Abstract
Radiation-induced malignancies (RIMs) are rare but well-documented late toxicities associated with exposure to radiation or radiotherapy. A keloid scar is a common benign proliferation of scar tissue which commonly develops at the site of an injury, such as on the earlobe after ear-piercing. While typically reserved for management of malignancies, radiotherapy is often utilized in the management of some benign conditions, including keloids. Given the benign nature of keloids, any theoretical late toxicity from radiotherapy, particularly a life-threatening toxicity such as a RIM, is particularly concerning. Here, we report a case of a 34-year-old male who presented with a radiation induced mucoepidermoid carcinoma of the parotid gland which developed in a previously irradiated field ten years after the patient received electron radiotherapy for a keloid of the earlobe. Using available literature, we estimate the risk of a RIM of the parotid gland from a typical course of radiotherapy to the earlobe as 0.007% per year.
Background
Radiation-induced malignancies (RIMs) are rare but well-documented late toxicities associated with exposure to ionizing radiation 1 or radiotherapy.2,3 The risk of a RIM, although low, is significantly greater in pediatric patients as the incidence of RIMs remains even up to 40 years after treatment. 3 The parotid gland is a common site for RIMs with mucoepidermoid carcinoma being the most common histology.4,5
Keloids are a common and benign dermatologic condition often seen in pediatric and adult populations. 6 Treatment of keloids is challenging as they tend to recur with recurrence rates as high as 50–80% after surgical excision. 7 Due to their tendency to recur, a variety of options are available to treat Keloids. These are often employed in the post-operative setting and include cryotherapy, radiofrequecy ablation, corticosteroid injection, and radiotherapy.8,9 Post-operative radiotherapy in the management of keloids is typically done with either kilovoltage photon (50 keV) or low energy (6 MeV) electrons en face with a dose ranging from 12 to 20 Gy in 3–5 fractions.10,11
Given the benign nature of keloids, any theoretical late toxicity from radiotherapy, particularly a life-threatening toxicity such as a RIM, is particularly concerning. Here, we report a radiation induced mucoepidermoid carcinoma of the parotid gland which developed ten years after an adolescent patient received electron radiotherapy for a keloid of the earlobe.
Case report
A 22-year-old African-American male, who had had his left ear pierced since adolescence, developed a keloid scar on the left earlobe immediately adjacent to the piercing site. This keloid was resected in 2009 and then followed with multiple corticosteroid injections. Two years later, at age 24, his keloid recurred. At that time, the keloid was noted to be 3 × 3.2 cm in size and extended to involve the posterior aspect of the left earlobe. The patient underwent a repeat excision which was followed by immediate post-operative radiation therapy, receiving 12 Gy given in 3 daily fractions of 4 Gy. Radiation was administered via a 6 MeV electron beam en face with treatment starting the same day as surgery. The treatment was well tolerated with no documented toxicities. The patient reports resolution of the keloid without recurrence.
Ten years later, in 2021, the patient (now 34 years old) noticed a nontender, soft lump present behind his left ear. He mentioned this lump to his primary care physician who monitored it for the following three months however the patient noticed the lump was growing significantly. He subsequently underwent a CT scan of the neck which showed a 2.3 cm nodule in the left superficial parotid gland. This prompted a referral to an Otolaryngologist who performed an ultrasound-guided core needle biopsy which identified a mucinous neoplasm positive for immune markers CK5/6, CK7, and p63 consistent with a low-grade mucoepidermoid carcinoma. The patient has no other medical history, takes no medications, has no known drug allergies, and has no pertinent surgical history. He is a daily marijuana smoker but denies any tobacco or alcohol use. He has no history of cancer in any first-degree relatives.
Soon after, the patient was referred to an oncologic head and neck surgeon. A MRI of the neck revealed an ill-defined heterogeneously-enhancing 3.6 cm superficial posterior left parotid gland mass with internal cystic changes. The mass appeared to be infiltrating the overlying cutaneous skin and was located just inferior to the external auditory canal, abutting the mastoid tip and insertion of the sternocleidomastoid muscle. Figure 1 contains representative images from this MRI scan. Notably, the tumor was immediately adjacent to the previously irradiated left earlobe. The patient also underwent a staging CT of the chest which was negative for metastasis. He then underwent superficial parotidectomy and left selective neck dissection, with surgical pathology reporting a low-grade mucoepidermoid carcinoma, sized at 3 cm in the greatest dimension, and with extra-parenchymal extension into the dermis identified. Surgical margins were negative and zero of eight lymph nodes were involved consistent with a pathologic stage III, T3N0, salivary gland cancer. Representative Axial (Left) and Coronal (Right) MRI T1 post-contrast MRI images showing left parotid tumor immediately adjacent to the left earlobe.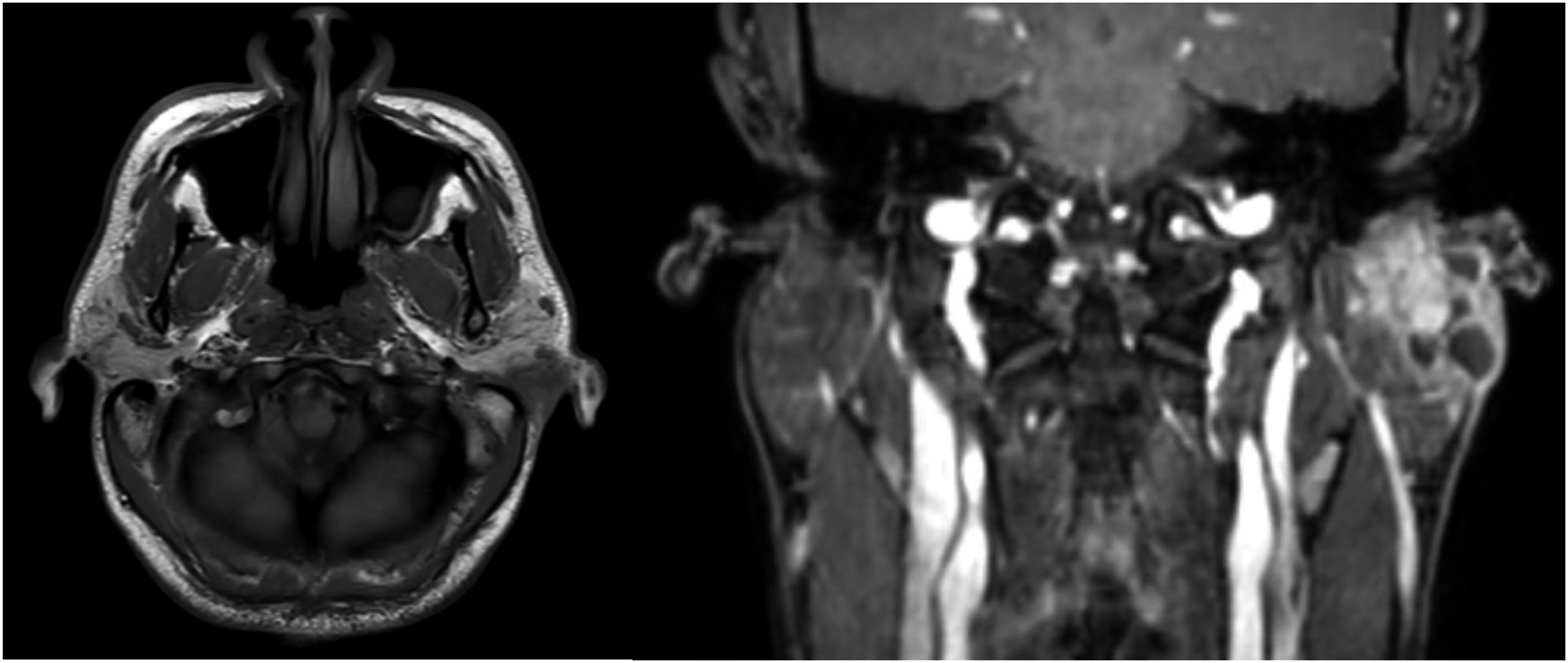
Post-operatively, this case was discussed at the institutional multidisciplinary tumor board and recommendation was for adjuvant therapy due to invasion of skin and thus referred to radiation oncology. He underwent CT simulation for radiation treatment planning and two weeks later adjuvant radiotherapy was initiated with a prescribed dose of 60 Gy administered in 30 daily fractions, targeting the surgical bed. However the patient only completed nine fractions (18 Gy) and was subsequently lost to follow-up.
Discussion
Salivary gland tumors are rare with an estimated incidence of 5 per 100,000 people per year. 12 An increased incidence of salivary gland tumors has been identified for patients exposed to ionizing radiation in the treatment of cancer 13 as well as in the treatment of benign conditions such as hyperthyroidism 14 and tinea capitis. 15 A retrospective analysis of nearly 3000 pediatric patients treated with ionizing radiation for benign conditions reported that 3% of these patients developed salivary gland tumors; however, the majority (75%) of these cases were benign neoplasms. 16 These pediatric patients were predominantly treated for enlarged tonsils and adenoids and no cases of keloid treatment were included.
RIM after radiation treatment for keloid is rare with only a limited number of case reports in the literature. A case series looking over a 100-year time frame (1901–2009) found only five documented cases of RIMs from radiation treatment of keloid, with only a limited documentation of dose or technique being available for most cases and with the extent of radiation field or dose being uncertain. 17 The majority of these RIMs developed in the skin in the radiated field although there were reports of development of suspected RIM in the breast and lung. There is one more recent documented case of a RIM which developed in a nearby organ at risk (OAR) from radiation treatment for a massive recurrent earlobe keloid, which developed in the parotid gland following multiple courses of radiotherapy. 18 RIMs after treatment of keloid using modern radiotherapy technique and dosages remain exceptionally rare. In fact, multiple registries investigating late toxicities in patients receiving radiotherapy for keloid failed to identify any induced malignancies in the radiation field.19-22
Radiation planning for keloid treatment is routinely done via a clinical setup with the beam angle and borders established with the patient in the treatment position on the linear accelerator couch.
10
A planning CT scan is typically not obtained for radiation treatment planning and dose to any nearby OARs is assumed to be negligible. As this patient subsequently developed parotid cancer and underwent radiation treatment post-operatively, he did undergo CT simulation for radiation treatment planning. In order to estimate the theoretical risk of RIM of the parotid gland from a keloid treatment, we simulated a 6 MeV electron field on this planning CT using an en face electron field to the contralateral (right) earlobe with a 5 mm bolus to a dose of 12 Gy given in 3 daily fractions of 4 Gy. Figure 2 contains representative images from this simulated keloid treatment. We estimate a typical keloid electron treatment (to 12 Gy in 3 fractions) will result in a maximum dose to the parotid of 102% of prescription dose (12.2Gy) with a mean parotid dose of 3.8 Gy. Representative Axial (Left) and Coronal (Right) CT images Depicting Radiation Treatment of Earlobe Delivering Dose to the Right Parotid (Outlined in Blue).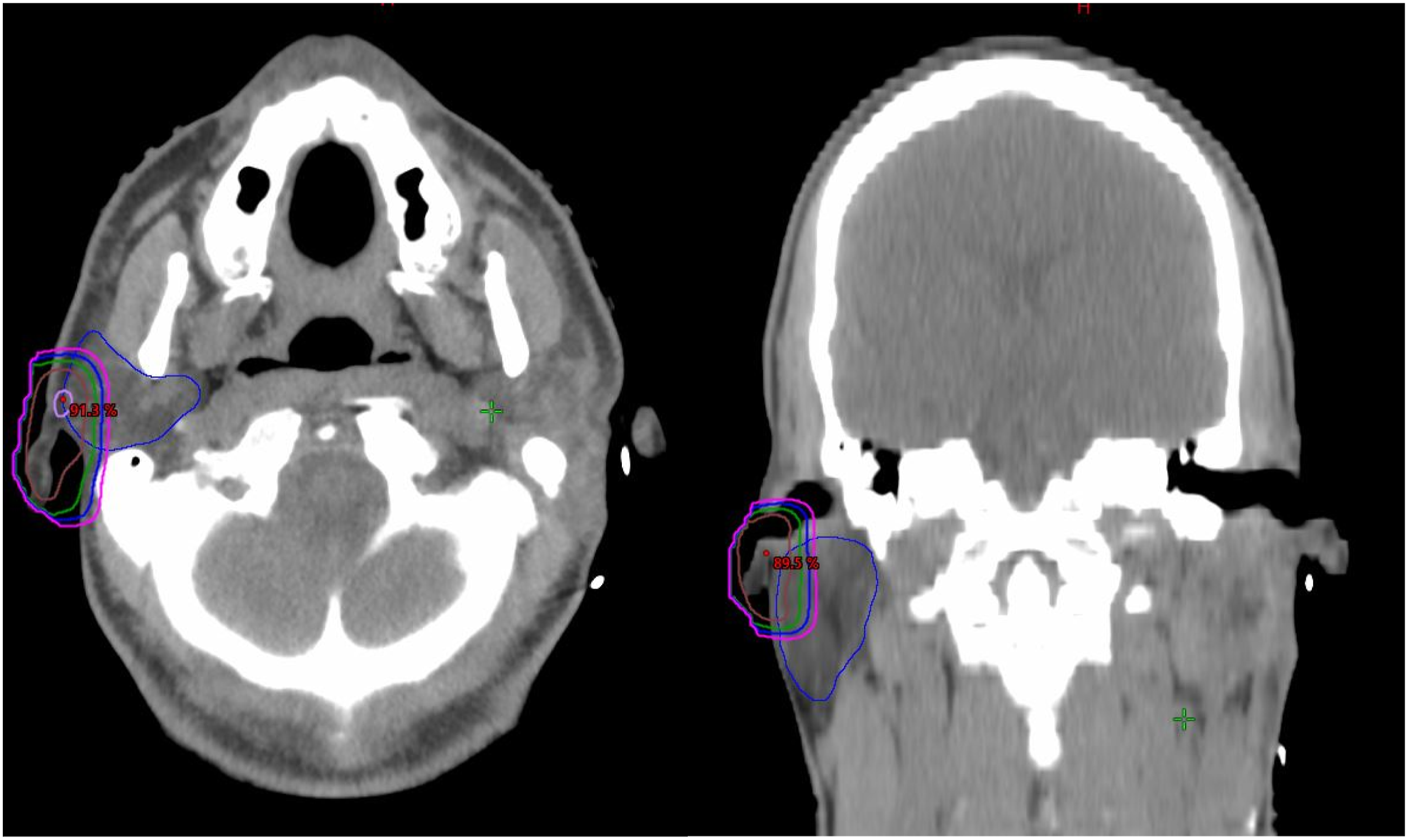
In order to estimate the increased risk for parotid malignancy from a routine keloid electron treatment to the earlobe, we assumed a linear relationship between mean parotid dose and risk of malignancy.16,23,24 Using prior reports of an increased relative risk per gray for all salivary gland neoplasms of 0.82 per gray, 16 with a mean ipsilateral parotid dose of 3.8 Gy this corresponds to a relative risk of 3.1. Using a baseline risk for salivary gland malignant neoplasms at 0.9 cases per 100,000 per year 12 and assuming three-quarters of all salivary gland neoplasms will involve the parotid gland, 25 we estimate the incidence of an ipsilateral parotid malignancy at .007% per year. The number of earlobe keloids needed to treat to induce a parotid cancer within 10 years is therefore roughly 14,000 patients.
Conclusions
Radiation therapy remains a widely utilized treatment in the management of keloid scars. Although rare, RIMs following a single course of radiotherapy, even at the lowest acceptable dose (12 Gy), to a small site (earlobe) can occur. When treating keloid patients with radiotherapy, care should be taken to minimize the dose and size of the radiation field to reduce dose to any non-target tissues. Patients should be consulted regarding the increased risk of RIM associated with radiotherapy, particularly in pediatric patients.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
