Abstract
Inflammatory myofibroblastic tumors (IMTs) are one of the most complex and rare neoplasms that have been discovered, with varying behavior in different cases. They mostly arise in the thoracic and abdominal cavity, the lungs, retroperitoneum, and extremities. They can also be detected in the head and neck area. IMTs have no age or gender preference which makes them tougher to anticipate. Head and neck IMTs are benign neoplasms with locally aggressive behavior and a low risk of metastatic spread. They have an unknown etiology, and they resemble malignant lesions radiologically. In this case report, we will review the case of a 40-year-old woman with an unusual IMT in the maxillary sinus that presented as a challenge in diagnosis and management.
Introduction
Inflammatory myofibroblastic tumor (IMT) has been, since 1994, defined as an intermediate-grade soft tissue myofibroblastic neoplasm by the WHO. 1 It consists of myofibroblasts, fibroblasts, and inflammatory cells due to which its etiology remains disputed and unclear. Due to the aforementioned varied components, IMT is also known as inflammatory pseudotumor, plasma cell granuloma, benign myofibroblastoma, xanthogranuloma, fibrous histiocytoma, and atypical fibromyxoid tumor. 2 IMT might present as a pure inflammatory process or as a myofibroblastic manifestation. Histologically, in a loose myxoid or edematous stroma, IMT is constituted of a varied admixture of fascicles of myofibroblastic spindle cells with a conspicuous polyclonal infiltrate of many plasma cells, lymphocytes, and acute inflammatory cells. 3
IMT is a very rare occurrence, observed in fewer than one person in a million. In the United States, between 150 and 200 persons are diagnosed with cancer each year. 4 There has been an equal prevalence of IMT in men and women, with no age risks; however, cases of extrapulmonary sites have been known to appear during childhood. 5,6 It can strike anyone at any age, from newborns to patients in the seventh decade of life. It affects a considerable number of young patients, accounting for 20% of all primary lung cancers in this age group. So far, less than fifty cases of head and neck IMT have been described, with the maxillary sinus being the least common. 7 IMTs of the head and neck have a 10%‐20% locoregional recurrence rate. Metastases are also documented, in about 5% of the cases. 8
IMT is mostly found in the lungs, abdomen, retroperitoneum, and extremities, while it is less common in the head and neck region. 9 Extrapulmonary IMTs in the head and neck area account for 14‐18% of all extrapulmonary IMTs, where they are most frequently found in the orbit. 4 They have been found in the larynx, tonsils, parapharyngeal region, nasal cavity, and maxillary sinus, among other places. 5,9,10 The most common signs and symptoms of sinonasal IMT include nasal obstruction, nasal mass or swelling, numbness, and trismus. 11
The pathogenesis, although unclear, is often indicated as an abnormal immune reaction by various reports. 1 It behaves like an aggressive lesion that causes a lot of damage. IMT is difficult to diagnose and treat; an inflammatory myofibroblastic tumor in the head and neck (HNIMT) accounts for 5% of all IMTs. 12 It is easily misinterpreted as malignant epithelial or mesenchymal spindle cell neoplasm. 5,9
We will be discussing a case of IMT arising within the maxillary sinus in a 40-year-old woman.
Case presentation
A 40-year-old housewife, a resident of Afghanistan, presented to otolaryngology clinic in December 2020 with right-sided facial swelling, gradually progressive for last 6 months along with ipsilateral, persistent nasal obstruction for 2 months. There were no complaints of rhinorrhea, epistaxis, postnasal drip, altered sense of smell, decreased hearing, visual problems, or headache. The facial swelling was associated with dull aching right-sided facial pain, aggravated on bending the head forward. On examination, there was obvious facial asymmetry, with a well-defined, non-reducible, non-fluctuant swelling on the right cheek, measuring approximately 4 × 5 cm in size. It was mildly tender, fixed to the underlying structures but not adherent to the overlying skin. A local rise in temperature was also noted in comparison to the surrounding tissue. Intraorally, a firm swelling was seen extending from the right superior buccogingival sulcus up to the hard palate, not crossing the midline.
On anterior rhinoscopy, deviated nasal septum on the right side of the nasal cavity was observed. Nasal endoscopy was performed. The rest of the examination was unremarkable.
Magnetic resonance imaging (MRI) showed a lesion with complete opacification of the right maxillary sinus which caused erosion of the anterior wall. PET CT (positron emission tomography—computed tomography) (Figure 1) scan of the patient revealed a 3.6 × 4.7 × 4.3 cm mass in the right maxillary sinus with SUV max of 5.57. Anterior infiltration of the premaxillary space caused a forward bulge of the overlying skin. Postero-laterally, the mass caused bony erosion of the posterior wall without extension into the infratemporal fossa. There was no metastatic lesion in the body. PET scan of patient showing lesion of 3.6 × 4.7 × 4.3 cm infiltrating mass in the right maxillary sinus with SUV max of 5.57.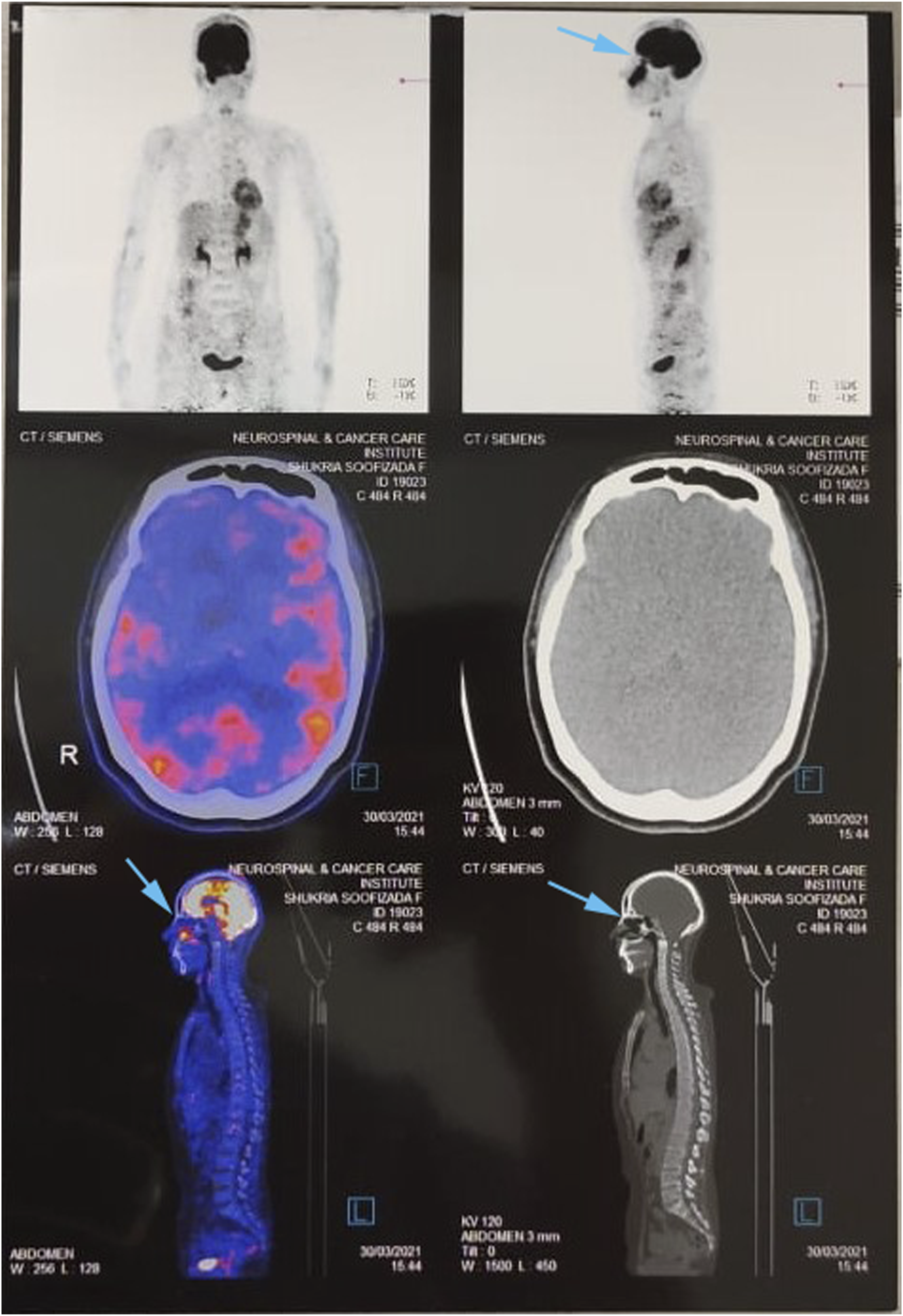
The patient underwent an endoscopic biopsy of the right maxillary sinus. Intraoperative findings included fleshy mass arising from the right maxillary ostium for which multiple biopsies were taken. The diagnosis was confirmed as a spindle cell neoplasm, favoring sarcoma: intermediate grade with myogenic differentiation (Figure 2). Histopathology indicated the mass to be alpha-smooth muscle actin (ASMA) patchy positive with high Ki-67, and positive P-63. All bone and soft tissue margins were clear. ASMA is an immunohistochemical antibody, identified in smooth muscle cells and myofibroblasts.
13
High magnification of the tumor showing sheets of spindle shaped neoplastic cells with elongated vesicular nuclei (arrow), moderate cytoplasm, and indistinct cell borders.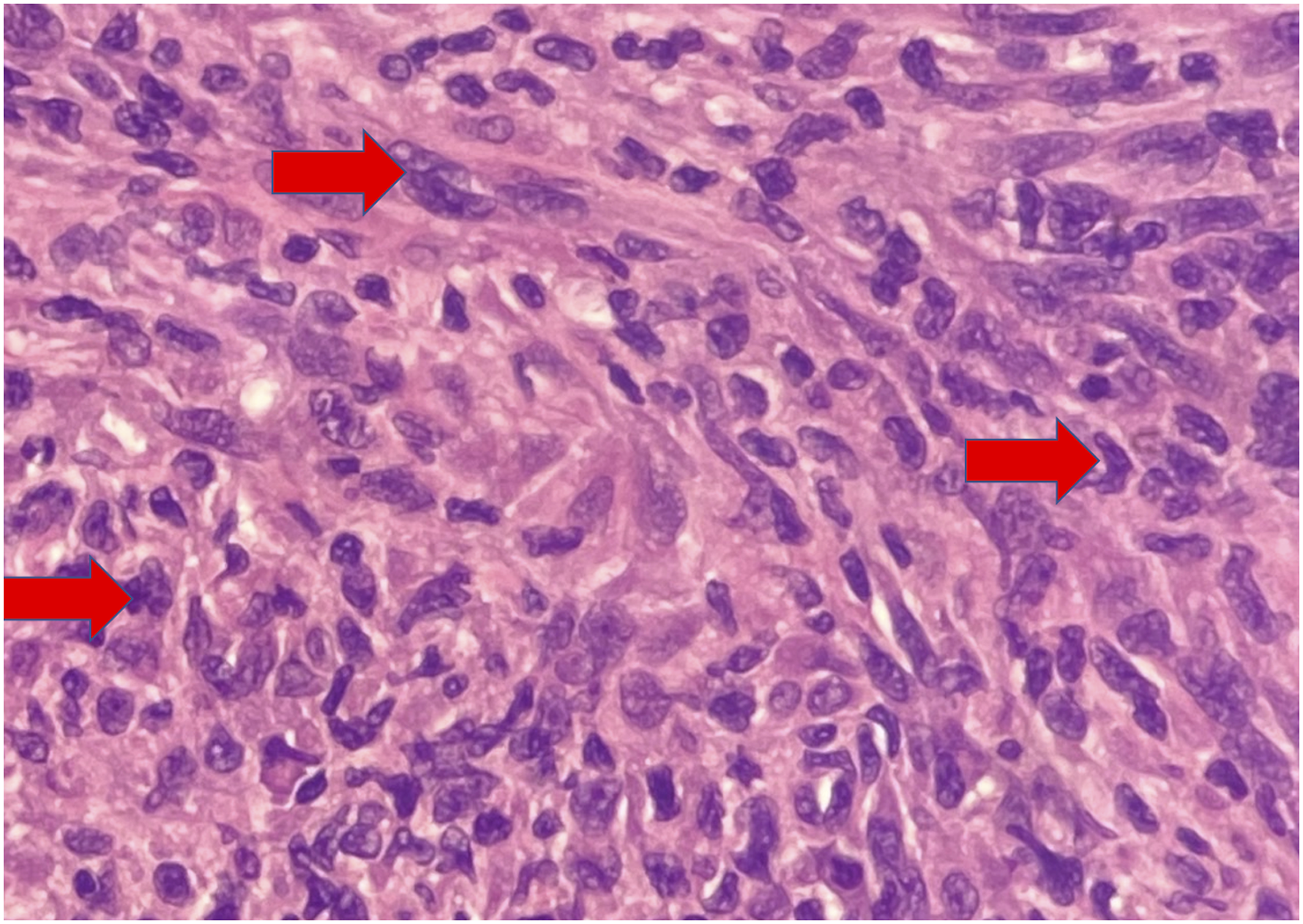
Ki-67 expression is tightly linked to tumor cell proliferation and growth, and it is commonly utilized as a proliferation marker in regular pathological studies. ALK was repeated twice but came back as “negative.” The anaplastic lymphoma receptor tyrosine kinase gene is abbreviated as ALK. This test detects ALK gene rearrangements in cancer cells. These modifications increase the likelihood that a person may react to targeted medication therapy. 14 After the initial biopsy, sarcomatoid carcinoma was also considered in the differentials; however, the former was favored as the final biopsy revealed inflammatory myofibroblasts. A multidisciplinary team meeting, involving the otolaryngology and head and neck surgery, radiology, oncology, and pathology services, agreed that the patient should undergo workup to rule out any metastasis, followed by surgical removal and concomitant chemoradiation (CCRT).
The patient underwent right total maxillectomy with reconstruction using anterolateral thigh (ALT) flap, through a Weber‐Fergusson incision and the lip-splitting approach. Intraoperative findings revealed a lesion extending superiorly into the right orbital cavity but not involving the orbital contents. There was no periorbital fat involvement, muscle involvement, or inferior orbital nerve involvement. There was an erosion of the bony floor but there was no involvement of the periosteum. The underlying mucosa appeared inflamed and granular. The patient remained stable throughout the hospital stay and was discharged on the fifth post-operative day. The functional outcome patient had normal speech with no nasal regurgitation of food and liquids. She could chew and swallow a soft diet at 6 months of follow-up. There was no local recurrence on clinical and nasal endoscopic examination on a 10-month follow-up.
Discussion
Inflammatory myofibroblastic tumor has been known to gather a considerable amount of attention from professionals owing to its peculiar nature concerning its rare occurrence as well as diagnostic, therapeutic, and pathophysiological challenges. Although not many, the cases reported with this diagnosis have one thing in common: the hurdles in achieving a clear identification of the diagnosis and exact etiology, due to the variations in the cytogenic expression of immunohistochemical markers. 15
ALK-1 is both diagnostically and therapeutically significant. It is amongst many cellular markers found to date such as desmin, vimentin, smooth muscle actin, cytokeratin, etc. ALK-1 differentiates IMT from spindle cell neoplasms also referred to as “inflammatory pseudotumors.” 15 The neoplastic tumors of IMT such as a calcifying fibrous tumor, nodular fasciitis, fibromatosis, and infantile fibrosarcoma are negative for ALK-1, and are more aggressive and metastasize at a far more rapid rate than the ALK-1‐positive neoplasms. 16
The surgical margins, tumor size, ALK-1 levels, and necrotic status all influence the prognosis of IMTs. 17 The disease prognosis is also influenced by its anatomic location. The nose and sinus regions are more aggressive in contrast to other regions of the body. Lung and bladder tumors have a better prognosis than other sites. 18
A small number of case reports have covered a variety of treatment options, yet there is no consensus guideline for the management of head and neck IMTs. 19 Currently, the treatment of choice is complete surgical excision. 10,15 Different combinations of surgery, steroid therapy, radiation, and chemotherapy, as well as observation alone, were employed in the literature. Some have also used NSAIDs, COX 2 inhibitors, and tyrosine kinase inhibitors alone or in combination. In one review, prednisone therapy was found to be associated with an improved overall survival rate (p = .046). However, more understanding of the treatment is required. 20 A study also reported a complete clinical-radiological response of an IMT of the maxillary sinus treated with steroids and radiotherapy. 21
Conclusion
Despite the availability of several alternative treatments such as chemotherapy, steroid administration, and radiation therapy, complete surgical excision of the tumor remains the dominant treatment modality for IMT of the maxillary sinus with adequate rehabilitation for a functional and good esthetic outcome. Our case has the distinction of having no relapse of the disease after only complete surgical excision without any steroid therapy or radiation, which to the best of our knowledge was not seen in any previous literature.
Footnotes
Acknowledgments
We would like to acknowledge the kind review and comments of Dr Asiya Kamber Zaidi, MBBS, MS, FWANS, Associate Fellow FACS Chair, W.A.M.S National Council of India and Dr Syed Akbar Abbas, MBBS, FCPS, Assistant Professor and Consultant as well as Director Otolaryngology and Head and Neck Surgery Aga Khan University Hospital, Karachi, Pakistan
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Verbal informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
Statement of Human and Animal Rights
All procedures in this study were conducted following the guidelines of the Helsinki Declaration.
