Abstract
Angiosarcoma is a rare primary tracheal-bronchus origin tumor which was only reported in a few cases. The diagnosis and treatment for such a rare tumor has long been a challenge. In this case, a male hemoptysis patient who was initially misdiagnosed with thyroid carcinoma with tracheal invasion was finally diagnosed with tracheal angiosarcoma. A thick-walled cystic nodular mass was found preoperatively in the right thyroid lobe overlying the tracheal angiosarcoma, which contributed to our initial misdiagnosis. Flap bleeding and necrosis occured after the surgery and patient continued to cough blood even when we excised the whole flap, which suggested the intramural growth and local invasion of the angiosarcoma. This study also reviewed previous literactures on airway angiosarcoma and discussed the treatment strategy.
Introduction
Angiosarcoma (AS) is a rare and highly aggressive malignant tumor originating from lymphatic or vascular endothelial cell. 1 It is an invasive tumor with a high rate of local recurrence and metastasis. 2 Generally, angiosarcoma can occur in any region of the body owing to its origin from endothelial cells. 3 The lung is the most affected by metastatic angiosarcoma. However, primary respiratory angiosarcoma has only been reported in few cases. 4
We reported a rare case with primary tracheal angiosarcoma. The patient underwent a total thyroidectomy as well as tracheal wall resection; supraclavicular artery island (SAI) flap was used to reconstruct the tracheal wall. The postoperative flap bleeding and necrosis was likely to be caused by angiosarcoma infiltration which was reported and discussed in detail.
Case Report
A 55-year-old man presented to our department of Otolaryngology Head and Neck Surgery for repeated hemoptysis for over 4 months. Though he had received the hemostatic medication and bronchial arterial embolization (BAE) in our respiratory department 2 months before this admission, his hemoptysis was not relieved. The patient was an occasional smoker with no history of drinking or illicit drug use. He was a farmer with no comorbidities and was not taking any medication on admission. Physical examination revealed a 5*6 cm tough, fixed, warm-to-touch right anterolateral neck mass. Thyroid bruit or cervical lymphadenectasis were absent.
Laboratory tests showed hemoglobin was 63 (120- 160) g/L. Lab coagulation test denied the existence of coagulopathy. Tumor markers were all within normal limits, including CEA, CYFRA21-1, NSE, and TPSA. The thyroid function tests revealed euthyroid status. Fine needle aspiration biopsy of the cervical mass indicated cyst fluid without any malignant findings. The thyroid color Doppler ultrasound found a 60 * 56 * 61mm hypoechoic area in the lower part of the right lobe, close to the trachea. Tumor shape was irregular with unclear border (Figures 1A and B). Contrast-enhanced cervical CT (computed tomography) revealed a cervical encapsulated mass (60 * 49mm) with liquid and calcifications squeezing adjacent structures (aerodigestive), apparent invasion was not reported (Figure 1C). Chest CT only revealed small areas of subtle ground-glass opacities in the right upper and bilateral lower lobes. (A and B) Preoperative Color Doppler ultrasound of the thyroid. (C) Preoperative contrast-enhanced cervical computed tomography of the neck. (D) Postoperative bronchoscopy 1 week after surgery.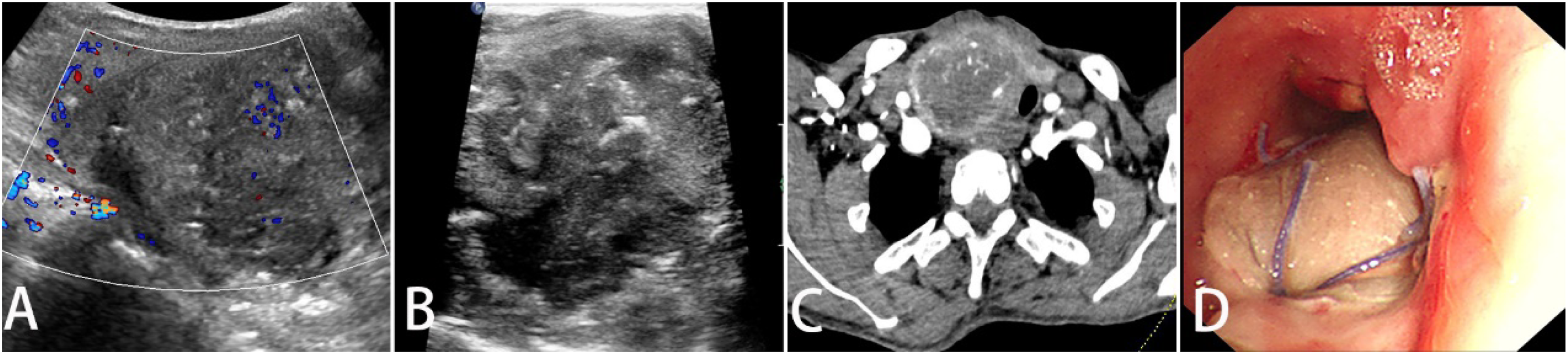
According to the patient’s medical history and test results, he was initially diagnosed with thyroid carcinoma with tracheal invasion. Radical thyroidectomy along with the resection of cancer-involved right lateral and posterior tracheal wall were performed. Intraoperative exploration found an 80 * 65 * 50 mm large, encapsulated, hemorrhagic tumor occupying the right lobe and a 25 * 15 * 8mm nodule in the trachea. A pedicled right SAI (Supraclavicular Artery Island) flap was used to reconstruct the tracheal defect. Tracheostomy was performed to maintain the airway patency. Intraoperative frozen pathology indicated right lobe nodular goiter with hemorrhage and cystic degeneration, while the tracheal wall mass was pathologically diagnosed as malignant tumor with uncertain origin. Hemoptysis was alleviated on the first day postoperatively and the hemoglobin rose from 63 g/L (before surgery) to 105 g/L on the sixth day after blood transfusion.
While awaiting the final pathology results, the patient began to cough blood from the tracheostomy tube on the 7th day after surgery, which was initially considered as flap bleeding. The bronchoscopy 1 week after surgery also revealed granulation tissue around the flap, supporting the diagnosis of flap hemorrhage (Figure 1D). Conservative management was implemented first. We performed multiple dressing changes and necrosis debridement, and eventually, we got the full flap excised. However, hemoptysis kept getting worse. Three weeks after the first surgery, the patient received an emergency surgery to stop the uncontrollable bleeding. Only granulation tissue was found on the right and posterior tracheal wall intraoperatively, no solid tumor was found.
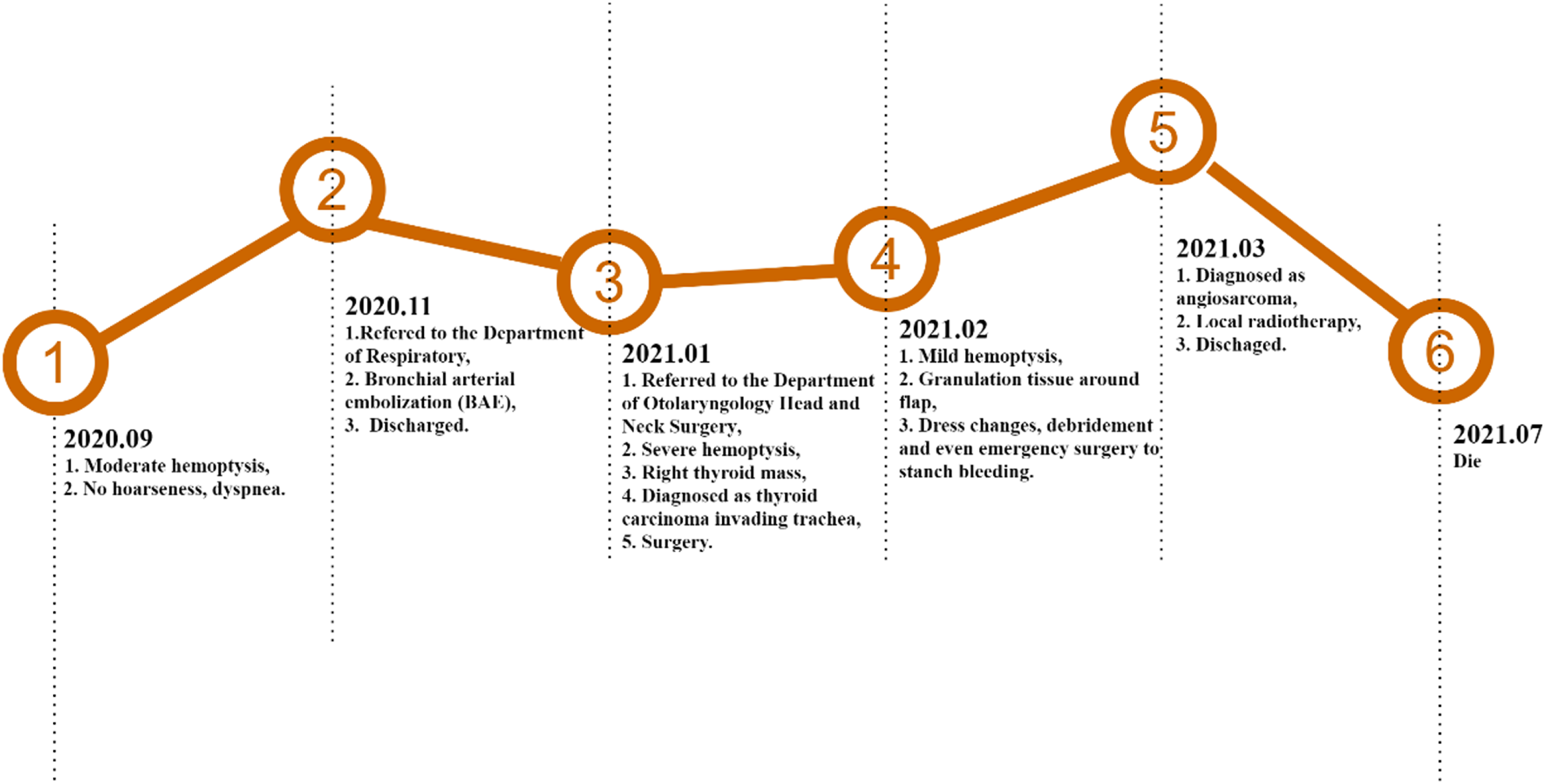
The final pathology results confirmed the diagnosis of tracheal angiosarcoma 28 days after the first surgery. The histological examination showed a soft-tissue neoplasm with a vascular channel lined by pleomorphic endothelial cells. Immunohistochemical staining (Figure 2) was diffusely positive for CD31 and the nuclear transcription factor Fli-1; focally positive for ERG, smooth muscle actin SMA, and P53; and negative for CD34. Therefore, the histology and immunohistochemistry were consistent with angiosarcoma. Though we scheduled local radiotherapy, the patient refused the treatment and insisted on discharge. Hemoglobin at discharge was 37 g/L. According to telephone follow-up, he died about 4 months after discharge. We made a detailed timeline to show the progress of his disease (Figure 3). Pathological manifestations of tracheal angiosarcoma. (A) Light microscope (HE, ×20). (B) CD31 positive by immunohistochemistry (×20). (C) ERG weakly positive by immunohistochemistry (×20). (D) Ki67 partially positive by immunohistochemistry (×20). (E) Fli-1 positive by immunohistochemistry (×20). (F) CD34 negative by immunohistochemistry (×20). The timeline of medical record of the reported case.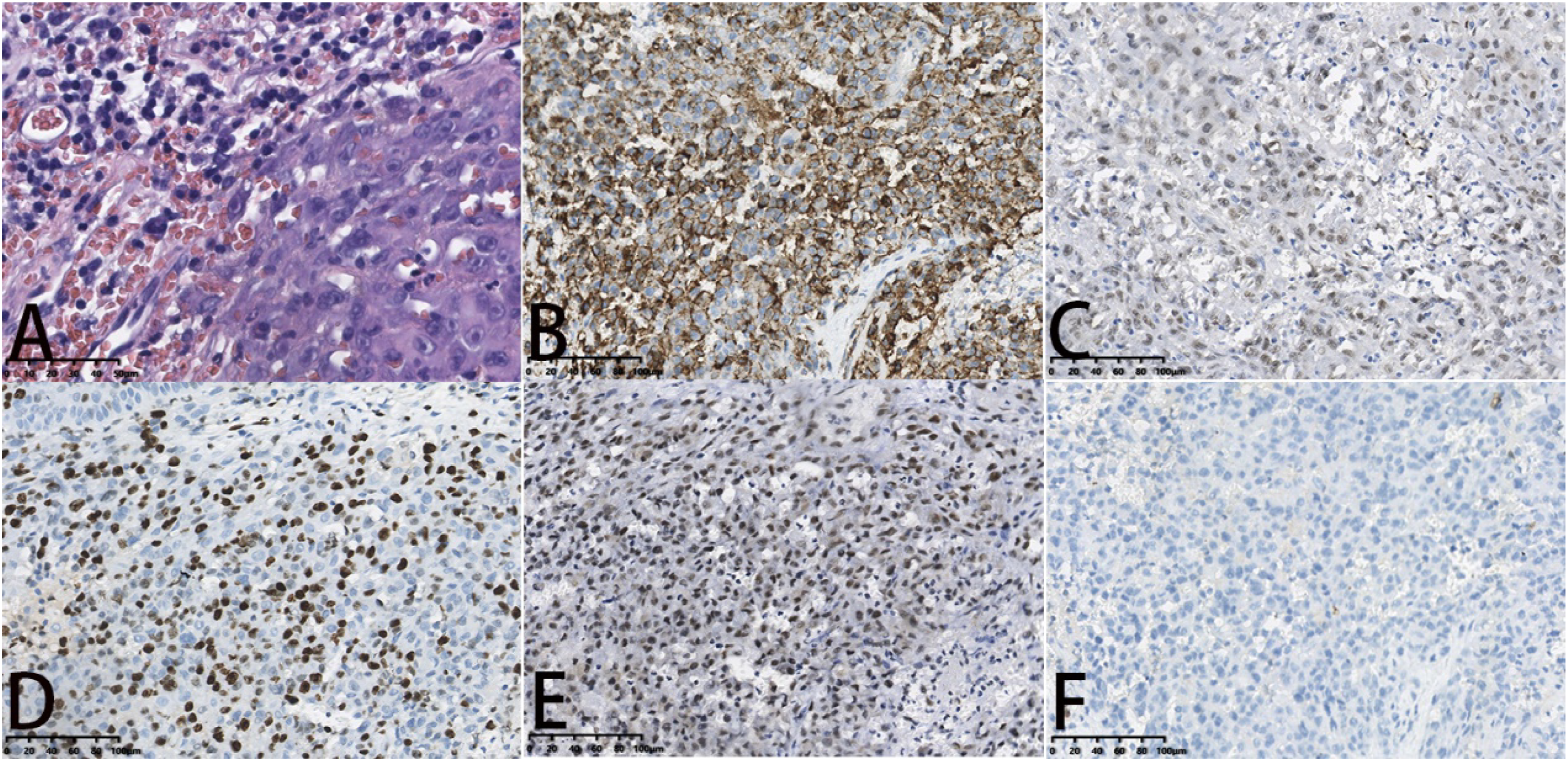
Discussion
Summary of Cases of Primary Trachea Angiosarcoma.

Alive: until 1 year after surgery
Early diagnosis of trachea angiosarcoma is hard because of the unspecific respiratory manifestations. Chest X-ray, chest CT, and bronchoscopy are helpful in dectecting the tumor, while histopathological and immunohistochemical examinations are essential for a definitive diagnosis. 4 In this case, the tracheal tumor lied beneath a huge nodular goiter, the endoscopy showed no obvious intraluminal tumor. So, it was initially misdiagnosed as an invasive thyroid cancer, and it was difficult to confirm the real causes of postoperative flap-associated bleeding and necrosis. According to our knowledge, the SAI flap may be a good option for selected head and neck reconstruction due to its reliability, ease of harvest, and favorable color match. Many large case series studies have praised its versatility and reliability in clinical settings, but the complications of SAI can be serious and can’t be ignored. Bleeding, flap necrosis and even total or partial flap loss were observed in some cases.10,11 Distal tip necrosis is common and could be attributed to an oversized flap or excessive tension applied during harvesting one inset under excess tension, which could be minimized by limiting flap dimension. 10 We didn’t find any report on tumor reccurence compromising the blood vessel thus causing complete skin necrosis. It is unlikely that the skin necrosis in our case was secondary to excessive flap harvesting, not only because the necrosis was not evident for 14 days after the operation but also because the bleeding was not stopped until the entire flap removal. Another piece of evidence that supported tumor-recurrence–caused flap necrosis is that once necrosed flap tissue was removed, flap bleeding decreased for one or two days during debridement.
Due to the rareness of angiosarcoma, no standardized therapy has been established yet. Surgery has been the mainstay treatment for locally confined disease. 4 However, for head and neck angiosarcoma, there is a high probability of local recurrence, even after extensive surgical excision. 12 The adjuvant radiotherapy following radical surgery was indicated to be the optimal combination for this disease.1,12 According to previous reports, 3 of five resected cases survived for more than 1 year after the surgery.6,8,13 This indicates that primary trachea angiosarcoma can be well managed by surgical resection if the tumor is localized with intraluminal growth as long as it’s detected in early stage. However, the present case documented the behavior of a malignant primary tracheal angiosarcoma, which is characterized by insidious intramural growth with extensive local invasion. So, we suggested the radiotherapy as the next-step treatment, but the patient refused any further treatment.
In summary, primary tracheal angiosarcoma is a rare malignant vascular tumor with an aggressive clinical course. Confirmatory diagnosis is made based on histopathological findings. The standard management of such disease remains to be established. Generally, the prognosis depends on the growth of the tumor. The angiosarcoma, as a differential diagnosis, should always be considered by the attending physician when treating patients with hemoptysis for unknown reason.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Written informed consent for publication of this paper was obtained from West China Hospital and all authors.
