Abstract
Neuroendocrine carcinomas (NECs) are poorly differentiated neuroendocrine tumors of the upper respiratory tract. We present an extremely rare case of nasopharyngeal large cell neuroendocrine carcinoma (LCNEC) synchronized with nasopharyngeal squamous cell carcinoma (SCC). Both SCC and LCNEC are associated with Epstein–Barr virus (EBV) infection, supported by the positive result of Epstein–Barr encoding region in-situ hybridization. Strong correlation is found between EBV infection and nasopharyngeal malignancies. Furthermore, the EBV status might be a crucial prognostic factor in nasopharyngeal LCNEC. EBV-positive LCNEC is effective to chemoradiotherapy, and may have preferable outcome than EBV-negative LCNEC arising in the nasopharynx or other sites. The recognition of the EBV status is important for patients to receive appropriate treatment.
Introduction
The etiology of NEC in the head and neck regions corresponds to the anatomic location. Laryngeal NECs are highly associated with tobacco use, whereas oropharyngeal and sinonasal NECs are most likely to be associated with human papillomavirus (HPV) infection. For NECs arising from other locations of the head and neck, the contributive factors are still unknown.4,6 A subset of sinonasal small cell neuroendocrine carcinoma (SCNEC) harbors a high risk of HPV.7,8 Moreover, a subset of oropharyngeal and sinonasal large cell neuroendocrine carcinoma (LCNEC) harbors a high risk of HPV, although most patients have smoking history. 9 Rare cases of nasopharyngeal LCNEC are Epstein–Barr virus (EBV)-positive cases.1,4 Clinical manifestations of sinonasal NECs include nonspecific symptoms of nasal obstruction, epistaxis, or facial pain. Extension into the nasopharynx, skull base, orbit, or brain might present in poorly differentiated NECs with symptoms, including palpable facial mass and exophthalmia. 10 Paraneoplastic syndrome, which is a syndrome of imbalanced secretion of antidiuretic hormone, has been stated.11,12 The lungs, liver, and bone are the most frequent locations for distant metastases. 10
Nasopharyngeal SCNECs seem to have worse prognosis than conventional NPCs have, and the absence of EBV further distinguishes these tumors from conventional NPCs. 13 Poorly differentiated NECs of the head and neck region are highly threatening malignancies associated with high rates of metastases, with 5-year survival rates of < 20% according to previous research.2,6 Aggressive trimodality therapy is the most effective strategy, although survival is still poor. 10 Surgery with postoperative radiotherapy or concurrent chemoradiotherapy is considered the primary treatment for localized disease. For patients with systemic disease, palliative chemotherapy and foremost supportive care are the good choices. 14
We present the case of a mix nasopharyngeal LCNEC and squamous cell carcinoma (SCC). To the best of our knowledge, this is the second case reported in literature review. A previous report of the case of HPV-associated small cell carcinoma with synchronous SCC in the nasopharynx was reported in 2021. 15
Case Report
A 36-year-old man presented with right neck level-V mass (1 × 1 cm) for 1 week with mild tenderness. Accompanied symptom included bilateral aural fullness for months. He had no other significant medical or family history.
Computed tomography (CT) scan revealed ill-defined enhancing mass lesions at the right nasopharyngeal roof, causing Rosenmuller fossa obliteration and nasopharyngeal narrowing (Figure 1A). Additionally, enlarged lymph nodes at the right jugular chain were noted (Figure 1B). Biopsy was performed for microscopic evaluation. Computed tomography (CT) reveals an ill-defined enhancing lesion at the right nasopharyngeal roof (A) and enlarged lymph nodes at the right jugular chain (B).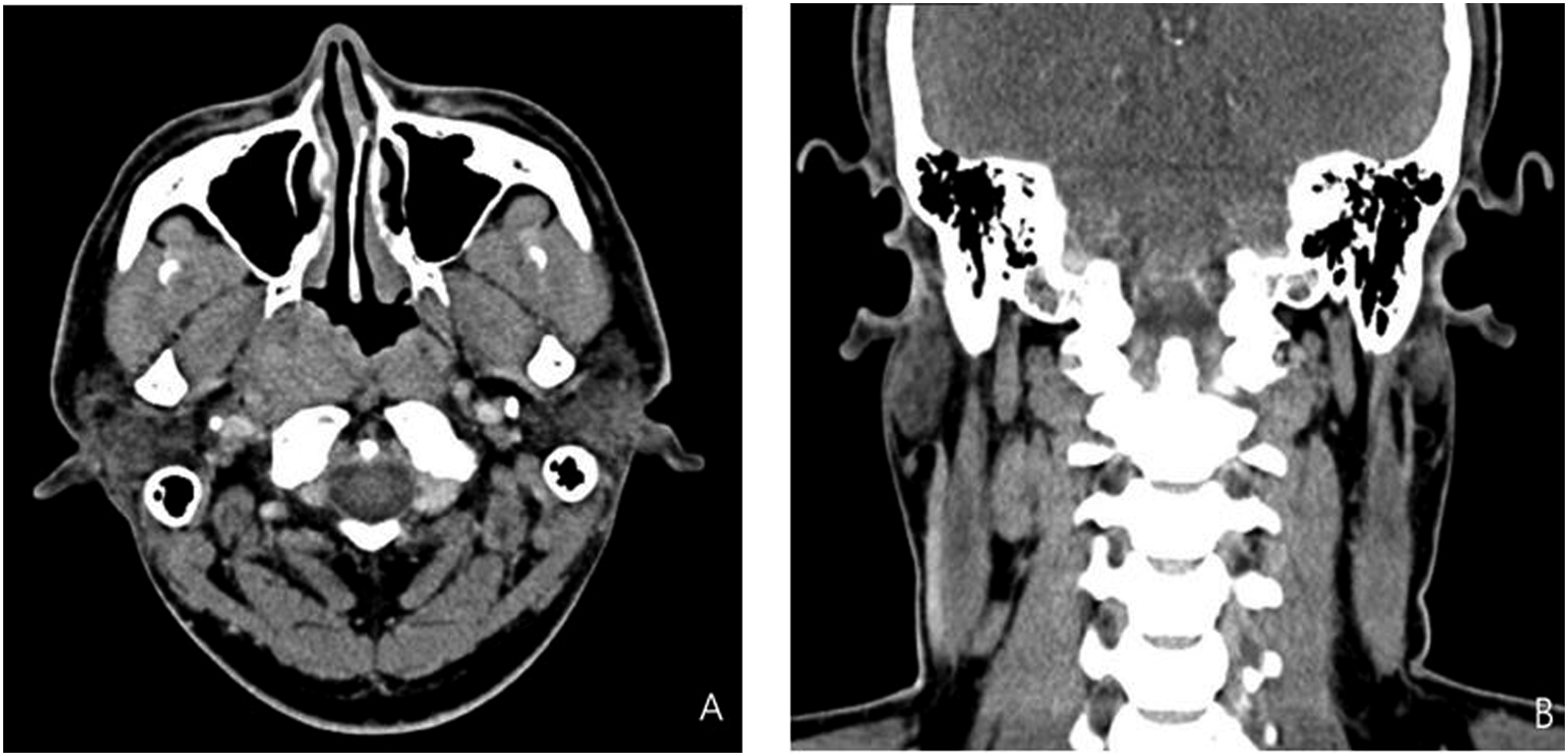
Microscopically, the tumor was composed of two components, including non-keratinizing SCC and LCNEC (Figure 2A). LCNEC showed a nested and trabecular pattern in the background of the desmoplastic stroma. Focal rosette formation was observed. Tumor cells had a moderate amount of cytoplasm, large pleomorphic nuclei, and 1–2 conspicuous nucleoli (Figure 2E). Ancillary immunohistochemical studies were performed. The SCC part was positive for p63, although it was negative for neuroendocrine markers (synaptophysin, chromogranin-A, CD56, and insulinoma-associated protein 1 [INSM1]) (Figure 2B,C). The LCNEC part was focally reactive with synaptophysin, CD56, and INSM1; however, it was negative for p63 and chromogranin-A (Figure 2B to H). Both SCC and LCNEC parts were diffusely positive for Epstein–Barr encoding region in-situ hybridization (Figure 2D). Histological and immunohistochemical morphology of mixed LCNEC and SCC, (A) H&E 100X, (B) p63 100X, (C) INSM1 100X, (D) EBER 100X).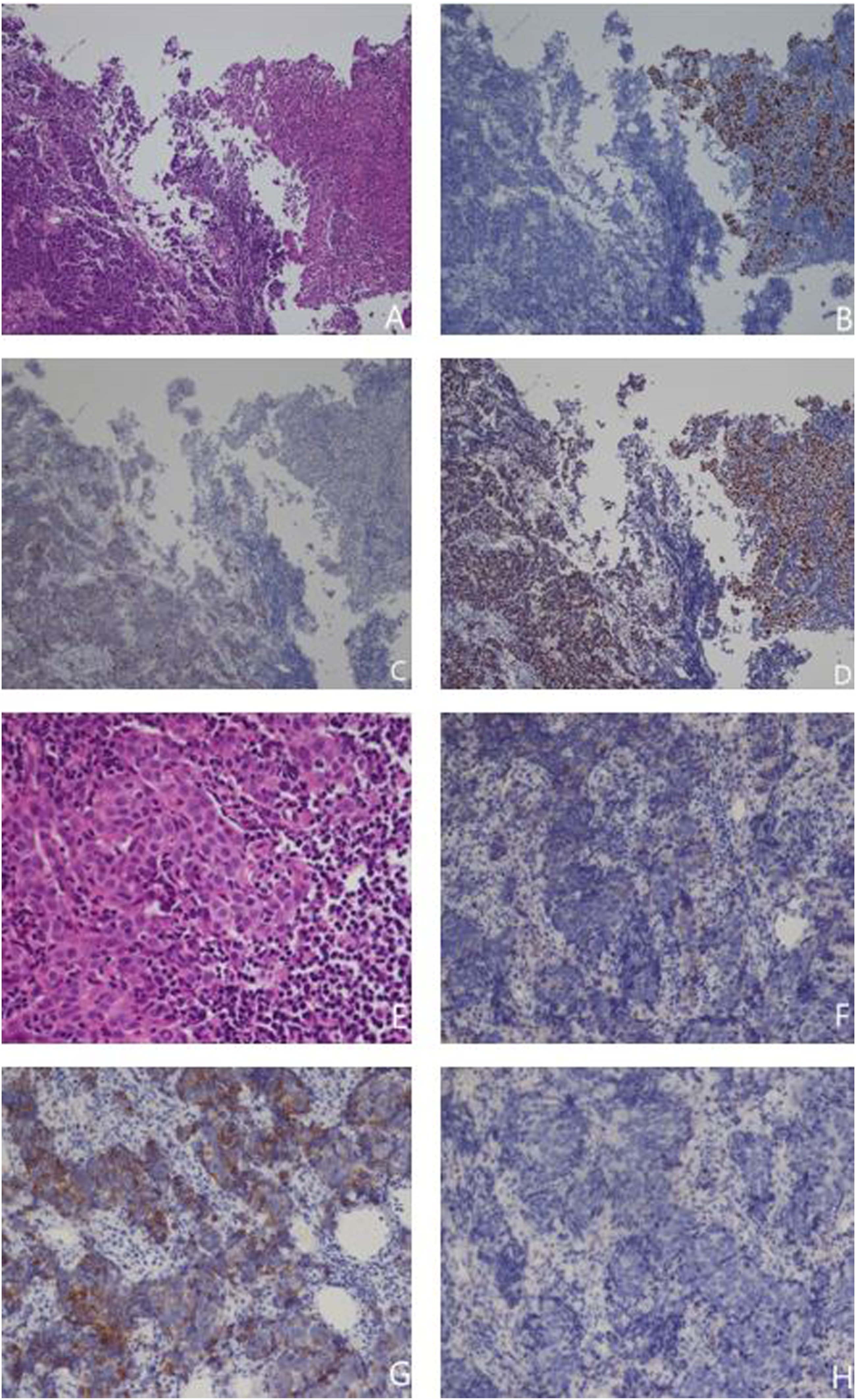
Discussion
The WHO classification of head and neck tumors classifies NENs into the following three categories based on cytomorphologic features and mitotic activity: well-differentiated NENs (grade 1 neuroendocrine tumors, typical carcinoid), which are tumors with minimal nuclear atypia and <2 mitoses/10 high-power fields (HPFs); well-differentiated NENs (grade 2 neuroendocrine tumors, atypical carcinoid), which are tumors with necrosis and/or 2–10 mitoses/10 HPFs; and poorly differentiated NECs, which include LCNEC and SCNEC, are tumors with >10 mitoses/10 HPFs. 5 SCNECs present with a very high nuclear-to-cytoplasmic ratio, evenly dispersed chromatin, and a lack of nucleoli. LCNECs have a relatively low nuclear-to-cytoplasmic ratio, coarse chromatin, frequent nucleoli and tumor necrosis, and immunoreactivity for neuroendocrine markers. 4 Traditional neuroendocrine markers, including synaptophysin, chromogranin, and CD56, are not frequently sufficiently sensitive or specific for diagnosis. INSM1 is a novel transcription factor that has recently demonstrated excellent sensitivity and specificity for neuroendocrine differentiation and may be used as a useful marker of neuroendocrine differentiation for head and neck tumors. 16
Distinguishing SCNECs from other small round cell tumors is important owing to their aggressiveness and poor response to therapy. However, this is often a challenge due to overlapping pathological features. Differential diagnosis includes NPC, basaloid squamous cell carcinoma (BSCC), olfactory neuroblastoma, melanoma, rhabdomyosarcoma, lymphoma, and Ewing sarcoma. Immunohistochemistry plays a remarkable role in distinguishing these entities, which differ in their management and prognosis. A limited panel, including pan-cytokeratin (AE1/AE3, positive in all epithelial tumors, with dot-like positivity in SCNEC), CK5/6 (positive in NPC and BSCC; negative in SCNEC), p63 (positive in all epithelial tumors except SCNEC), and HLA-DR (positive in NPC), could discriminate between most cases. While staining with neuroendocrine markers helps identify SCNEC, it is often focal or of weak intensity and may even be negative of neuroendocrine markers. Finally, another obstacle to diagnosing SCNEC is the lack of viable tumor tissue in small biopsies, leading to the need for multiple biopsies before final diagnosis is done. 17
NEC of the nasopharynx is very rare, and the majority are poorly differentiated NEC. EBV infection is strongly related to nasopharyngeal malignancies, including NPC and NK/T-cell lymphoma; this suggests that the virus has a unique tropism for the nasopharynx and is capable of infecting cells that can create histologically and immunophenotypically distinct entities. Non-keratinizing differentiated and undifferentiated NPCs are strongly associated with EBV, whereas basaloid and keratinizing NPCs are not related to EBV in nonendemic regions. 6 Moreover, limited reports are available suggesting that EBV-associated LCNEC also has a tendency to disseminate early to cervical lymph nodes, and patients often present with bilateral neck masses. 4 EBV-positive NPCs show remarkably improved response to treatment and better survival than EBV-negative malignancies. Furthermore, the EBV status might be a crucial prognostic factor in nasopharyngeal LCNEC. EBV-positive LCNEC is effective to chemoradiotherapy, and may have preferable outcome than EBV-negative LCNEC arising in the nasopharynx or other sites. Moreover, poorly differentiated NECs of the head and neck are associated with poor prognosis; it is unclear if the presence of EBV alters the outcome in these patients. EBV-positive tumors may respond favorably to local radiotherapy even in patients who present with extensive nodal disease. 4
Nasopharyngeal mixed neuroendocrine-non-neuroendocrine neoplasms (NP-MiNENs) are extremely rare with very few cases in the published literature. 5 In 2021, a patient diagnosed with secondary nasopharyngeal mixed adeno-NEC following radical radiotherapy for NPC was reported. 18 Histologically, these tumors have neuroendocrine and non-neuroendocrine tumor components. The neuroendocrine component is usually composed of NEC, and the non-neuroendocrine component can majority be represented by adenocarcinoma, SCC, adenosquamous carcinoma, or inverted papilloma. Nasopharyngeal MiNENs are typically aggressive cancers, and they are treated in the same way as poorly differentiated NECs are treated, with poor overall survival despite multimodal systemic therapy.
Nasopharyngeal NEC is a very rare entity with poor prognosis. EBV infection is associated with nasopharyngeal LCNEC, whereas no EBV-associated SCNEC has been reported yet. Compared with EBV-negative LCNECs, excellent response to chemoradiotherapy has been reported in EBV-associated nasopharyngeal LCNECs. NP-MiNENs are extremely rare with poor overall survival despite multimodal systemic therapy. The recognition of this rare entity and EBV status are important for patients to receive appropriate treatment.
Footnotes
Acknowledgments
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
The study was approved by the Ethics Committee of Mackay Memorial Hospital, Taipei, Taiwan (22MMHIS011e).