Abstract
Plasmablastic lymphoma (PBL) is a rare type of non-Hodgkin lymphoma frequently found in the context of immunosuppression and infection with human immunodeficiency virus (HIV) and/or Epstein-Barr virus (EBV). A 33-year-old immunocompetent male presented with recurrent episodes of epistaxis and a growing intranasal mass. Excisional biopsy of the mass revealed an immunohistochemical profile diagnostic of PBL. Upon completion of chemoradiation, he underwent a transnasal endoscopic mucosal flap tissue rearrangement to restore patency for both functional and surveillance purposes. There was no endoscopic evidence of residual or recurrent disease. However, 8 months later, he was found to have a relapse involving the skin. The nasal cavity is one of the most common sites affected by PBL. Involvement of the nasal cavity may present with symptoms of persistent epistaxis accompanied by an enlarging mass. A plasmablastic immunophenotype in combination with HIV or EBV positivity can aid diagnosis.
Keywords
Introduction
Plasmablastic lymphoma (PBL) is a rare, aggressive type of non-Hodgkin lymphoma diagnosed by a plasmablast immunophenotype and absent mature B-cell markers. It is frequently associated with immunocompromised, human immunodeficiency virus (HIV), or Epstein-Barr virus (EBV)-infected individuals. Mucosal sites are most commonly affected, including the nasal, oral, and gastrointestinal cavities. Secondary cutaneous involvement due to relapse of PBL is exceedingly rare, with only one other case reported in the literature. We present a case of a 33-year-old immunocompetent male, with HIV-negative and EBV-positive status, who was initially diagnosed with PBL of the nasal cavity and was found less than one year later to have a relapse involving the skin.
Case Presentation
A 33-year-old male presented for intermittent epistaxis with prolonged bleeding lasting several hours. He noted symptoms of fatigue one-month prior to the onset of nose bleeds in addition to a small lump in his left nostril that was gradually enlarging. He had associated symptoms of congestion, facial swelling, and sinus pain.
Physical examination showed a gross asymmetry of the nose, left larger than right. A large, exophytic mass in the left nasal cavity was evident by endoscopy. Histopathologic features from an excisional biopsy of the intranasal mass included a dense sheet-like infiltrate within the dermis, composed of relatively uniform mononuclear cells with a plasmacytic appearance (Figure 1). The nuclei were round, with dispersed chromatin, and a small nonprominent nucleolus. There was diffuse expression of plasmacytic markers (CD138, CD79a, and MUM1) with a subset expressing CD43 and CD56. Cells were negative for CD20 and PAX-5 and demonstrated a lambda light chain restriction. An Epstein-Barr encoding region in situ hybridization study was diffusely positive. Serology was negative for HIV.
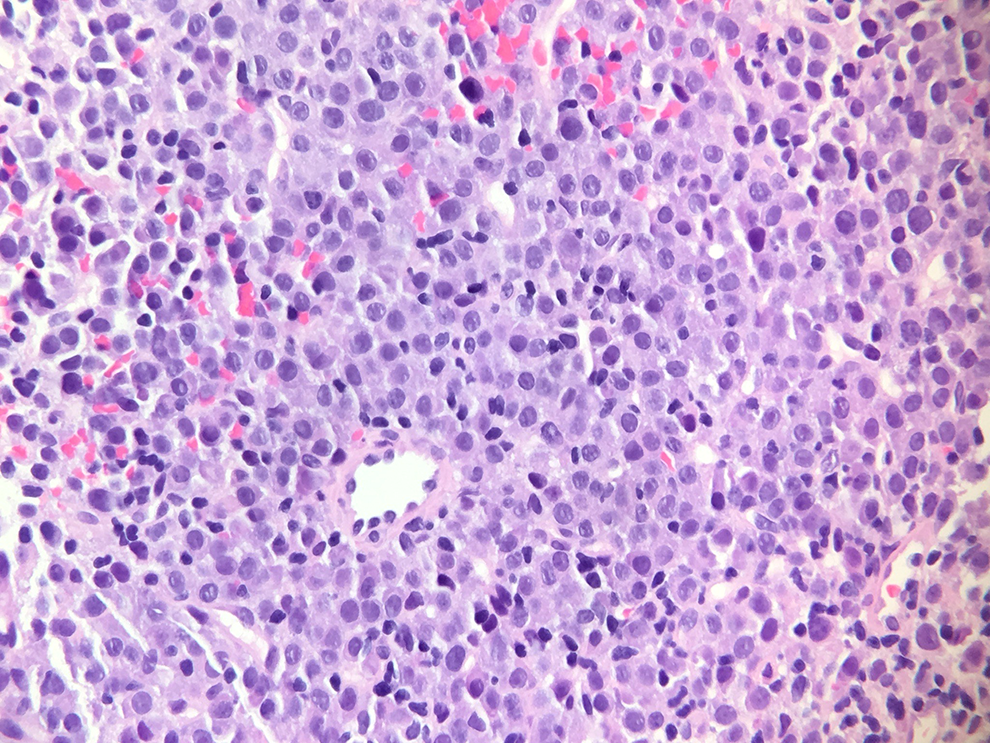
Hematoxylin and eosin (H&E) stain ×40 magnification. Dense sheet–like infiltrate of uniform mononuclear cells with round nuclei, dispersed chromatin, and a small nonprominent nucleolus.
Based on these findings, the patient was diagnosed with PBL of the nasal cavity. Treatment included 6 cycles of dose-adjusted etoposide, prednisone, vincristine, cyclophosphamide, doxorubicin (DA-EPOCH) chemotherapy with intrathecal methotrexate.
After completion of chemoradiation, the patient continued to note left facial swelling and nasal airway obstruction; a left tissue mass was demonstrated by anterior rhinoscopy. Nasal endoscopy was performed as a separate procedure to further evaluate for tumor recurrence and showed a left nasal obstruction due to adhesions post treatment, but no endoscopic evidence of residual or recurrent lymphoma. He underwent a transnasal endoscopic mucosal flap tissue rearrangement to restore patency, both for function and surveillance purposes.
Eight months after diagnosis, dermatology was consulted for asymptomatic lumps on his right upper back and scalp (Figure 2). Skin findings on physical examination showed subcutaneous nodules on the upper back, left shoulder, right vertex of the scalp, bilateral thighs, and right upper buttock. The nodules were mobile and rubbery with overlying erythema and telangiectasias. Interestingly, these became more indurated with time. A punch biopsy of the nodule on his scalp demonstrated an immunohistochemical profile that was consistent with the patient’s original biopsy from the intranasal mass. Consequently, he was found to have relapsed PBL. Treatment included weekly bortezomib monotherapy, with options to pursue lenalidomide or ICE chemotherapy if refractory.
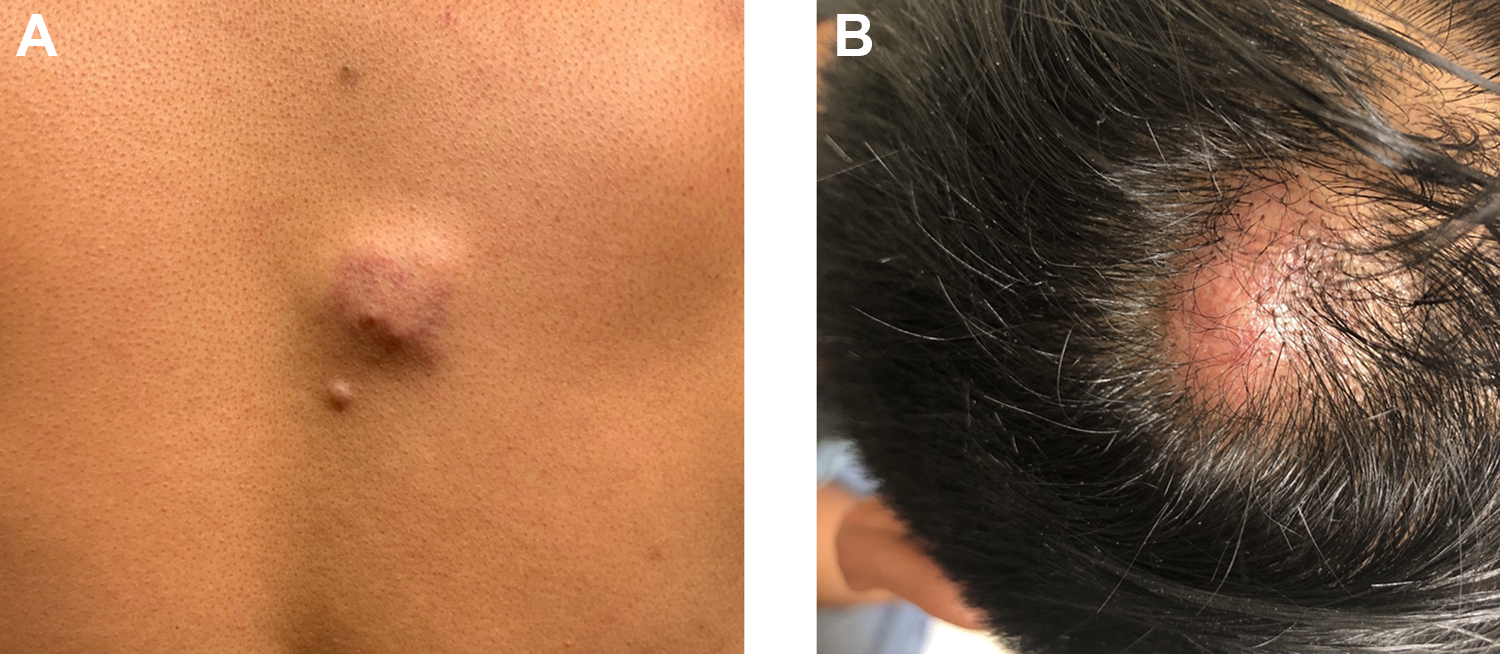
A, Subcutaneous nodule with overlying telangiectasias on the back. B, Erythematous nodule on the vertex of the scalp.
Discussion
Plasmablastic lymphoma is a rare, aggressive subtype of diffuse large B-cell lymphoma. 1 It is characterized by a proliferation of large neoplastic immunoblasts that express plasma cell differentiation antigens but lack mature B-cell markers. 2 The cancer often arises in the setting of immunosuppression, such as posttransplant, autoimmune diseases, and acquired immunodeficiency syndrome–defining conditions. 1 When it is reported in the immunocompetent, it is often diagnosed at an older age.1,2
Human immunodeficiency virus and EBV infections are strongly associated with PBL and may influence the prognosis of disease. 1 EBV status and the presence of myc aberration can affect treatment response and overall survival.1,3 Pathogenic mechanisms of the viral infection include immune checkpoint modulation, inhibition of B-cell apoptosis, and increased susceptibility to proto-oncogenes such as c-myc. 4
Cutaneous PBL is rare. Only one other report has described skin findings secondary to relapse from a different primary source (the oral cavity), as was the case in our patient. 5 The most common clinical presentation involves erythematous to violaceous papules or nodules, occasionally associated with ulceration and necrosis. From the published studies of cutaneous PBL, 19 (64%) were male and 10 (36%) were female, with a median age of 52 years. The majority were associated with some form of immunosuppression: 4 (15%) were associated with posttransplant lymphoproliferative disorder, 15 (52%) were HIV positive, and 19 (67%) were EBV positive (Table 1).
Cases of Cutaneous Plasmablastic Lymphoma.
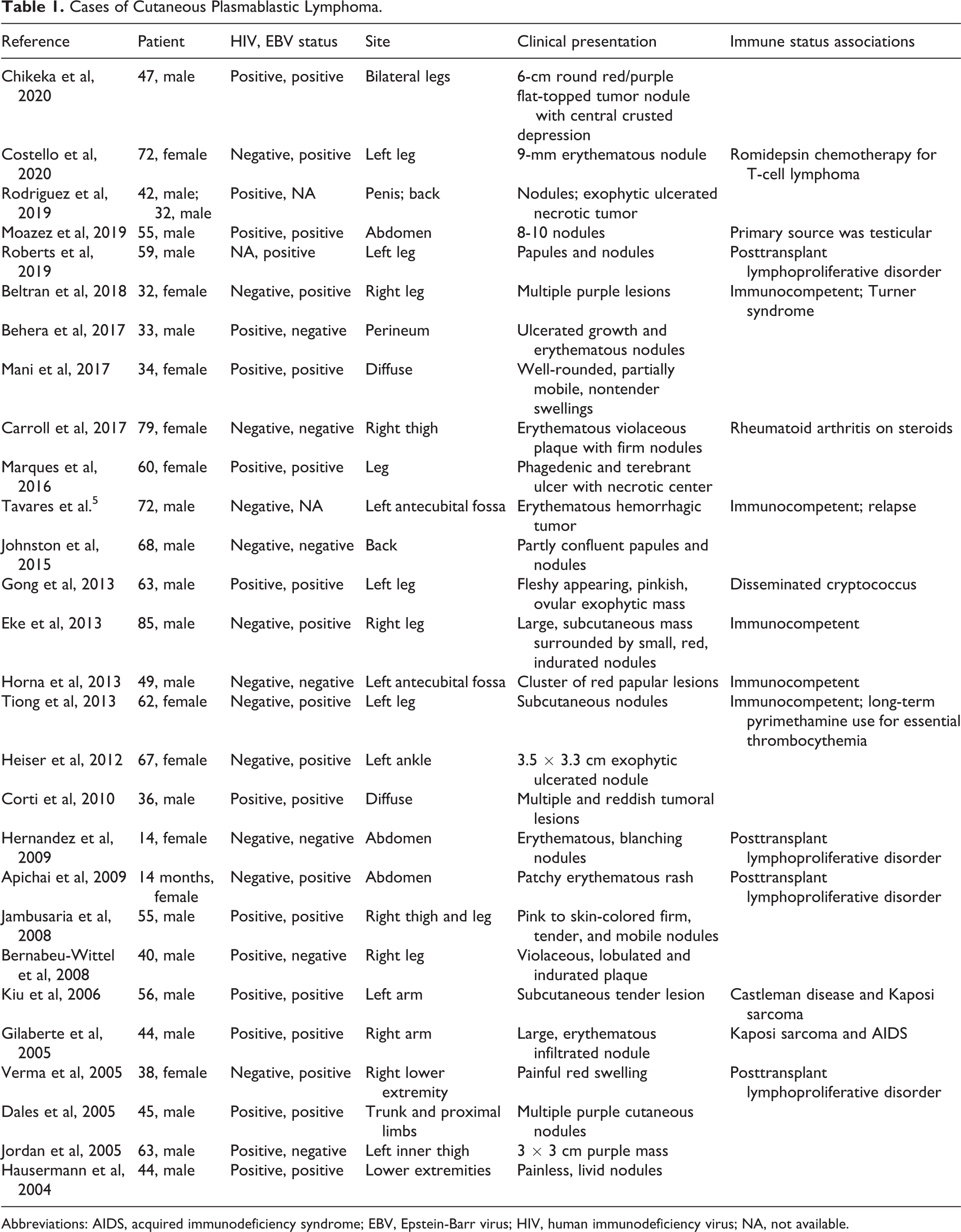
Abbreviations: AIDS, acquired immunodeficiency syndrome; EBV, Epstein-Barr virus; HIV, human immunodeficiency virus; NA, not available.
Histopathologic features include large plasmablasts characterized by moderately abundant cytoplasm, central oval nuclei, and prominent nucleoli. The immunophenotype is positive for many plasma cell–associated markers including CD79a, IRF-4/MUM-1, BLIMP-1, SBP-1, VS38, CD38, and CD138. However, it lacks mature B-cell markers such as CD19, CD20, and PAX-5. The marker of proliferation Ki-67 is often high.1-3 The protooncogene c-myc is frequently overexpressed and more commonly found in association with EBV positivity. 2
The plasmablast immunophenotype may also be seen in other types of lymphomas and myelomas, making diagnosis challenging. A differential diagnosis should consider myeloma, plasmacytoma, primary effusion lymphoma, DLBCL associated with HHV8 (Castleman disease), and other large B-cell lymphomas which can exhibit anaplastic, plasmacytic, or plasmablastic morphology. 1 A high proliferation index, lack of mature plasma cells, and association with EBV and/or HIV infection are features that support the diagnosis of PBL among others. 1
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
