Abstract

Report
A 14-year-old female with no significant medical history presented with a 4-month history of a rapidly growing right cheek mass. She had intermittent temporomandibular joint discomfort and paresthesia in the mandibular division of the trigeminal nerve. Facial nerve function was intact. Fine needle aspiration (FNA) of the mass was indeterminant and computed tomography showed a 5-cm right parotid mass that extended to the parapharyngeal space (Figure 1). Representative CT with contrast image, axial view. There is a 5-cm tumor (white arrow) involving the entire right parotid gland and extending into the parapharyngeal space.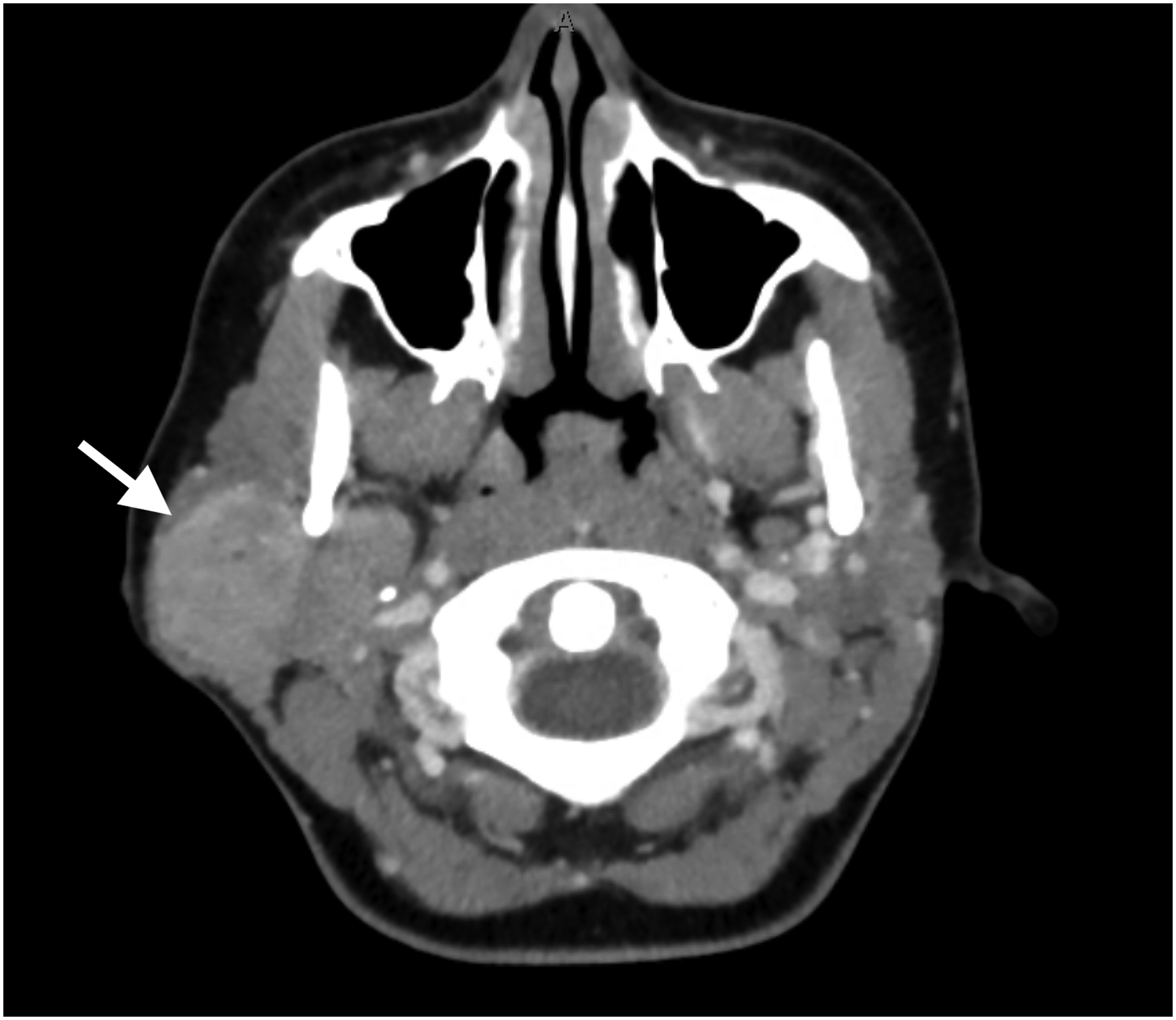
A total right parotidectomy was performed. Surgery was challenging given the size of the tumor, the location in the deep lobe, and the increased neovascularization. Additionally, the branches of the facial nerve stretched over the inferior, lateral, and anterior surfaces of the tumor, but were preserved. Postoperatively, the patient had near complete facial palsy that recovered fully 1 month after surgery.
Histopathological examination revealed a hypercellular tumor composed primarily of sheets and nests of round blue cells separated by fibrous septa that infiltrated and effaced the affected parotid glandular tissue (Figure 2). On immunohistochemical staining, the tumor was diffusely positive for AE1/AE3 cytokeratin and CD99. Finally, the translocation EWSR1-FLI1 (t(11; 22) (q24; q12)) identified on the fluorescence in situ hybridization (FISH) analysis confirming the diagnosis of adamantinoma-like Ewing sarcoma (ALES). Histological appearance of the tumor (H&E stain, high power). Yellow arrow: Fibrous septa consistent with Ewing sarcoma family of tumors; red arrow: Hypercellular sheets/nests of round cells with high nuclear-to-cytoplasmic ratio.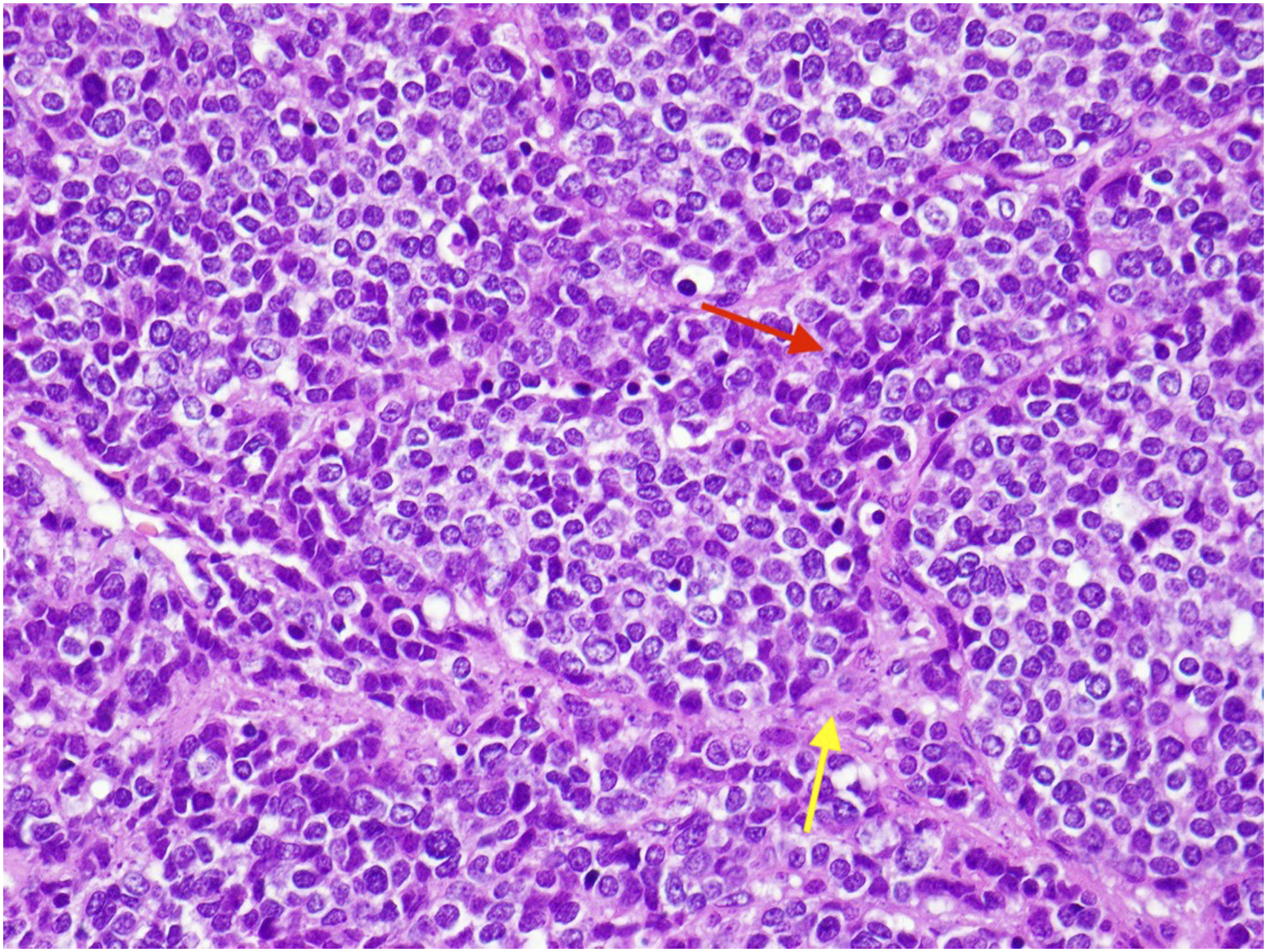
The patient was referred to oncology for further treatment. Initial staging workup, including a PET/CT and dedicated CT chest and bone marrow biopsies, was negative for metastatic disease. The patient was started on an interval-compressed chemotherapy regimen of vincristine, doxorubicin, and cyclophosphamide alternating with ifosfamide and etoposide every 2 weeks followed by radiation therapy at the primary tumor site.3,8
Ewing sarcoma (ES) is an aggressive pediatric primary bone tumor with a high propensity to metastasize.1,2 Genetic analysis has identified a non-random translocation between the EWSR1 gene (22q12) and the ETS gene family, most commonly the FLI1 gene(11q24) as the pathognomonic feature of ES. 3 This has led to the discovery of other soft tissue tumors that share this translocation and present with histopathology similar to ES, all of which have been grouped as the Ewing sarcoma family of tumors (ESFT).4,5 Today, 15% of ESFTs are estimated to be extraosseous, with 3 to 9% derived from the head and neck.1,6,7 ESFTs affect primarily the pediatric population. Approximately 3 cases per 1,000,000 children are reported each year in the United States, with 75-85% arising from bone.1,9 Even more rare is ALES, a type of ESFT. In 2019, Rooper et al. reported on 26 patients with ALES in the literature, 19 (73%) in the head and neck. 10 More specifically, ALES arises from glandular tissue, particularly the parotid glands.11-13 This is unique to ALES as other forms of head and neck ESFT rarely arise from the salivary glands. 7 Additionally, ALES typically affects adults. 11 As more cases of ALES are identified, additional studies are needed to better understand the clinical and demographic characteristics of ALES.
Although parotid gland lesions are uncommon in children, the differential diagnosis includes both non-neoplastic (e.g., viral, cystic, and granulomatous) and neoplastic etiologies14,15 In children, tumors make up an estimated 39% of parotid lesions, 16 with pleomorphic adenomas and mucoepidermoid carcinomas being the most common benign and malignant neoplasms, respectively. Unlike in adults, up to 50% of pediatric parotid tumors are malignant. Parotid tumors are generally painless and insidious in nature, with an average presentation time of 812 months. 14 This is different from ESFTs which are more aggressive, 3 as was the case in this child who presented with a rapidly growing mass over 4 months.
Treatment almost always includes superficial or total parotidectomy, especially when the result of fine needle aspiration is indeterminate or suspicious of malignancy. Parotidectomy is safer than tumor enucleation and has better outcomes. 17 In a pediatric patient, there is an increased emphasis on functional preservation: the surgeon should try to save a functional facial nerve. Most pediatric parotid tumors can be treated with adjuvant chemotherapy and/or radiation after surgery. Indications for facial nerve sacrifice include cases where function was already compromised by tumor invasion or where complete encasement of the nerve by the tumor prevents adequate dissection and excision. 14 Facial nerve sacrifice may also be considered in salvage surgery, where complete resection is the only available therapeutic option.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
