Abstract
Plasmablastic lymphoma (PBL) is a rare and aggressive form of mature B cell neoplasms almost exclusively identified in patients infected with the human immunodeficiency virus (HIV). The small number of HIV-negative PBL cases reported in the literature to date is composed of single case reports and small case series which characteristically are present involving the oral cavity mucosa or gingiva. We present a 72-year-old HIV-negative Australian patient without any cause of immunodeficiency, with an isolated left maxillary sinus PBL.
Introduction
Plasmablastic lymphoma (PBL) was initially described in 1997 by Delecluse et al as a rare and aggressive HIV-associated form of mature B cell neoplasm with plasmacytic differentiation. 1 Plasmablastic lymphoma was later associated with solid organ transplantation and other immunosuppressed patients.
The term PBL should be restricted to cases with this histology and phenotype and not confused with plasma cell myeloma showing immature features. 2 Plasmablastic lymphoma constitute a subgroup of lymphomas with heterogenous clinical, histological, and genetic features that represent a diagnostic challenge to pathologists 3 and a clinical challenge due to its rarity and consequently the lack of specific guidelines and optimal treatment strategies. 4
Common sites for PBL commonly presents in extranodal regions, commonly confined to the oral cavity mucosa and gingiva, followed by digestive tract and skin. 5 The incidence of PBL is estimated in 2% to 12% of all HIV-associated lymphomas; however, the incidence in the immunocompetent population is still unknown due to its rarity with only a handful of PBL cases reported to date in immunocompetent individuals. 3
To date, PBL has been only partially characterized based on sporadic reports describing the condition, almost exclusively in immunodeficient patients with predominance of extranodal disease involving the oral cavity. In this report, we present the first Australian case with an unusual combination of PBL arising from within the maxillary cavity of an immunocompetent patient.
Case Report
A 72-year-old male was referred to the Otolaryngology, Head and Neck surgery Department at the Royal Darwin Hospital, Northern Territory for clinical assessment. He experienced unilateral left nasal obstructive symptoms and ipsilateral facial pain and numbness for 3 months. Prior to this, he had been treated for chronic sinusitis with multiple courses of oral antibiotics without improvement. He presented to the emergency department with acute onset diplopia without visual deterioration. He reported no fever, night sweats, or unintentional weight loss.
On physical examination, the patient had left eye proptosis with normal pupillary reflexes. Visual acuity 6/9 bilaterally. A nasal flexible endoscopy showed a large unilateral mass within the left nasal cavity. The rest of ear, nose, and throat examination was unremarkable.
Subsequent computerized tomography (CT) scan and a magnetic resonance imaging (MRI; Figure 1) of face and paranasal sinuses revealed a 46 × 46 × 60 mm3 left maxillary mass with an area of heterogeneous attenuation and a fluid present inferiorly. The mass had eroded through the medial wall of the maxillary sinus and through the roof into the floor of the orbit and was contiguous with the sphenoid sinus and posterior ethmoid air cells. No extension into the anterior cranial fossa was noted. Proptosis of the left eye is observed with normal extraconal muscles, but the optic nerve appeared tense.
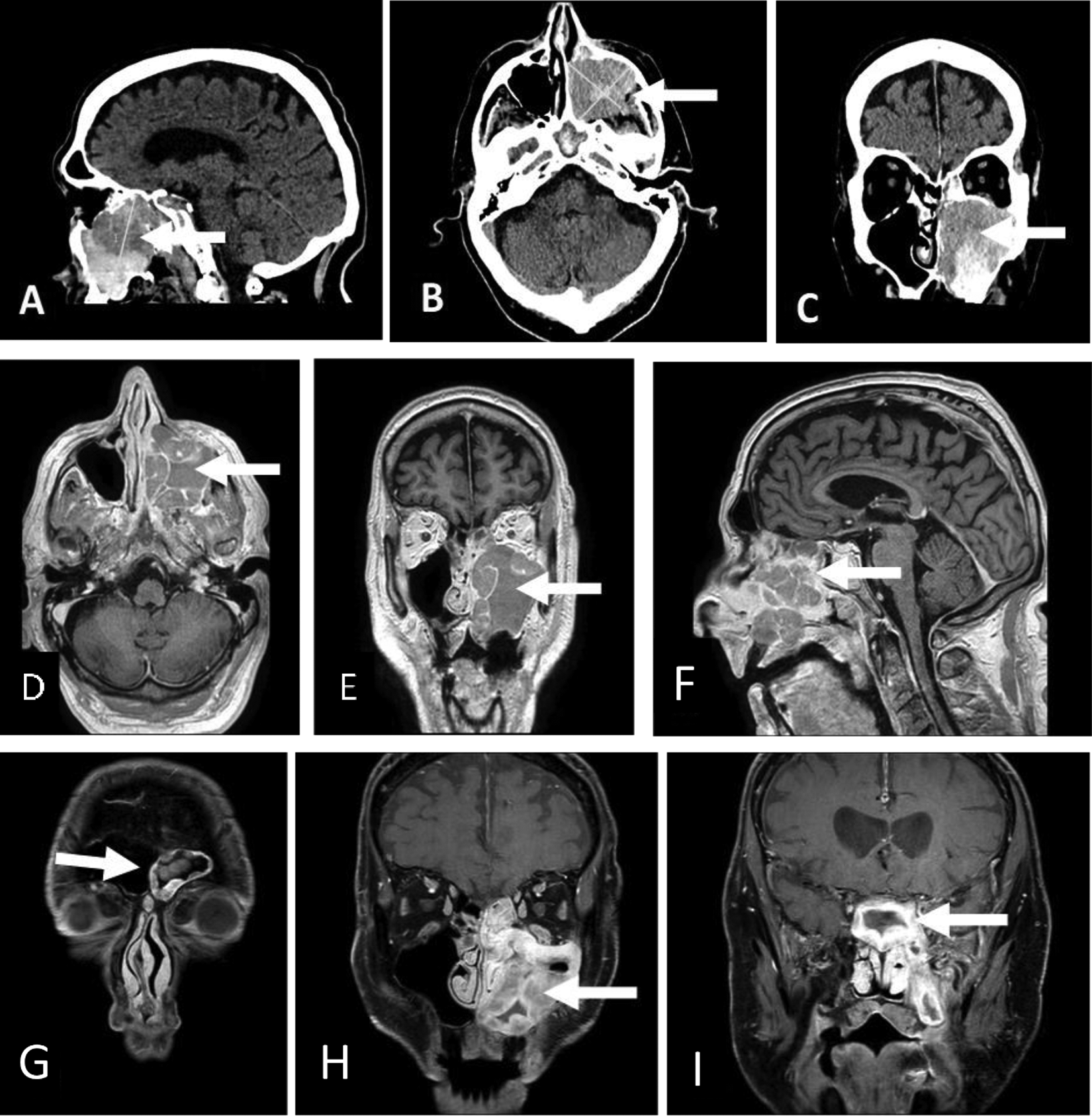
Computerized tomography Image of the brain and orbits sagittal (A), axial (B), and coronal views (C). MRI of brain and paranasal sinuses prior therapy axial (0), corona! (E), and sagittal views (F). Showing a large expansile soft tissue mass centered within the left maxillary sinus with extensive bone erosion (arrow). MRI posttreatment showing progression of the disease into the ipsilateral frontal walls of maxillary sinuses (G), middle turbinates (H), and into sphenoid sinus (I). MRI indicates magnetic resonance imaging.
At surgery, endoscopic view confirmed a mass extending through the left maxillary ostium into the middle meatus with polypoid changes of the surrounding mucosa and bony expansion of the maxillary sinus into the inferior meatus (Figure 2). Initial biopsy results suggested no evidence of malignancy. A second biopsy from deeper tissues within the maxillary sinus was undertaken. Histology showed presence of fibrous tissue and occasional minor salivary gland. These were infiltrated by atypical population of cells with morphology ranging from mature plasma cell like to highly atypical plasma blast like cells. Immunohistochemistry demonstrated the atypical lymphoid cells negative for B cell marker CD20 as well as PAX5. Positive immunoexpression was demonstrated for plasma cell associated markers including CD38, CD138, MUM1, and CD56. K67 showed a remarkably high proliferation rate greater than 90%. The EBER-ISH was performed and diffuse-positive staining was demonstrated throughout the tumor cell population (Figure 3). These findings confirmed the diagnosis of PBL.
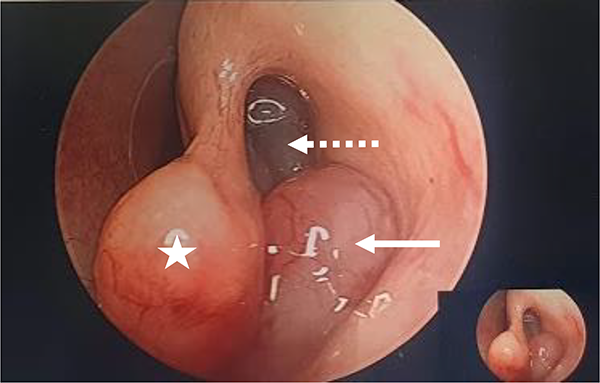
Endoscopic view of left maxillary ostium. Shown are the left middle turbinate (*) and polypoid lesion extending through the maxillary ostium (Arrow) with surrounding polypoid mucosal changes (dotted arrow).
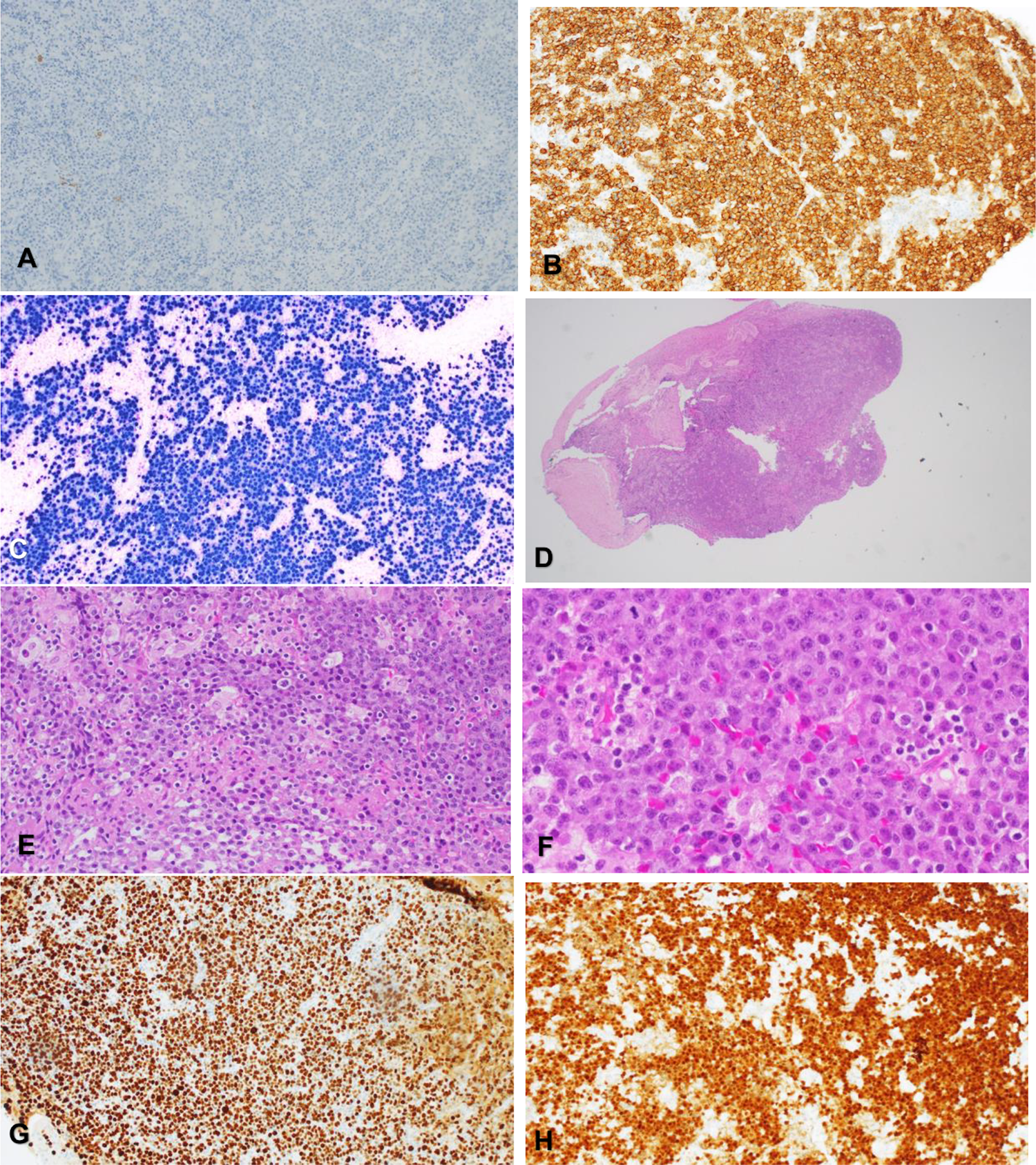
A, CD20 and PAX5 were negative B cell markers in the tumor cells. B, CD38. Plasma cell–associated marker CO38 is positive diffusely in the tumor cells. C, EBV is strongly and diffusely positive in the tumor cell population. D, H&E 20×. There is a proliferation of highly atypical cells with morphology ranging from plasma cell like to atypical plasmablastic like cells. E, H&E 200×. High power view of the atypical lymphoid cells with mature plasma cells and plasmablastic like cells appearance. F, H&E 400×. An area featuring more of plasma cell like atypical cells. G, Ki67. High proliferation at greater than 90%. H, MUM1. Diffuse positivity is seen for MUM1 in the atypical population. H&E indicates hematoxylin and eosin.
Nuclear medicine testing with gallium body scan with SPECT CT and a bone marrow aspirate with trephine morphology show no definitive evidence of plasma cell dyscrasia in the bone marrow. Testing for HIV by a p24 antigen immunoassay was negative.
The case was discussed in the lymphoma multidisciplinary meeting interstate (Royal Adelaide Hospital) where the recommendation of to start VELCADE (bortezomib) + EPOCH (etoposide, prednisone, vincristine oncovin), cyclophosphamide and doxorubicin hydrochloride (hydroxydaunorubicin hydrochloride) as primary treatment with additional HD-MTX (high-dose methotrexate) and radiotherapy (RT) to the local site was given.
Unfortunately, after 2 cycles of bortezomib/EPOCH and 3 cycles chemotherapy with HD-MTX patient needed admission for fluid overload, diarrhea, and liver enzyme derangement.
Magnetic resonance imaging was performed to assess the response to treatment (Figure 1) showed significant local disease progression now involving all sinuses and local bone destruction. However, no evidence of tumor extension into the anterior or medial cranial fossa and no intracranial collection. Based on these findings the treatment strategy is being reevaluated at the time of this report.
Discussion
Plasmablastic lymphoma is a rare and rapidly progressive variety of diffuse large B-cell lymphoma with male predominance, originally reported exclusively in the jaw and oral mucosa of immunosuppressed individuals. 1 At the time of writing, the authors are aware of 4 reported cases of PBL arising in the maxillary sinus in immunocompetent patients. The present report describes the first case in Australia (Table 1).
PBL Report.
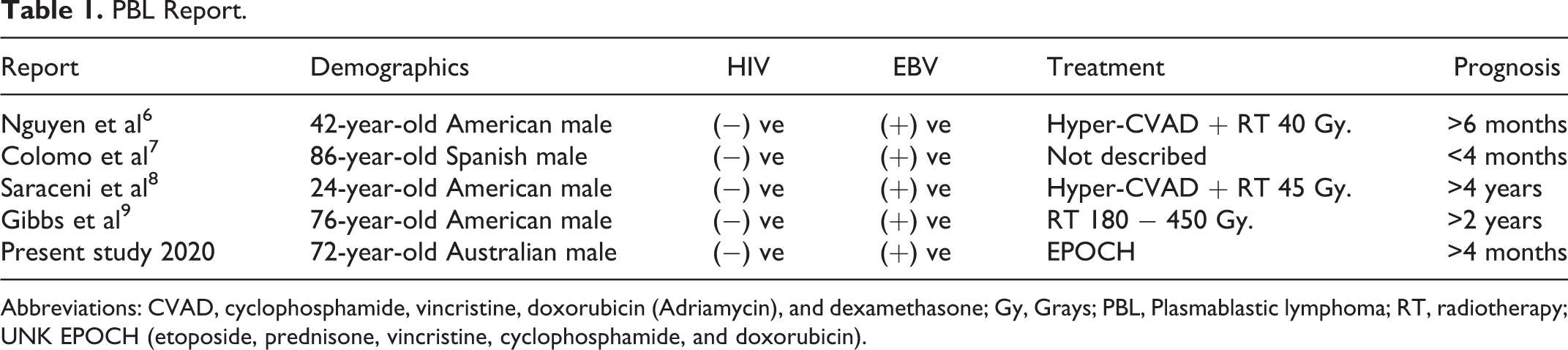
Abbreviations: CVAD, cyclophosphamide, vincristine, doxorubicin (Adriamycin), and dexamethasone; Gy, Grays; PBL, Plasmablastic lymphoma; RT, radiotherapy; UNK EPOCH (etoposide, prednisone, vincristine, cyclophosphamide, and doxorubicin).
Diagnosis of PBL represents a challenge requiring high degree of clinical suspicion. Unilateral sinonasal symptoms including facial pain, swelling, numbness, orbital symptoms of compression (eg, diplopia, vision acuity changes) with or without blood-stained discharge should point toward a suspicion of neoplastic pathology until proven otherwise. 10 Radiology investigations to include CT scan and/or MRI to ascertain disease extent and bone destruction should confer further suspicion. 11
Immunophenotyping which helps differentiating PBL from other similar malignancies is essential to achieve accurate pathologic diagnosis. Immunophenotype stains of PBL have a characteristic pattern of terminally differentiated B lymphocytes with plasma cell markers CD38, vs38C, multiple myeloma oncogene-1 (MUM1), and syndical-I (CD138). 12
Although the pathophysiology is not well understood, somatic hypermutation of the post germinal center of terminally differentiated B cells 13 and some chromosomal aberrations including recurring rearrangement involving MYC have been identified. 14 In addition, a strong association between PBL and EBV infection has been established 15 based on the expression of EVB/encoded RNA (EBER). 16
Treatment regimens have been largely varied without establish consensus, although literature suggest overall response of PBL to chemotherapy. 13 Standard chemotherapy combinations of cyclophosphamide, doxorubicin, vincristine, and prednisone (CHOP) are not considered efficacious enough to be considered adequate therapy, and NCCN guidelines’ recommended chemotherapy regime is etoposide, prednisone, vincristine (oncovin), cyclophosphamide, and doxorubicin hydrochloride (EPOCH). High-dose therapy with autologous stem cell rescue in first complete remission in select high-risk patients can be considered. 17 Survival is variable ranging from 4 to 12 months. 9
Our case is a rare condition in an unusual site in an immunocompetent patient. His diagnosis was a challenge for primary and specialist clinicians and pathologists. Although no malignancy was found in his first biopsy, clinical and radiological suspicion led to a second more invasive biopsy from within and in the walls of the involved maxillary sinus. With aid of immunohistochemistry plasma cell–associated markers, CD38, CD138, MUM-1, and CD56 were identified. Ki-67 and EBV in situ hybridization shown to be strongly and diffusely positive throughout the tumor cell population eventuating in a diagnosis of PBL. To be certain, second opinion was sought from a tertiary specialist laboratory before a final histopathological consensus diagnosis was released. Currently, our patient is 5 months posttreatment and unfortunately repeat MRI post-EPOCH therapy shows progressive disease.
Conclusion
Since the first description in 1997 of a rare, HIV-related B-cell Lymphoma and its predilection for confinement to oral cavity and gingiva, a small subset of reported cases, including the present case, have shown that PBL can arise in atypical ENT sites such as the sinuses in immunocompetent patients.
Close working with associated specialties is essential to arrive at a diagnosis. A learning pint is the fact that repeated biopsies are indicated where there is clinical and radiological suspicion even when the lesion displays benign characteristics on endoscopic appearance, as seen in this case. Knowledge of disease natural history will help clinicians provide appropriate patient counseling.
Footnotes
Authors’ Note
Written informed consent was obtained from the patient for publication of this report and any accompanying images.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
