Abstract
Otoscopic Clinic
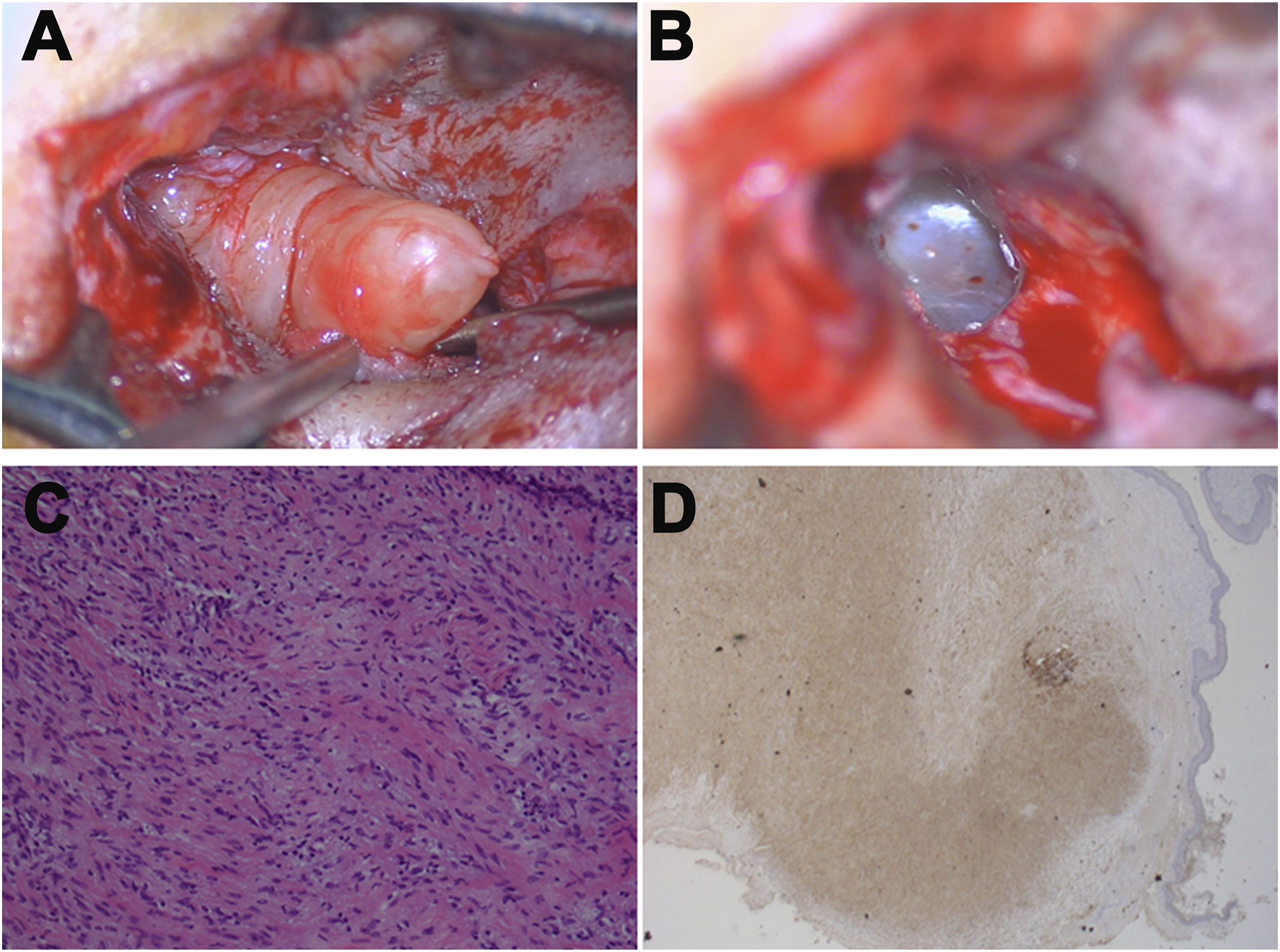
A 40-year-old man without systemic disease presented at our clinic with right-sided hearing impairment that he had experienced for 2 years. Otoscopy showed a dense and painless tumor occupying the space over his right external auditory canal (EAC) and extending inferiorly into the cartilaginous section of the EAC (Figure 1A). Pure-tone audiometry revealed conductive hearing loss at the level of 35 dB with a 20 dB air–bone gap over the right ear. High-resolution computed tomography (CT) of the temporal bone demonstrated a mild contrast-enhanced soft-tissue nodule of approximately 1.2 × 0.7 cm2 in the right EAC with intact tympanic membranes, a bony wall, and ossicular chains (Figures 1B and 1C). The tumor was excised under general anesthesia by making an endaural incision under microscopy. The incision was extended inferiorly because the tumor arose subcutaneously from the inferior wall of the cartilaginous part of the EAC. The tumor, measuring 2.3 × 0.8 × 0.7 cm3, was well circumscribed, and the eardrum was intact after the tumor was removed (Figures 2A and 2B). Histopathology showed a myxoid matrix including spindle cells and informal collagen bundles, with positive immunostaining for S100 consistent with pictures of neurofibroma (Figures 2C and 2D). The incision was loosely sutured using nylon, and the wound over the EAC was packed with Gelfoam for 2 weeks and left to heal. The patient’s hearing recovered without EAC stenosis or obstruction. No evidence of recurrence had appeared for 3 years postsurgery. (A) Upon otoscopic examination, a dense and painless mass occluded the right EAC. (B) Axial and (C) coronal view in high-resolution CT of the temporal bone revealing a soft-tissue nodule over the right EAC. Abbreviations: EAC, external auditory canal. (A) Intraoperative view of the fusiform-shaped tumor. (B) Tympanic membrane visible after removal of the tumor. (C) Hematoxylin and eosin staining revealing myxoid matrix containing neoplastic cells and informal collagen bundles (X200 magnification); (D) Positive immunostaining for S100 (×40 magnification).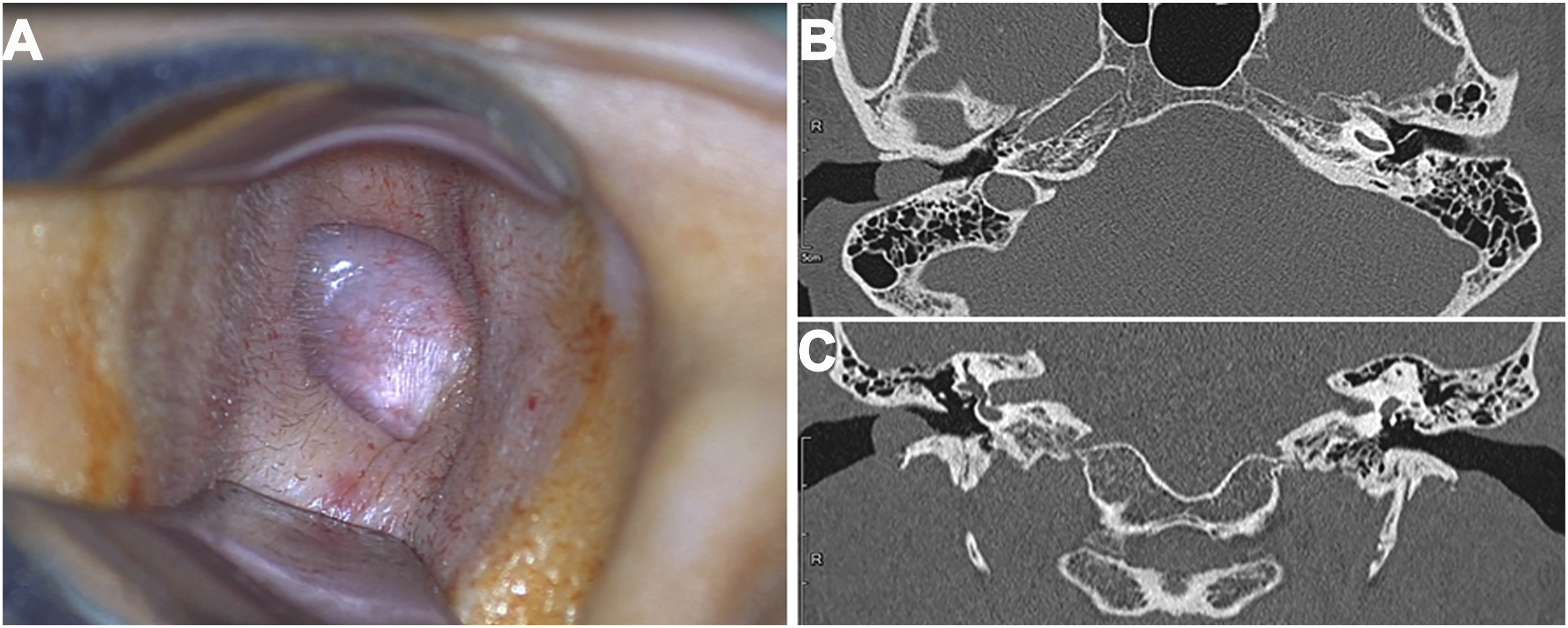
Neurofibromas, relatively benign tumors within the nervous system, are derived from perineural cells such as Schwann cells, fibroblasts, and supporting cells. 1 They can be found in any part of the body where nerve cells a and are classified as either solitary or multiple. 2 Histological subtypes of neurofibromas include localized, diffuse, pigmented, and plexiform. Plexiform neurofibromas are more difficult to treat and have greater potential of transforming into malignant tumors compared with the other subtypes. 2,3 Patients who are not diagnosed as having neurofibromatosis (NF), a widespread inherited disorder that can has 2 types (NF1 and NF2), may have a single or few neurofibromas. 4 Our patient had a solitary, sporadic, and localized cutaneous neurofibroma arising from the EAC but did not present any other symptoms of NF1.
Although EAC neurofibromas are superficial lesions that can be clinically evaluated, CT or magnetic resonance imaging (MRI) are preferable evaluation modalities. 3,5 CT can help doctors determine the extent and bony EAC involvement of the lesion, and MRI can detect highly vascular lesions and differentiate among the local, plexiform, and diffuse varieties of neurofibromas. 5 The differential diagnosis includes schwannoma, neuroma, myxoma, lipoma, and malignancy peripheral nerve sheath tumors. 3
Neurofibromas are treated through surgical removal. Studies have suggested that subtotal resection or palliative treatment is preferred over complete excision because the tumors are often ill defined, diffuse, and frequently recurring, especially rare, malignant neurofibromas. 2,6 By contrast, some recently published reports have presented successful cosmetic and overall outcomes after complete eradication. 1,6 Most EAC tumors can be excised through a transcanal or transmeatal approach under microscopy, but larger lesions should be excised by performing endaural incision or postauricular incision. 2 Several authors have advised the use of wide meatoplasty to prevent reobstruction. 5 In the present case, we removed the entire well-defined tumor and some skin and performed the primary suture without meatoplasty or skin graft. No recurrence, EAC stenosis, or obstruction was found during the 3-year follow-up appointment.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Tri-Service General Hospital, Grant Number TSGH-D-110081.
Patient’s Consent
The patient’s identity is not identifiable in the figures or research information. The patient consent statement is not included.
