Abstract
Plasmablastic lymphoma (PBL) is an aggressive, rare variant of B-cell lymphoma typically associated with human immunodeficiency virus and other immunocompromised populations. Most commonly found in the oral cavity, PBL can occasionally originate in the sinonasal tract. Diagnosis of PBL is difficult due to overlapping features with other malignancies; however, early detection and treatment are imperative given its aggressive clinical course. When in the sinonasal tract, the diagnostic process can be further complicated if the patient has a history of recurrent nasal polyposis. Described is the case of a 57-year-old immunocompetent male who initially presented with benign nasal polyposis, only to return a year after sinus surgery with a unilateral sinonasal mass consistent with PBL. As literature has yet to characterize this phenomenon, this article presents the first case reported of sinonasal PBL arising in the setting of recurrent nasal polyposis. This case emphasizes the importance of investigating sinonasal masses showing laterality, maintaining a high index of suspicion for malignancy, and keeping close surveillance of the patient after treatment of PBL.
Keywords
Introduction
Plasmablastic lymphoma (PBL) is a rare subtype of non-Hodgkin’s lymphoma that is associated with an aggressive clinical course, poor prognosis, and frequent relapse. While classified as a variant of B-cell lymphoma, this disease originates during an activated B lymphocyte’s differentiation toward a plasmablast. Subsequently, the tumor cell loses its B cell immunophenotype, yielding the markers and morphology similar to a plasmablast. The incidence of PBL is higher in immunocompromised populations, most notably people with human immunodeficiency virus (HIV), who make up 69% of cases, whereas about 20.6% of cases are immunocompetent. 1,2 However, a patient’s HIV status likely does not confer any prognostic differences, with median overall survival at 9–12 months. 3 Studies have also found evidence of Epstein–Barr virus (EBV) in 50% of HIV-negative cases, suggesting a role in PBL’s pathogenesis. 4
This unique B cell malignancy has been described most often in the oral cavity, lymph nodes, other extranodal sites, and, in 12% of HIV-negative cases, the sinonasal cavity. In this same study, 60% of these HIV-negative patients presented in either stage 3 or 4. 5 Since the first reports of PBL by Delecluse et al in 1997, 6 PBL has not been described in the setting of nasal polyposis. As difficult as it is to diagnose PBL accurately with its similar immunoprofile to other malignancies, when it presents in between episodes of nasal polyposis, it can hinder the diagnostic process and potentially delay time sensitive treatment. This article presents the first case reported of a sinonasal PBL emerging in the setting of recurrent nasal polyposis and highlights the lessons to be learned for future patients.
Case Report
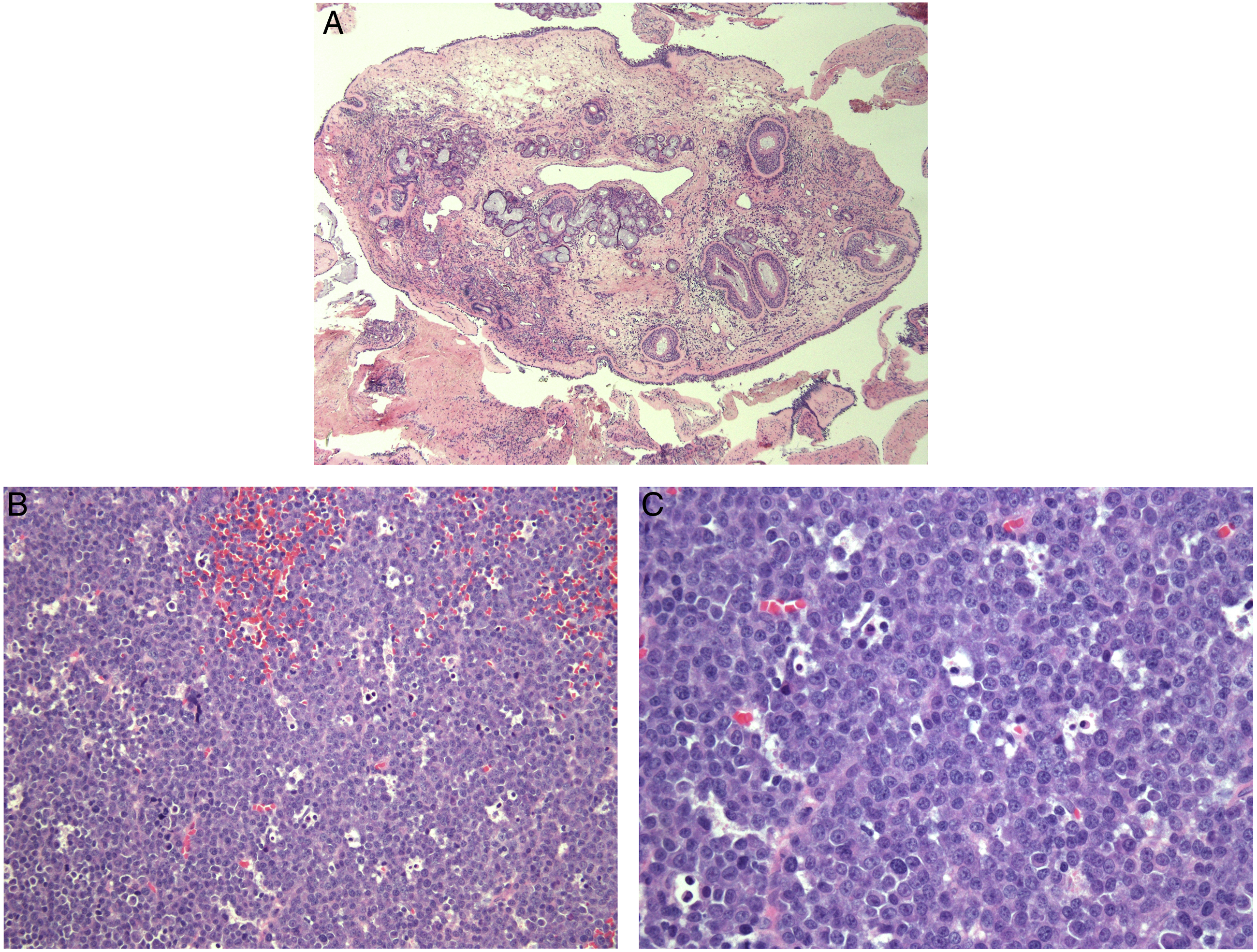
A 57-year-old male presented with a longstanding history of nasal obstruction and recurrent sinusitis. There was no history of HIV, organ transplantation, or systemic immunosuppression. He complained of persistent bilateral nasal blockage despite treatment with multiple antibiotics, nasal steroids, and antihistamines. Allergy testing revealed severe weed allergies. Computed tomography (CT) of the sinuses demonstrated bilateral polypoid disease in the maxillary sinuses and nasal airways (Figure 1A). The patient then underwent his first endoscopic sinus surgery and polypectomy, finding diffuse nasal polyposis in the ethmoid, maxillary, sphenoid, and frontal sinuses bilaterally. Bilateral sinus contents were consistent with benign, chronically inflamed respiratory mucosa. (Figure 2A). (A) Coronal section of a sinus CT, showing bilateral polypoid disease in the maxillary sinuses and nasal airways. (B) Repeat sinus CT 1.5-year status post sinus surgery. Coronal section reveals a 5.1 × 4.5 cm, left-sided polypoid mass involving the maxillary and ethmoid sinuses. The mass is seen causing a demineralization of the left maxillary sinus floor and nasal septum, destruction of the middle and inferior turbinates, and bowing of the septum to the right. (C) Repeat sinus CT 1-year status post removal of PBL tumor, demonstrating diffuse soft tissue density within the maxillary sinuses and remineralization of the left maxillary sinus floor. (A) Hematoxylin and eosin staining of benign nasal polyps, showing presence of mature lymphocytes, plasma cells, and eosinophils (40×). (B) Histopathological appearance of plasmablastic lymphoma with H&E staining, displaying large sheets of dyshesive cells with eccentric nuclei and prominent nucleoli, scant cytoplasm, and scattered tingible macrophages, ultimately imparting the “starry sky” pattern (200×). (C) Mature section of PBL with H&E staining, exhibiting plasmacytic morphology including eccentric nuclei, prominent nucleoli, and pale perinuclear cytoplasm (400×).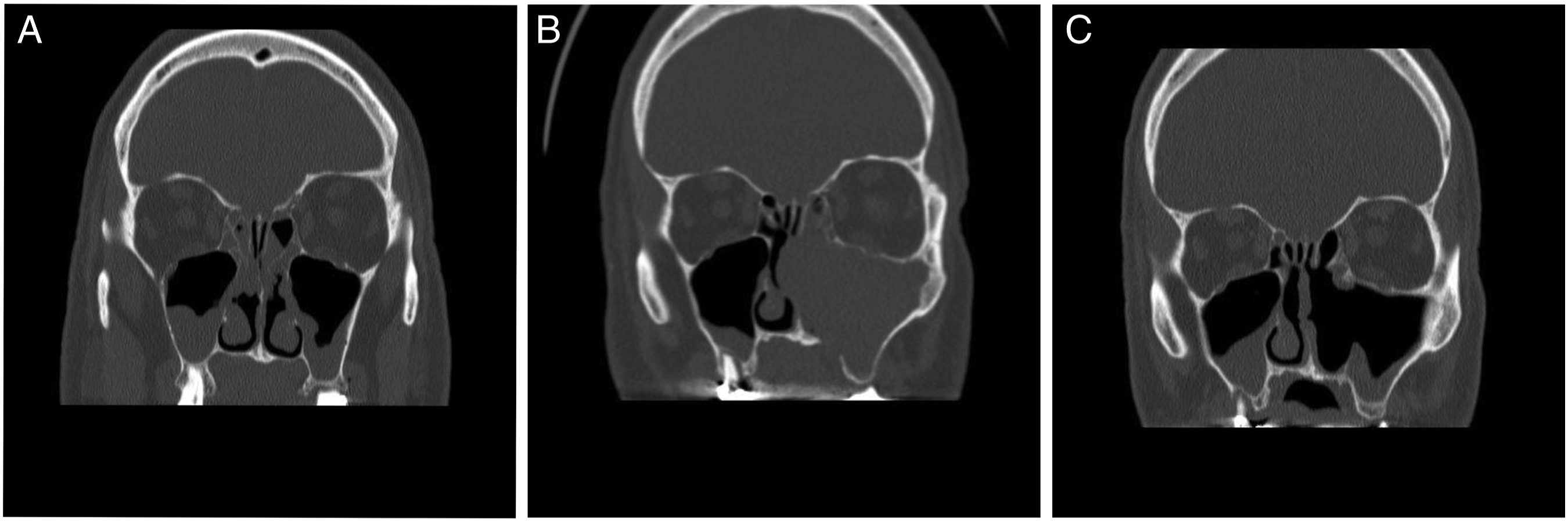
After one and a half years of improved nasal breathing, the patient began to experience recurrence of symptoms with the development of nasal congestion and anosmia. Over the next months, his symptoms worsened to complete left-sided nasal obstruction and intermittent night sweats. On reevaluation, nasal endoscopy showed polypoid disease in the left osteomeatal complex and complete patency of the right osteomeatal complex. During examination, these polyps bled when palpated. Symptoms were refractory toward PO ciprofloxacin and methylprednisolone. Repeat sinus CT indicated a 5.1 × 4.5 cm unilateral soft tissue mass involving the left sinonasal cavity including the frontal recess, ethmoid, and maxillary sinuses (Figure 1B). No orbital or intracranial involvement was noted.
Given that the patient’s symptoms returned and his polyps possessed an unusual presentation, he underwent a second endoscopic sinus surgery and polypectomy. This time, the polypoid tissue in the left ethmoid and maxillary sinuses possessed a rubbery, friable appearance. Hematoxylin and eosin (H&E) staining of the specimen revealed sheets of medium-sized cells with hyperchromatic nuclei (Figure 2B and 2C). The cells were dyshesive, not forming any particular architecture. Abundant mitotic activity was observed with scattered tingible body macrophages, imparting the “starry-sky” appearance. Immunohistochemical (IHC) studies found that the cells were immunoreactive toward MUM-1, CD138, and diffusely positive for in situ EBV-encoded RNAs (EBER). IHC staining was negative toward CD20, CD45, CD10, BCL-1, BCL-2, BCL-6, TDT, and PAX5. IHC stains for S100, Melan-A, cytokeratin OSCAR, and cytokeratin AE-1/AE-3 were negative. Ki-67 showed a proliferation rate of 50%. Fluorescence in situ hybridization (FISH) studies revealed the neoplasm to be negative for IGH/MYC, BCL6, and IGH/BCL2 translocations. The patient’s beta-2 microglobulin was 1.6 mcg/mL, white blood cell count was 4900 cells/uL, erythrocyte sedimentation rate was 29 mm/hour, and HIV screen confirmed a negative status. Last, the bone marrow biopsy was consistent with normal hematopoiesis and no neoplastic infiltrate. This ruled out plasma cell myeloma from the differential and confirmed the diagnosis of plasmablastic lymphoma of the sinonasal tract.
The patient’s benign nasal polyp histology from the year prior was reevaluated and no plasmablastic morphology was appreciated. PET scan showed involvement in the right eighth rib as well as the subtrochanteric portion of the left hip, indicating a stage IV PBL. Two months after surgery, he received 6 cycles of dose-adjusted etoposide, prednisone, vincristine, cyclophosphamide, and doxorubicin (DA-EPOCH) and 2 treatments of intrathecal methotrexate. After 4 months of chemotherapy, the patient had a repeat PET scan, which showed resolution of hypermetabolic activity in the left maxillary sinus. Two months following chemotherapy, the patient was started on consolidative 3600 cGy of Intensity Modulated Radiation Therapy to the left maxilla. Three months after chemoradiation, CT and PET imaging of the sinuses, chest, abdomen, and pelvis confirmed complete metabolic remission of the left maxillary sinus tumor with no evidence of residual lymphoma in the extranodal sites.
Every 3 months, the patient underwent repeat nasal endoscopies. One year following removal of the PBL, the patient had a repeat CT of the sinuses, exhibiting diffuse soft tissue density within the maxillary sinuses, suggestive of a return in his polyps (Figure 1C). Considering his history, the patient had a repeat polypectomy of the ethmoid and maxillary sinuses. However, this time the pathology was consistent with benign sinonasal mucosa with chronic inflammation and fibrosis. There was no evidence of a relapsing lymphoma in the sinonasal tract, indicating another episode of recurrent polyposis. The patient is currently followed every 6–12 months with routine nasal endoscopies and annual CT imaging of his sinuses, with no recurrence of PBL. The only reported residual side effect from his treatment is peripheral neuropathy of the hands and feet. He continues to manage his sinuses with daily saline rinses and antihistamines.
Discussion
Plasmablastic lymphoma (PBL) poses serious diagnostic and therapeutic challenges. It has been well established that unilateral sinonasal masses carry an added concern for malignancy as opposed to bilateral masses. However, unilateral masses are still typically benign in nature. One retrospective study found that only 4.1% of single-sided sinonasal masses were malignant, with the vast majority of them being benign polyps secondary to chronic inflammation. 7 Nonetheless, in the presence of a unilateral sinonasal polyp, clinicians should not discount the possibility of cancer and follow the steps to rule out neoplastic disease. Physicians also frequently encounter patients with relapses in their nasal polyps. Classically, allergies foster a chronic inflammatory environment conducive for these sinonasal protrusions to return. Studies have noted a 60% recurrence rate in sinonasal polyposis following endoscopic sinus surgery, of which 73% of those patients with recurrent polyps after surgery had environmental allergies. 8
With these statistics of benign unilateral polyps and recurrence rates in mind, it is understandable to assume that the patient simply developed another benign sinonasal polyp secondary to his allergies. Being one and a half years following bilateral sinus surgery with no worrisome pathological findings, clinicians may be inclined to continue symptom management such as with antihistamines or nasal steroids. If the unilateral disposition of this mass on nasal endoscopy had not prompted a precautionary CT of the sinuses, this lymphoma would have continued to proliferate and could have worsened our patient’s prognosis. Imaging is especially important if systemic steroids are administered since they can partially treat lymphoma. As studies have shown that the median overall survival of an untreated HIV-negative PBL patient is 3 months, it is crucial to diagnose this disease as soon as possible. 1 Like most PBL cases reported in literature, by the time this lymphoma was detected, it had already progressed to stage IV. 5 Clinicians should maintain a high index of suspicion for malignancy when presented with a unilateral sinonasal mass, despite typical signs of recurrence in nasal polyps.
In the instance of finding a unilateral mass in a patient with a history of recurrent nasal polyps, it is important to exclude lymphoma from the differential. Following removal of the mass, scrutinizing the polyp for morphological and genetic aberrations is recommended as the first line of investigation. Our patient’s neoplasm satisfied all current diagnostic criteria for PBL, including plasmablastic morphology (Figure 2C), loss of the B cell markers CD20 and PAX5, and gain of the plasmablast markers MUM1 and CD138. This immunoprofile can overlap with melanoma, carcinoma, and plasma cell myeloma, so it is important to rule these out with IHC staining for Melan-A, IHC staining for cytokeratin, and a bone marrow biopsy, respectively. Correlated with pathology and clinical findings, the presence of EBV also favors the diagnosis of PBL, even in immunocompetent patients. It would be beneficial for clinicians to remember this association of PBL and EBV in immunocompetent patients and ask for a possible history of mononucleosis. The leading theory is that EBV’s latent infection likely reactivates in the germinal center of lymph nodes, infects B cells, inhibits apoptosis, and thus allows oncogenic aberrations to ensue. 4,9,10 It is thought that EBV’s latent capabilities may contribute to the high PBL relapse rate of 40% seen in patients treated with DA-EPOCH, invoking the need for closer surveillance of the patient after remission. 11 A year after excision of our patient’s PBL, he developed additional polypoid masses in the sinonasal tract (Figure 1C). Since we were monitoring him every 3 months with routine nasal endoscopies, there was no hesitancy to return to surgery. Interestingly enough, this was another bout of benign recurrent nasal polyposis. To our knowledge, this unique chronology of nasal polyp and PBL presentation has not been documented before. Given the high relapse rates of both nasal polyps and PBL, closely monitoring the patient after remission was important, including routine nasal endoscopies, sinus CTs, and if needed, tissue biopsies.
In line with ensuring the well-being of patients, the pathological findings of a PBL tumor hold a great deal of clinical utility in hopes of improving treatment. Numerous studies have elucidated how a Ki-67 above 80% or genetic aberrations of the myc oncogene confers a significantly worse prognosis. 3,12 However, our patient had a proliferation index of 50% with no myc aberrations. Thus, our patient was projected to have a better outcome based on current prognostic models, yet he still presented with an already metastasized disease. This raises the question, what oncogenes were contributing to the carcinogenesis of this PBL? PBL’s lack of CD20 restricts the use of rituximab, an anti-CD20 monoclonal antibody, that has been successful with other B cell malignancies. The standard of care for PBL has been DA-EPOCH for the past decade. A number of studies have demonstrated involvement of STAT3. One genomic analysis reported that 64% of PBL cases had gain of function mutations in STAT3 while another revealed that EBV-positive cases possessed more STAT3 mutations. 13,14 In vitro assays with a pan-JAK inhibitor were noted to decrease levels of activated STAT3 and thus lowering cell viability. 15 Despite the rarity of PBL impeding large population trials, small scale studies have shown success with lenalidomide, a multifaceted immunomodulator that can inhibit STAT3 phosphorylation. 16 In an analysis of 173 PBL patients, those who received lenalidomide adjuvant to EPOCH did not experience relapse. 17 Our patient was not assessed for STAT3 mutations. Going forward, it would be advantageous for pathology to assess for STAT3 aberrations to potentially warrant an immunotherapy that exploits this mechanism. Furthermore, knowing a tumor’s genetic aberrations could lead to a better prognosis for the patient.
Conclusion
This study reports the first documented case of a patient who developed sinonasal PBL amidst recurrent polyposis, suggesting clinicians should maintain a high index of suspicion, never assuming a polyp is benign. With patients who have a history of recurrent nasal polyposis, clinicians should stay vigilant of sinonasal symptoms and laterality. Given the high relapse rates of both nasal polyps and PBL, close follow-ups are advisable, with routine nasal endoscopies and CTs of the sinuses.
Footnotes
Acknowledgments
The authors would like to thank Dr. Michael Hitchcock, Dr. Sejong Chun, and Dr. Gregory Priebe for their support. We are also grateful for the patient’s willingness to share his story.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Written informed consent was obtained from the patient for their anonymized information to be published in this article.
