Abstract
Objective:
To highlight that dural arteriovenous fistula (DAVF) can exist in subjects with unilateral vascular pulsatile tinnitus (PT), positive internal jugular vein (IJV) compression tests, and radiologic evidence of sigmoid sinus wall anomalies (SSWA).
Methods:
A retrospective analysis was conducted on 80 subjects with PT as the primary symptom and intracranial DAVF detected based on magnetic resonance angiography results. Among them, IJV and retroauricular compression tests and temporal bone computed tomography (CT) were performed. The moth-eaten sigmoid plate sign and DAVF-induced SSWA were defined, and their correlation with PT duration was studied.
Results:
Significant differences were observed between ipsilateral IJV and retroauricular compression outcomes in DAVFs located at the transverse-sigmoid sinus (P < .001) and hypoglossal canal (P = .026). Among 71 subjects with CT data, the moth-eaten sign was found in 29 of 37 subjects (sensitivity 78.4%, 95% CI 0.63-0.89) with DAVFs at the transverse-sigmoid sinus. SSWA and jugular bulb (JB) anomalies were observed in 40.8% of subjects. PT duration significantly differed between subjects with SSWA and those without SSWA (P < .0026).
Conclusion:
The presence of SSWA on CT and a positive IJV compression test should not be considered conclusive for diagnosing venous PT. The “moth-eaten sigmoid plate” sign on non-contrast CT and positive retroauricular compression are strong indicators of DAVF as the primary cause of PT.
Keywords
Introduction
Sigmoid sinus wall anomalies (SSWA) and intracranial dural arteriovenous fistulas (DAVFs) are common causes of vascular pulsatile tinnitus (PT), a condition in which patients perceive blood flow sounds synchronized with their heartbeat.1,2 SSWA includes sigmoid sinus wall dehiscence and diverticulum, while DAVF involves an abnormal connection between arteries and veins within the dura mater, accounting for 10% to 15% of all intracranial arteriovenous malformations.1,3,4 Both conditions are amenable to surgical treatment, although DAVF primarily relies on interventional procedures. PT is a common symptom of intracranial DAVF, and up to 70% of transverse-sigmoid sinus DAVFs present with PT. 1 Besides PT, intracranial DAVF can lead to intracranial hemorrhage, venous thrombosis, and increased intracranial pressure. The existing literature provides comprehensive documentation of these conditions and their treatment approaches.2,4-7
In prior studies, numerous diagnostic algorithms have been devised to distinguish between venous and arteriovenous PT by conducting the ipsilateral internal jugular vein (IJV) compression test. This test typically elicits a response in subjects with SSWA upon the compression of the ipsilateral IJV, a reaction that is generally not observed in DAVF.1,8 However, the IJV compression method is not consistently reliable for all PT patients, necessitating additional objective diagnostic techniques, such as the water occlusion test and trans-canal recording technique, to aid in PT diagnosis.9,10
The mechanisms of PT caused by SSWA and DAVF differ. Symptomatic PT generally arises from an objective acoustic source with specific transmission pathways to the inner ear, enabling its perception by the cochlea. While both SSWA- and DAVF-related PT are generated by blood flow kinetic energy, 11 SSWA-related PT originates from dural venous sinus flow or intrasinus vortex (which may or may not be enhanced by transverse sinus stenosis), transmitting primarily via an air-conduction route through temporal bone dehiscence.2,6 In contrast, DAVF-related PT may result from both enhanced shunt flow and/or venous reflux/turbulence at various locations near the petrous bone, transmitting predominantly via the air and/or bone conduction route to the cochlea.12,13
An increasing trend has been observed in our PT clinic where patients who have undergone sigmoid sinus wall resurfacing/compression surgery at other facilities are subsequently diagnosed with DAVF-induced PT after the procedure fails to resolve their symptoms, despite the presence of SSWA. This study aimed to demonstrate that the physical and radiologic outcomes for PT caused by SSWA and DAVF can overlap, necessitating a thorough investigation to differentiate between PT caused by SSWA and PT caused by “DAVF mimicking SSWA.” We hypothesize that a positive IJV compression response combined with CT evidence of SSWA is not sufficient to identify candidates for SSWA surgery. To that end, the feeding artery of a DAVF involving the transverse sinus may cause sigmoid plate erosion and produce signs of jagged and mosaic edges on the sigmoid plate, distinct from SSWA. Understanding the diagnostic and radiologic nuances in differentiating PT caused by SSWA from that caused by DAVF with SSWA is crucial to prevent the mismanagement of DAVF-related PT as SSWA-associated venous PT.
Patients and Methods
Patient Clinical Data and Diagnostic Algorithm
This retrospective study comprises 80 outpatients diagnosed with DAVF-induced PT at the PT clinic of Fudan Eye & Ear Hospital consecutively from January 2016 to August 2024. Patients regardless of positive or negative IJV tests were ordered to undergo non-contrast CT and magnetic resonance angiography (MRA)/venography.
Subsequently, all subjects completed MRA test, while a total of 10 subjects declined CT scans due to personal health concerns, CT radiation, time constraint, and economic reasons. Upon the diagnosis of DAVF, all subjects were immediately referred to the interventional neuroradiologists of other tertiary hospital for digital subtraction angiography (DSA) examination and further treatment. Exclusions included PT caused by SSWA without the evidence of DAVF, temporal bone neoplasms, superior semicircular canal dehiscence, otosclerosis, anemia, and thyroid dysfunction. Among the 71 subjects with confirmable follow-up consultations, endovascular treatment was administered for PT, resulting in no recurrence in 64 cases, which accounts for 90.1% of the total. The remaining 7 subjects experienced a recurrence of PT; however, due to interventional surgery performed at other facilities, we are unable to report the surgical procedures. None of the subjects reported severe or remarkable complications.
Pure-tone audiometry was conducted on 28 subjects to screen for hearing loss. The remaining subjects declined to participate in the pure-tone audiometry test, citing their self-assessed absence of hearing loss as the reason. The air-conduction pure-tone average was determined at 0.125, 0.25, 0.5, 1, 2, 4, and 8 kHz. Low-frequency hearing imbalance was characterized by the following: (a) a difference of 10 decibels or greater in low-frequency thresholds across at least 2 sequential audiometric frequencies (0.25 and 0.5 kHz), or a difference exceeding 20 decibels at any single frequency (eg, 0.25 or 0.5 kHz), as identified through pure-tone audiometry and (b) the presence of higher low-frequency pure-tone thresholds (below 1 kHz) in the ear with PT compared to the ear without PT.
Ultrasonographic IJV Probe Compression and Retroauricular Compression
Ipsilateral ultrasonographic IJV probe compression was performed with subjects positioned in a supine position. For IJV compression test, a transducer LA-523 with a center frequency of 4 to 13 MHz (MyLab ClassC, Esoate SpA, Genova, Italy) was placed over the upper IJV at the level of the mandibular angle. Retroauricular compression was carried out by first identifying the bifurcation of the carotid artery and tracing it to the lower retroauricular region. The applied pressure should be adequate and gentle enough to cause collapse of the IJV and the ~1 cm from the root of the external carotid artery on the occipital artery under ultrasonographic visualization.
The change in PT during compression tests were categorized into 2 tiers: (a) persistent (negative), meaning no change to PT and (b) responsive (positive), meaning PT reduced or disappeared after compression.
Protocols of Radiologic Modalities and Definitions
Non-contrast enhanced CT examination was performed using a SOMATOM Definition Flash CT scanner (Siemens AG, Munchen, Germany) with settings of 192 mA and 120 kV. The “moth-eaten sigmoid plate” sign of sigmoid plate was identified as appearance of unsmoothed and rough edges between the osseous and vascular wall lining of the petrous surface and sigmoid sinus lumen, with or without osteolytic changes (increased radiolucency) at the mastoid cortex and/or sigmoid plate at least 3 consecutive axial CT images. Dehiscence is defined as the absence or discontinuity of bone structure in the sigmoid plate, leading to the appearance of a direct contact between sigmoid sinus wall and air. A diverticulum is defined as an outpouching of the sigmoid sinus vessel wall that protrudes into the mastoid air cells and/or cortex, with an irregular trajectory of the vascular silhouette. 2
Routine MRA and specific sequences for PT are detailed in Supplementary Material 1. MRA was performed when subjects were highly suspected of having DAVF-related PT, with the location of DAVF involving the transverse sinus identified. The location of DAVF is categorized into transverse-sigmoid sinus, hypoglossal canal, JB, middle cranial fossa, cavernous sinus, and parotid regions. An additional susceptibility-weighted imaging method is applied when DAVF is suspected on the 3D time-of-flight sequence.
Statistical Analysis
Statistical analysis was performed using Origin 9.1.0 Pro (OriginLab Corporation, Boston, USA). The Shapiro-Wilk test was employed to assess the normality of continuous data. Depending on the normality, either a 2-sample t-test or the Mann-Whitney U test was applied. Categorical data were evaluated using either the chi-squared test or Fisher’s exact test. A P < .05 was considered statistically significant.
Results
Descriptive Characteristics and Locations of DAVF
The average age of all subjects was 48.4 ± 13.3 years (range: 22-78 years). The cohort consisted of 37 males and 43 females. The duration of PT averaged 18.7 ± 23.9 months (range: 1-120 months). Among the 80 subjects with DAVF in which PT was the primary symptom, 71 subjects (88.8%) had unilateral PT, while 9 subjects (11.2%) had bilateral PT. All unilateral PT cases corresponded to the side of the DAVF lesion. Of the 9 subjects with bilateral PT, 3 had DAVFs at the transverse-sigmoid sinus, 3 at the hypoglossal canal, 2 at the cavernous sinus, and 1 had DAVFs at both the right transverse sinus and the left hypoglossal canal.
Among the 80 DAVF cases, the transverse-sigmoid sinus was the most common site, accounting for 42 cases (52.5%). The second and third most common sites were the hypoglossal canal with 20 cases (25%) and the middle cranial fossa with 9 cases (11.3%).
Besides PT, a total of 5 subjects reported headaches, with 4 subjects having DAVF involving the transverse-sigmoid sinus and 1 with DAVF in the hypoglossal canal. Additionally, a subject with a cavernous sinus DAVF reported experiencing moderate bilateral ocular pain.
Out of the 28 subjects who underwent pure-tone audiometry, 2 individuals (comprising 7.7%) were identified with ipsilateral low-tone hearing asymmetry (below 1 kHz). Both subjects with bilateral PT were excluded from this count. Among those with hearing asymmetry, 1 had a transverse-sigmoid DAVF, presenting a 25 dB peak difference, while the other had a cavernous sinus DAVF, showing a 15 dB difference.
IJV/Retroauricular Compression Outcomes
The outcomes of ipsilateral IJV and retroauricular compression tests are detailed in Table 1. Significant differences were observed between the outcomes of ipsilateral IJV and retroauricular compression in DAVFs located at the transverse-sigmoid sinus (Fisher’s exact test, P < .001) and the hypoglossal canal (Fisher’s exact test, P = .026).
Outcomes of Pulsatile Tinnitus Changes During Ipsilateral Internal Jugular Vein and Retroauricular Compression of 80 DAVF Subjects.
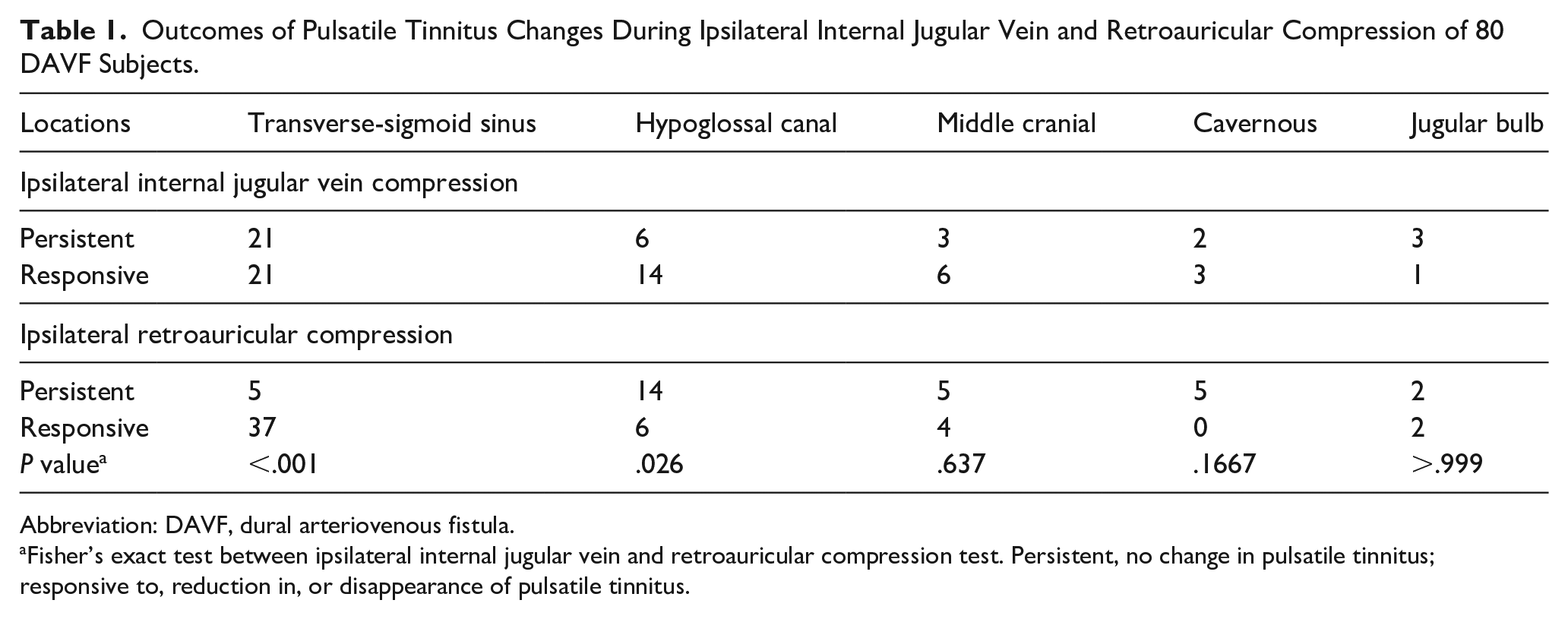
Abbreviation: DAVF, dural arteriovenous fistula.
Fisher’s exact test between ipsilateral internal jugular vein and retroauricular compression test. Persistent, no change in pulsatile tinnitus; responsive to, reduction in, or disappearance of pulsatile tinnitus.
“Moth-Eaten Sigmoid Plate” Sign and SSWA
Non-contrast CT imaging was performed on 71 of the 80 DAVF subjects. The moth-eaten sigmoid plate sign was identified in 35 subjects (49.3%; Table 2). Specifically, the sign was found in 29 of 37 subjects with DAVFs at the transverse-sigmoid sinus (sensitivity 78.4%, 95% CI 0.63-0.89), 2 of 4 subjects with DAVFs at the JB region (sensitivity 50.0%, 95% CI, 0.15-0.85), and 3 of 17 subjects with DAVFs at the hypoglossal canal (sensitivity 17.6%, 95% CI, 0.06-0.41; Figure 1).
Moth-Eaten Sigmoid Plate Sign and Duration of PT Caused by DAVF.
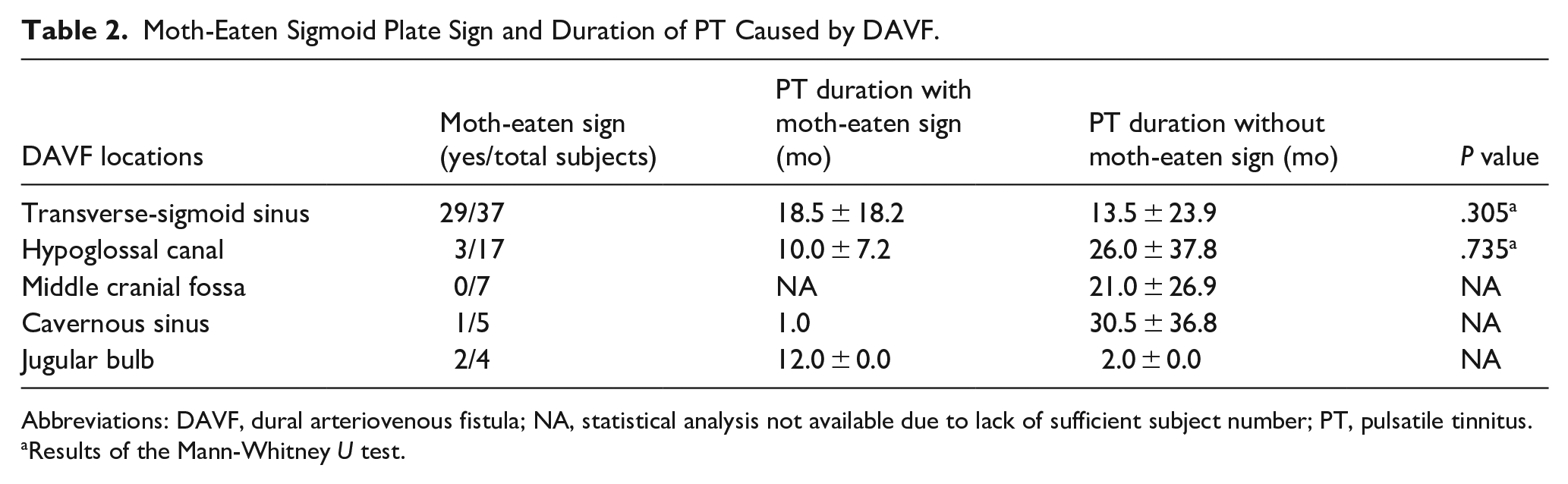
Abbreviations: DAVF, dural arteriovenous fistula; NA, statistical analysis not available due to lack of sufficient subject number; PT, pulsatile tinnitus.
Results of the Mann-Whitney U test.
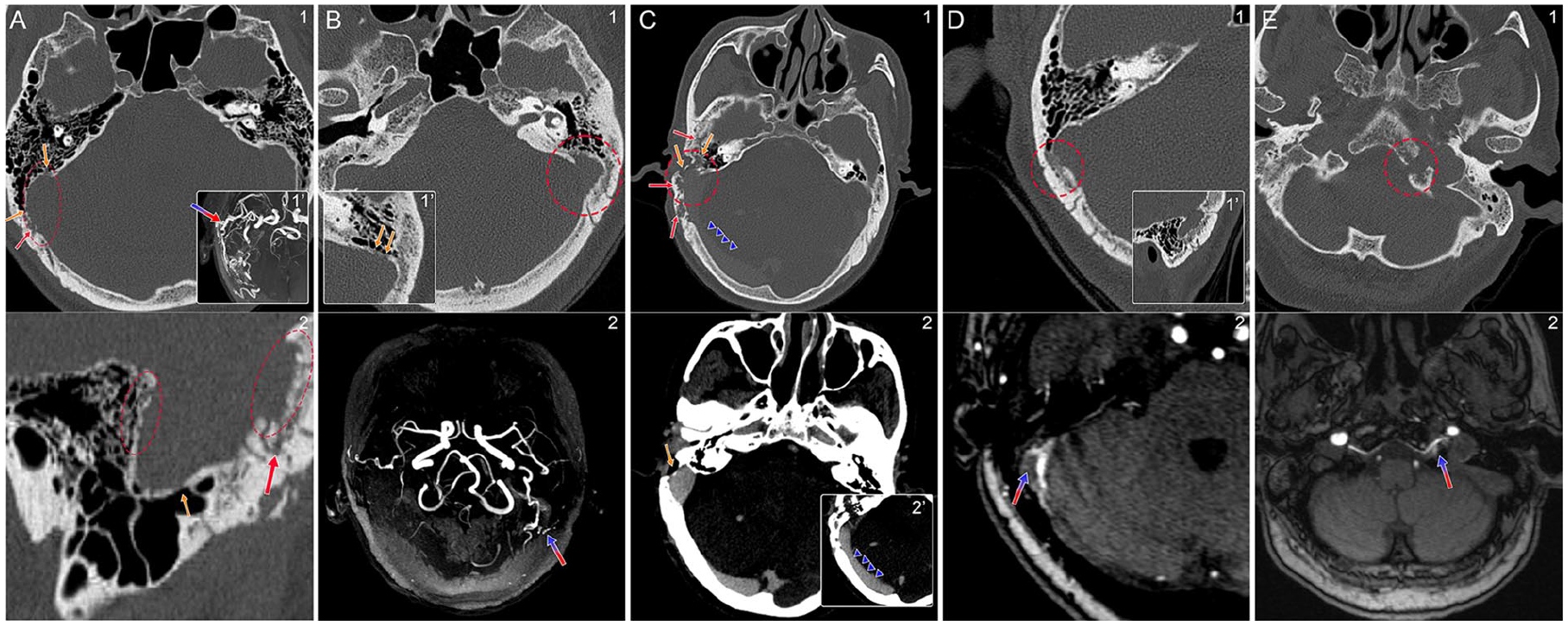
The newly-described “moth-eaten sigmoid plate” sign and CT indicators for DAVF. The blue-red arrow marks the location of the DAVF; the red dotted rectangle/circle highlights the moth-eaten sigmoid plate sign; the red arrow points to bone-penetrating vascular structures; and the yellow arrow indicates dehiscence or diverticulum of the sigmoid sinus wall. (A, 1) Axial non-contrast CT image showing the moth-eaten sigmoid plate sign on the right side and the transverse-sigmoid sinus DAVF. Notice the jagged and rough edges (red dotted rectangle) and the DAVF-induced SSWA, where erosive changes are observed on the sigmoid plate (yellow arrows). (1′) Axial MIP image of the DAVF. (2) Sagittal non-contrast CT image showing the moth-eaten sigmoid plate sign, sigmoid plate dehiscence, and penetrating vascular structures. (B, 1) Axial non-contrast CT image revealing the moth-eaten sigmoid plate sign and SSWA. (1′) Note the dehiscent changes in the sigmoid plate. (2) Axial MIP image from MRA showing the transverse-sigmoid sinus DAVF. (C, 1, 2, 2′) Axial contrast-enhanced CT images displaying a pronounced right-sided moth-eaten sigmoid plate sign, enlarged bone-penetrating vasculature, severely-encroached mastoid cortex, and a dilated transverse sinus (blue triangles) resulting from the transverse-sigmoid sinus DAVF. (D, 1) Axial and (1′) sagittal non-contrast CT images showing the right-sided moth-eaten sigmoid plate sign and lateral encroachment of the mastoid cortex. (2) Axial MRA image illustrating the transverse-sigmoid sinus DAVF. (E, 1) Moth-eaten sigmoid plate sign located at the left hypoglossal canal, accompanied by sclerotic changes in the clivus and sphenoid bones. (2) Axial MRA image revealing the left DAVF at the hypoglossal canal. DAVF, dural arteriovenous fistulas; MIP, maximum intensity projection; MRA, magnetic resonance angiography; SSWA, sigmoid sinus wall anomalies.
Among the 71 subjects with CT data, SSWA and JB anomalies were observed in 29 subjects (40.8%; Table 3). Of these, 21 subjects had DAVFs at the transverse-sigmoid sinus, 6 subjects had DAVFs at the hypoglossal canal, and 2 subjects had DAVFs at the jugular foramen region. There was a significant difference in the duration of PT between subjects with SSWA (average: 29.5 ± 25.4 months, range: 2-84 months) and those without SSWA (average: 5.9 ± 5.3 months, range: 1-18 months) (2-sample T-test, P < .0026) among subjects with DAVFs at the transverse-sigmoid sinus. A total of 19 (54.3%) subjects had both moth-eaten sign and SSWA simultaneously.
Sigmoid Sinus Wall Anomalies and Duration of Pulsatile Tinnitus Caused by DAVF.
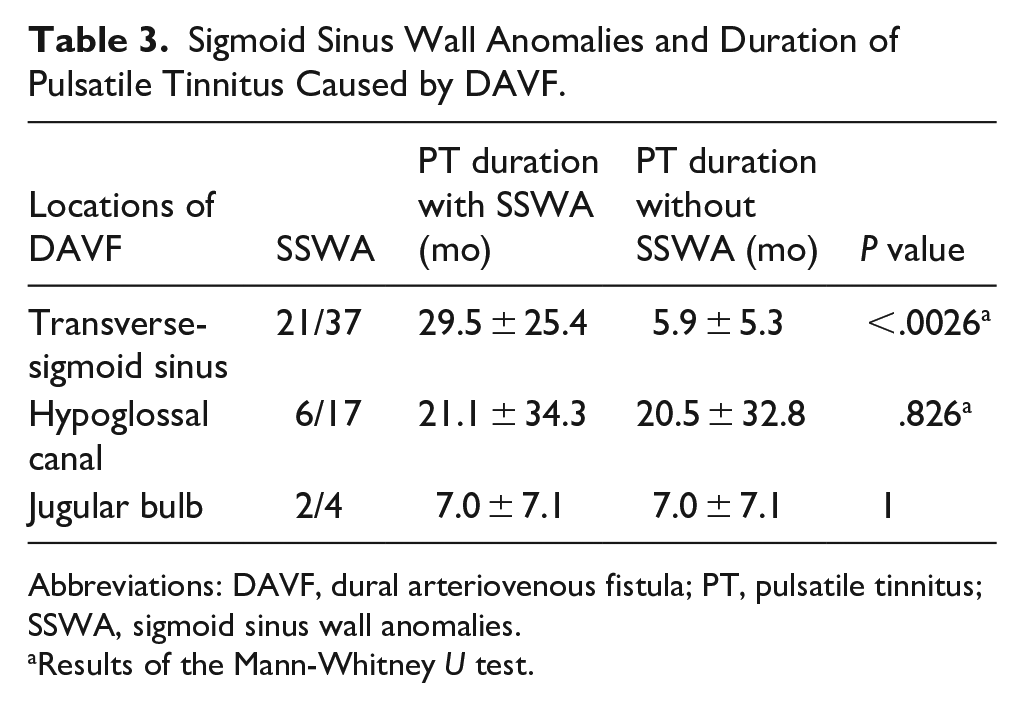
Abbreviations: DAVF, dural arteriovenous fistula; PT, pulsatile tinnitus; SSWA, sigmoid sinus wall anomalies.
Results of the Mann-Whitney U test.
Those with DAVF who had negative IJV compression outcome and SSWA but without moth-eaten sigmoid plate sign are shown in Figure 2.
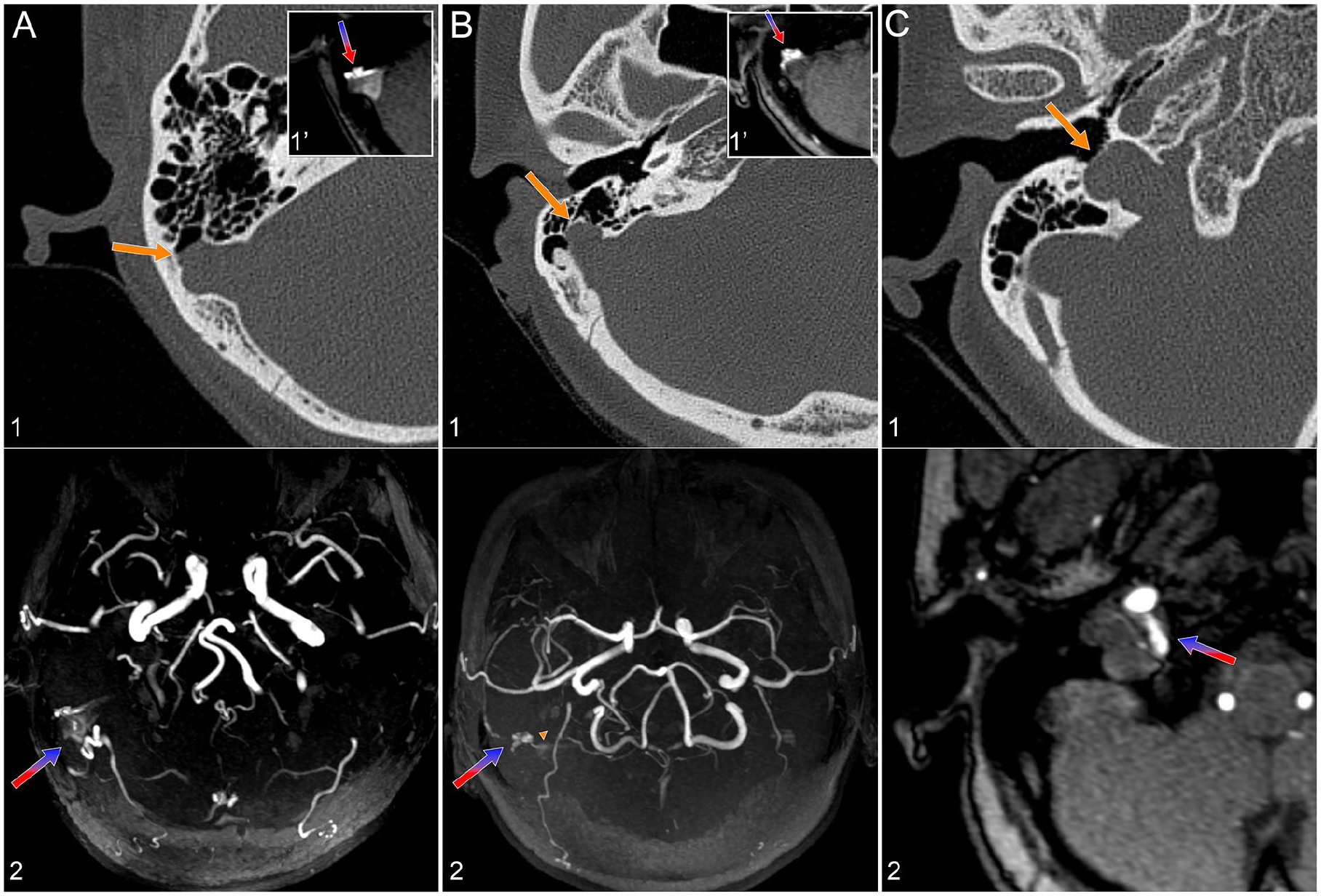
DAVF mimicking SSWA. All subjects had positive IJV tests. The blue-red arrow marks the location of the DAVF, while the yellow arrow indicates dehiscence or a diverticulum of the sigmoid sinus wall. (A, 1) A right-sided DAVF causing a diverticulum without the moth-eaten appearance, where ipsilateral occipital artery vessel runs into the sigmoid sinus. (1′) MRA slice showing the DAVF. This right-sided DAVF causes a diverticulum without the moth-eaten appearance. (2) Axial MIP image from MRA displaying the transverse-sigmoid sinus DAVF. (B, 1) Another right-sided DAVF causing an anteriorly-protruding diverticulum without the moth-eaten appearance, where the dural branch of the ascending pharyngeal artery (yellow triangle) runs into the sigmoid sinus. (1′) MRA slice showing the DAVF. (2) Axial MIP image from MRA illustrating the transverse-sigmoid sinus DAVF. (C, 1, 2) A right-sided DAVF located at the hypoglossal canal with right jugular bulb dehiscence, without the moth-eaten appearance. DAVF, dural arteriovenous fistulas; IJV, internal jugular vein; MIP, maximum intensity projection; MRA, magnetic resonance angiography; SSWA, sigmoid sinus wall anomalies.
Discussion
An algorithm for screening venous PT using postive IJV compression test and evidence of SSWA on CT cannot reliably identify the underlying cause of PT induced by DAVF. Notably, more than 50% of DAVF subjects experience either complete or partial reduction in PT sensation during IJV compression, irrespective of DAVF location. In addition to DAVFs at the transverse-sigmoid sinus, SSWA may also be present in subjects with DAVFs at the hypoglossal canal and JB regions. These outcomes may confuse clinicians in determining the true cause of PT if otologists rely solely on CT and are unfamiliar with ruling out DAVF despite the presence of SSWA.
In addition to the comprehensive analysis of physical examination techniques summarized in Cummins’ recent article, 13 2 techniques introduced here aim to screen for DAVF in individuals presenting with PT as the primary symptom, before contrast-enhanced CT and MRA are performed: (a) retroauricular compression and (b) the currently-identified “moth-eaten sigmoid plate” sign observed via non-contrast CT. The external carotid artery branches traverse the retroauricular region, and by collapsing its vascular wall, PT may be reduced or eliminated. This method is applicable across all DAVF regions causing PT, as the primary feeding arteries of DAVFs commonly originate from the external carotid artery (up to 80%), which the middle meningeal artery is usually involved.14,15 Additionally, contrast-enhanced CT can reveal asymmetrically-enlarged feeder arteries, typically the external carotid artery branches such as occipital artery and meningeal artery, turtous and shaggy vasculature, transcalvarial channels, or enlarged dilated venous structures and cortical veins near DAVF site.16,17
The “moth-eaten sigmoid plate” sign observed along the border of the sigmoid sinus vascular wall/sigmoid plate and the mastoid cortex is a highly-indicative CT marker of DAVF involving the transverse-sigmoid sinus, accounting for nearly 80% of cases with transverse-sigmoid sinus DAVF. 15 This specific sign differs from osteolytic temporal boen neoplasms, osteonecrotic changes and diffuse/polyostotic involvement of calvarium/skull base such as Paget’s disease that cause more extensive and deeper alterations to the entire bone structure rather than being confined to areas adjacent to the sigmoid sinus wall and mastoid cortex. 18 Although the precise mechanism behind moth-eaten sigmoid plate sign’s emergence is unclear, our data suggest that it can develop within weeks and is independent of PT duration. This indicates that DAVF, along with the proliferation of numerous small vascular structures penetrating the mastoid cortex, may precede the onset of PT sensation. With this evidence, it is hypothesized that high pressure penetrating vascular structures, venous reflux, and increased intracranial pressure could be critical factors leading to osteolytic changes in the sigmoid plate, ultimately causing SSWA. These changes likely occur before the onset of PT and may share the same pathogenesis as SSWA of venous etiologies, with transverse sinus stenosis or increased intracranial pressure being contributory factors. 19 The moth-eaten sign, along with sclerotic bone appearances, can also be observed at the sphenoid bone-clivus and hypoglossal canal regions, 20 although it is significantly less common compared with cases of DAVF occurring at the transverse-sigmoid sinus regions. Given that DAVF-induced SSWA and the moth-eaten sign coexist in 54.3% of subjects, it is postulated that the 2 may share an identical pathogenesis. Since DAVF-induced SSWA is time-dependent, with an average longer PT duration, it can be extrapolated that SSWA is a consequence of the moth-eaten sign, possibly due to flow impingement and aseptic inflammation of the bone structure, ultimately leading to bone loss.
True SSWA-induced venous PT does not coexist with the moth-eaten sigmoid plate sign. However, it is note worthy that 4 subjects exhibited SSWA without the moth-eaten sign, and 1 with JB dehiscence showed no discernible differences from cases of true SSWA-induced venous PT. Therefore, clinicians should consider the possibility of DAVF-induced PT even in the absence of the moth-eaten sign. In such cases, DAVF-induced SSWA can lead to the misdiagnosis of venous PT, potentially resulting in inappropriate transmastoid sigmoid sinus wall resurfacing surgery if advanced radiologic modalities such as dynamic MRA, dynamic CT angiography, and DSA are not employed. 16
The current study observed a significantly-lower rate of ipsilateral low-frequency hearing loss at 7.7%, contrasting with the 50% reported by An et al. 1 The 2 subjects with DAVF at different locations, who exhibited hearing loss on pure-tone audiometry, suggest that either air or bone conduction could be responsible for the masking effect. PT from cavernous DAVF is less likely to be transmitted via air conduction due to the absence of an aerial space as a medium. Given that PT caused by DAVF can be recorded through external canal recording or auscultated with a stethoscope, 1 it is reasonable to infer that both pathways might contribute to low-to-middle frequency hearing loss due to the masking effect of PT. This effect is analogous to that observed in individuals with SSWA. 5
Finally, some unfortunate yet noteworthy cases highlight the challenges in interventional catheter insertion at the DAVF site, which becomes nearly impossible due to the overcompression and collapse of the sigmoid sinus following multiple transtemporal surgeries erroneously aimed at treating venous PT (Figure 3). Given that PT typically responds positively to ipsilateral IJV compression in DAVF patients, some may consider further compressing the sigmoid sinus during revision surgery using robust materials like bone wax if initial dehiscence resurfacing fails. However, this approach can complicate subsequent interventional procedures and possibly induce the risk of DAVF progression, necessitating additional open surgery to retrieve all inserted materials, and PT cannot be altered simply by operating on the sigmoid plate alone.
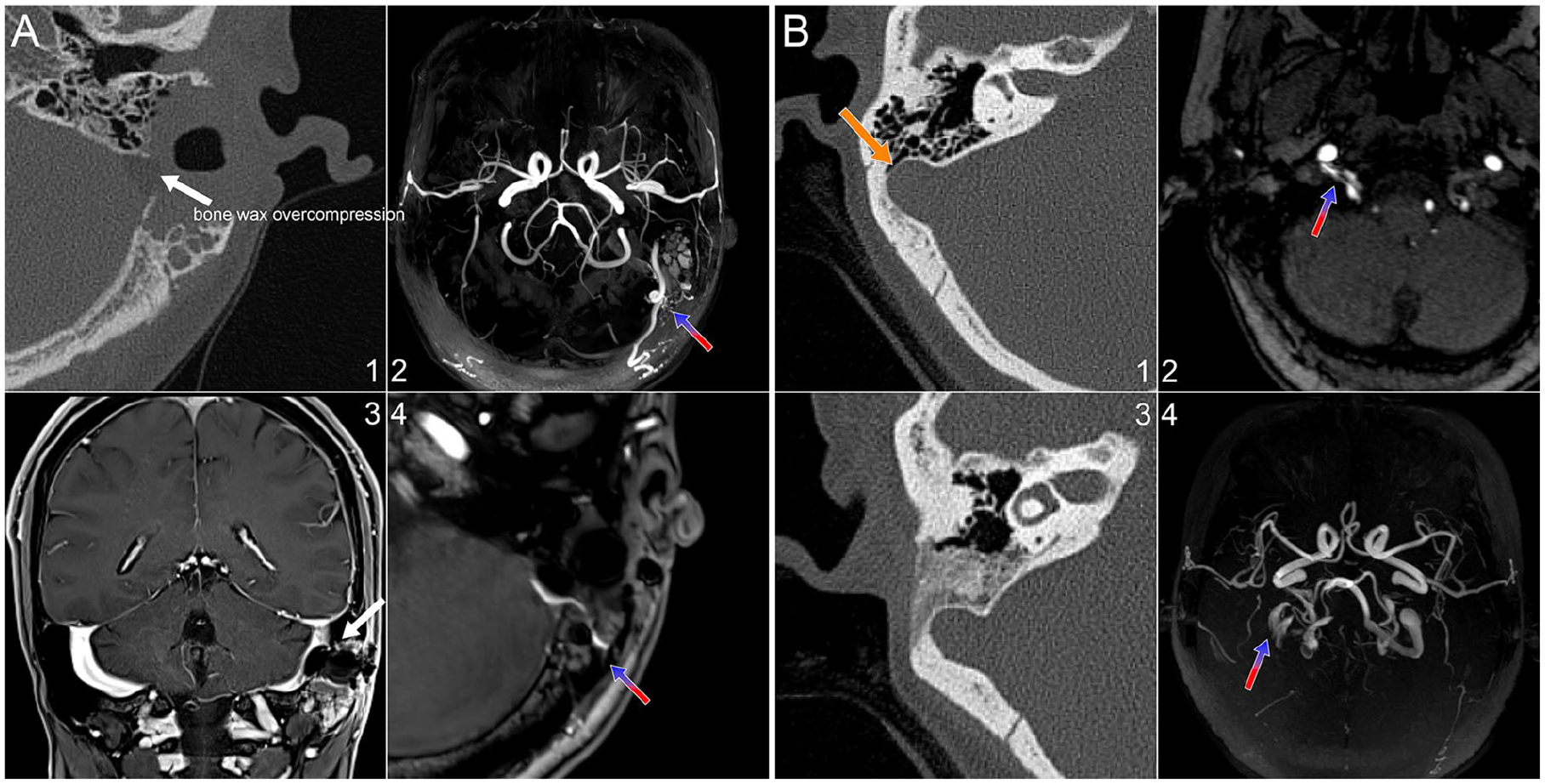
DAVF cases involving patients who underwent transtemporal resurfacing surgery, mistakenly aimed at treating venous PT, encountered at our PT clinic. All subjects had positive IJV tests. All subjects with DAVF were rapidly referred to interventional radiologists. The blue-red arrow marks the location of the DAVF. (A, 1-4) One subject with a left-sided DAVF underwent 2 resurfacing surgeries at another medical facility. The first surgery addressed dehiscence, but PT recurred. The surgeon then performed a second surgery, completely collapsing the sinus wall using bone wax and hard materials since the IJV compression test remained positive. PT recurred a few days after the surgery, leading the patient to seek treatment at our clinic. Upon evaluation, it was revealed that an underlying transverse-sigmoid sinus DAVF was the true cause of the PT. An interventional neuroradiologist later assessed this patient, noting limited venous access due to the absence of a sigmoid sinus entrance and a type 3 torcula herophili. The only remaining option for treatment was transarterial approach via the middle meningeal artery. Additionally, it was noted that such surgeries might increase the risk of DAVF progression. (B, 1, 2) A patient with a DAVF located at the hypoglossal canal was misdiagnosed at another facility as having sigmoid sinus wall dehiscence. Despite positive IJV compression, the patient underwent resurfacing surgery. (3, 4) PT worsened postoperatively, and the DAVF persisted. DAVF, dural arteriovenous fistulas; IJV, internal jugular vein; PT, pulsatile tinnitus.
First, this study faces limitations primarily in grading DAVF based solely on MRA data, which hinders accurate classification. The study concentrated on the diagnostic indicators of DAVF, not treatment outcomes; hence, the interventional results were reported based on consultations rather than on comprehensive medical records and imaging data. This was due to the absence of an interventional radiology department within our institution. Second, the sample size, particularly for DAVF in the JB, is insufficient, potentially limiting the physical examination outcomes. Third, while low-frequency hearing loss and intracranial hypertension can overlap in SSWA or DAVF patients, our study did not assess papilledema or perform lumbar punctures due to the rarity of these symptoms as the sole presentation in patients with PT and the rapid referral of such cases to other tertiary hospitals for further evaluation. This omission may prevent definitive identification of increased intracranial hypertension. Lastly, the study’s scope, restricted to PT and DAVF patients, precludes the establishment of a control group without PT for specificity calculation due to DAVF’s infrequency and unique clinical profile. Despite these limitations, the sensitivity findings suggest the moth-eaten sigmoid plate sign’s diagnostic potential in DAVF.
Conclusion
DAVFs involving the transverse-sigmoid sinus and hypoglossal canal can cause SSWA/JB abnormalities, potentially mimicking the clinical and non-contrast CT findings typically seen in SSWA-venous PT. Therefore, the presence of sigmoid sinus dehiscence/diverticulum on CT and a positive IJV compression test should not be deemed definitive for a venous PT diagnosis. Consequently, routine MRA and other appropriate vascular imaging modalities are essential for detecting underlying DAVFs in patients with vascular PT. Furthermore, a positive retroauricular compression and the “moth-eaten sigmoid plate” sign on CT are indicative of DAVFs affecting the transverse sinus and/or other locations, aiding otolaryngologists in distinguishing between DAVF and venous PT.
Supplemental Material
sj-docx-1-ear-10.1177_01455613251342851 – Supplemental material for Intracranial Dural Arteriovenous Fistula Can Mimic Sigmoid Sinus Wall Anomalies Induced Pulsatile Tinnitus: Caution Before Considering It’s Venous
Supplemental material, sj-docx-1-ear-10.1177_01455613251342851 for Intracranial Dural Arteriovenous Fistula Can Mimic Sigmoid Sinus Wall Anomalies Induced Pulsatile Tinnitus: Caution Before Considering It’s Venous by Yue-Lin Hsieh, Xu Liu, Shenjiang Wang and Wuqing Wang in Ear, Nose & Throat Journal
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Science and Technology Innovation Plan of Shanghai Science and Technology Commission (no. 22Y11902200) to Wuqing Wang.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
