal stenosis after CHEP is rare and often ignored.22,23
Causes of Laryngeal Cavity Laryngeal Stenosis
Combining the findings of this study with previous results, the following are the authors’ suspected causes of laryngeal stenosis.
Radiotherapy Factors
We believe that radiotherapy is one of the main causes of laryngeal cavity stenosis after SCPL-CHEP in mid-stage and advanced laryngeal cancer. In Jayawardena’s report, the author found 3 patients with advanced head and neck tumours who had difficulty breathing due to laryngeal stenosis after radiation therapy. 24 In Incandela’s studies, radiotherapy was a leading cause of laryngeal stenosis (20%), second only to laryngeal surgery. 25 Zhou Liang, 16 Nakayama 17 and Chevalier 18 reported that when not receiving postoperative adjuvant radiotherapy, almost no patients had laryngeal stenosis, and the success rate of tube extraction was nearly 100%. However, in Wu Xuan’s research, 40.9% (9/22) of all patients had laryngeal stenosis caused by radiotherapy after partial laryngectomy, while only 13.8% (16/116) of all patients were without postoperative radiotherapy. 26 Accordingly, it can be seen that radiotherapy stimulation plays an important role in the cause of postoperative laryngeal stenosis.
Radiotherapy can cause tissue swelling, fibrinoid necrosis and the extensive generation of granulation tissue in the glottic range. Oedema, hyalinisation and granulation tissue hyperplasia were present at the affected site, and infiltration by macrophages, plasma cells and fibroblast cells was also observed in the granulation tissue. They can extensively block the airway and cause laryngeal stenosis. 27
In the present study, 7 patients with mid-stage and advanced laryngeal cancer with unsuccessful decannulation underwent radiotherapy after SCPL-CHEP, leading to severe oedema and scarring at the wound site. During the healing process, the residual interarytenoid area and the repaired area of the arytenoid cartilage removal site underwent an oedema reaction. We hypothesised that due to further stimulation by radiotherapy, the patients were prone to postoperative oedema aggravation and fibrotic connective tissue hyperplasia, which caused laryngeal stenosis.
Infective Factors
Infection is a common complication of SCPL-CHEP. Often, patients have gastroesophageal reflux disease due to long-term intubation or surgical reasons, which may cause local infection and lung infection.22,28 Simultaneously, infection can cause granulation tissue hyperplasia and an increase in fibroblasts. It is common knowledge that granulation tissue and scar tissue hyperplasia are the main causes of laryngeal stenosis.
Surgical Factors
Laryngeal wound defect repair and laryngeal cavity reconstruction directly affect the post-surgical occurrence of laryngeal stenosis of the laryngeal cavity. Incorrect alignment between the hyoid bone and annular cartilage may be one of the causes of postoperative laryngeal stenosis. Eduardo M. Diaz argued that incorrect alignment between the two resulted in a high probability of laryngeal stenosis after surgery. 21 Moreover, because this operation typically removes one side of the arytenoid cartilage, granulation and scar hyperplasia occur in this area after surgery. Since some patients have scars, excessive hyperplasia scarring occurs in this area, leading to the formation of a ‘scar lid’, which blocks the laryngeal cavity.
Other Factors
Postoperative stenosis after SCPL-CHEP may also be associated with patients’ personal constitutions. 29 For example, 2 patients in this study had scarring. Excessive hyperplasial scarring of the laryngeal wound after partial resection of the larynx may also be one of the important factors of laryngeal stenosis.
Clinical Treatment of Laryngeal Stenosis
In our hospital, patients with early larynx cancer are treated mainly with vertical hemilaryngectomy since SCPL-CHEP is mainly used for patients with mid-stage and advanced laryngeal cancer. Most patients need radiotherapy to reduce the probability of tumour recurrence after SCPL-CHEP.19,20 If the laryngeal tissue appears to have hyperplasia after surgery, we perform a biopsy to determine whether it is hyperplasia rather than recurrence.
To avoid secondary open surgery, conventional throat shearing and narrow scar resection are performed using laryngoscopy under total anaesthesia. If the effect is not good, or the new organism recurs, low-temperature plasma technology with supported laryngoscopy can be used to remove and ablate the tissue in the narrowed area. Most patients get satisfactory results using this method. However, if the effect is poor and the treatment is ineffective, a laryngeal T-tube can be added to expand the laryngeal cavity.
We can also use different treatments according to the anatomical locations of the patient’s new larynx organism. Using suspension laryngoscopy, most narrow areas can be satisfactorily exposed when the depth of anaesthesia and muscle loosening are both good and the surgeon’s technique and proficiency are excellent. Then, we can remove both hyperplastic granulation and scar tissue using CO2 laser or cryogenic plasma techniques. The surgical injury is smaller and less influential in patients who have experienced SCPL-CHEP trauma. According to our study, this treatment can effectively reduce patient trauma from secondary surgery and facilitate postoperative recovery in patients with mid-stage and advanced laryngeal cancer. If the patient reappears with laryngeal stenosis, we can add a laryngeal T-tube to expand the laryngeal cavity. For example, in this study, 2 patients had hyperplastic scarring and granulation tissue removed via laryngoscopy. However, because the patient’s surgical wound had less normal mucosa that could not be effectively epithelial in addition to scarring, we performed a laryngeal T-tube implantation.
The limitation of this study is that the sample size was relatively small, with only 7 patients in 8 years, so some of the results may contain large deviations. Thus, further studies with larger sample sizes are needed.
In conclusion, SCPL-CHEP can preserve a patient’s postoperative pronunciation function while completely removing laryngeal cancer lesions. The most recent phonatory rehabilitation after laryngectomy allows excellent vocal results even with total laryngectomy. On the other hand, organ preservation and the absence of a permanent tracheostomy improve patients’ quality of life, and the correct selection of patients also plays a fundamental role.30,31 It is undoubtedly a huge improvement for patients who have cancer cells simultaneously invading the glottic and supraglottic areas and who require total laryngectomy in the traditional sense. Coverage of the wound, accurate ring–tongue alignment and active anti-infection treatment after surgery are of great significance in preventing postoperative laryngeal stenosis. For some patients with laryngeal cancer simultaneously invading the glottic and supraglottic areas and who require postoperative radiotherapy, the probability of postoperative laryngeal stenosis will increase. Once laryngeal stenosis occurs and cannot be decannulated, the recurrence of the tumour should first be clarified. Under the premise of excluding tumour recurrence, the use of different surgical methods, initially under supported laryngoscopy, could remove the larynx organism to prevent secondary open injuries. If the curative effect is not good, then dilatation of the laryngeal stenosis or even laryngeal T-tube insertion can achieve satisfactory decannulation results.
Footnotes
Authors’ Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Yunnan Provincial Department of Science and Technology Grant. (40219004, 202101AY070001-051 and 2019J1185) and “Follow-up Fund Support for Famous Doctors” for high-level Talents in Yunnan Province.
Ethical Approval
This study was conducted in accordance with the Declaration of Helsinki and approved by the ethics committee of THE 1st Affiliated Hospital of Kunming Medical University, Informed consent has been obtained from all individuals included in this study.
Data Availability
All data generated or analysed during this study are included in this published article
