Abstract

Keywords
Significance Statement: We herein report an interesting case of objective pulsatile tinnitus secondary to the transverse-sigmoid sinus dAVFs and the jugular foramen dAVFs supplied by multiple arteries. The jugular foramen is an uncommonly rare location for dAVFs and difficult to diagnose and treat. 4D-CE-MRA may be a reliable tool for the characterization of intracranial dAVFs with respect to the fistula location, main arterial feeder, and venous drainage.
Less than 10% of all cases presenting with tinnitus have pulsatile tinnitus, defined as a sound synchronous to the heartbeat. 1 In case of objective tinnitus, which can be heard by the clinician, there are somatosounds near the ear generated by vascular or other disorders. Intracranial dural arteriovenous fistulas (dAVFs) are extremely rare developmental lesions and can cause unbearably objective pulsatile tinnitus. 2 These fistulas are abnormal arteriovenous anastomoses located along the dura or within a dural sinus, characteristically at the transverse-sigmoid junction. We herein report an interesting case of objective pulsatile tinnitus secondary to the transverse-sigmoid sinus dAVFs and the jugular foramen dAVFs supplied by multiple arteries. Four-dimensional contrast-enhanced magnetic resonance angiography (4D-CE-MRA) was performed for pretreatment evaluations including their locations, main arterial feeders, and venous drainage patterns.
A 49-year-old male patient complained of pulsatile tinnitus in the left ear, progressively increasing over a 2-year period. Although it was bothersome during the day, it was especially troublesome at night and disturbed his sleep. The tinnitus could easily be decreased by ipsilateral compression of the neck. The patient had no history of traumatic head and neck injury, infection, and previous surgery. A bruit was heard on auscultation of the left retroauricular region. Given the high suspicion for a vascular anomaly of his objective pulsatile tinnitus, 4D-CE-MRA (Figure 1) at 3.0 T was carried out for the evaluation of temporal flow dynamics. In the transverse-sigmoid junction and jugular foramen dAVFs, both readers judged that the occipital artery was the main arterial feeder, and Borden-Shucart classification was type I, analogous to Cognard type I and IIa lesions.
3
Superoinferior (top row), anteroposterior (second row), and left lateral (bottom row) projections of maximum intensity four-dimensional contrast-enhanced magnetic resonance angiography (4D-CE-MRA) images reconstructed with the multiple dural arteriovenous fistulas (dAVFs). There were early depictions of the left transverse and sigmoid sinus, internal jugular vein, and vertebral artery venous plexus at the early arterial (left) to the arterial phase (middle). The left transverse-sigmoid junction dAVFs (big circles) were fed by multiple dural branches of the left occipital artery (dashed arrow), posterior auricular (solid arrow), middle meningeal artery (clear arrow), and meningeal pituitary trunk (small solid arrow). The left jugular foramen dAVFs (small circles) were mainly fed by small branches of some external carotid arteries (dotted arrow) ascending along the internal jugular vein. Antegrade venous drainage directed into the left sigmoid sinus. The mild retrograde venous flow through the transverse sinus to the torcular herophili (arrowheads), without cortical venous drainage, was also depicted. In the multiple dAVFs, both readers judged that the left occipital artery was the main arterial feeder, and Borden-Shucart classification was type I, analogous to Cognard type I and IIa lesions.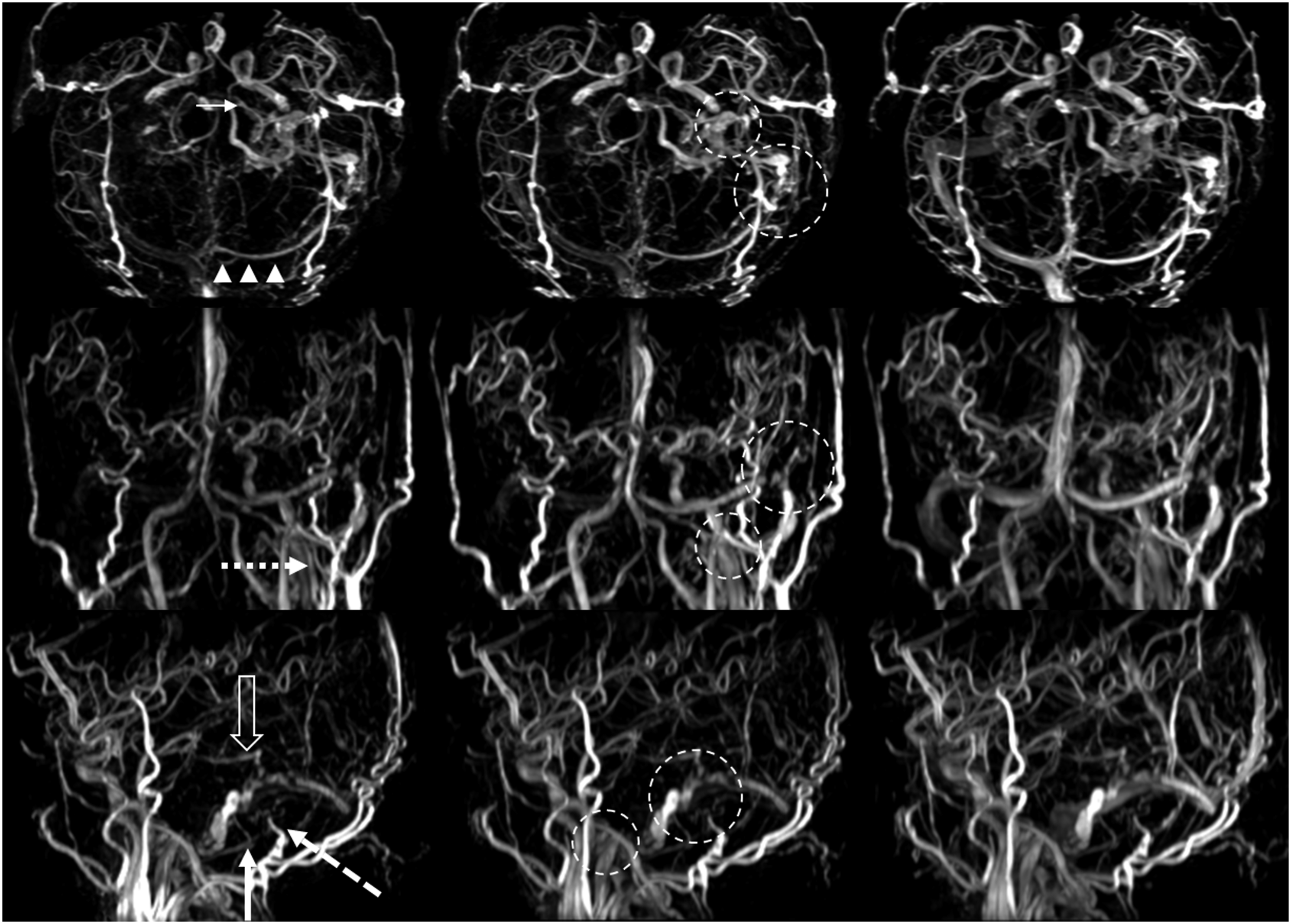
According to the risk of hemorrhage, the multiple dAVFs of the patient were low-grade dAVFs without an increased risk of hemorrhage.
3
Therapy was indicated for his intolerable symptom of pulsatile tinnitus. The multidisciplinary treatment plan prescribed transarterial liquid embolization for the main arterial feeder in anticipation of the remarkable reduction of the dAVFs flow. Transfemoral cerebral digital subtraction angiography (DSA) was performed under general anesthesia to verify the venous drainage pattern and the treatment plan. The agreement between 4D-CE-MRA and DSA findings was good to excellent with respect to the fistulas locations, main arterial feeders, and venous drainage patterns (Figure 2A and 2B). The shunt of transverse-sigmoid junction dAVFs was almost disappeared (Figure 2C and 2D) after using Onyx to embolize the main dural branch of occipital artery. However, the jugular foramen dAVFs could not be embolized, due to the small and tortuous feeders from the left occipital artery and posterior auricular, which could not be catheterized distal enough. After the partial embolization, the abnormal flow within jugular bulb was remarkably reduced. We informed the patient of external manual carotid compression to promote and support spontaneous regression of the lesions. There was no auscultated sound at the left retroauricular region following the procedure. The patient’s pulsatile tinnitus was obviously declining but still persistent at the 3-month follow-up. Anteroposterior (upper) and lateral (lower) projections of transfemoral cerebral angiography during the arterial phase were shown before (A and B) and after (C and D) partial embolization of the left dural branches of occipital artery. A and B: The transverse-sigmoid junction dural arteriovenous fistulas (dAVFs) (big circle) were fed by multiple dural branches of the left occipital artery (dashed black arrow), posterior auricular (solid black arrow), middle meningeal artery (clear arrow), and meningeal pituitary trunk (dashed white arrow). The jugular foramen dAVFs (small circle) were mainly fed by dural branches of the left occipital artery (solid white arrow). Mild retrograde venous flow into the transverse sinus (white arrowheads) without cortical venous drainage was depicted. C and D: The shunt of transverse-sigmoid junction dAVFs was almost disappeared after using Onyx (black arrowhead) to embolize the main dural branch of occipital artery. The jugular foramen dAVFs could not be embolized, due to small persistent branches of the occipital artery (solid white arrow), and particularly because of multiple feeders from the posterior auricular (solid black arrow), which could not be catheterized distal enough because of their small size and their tortuosity.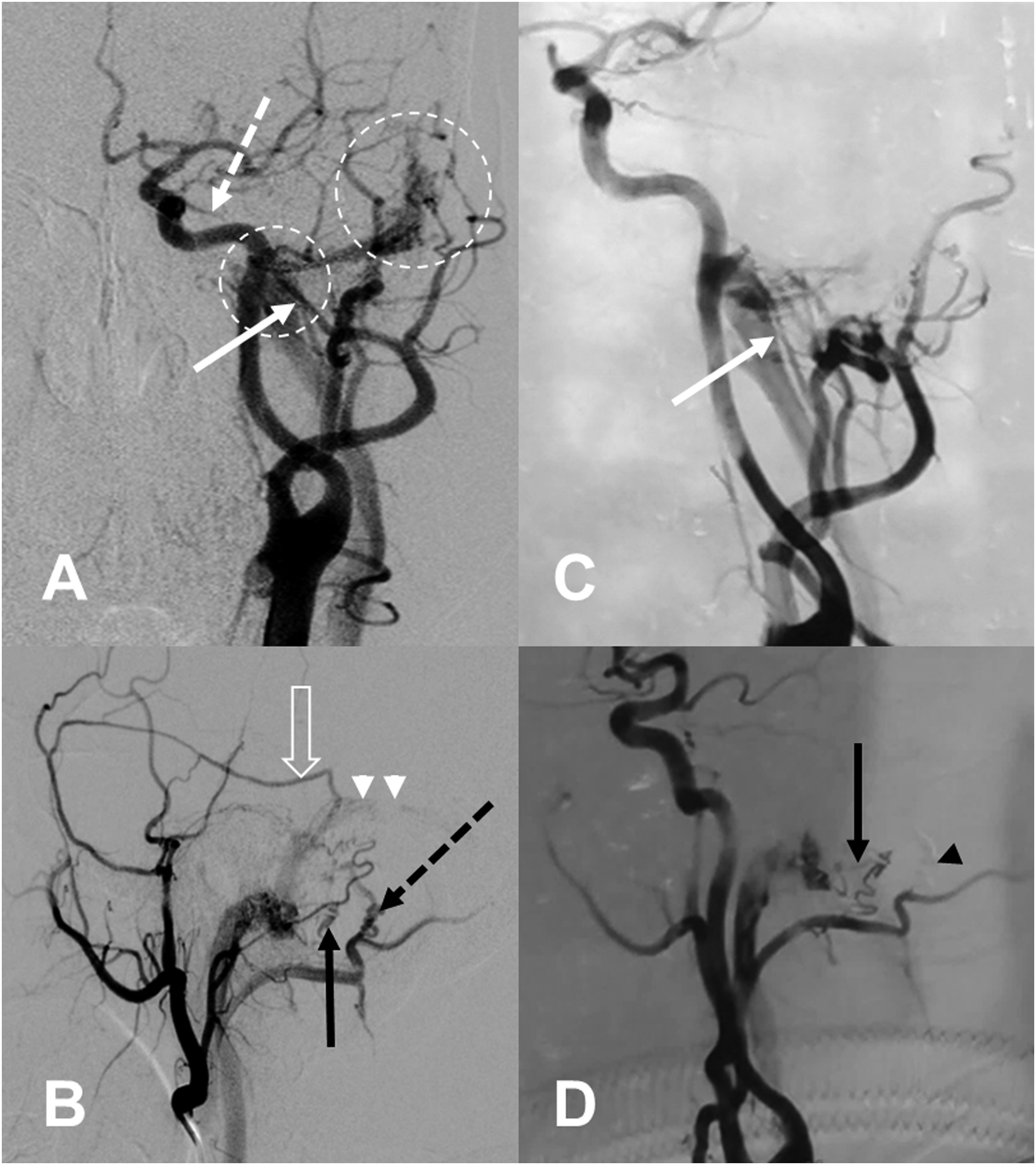
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grant from the S &T Program of Hebei (H2020505016) and Medical Science Research Program of Hebei (20190182).
