Abstract
Introduction
Several antiresorptive drugs are used to reduce bone density loss and to decrease fracture incidence in postmenopausal women for a long period. They are also indicated in patients with multiple myeloma (MM) and other cancer patients with bone metastases. Denosumab is a human monoclonal antibody against RANKL (receptor activator of nuclear factor kappa-B ligand). Denosumab prevents the fusion of preosteoclasts into an osteoclast by competitive inhibition of their receptors. Another group of antiresorptive medications are bisphosphonates (BPs). Bisphosphonates reduces bone resorption by inhibiting osteoclast function and initiating their apoptosis, and they form toxic ATP analogues and chemically bind to bone minerals. Denosumab has been introduced into clinical practice (FDA approval in June 2010) as an alternative to BPs, with premise of its lower rate of side effects. Unlike BP, denosumab does not cause post-application flu-like syndrome, does not irritate the gastrointestinal tract, and is not nephrotoxic. After initial optimism, it became clear that denosumab has the same skeletal side effects as BP. These are mainly jaw osteonecrosis and atypical fractures of the femur (ATFF). Jaw osteonecrosis is an area of necrotic bone exposed intraorally or extraorally that persists for more than 8 weeks. At first, it was only referred to patients taking BPs, and the disease was called bisphosphonate-related osteonecrosis of the jaw (BRONJ). Now, other substances causing osteonecrosis are known, especially denosumab, antiangiogenic drugs, and kinase inhibitors, and therefore the condition is now called medication-related osteonecrosis of the jaw (MRONJ). 1 Although MRONJ is the most common skeletal adverse effect of antiresorptive therapy, other bones could also be affected. Atypical fractures of the femur occur in patients treated with BP as a low-energy subtrochanteric or proximal diaphyseal femoral fracture. It is typically horizontal and starts in the lateral cortex. It has also been described in patients using denosumab. 2
Several cases of bisphosphonate-associated ear canal osteonecrosis (BPECO) have been described in recent literature. 3 -9 Recently, there were some published cases of osteonecrosis of the ear canal in patients receiving denosumab 10,11 and also one case with a patient receiving a protein kinase inhibitor. 12 It commonly presents as an exposed bone of the external auditory canal and in advanced stages, destruction of the middle ear and propagation to the skull base could occur. The quality of life of the patient severely deteriorates, and treatment of these advanced stages is challenging.
We present a unique case of denosumab-related external auditory canal osteonecrosis and its successful surgical treatment in the early stage of the disease.
Case report
In December 2014, a 68-year-old patient was referred to the oral, jaw, and facial surgery clinic by a treating oncologist with suspicion of MRONJ. The patient underwent a comprehensive oncological treatment of breast cancer. Therapy included left mastectomy with axillar dissection, radiation therapy, and aromatase inhibitor. In March 2011, the patient started on denosumab (120 mg subcutaneously) once every 3 months. The last dose of denosumab was administered in September 2014.
A clinical examination confirmed a necrotic bone in the right molar region of the mandible under a partial overdenture. Necrosis was painless and did not show signs of inflammation. Therefore, it was classified as stage 1 of MRONJ by the American Association of Oral and Maxillofacial Surgeons. 1 During examination, the patient also complained of a 4-month–lasting discharge from the right ear, which was repeatedly unsuccessfully treated with ear drops.
Denosumab was discontinued and the necrotic bone of the mandible was resected under local anesthesia and antibiotic coverage (amoxicillin with clavulanic acid 875 mg/125 mg BID, beginning 1 week before and lasting 2 weeks after surgery). Since only the surface of the mandibular alveolus was necrotic, its resection could continue to bleeding edges and subsequent soft tissue hermetic closure did not cause problems. Healing in the oral cavity took 3 weeks without complications. Histopathological examination showed fragments of the necrotic bone with clusters of branching microbial filaments (Actinomyces). Consequently, the patient had been sent to the ENT department. The otoscopy revealed a suppurating skin defect on the floor of the right auditory canal. The defect was located approximately above the border of the bony and cartilaginous part of the auditory canal. Around the wound, there was adhering incrustation. The tympanic membrane has a normal appearance.
The necrotic bone was removed and the defect was covered with a free skin graft (from the retroaurical region) under general anesthesia. Postoperative healing was again uneventful. The bone sample from the defect was fragmented, so histopathological examination was not possible. The patient has been asymptomatic and has no signs of recurrence of both carcinoma and bone necrosis for 5 years.
Discussion
Overview of MRECO cases.
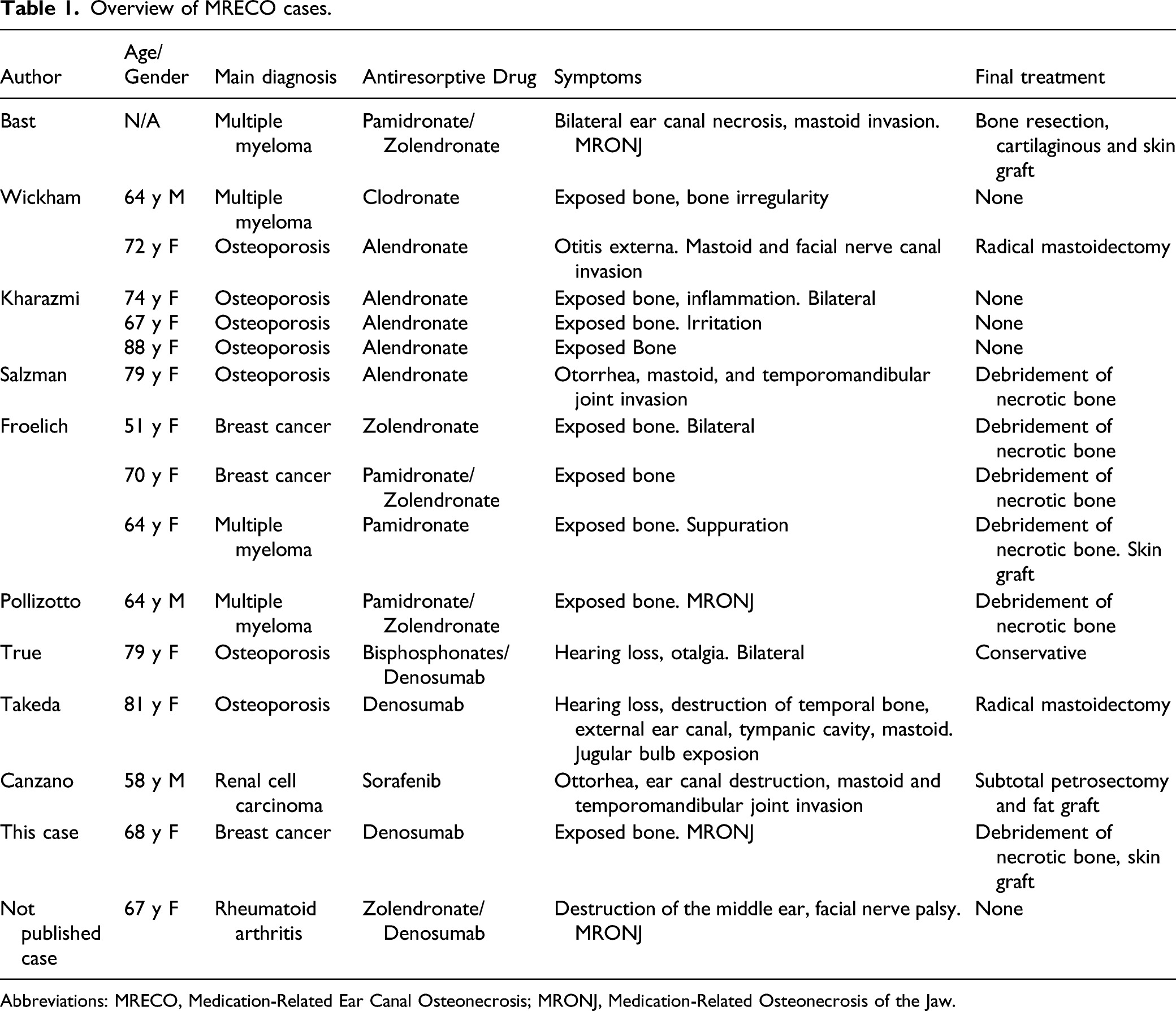
Abbreviations: MRECO, Medication-Related Ear Canal Osteonecrosis; MRONJ, Medication-Related Osteonecrosis of the Jaw.
Osteonecrosis of the external auditory canal is certainly quite rare. However, it is necessary to consider this complication in differential diagnosis, as the quality of life of an affected patient could be dramatically impacted. The authors of this article had previously treated another patient who suffered complete destruction of the middle ear caused by osteonecrosis. The disease resulted in hearing loss and facial nerve paresis on the affected side (the case had not been published as the patient died early from cardiac comorbidity)—Figure 1. Destruction of the posterior wall of the auditory canal. CT transversal section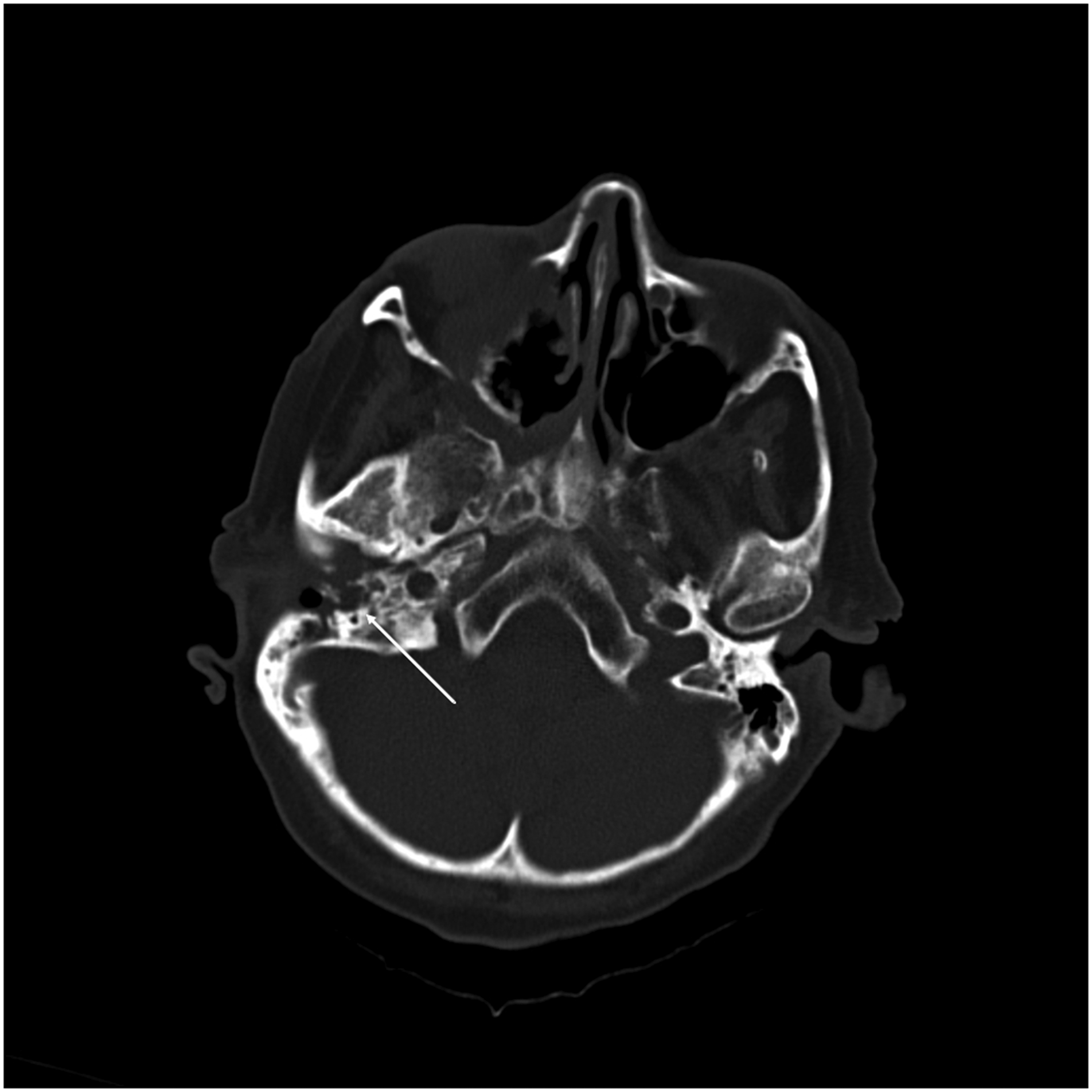
Due to the analogy with MRONJ and the existence of denosumab-related necrosis of the ear canal, we propose to unify the terminology on medication-related ear canal osteonecrosis (MRECO). Diagnostic criteria include necrotic bone of the ear canal exposed for 8 weeks in the patient using risk medication (especially BP, denosumab, bevacizumab, temsirolimus, and sunitimib), absence of previous radiotherapy in the temporal area, exclusion of metastases, and cholesteatoma. 15
MRECO stages.

Abbreviations: MRECO, Medication-Related Ear Canal Osteonecrosis; TMJ, Temporo-Mandibular Joint.
Because only a few cases of MRECO have yet been described, conclusions on the most appropriate treatment strategy are difficult. Based on the analogy with MRONJ, therapy should be initiated conservatively by systemic administration of antibiotics and local therapy with antiseptic rinses. In insufficient response to treatment, complete resection of the necrotic bone with free graft overlap should follow. In advanced stages, radical resection of affected structures with subsequent reconstruction is considered necessary. The purpose of the surgical procedure is to remove all necrotic bones until fresh bleeding edges are reached. Prolonged antibiotic administration until complete healing of the defect is commonly indicated. On the contrary, a cessation of antiresorptive/antiangiogenic therapy (so-called drug holidays) is not universally recommended. It must be individually decided based on the general condition of the patient and the risks and benefits of discontinuing therapy evaluation.
Due to the rarity of MRECO, all-encompassing prevention is useless. Detection of this complication and ensuring further professional treatment remains in the hands of specialists prescribing this risk medication. They should actively ask for signs of MRECO, particularly a history of ear discharge, and eventually refer such a patient to an Ear, Nose and Throat (ENT) department. ENT specialists should consider the possibility of osteonecrosis when treating patients with chronic ear disorders. Similarly, oral surgeons should be aware of MRECO when treating patients with MRONJ who simultaneously suffer from ear problems.
Conclusions
The article pointed out the benefit of early surgical management of MRECO in comparison to the late approach.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research of this article: This work was supported by Internal grant of Palacky University IGA LF 2018-006 and Ministry of Health of the Czech Republic Grant DRO (FNOl, 00098892).
