Abstract
Small cell carcinoma (SCC) in the nasal cavity and sinuses is extremely rare. The clinical data of 15 patients with primary SCC in nasal cavity and sinuses were analyzed retrospectively. All patients were treated with surgery, radiotherapy, and chemotherapy. Of the 15 patients, 2 patients are alive for more than 6 years, and 5 patients died after the median follow-up period (11 months). Most of our patients represent the later stage (73% presented at stage III or IV) and had surgery combined with radiotherapy and chemotherapy; however, nearly half of patients have tumor recurrence and/or distant metastasis. SCC of nasal cavity and sinuses often invades surrounding tissues, and the long-term curative rate is generally low. Early diagnosis and comprehensive treatment are key to improve survival. Although the overall survival time of SCC is not optimistic, it is still recommended that patients take comprehensive treatment.
Introduction
Small cell carcinoma (SCC) is one of the most aggressive types of neuroendocrine carcinomas. The SCC is microscopically defined by small to intermediate sized tumor cells, large number of apoptotic cells, focal necrosis, high mitotic rate, and lack of neurofibrillary stroma. 1 In addition, SCC often stains positive for at least one of neuroendocrine markers such as synaptophysin, CD 56, and chromogranin A. 1 The larynx, salivary glands, and the sinonasal region are the most common sites for SCC of the head and neck. 1 –3 SCC of the nasal cavity and paranasal sinuses is an extremely rare neuroendocrine tumor 1,2 that can occur in various anatomical regions and overall has a poor prognosis. Its histological appearance is similar to small cell lung carcinoma. 3 SCC is highly invasive and tends to spread far and/or locally. Treatment options include surgery, radiotherapy, chemotherapy, or a combination of these methods. 2 Since this kind of tumor is rare, there are no specific suggestion or treatment guidelines for its management. We retrospectively analyzed the cases of primary SCC in the nasal cavity and paranasal sinuses in recent 10 years at Xijing Hospital, and summarized the treatment experience of the disease.
Materials and Methods
We conducted a retrospective, case-only descriptive analysis of data from Xijing hospital. We found and confirmed 15 patients with primary SCC of the nasal cavity and paranasal sinuses who were treated in the Department of Otolaryngology, Xijing Hospital from January 2010 to December 2020, of which 11 were male and 4 were female. The average age of patients was 48.5 years (ranging from 20 to 73 years). All patients were diagnosed with SCC by pathology. Each pathological diagnosis report is verified by one initial pathologist and two senior pathologists before it is released. All of the patients had undergone CT and MRI for the paranasal sinuses and chest, ultrasound for neck lymph nodes and abdominal organs, and routine laboratory examination. According to the extent of tumor invasion, we classified the tumor stages of these patients based on American Joint Committee on Cancer (AJCC) sinonasal tumor grading standards (eighth edition). 3 By reviewing case records, the basic information, disease characteristics, treatment process, and outcome of patients were summarized. All patients were treated with radiotherapy and chemotherapy combined with surgery. Among them, 5 cases received radiotherapy and chemotherapy before operation, 9 cases received radiotherapy and chemotherapy after operation, and 1 case received radiotherapy and chemotherapy before and after operation. All patients had at least one postoperative follow-up record, and all patients were confirmed by telephone/WeChat app. The total survival time was calculated as the interval from diagnosis to death or the last follow-up time (June 2021). The log rank test 4 was used for exploratory analysis to evaluate the univariate effects of location and treatment on these two survival endpoints.
Result
Patient characteristics of sinonasal small cell carcinoma.
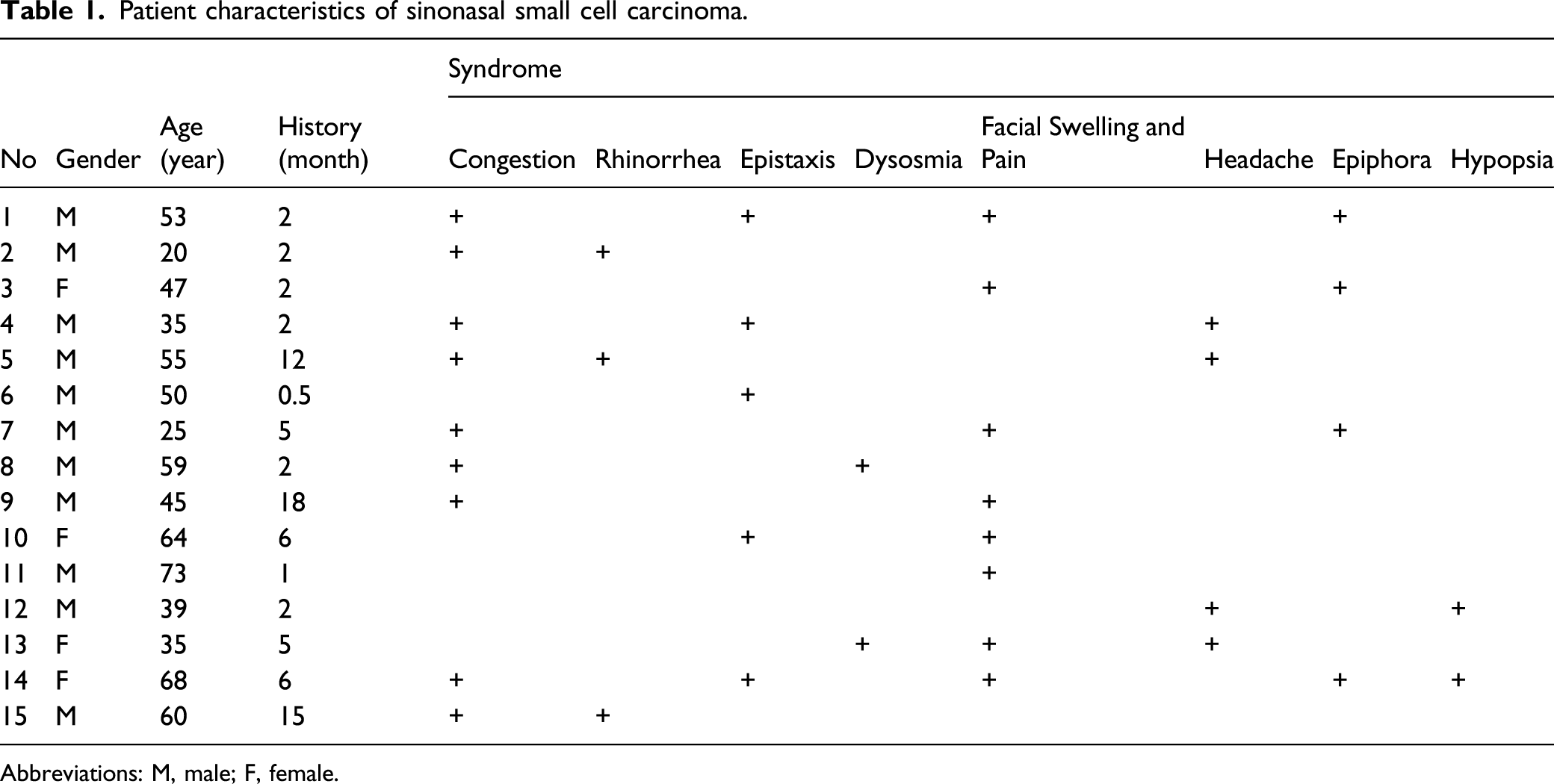
Abbreviations: M, male; F, female.
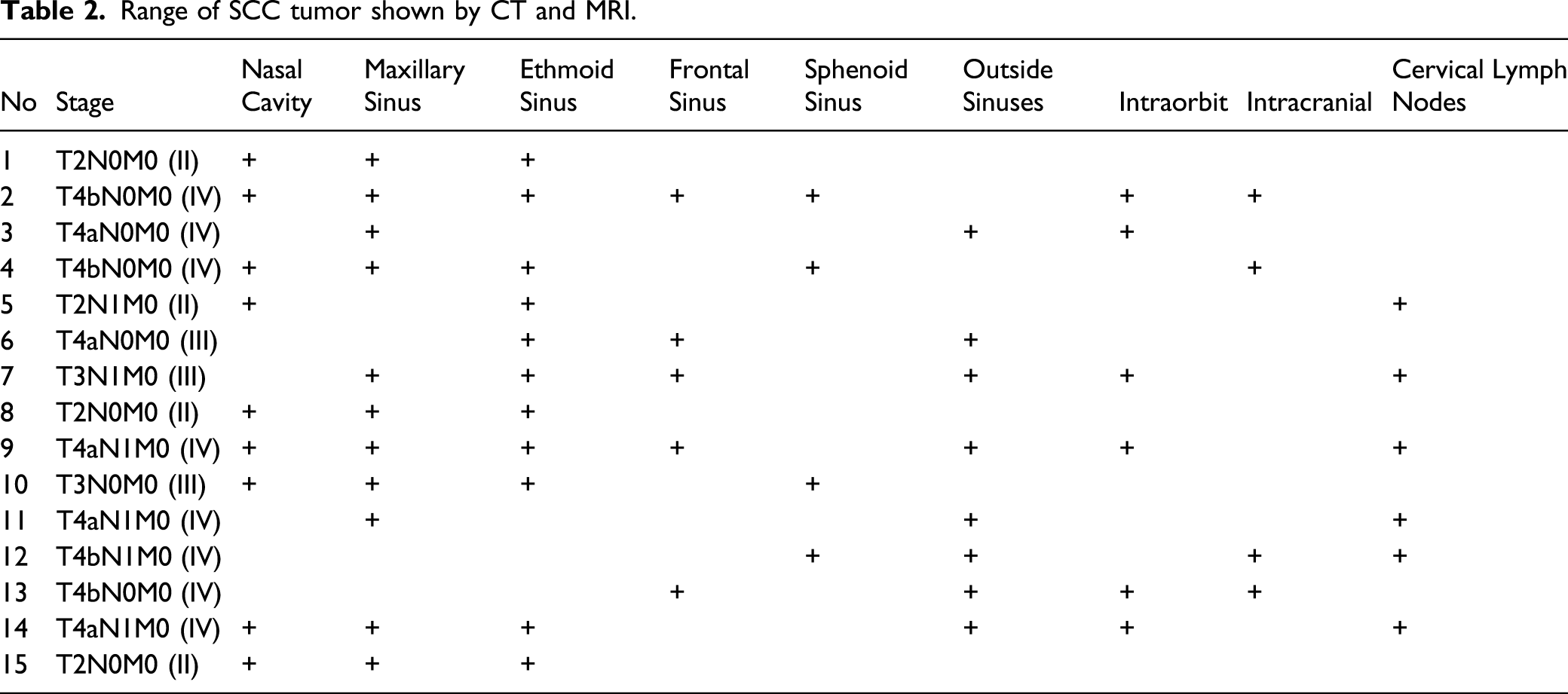
Range of SCC tumor shown by CT and MRI.
Treatment strategy and outcome of these patients.
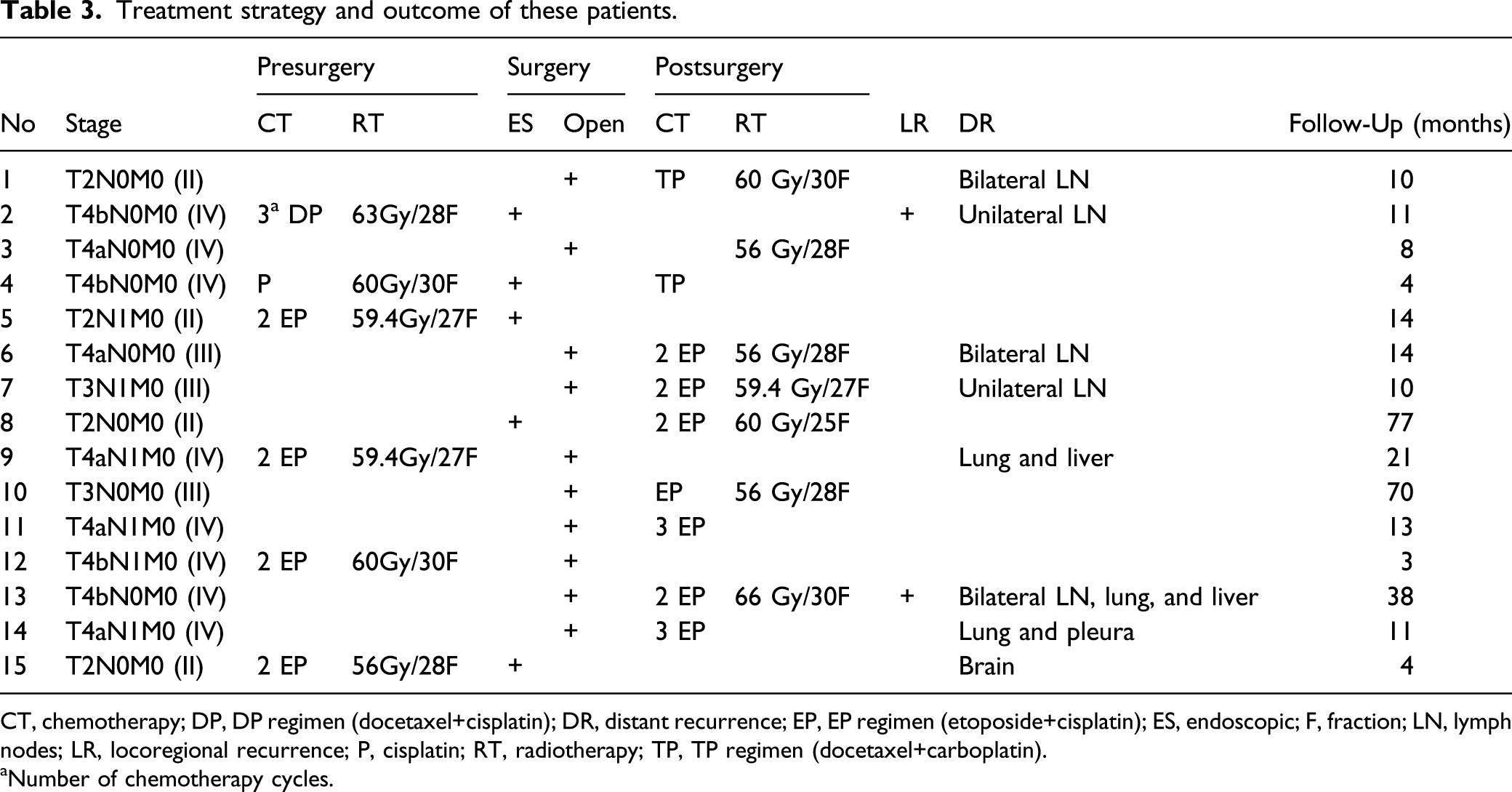
CT, chemotherapy; DP, DP regimen (docetaxel+cisplatin); DR, distant recurrence; EP, EP regimen (etoposide+cisplatin); ES, endoscopic; F, fraction; LN, lymph nodes; LR, locoregional recurrence; P, cisplatin; RT, radiotherapy; TP, TP regimen (docetaxel+carboplatin).
aNumber of chemotherapy cycles.
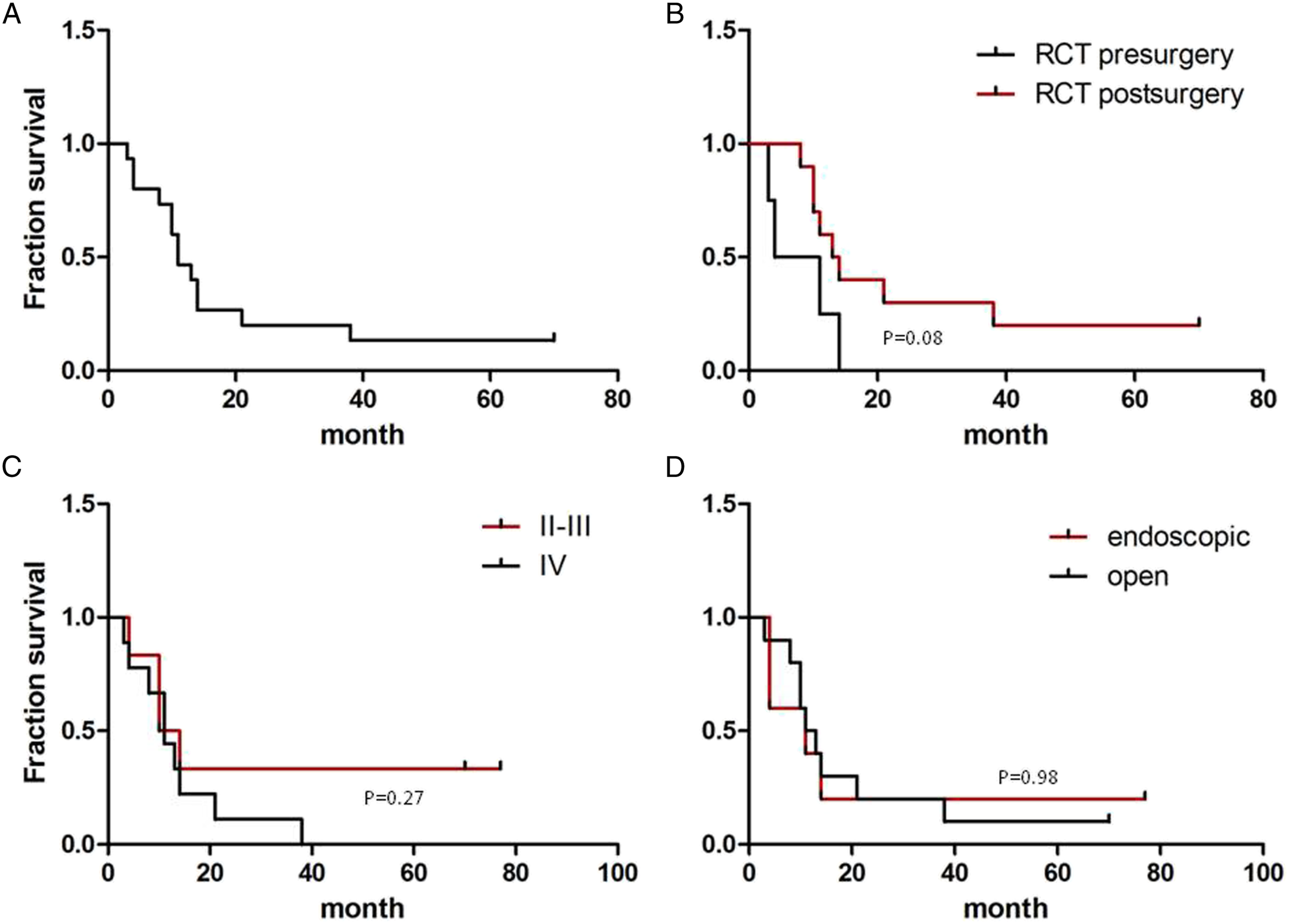
Survival characteristics of patients with SCC. A The overall survival time of 15 patients with SCC; B, the comparison of survival time between preoperative and postoperative chemoradiotherapy groups; C, comparison of survival time of patients with different tumor stages; D, comparison of survival time between endoscopic surgery and open surgery groups.
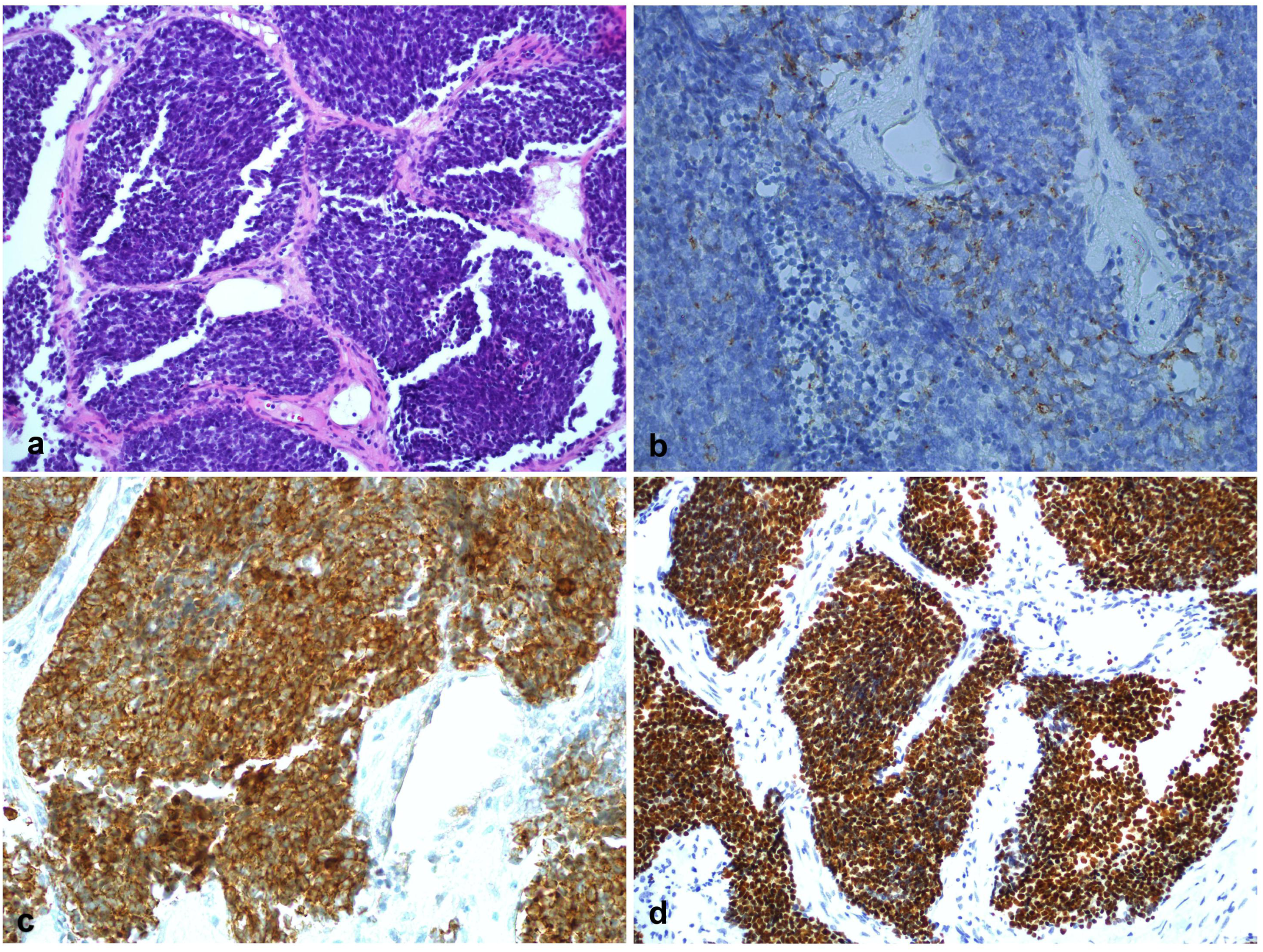
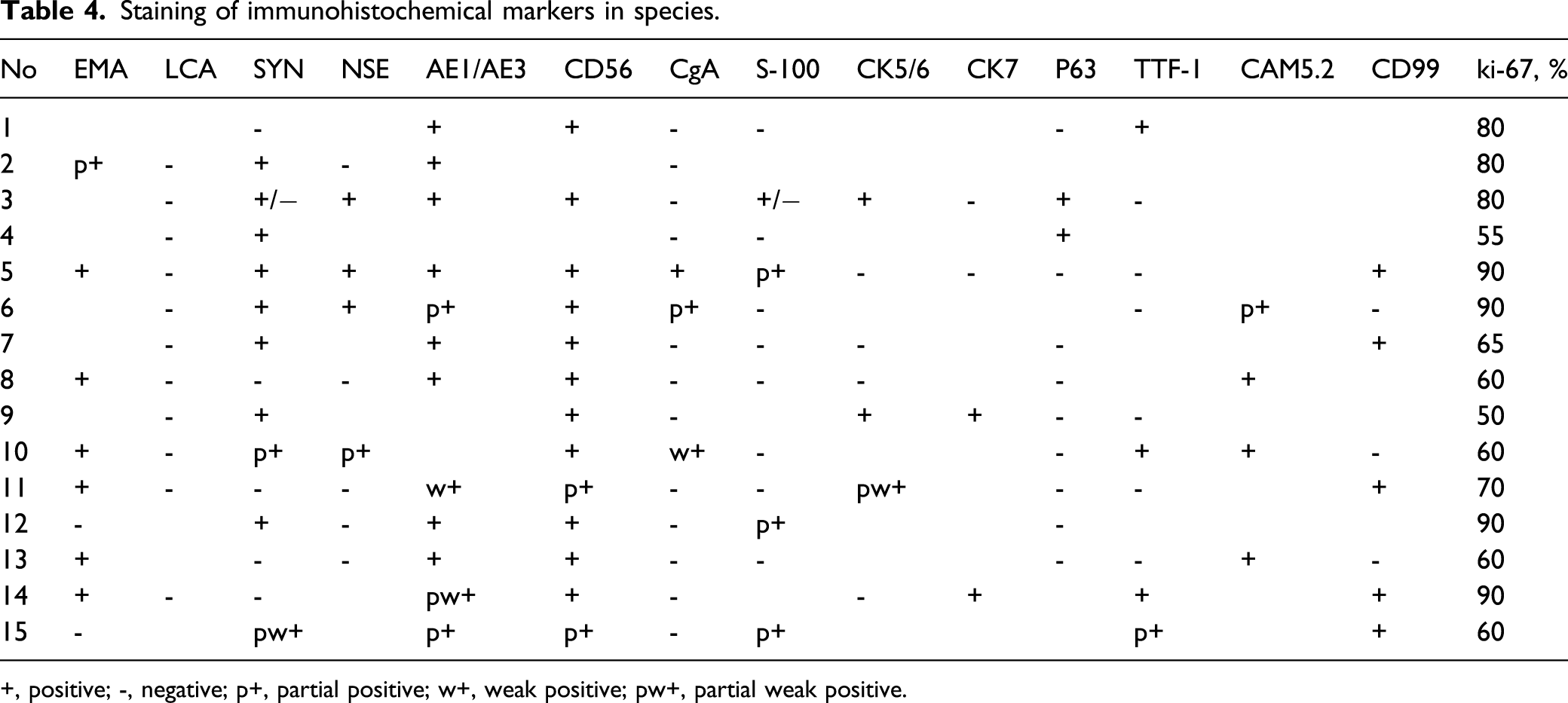
The general manifestations of pathological tissues are as follows: heterotypic cells in fibrous tissues are arranged in clusters, with enlarged nuclei, deep staining, uniform size, less cytoplasm, and infiltrating growth. To summarize and analyze the staining of main immunohistochemical markers in tumor specimens, among which CD56, AE1/AE3, synaptophysin (SYN), EMA, and CAM5.2 were positive in most of specimens (Figures 2 and 3), while LCA, CgA, S-100, and P63 were negative in most of time, and other markers could be positive or negative. The ki-67 index of tumor specimens was higher than 50%, and some tumors were as high as 90%, with an average of 72%, which indicated that SCC had a rapid growth rate and a high malignant degree (see Table 4). Pathology of patient No. 8, showing histologic features of small cell carcinoma with clusters of small to medium sized cells with minimal cytoplasm, hyperchromatism, and nuclear molding figures. A–C, hematoxylin and eosin, original magnification ×10 (A), ×20 (B) and ×40 (C). Ki-67 index is about 60% (D). Histologic features of patient No. 10 show neoplastic cells with a high nuclear to cytoplasmic ratio, scant cytoplasm, molding and delicate nuclear chromatin (A, hematoxylin and eosin, original magnification, ×20). CgA (B) shows a weak positive reaction, whereas CAM5.2 (C) and TTF-1 (D) show a strong cytoplasmic reaction. Staining of immunohistochemical markers in species. +, positive; -, negative; p+, partial positive; w+, weak positive; pw+, partial weak positive.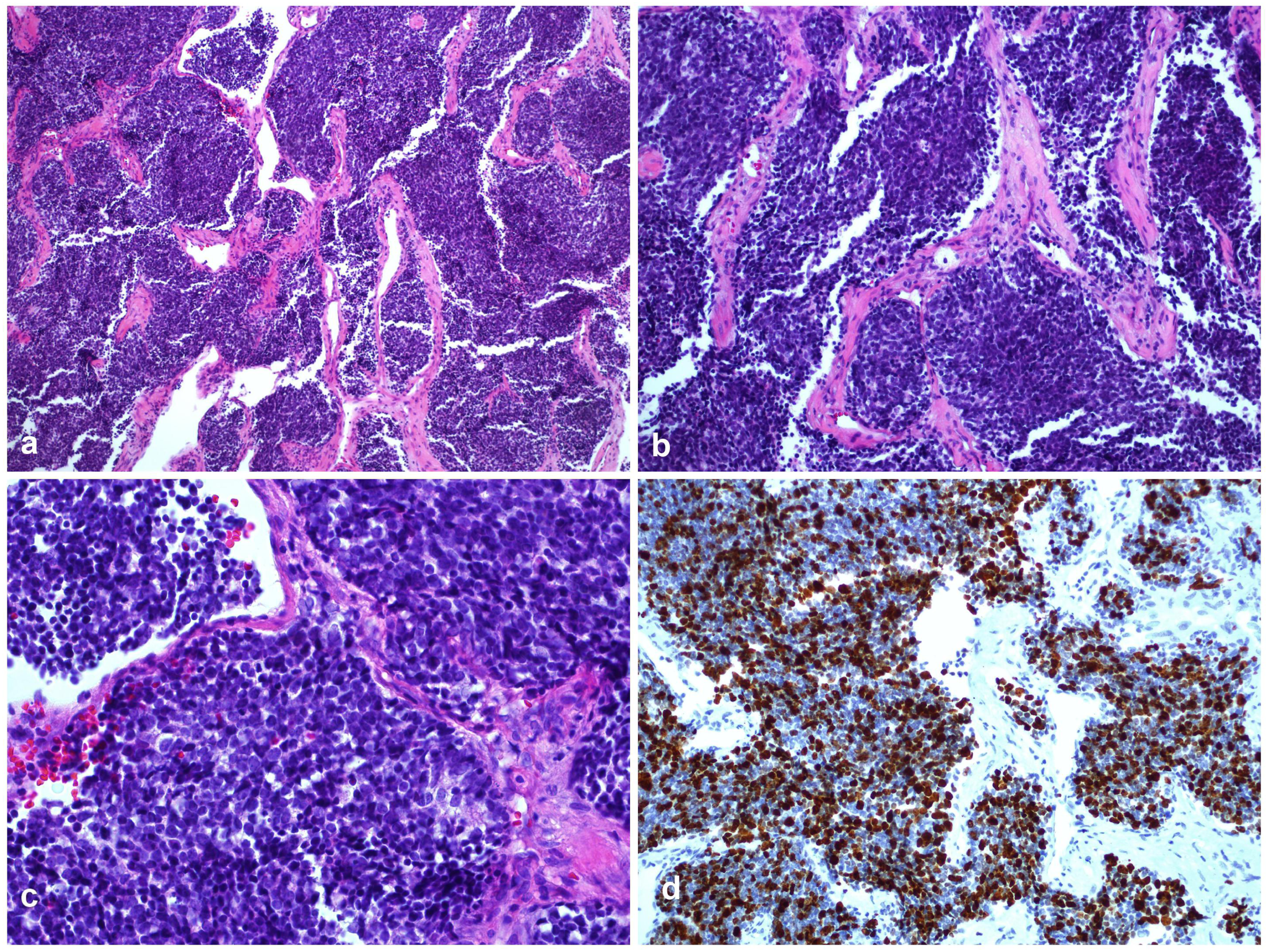
Discussion
Primary SCC of the head and neck is rare, accounting for about 2.5% of the head and neck tumor, 4 and the proportion of sinuses and skull base is even lower. 1 Small cell carcinoma of paranasal sinuses has strong invasiveness, grows fast, has strong local infiltration and metastasis, and most of them have broken through a single anatomical area and invaded surrounding tissues when they were discovered. 1 Patients often visit a doctor because of facial swelling and pain, headaches, nasal bleeding, and vision problems in the early stages, but these clinical manifestations have no diagnostic specificity, which often does not arouse the vigilance of patients and doctors. In this group of patients, the average time from symptom to visit was 5.3 months, with a median of 2 months, but 40% of patients had cervical lymph node metastasis at the time of first visit. According to literature reports, about 80% of patients with SCC of the head and neck have found cervical lymph node metastasis at the time of diagnosis. 5,6 We only counted the patients who were hospitalized and operated in our department, excluding those patients with distant metastasis that were not surgically treated; hence, data of early distant metastasis of SCC are unknown.
Although the final pathological diagnosis of these patients is SCC, there are still differences in the positive expression of immunohistochemical markers (Figure 3). In this study, CD56, AE1/AE3(CK), EMA, and SYN were the most positive markers in tumor specimens, while CgA, Neuron-Specific Enolase (NSE), CK, and SYN were the most positive markers in the literature. 2,7 The ki-67 index of SCC is very high (Figure 2), which indicates that the tumor proliferation is fast and the prognosis of tumor might be poor. However, the ki-67 index cannot fully reflect the tumor characteristics, and the ki-67 index does not significantly affect the survival curve of patients. The number of cases of SCC of nasal cavity and paranasal sinuses is small, and the relationship between immunohistochemical markers and tumor growth characteristics and treatment effect is still uncertain.
At present, clinical comprehensive treatment is often followed with reference to the radiotherapy and chemotherapy scheme introduced in 2003. 1 Our data show that preoperative radiotherapy and chemotherapy or postoperative radiotherapy and chemotherapy have no significant difference in the overall survival curve of the disease. The literature holds that there is minimal difference in overall survival rates between radiotherapy and chemotherapy alone and surgery plus radiotherapy and chemotherapy, although the survival rate of patients with chemotherapy alone seems lower. 1 New chemotherapy drugs, such as taxanes, camptothecin derivatives (topiramate and irinotecan), and gemcitabine, have shown anti-SCC activity, 8 –10 which may help to improve the outcome of this disease. New radiotherapy techniques, such as intensity-modulated radiotherapy and proton and heavy ion radiotherapy, may increase the efficiency of radiotherapy and reduce the damage of surrounding tissues. 11 Immunotherapeutic and targeted drugs may improve survival rate of the patients. 10 Therefore, further research is needed to optimize the treatment plan.
Some patients have SCC of stage II or III, and there is no significant difference in survival curve compared with patients with SCC of stage IV (Figure 1). For SCC of the nasal cavity and paranasal sinuses, TNM classification system or other classification systems 3 have limited value in predicting prognosis and helping to choose treatment. 1,2 We believe that surgery should be the main treatment for small cell carcinoma, regardless of the stage of tumor, as long as there is an opportunity to remove the tumor. Our results show that endoscopic surgery or open surgery has no significant effect on the survival curve of the disease (Figure 1). Therefore, if the patient's conditions permit, endoscopic surgery can be chosen. On the one hand, the image navigation technology is helpful in designing and guiding the surgery, and the endoscopic view can help the surgeons clearly identify the tumor boundary to operate on and important anatomical structure to preserve. On the other hand, the facial cosmetic result could increase the patient's confidence in overcoming the disease.
This group of SCC patients had no paraneoplastic syndrome during treatment, so no hormone-related examination was performed. Previous literature reported that the incidence of ectopic hormone production was low, 1,4,12 so it was not included in routine examination items.
The median survival time of this group of patients was 11 months, which was similar to the literature. 6 However, the 5-year survival rate was only 2/15, far lower than the 50% reported in the literature. 2 For economic and cultural reasons, our patients may choose to give up active treatment and enter hospice care after tumor recurrence and distant metastasis, which may also be the reason for the low 5-year survival rate of these patients. As a result, while optimizing the treatment plan, we should also improve patients' faith in their ability to survive cancer and extend survival time as much as possible on the basis of increasing quality of life, which will be the future focus of our treatment.
Conclusion
Small cell carcinoma of the nasal cavity and paranasal sinuses is a rare but aggressive clinical entity with poor overall survival. The tumors grow rapidly and have no special clinical manifestations in early stage. It remains challenging diagnostically by morphology under microscope as well as with advances in immunohistochemical testing due to immunohistochemical markers variability. We present here a small group of sinonasal SCC from a single medical center. Most of our patients represent the later stage (73% presented at stage III or IV) and had surgery combined with radiotherapy and chemotherapy; however, nearly half of patients have some form of recurrence within the follow-up years. The 5-year survival of this group is much lower than reported in the literature, so investigation is needed to determine optimal treatment strategies in the future.
Footnotes
Acknowledgments
We are thankful to pathologist Jianfeng Wu for his help in pathology figures and knowledge support and to Mr Dingshun Liang for supervising the language editing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Shaanxi Provincial Department of Science and Technology key industry innovation chain (Group) social development field. Fund serial number: 2021ZDLSF02-12. This work was also supported by Xi'an Health Committee research project (SN:2020yb73).
