Abstract
Chondromesenchymal hamartoma (CMH) is a rare, benign lesion of the nasal cavity, paranasal sinuses, and skull base, composed of islands of hyaline cartilage in a myxoid background. The vast majority of CMH cases are infants and young children. According to the world literature, nasopharyngeal involvement of CMH is extremely rare. In all cases, the lesions were masses protruding from the nasal cavity or paranasal sinuses to the nasopharynx. We hereby report 2 adult male patients with masses completely situated in the nasopharyngeal space. In the first patient, the tumor originated from the posterior edge of the nasal septum and in the second one, from the posterolateral wall of the nasopharynx, adjacent to the pharyngeal orifice of the Eustachian tube. In both patients, the lesion was excised endoscopically, and histopathological analyses were consistent with a diagnosis of CMH. To our knowledge, those are the only cases of CMH completely situated in the nasopharynx.
Introduction
Hamartomas are disorganized lesions of tissues indigenous to the area in which they arise. 1 Although some authors considered them to represent the results of developmental anomalies, the exact pathogenesis of different forms of hamartomas is not known. 1 The term hamartoma has Greek roots and is derived from the words hamartia (defect, error) and oma (tumor-like growth). 1 These lesions are best classified according to their predominant histological characteristics into epithelial or mesenchymal. 2 Among epithelial hamartomas of the sinonasal region are respiratory epithelial adenomatoid hamartoma and seromucinous hamartoma, while the most common type of mesenchymal hamartomas is chondromesenchymal hamartoma (CMH). 2 Nasal CMH is a rare, benign tumor in infant and young children, found usually in the nasal cavity, paranasal sinuses, orbit, and skull base. 3 Only 50 cases have been reported to date, of whom only several were adult patients.3,4
Chondromesenchymal hamartoma is extremely rare in the nasopharynx, and there have been only a few case reports in the literature. In all the cases, the lesions were masses protruding from the nasal cavity or paranasal sinuses to the nasopharynx.5-8 In this work, we report 2 new cases of nasopharyngeal CMH in adult patients, and to our knowledge, those are the only cases of CMH completely situated in the nasopharyngeal space.
Case Reports
Case 1
A 42-year-old male presented with a 3-year history of slowly progressive bilateral nasal obstruction, snoring, intermittent mucopurulent anterior and posterior nasal discharge, and mild postnasal hemorrhage. Except for arterial hypertension, his other medical history was unremarkable. An endoscopic examination showed the presence of a lobulated, smooth, yellow-pink polypoid lesion that originated from the posterior edge of the nasal septum. A coronal plane computed tomography (CT) scan of the paranasal sinuses (Figure 1A) demonstrated a soft-tissue mass which filled the nasopharynx. In order to diagnose the vascular anomalies and tumors, we performed a contrast-enhanced CT scan with angiography. The imaging showed the mass attached to the posterior edge of the nasal septum, of size 41.6 by 24.8 mm, with few demonstrable feeding vessels, and without significant enlargement. These findings were therefore inconsistent with a diagnosis of a vascular lesion or angiofibroma (Figure 1B). In addition, no calcifications or necrosis were observed within the lesion, and there were no abnormalities in the nasal cavity and paranasal sinuses. We then performed the complete endoscopic excision of the pedunculated lesion under general anesthesia. Macroscopically, the tumor appeared to be a soft, polypoid, yellow-pink mass no longer than 42 mm (Figure 2). Histological examination showed pseudostratified respiratory epithelium surfacing a stroma consisting of a highly cellular, benign spindle cell proliferation with variably sized nodules of hyaline cartilage, vascularized by normal vessels (Figure 3). These findings were consistent with a CMH diagnosis. At follow-up 12 months after the excision, the patient showed good recovery, without the residue. Four years after the excision, the patient was without disease recurrence.
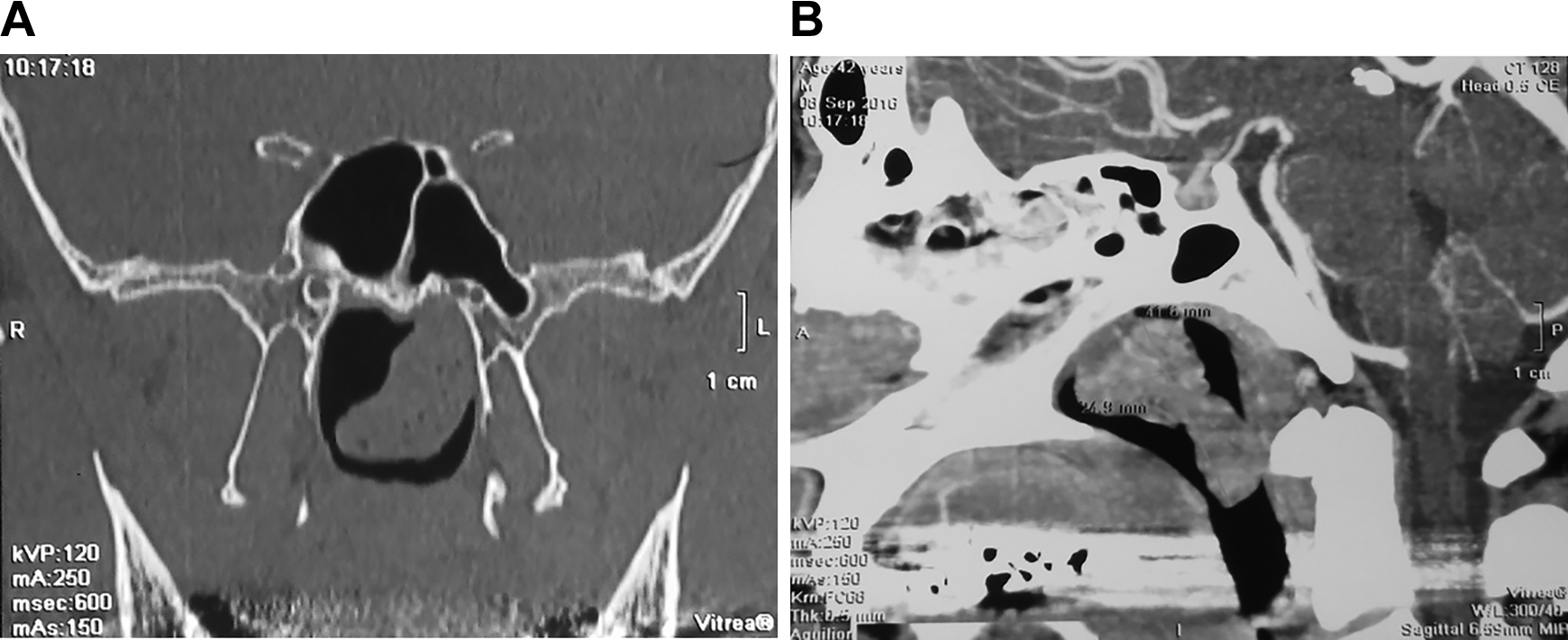
A, Coronal plane of the CT scan showing the soft-tissue mass filling the nasopharyngeal space. B, Sagittal plane of a contrast-enhanced CT scan with angiography showing the lesion attached to the posterior edge of the nasal septum, with dimensions of 41.6 × 24.8 mm with only few demonstrable feeding vessels, without significant enlargement, excluding the diagnosis of a vascular lesion or angiofibroma. No calcifications or necrosis was seen within the lesion. CT indicates computed tomography.
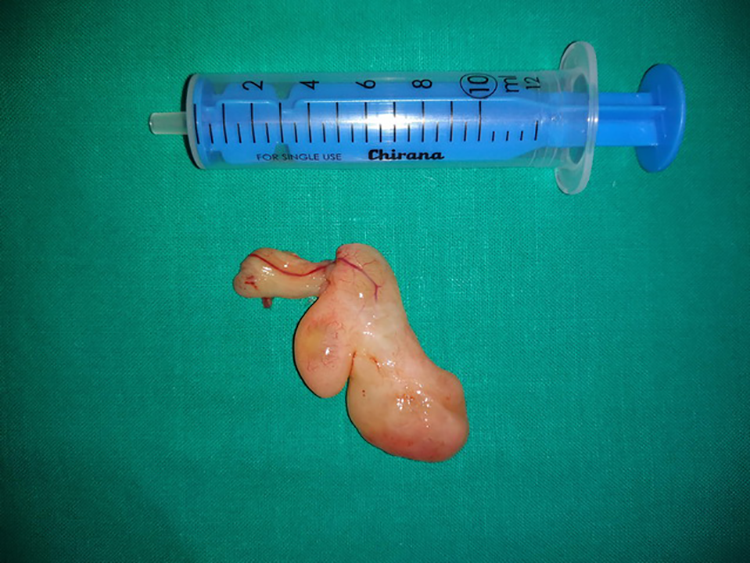
Macroscopically, the excised tumor appeared to be a soft, polypoid, yellow-pink mass with maximum dimension of 42 mm.
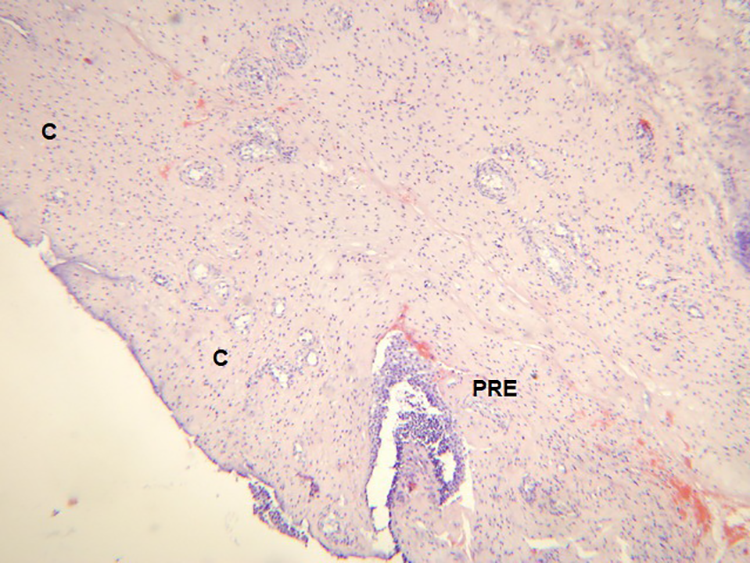
Histopathological examination showing the pseudostratified respiratory epithelium surfacing a stroma consisted of a highly cellular, benign spindle cell proliferation with variably sized nodules of hyaline cartilage, vascularized by normal vessels (hematoxylin & eosin stain, magnification ×50). C indicates hyaline cartilage; PRE, pseudostratified respiratory epithelium.
Case 2
A 48-year-old male was admitted to our ENT department with a 1-year history of slowly progressive right-sided nasal obstruction, mucopurulent postnasal drip, and the sensation of a foreign body in the throat. The patient also had a history of recurrent right-sided otitis media with effusion, with the first episode reported 6 months prior. Endoscopic examination revealed the presence of a smooth, polypoid, pale-pink mass attached to the posterolateral wall of the nasopharynx, adjacent to the pharyngeal orifice of the Eustachian tube, which also showed on the CT scan of the skull base (Figure 4). Surgical excision of the lesion was performed via the endoscopic endonasal approach. The removed oval, polypoid mass was approximately 15 by 10 mm. Our histopathological examination showed the mass consisted of variably sized nodules of hyaline cartilage, set within a fibromyxoid background, with the presence of dilated seromucinous glands as well as mature adipose tissue components (Figure 5A). The lesion was covered by respiratory epithelium. Multiple islands of mature cartilage contained benign chondrocytes without cellular atypia, mitotic activity, or necrosis (Figure 5B). On immunohistochemical analysis, the stromal cells and chondrocytes were diffusely positive for S-100. The histologic pattern was that of CMH. A recent follow-up at 4 months revealed no recurrence, and the patient’s hearing was normal.

Sagittal plane of the CT scan showing a polypoid mass (arrow head), attached to the posterior wall of the nasopharynx. CT indicates computed tomography.
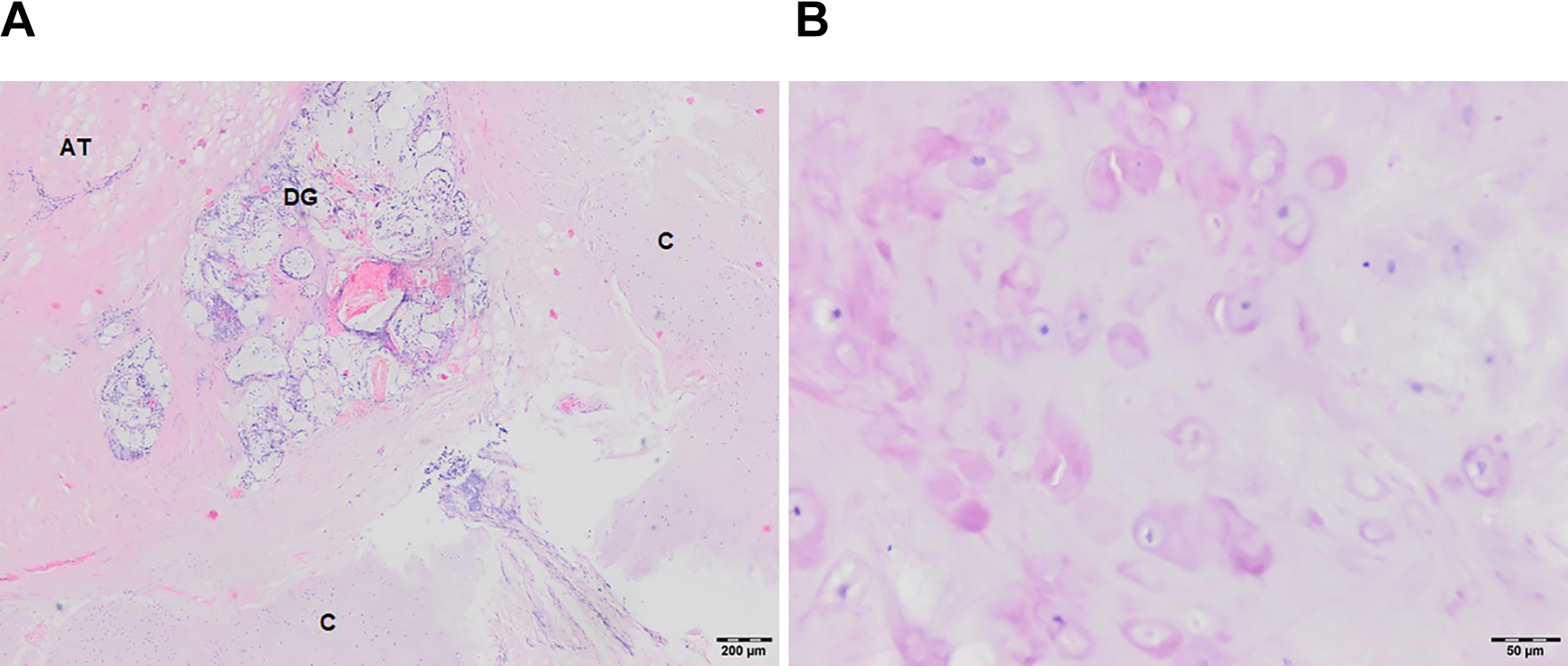
A, Histopathological examination showing variably sized nodules of hyaline cartilage, set within a fibromyxoid background, with presence of dilated seromucinous glands as well as mature adipose tissue components (hematoxylin & eosin stain, magnification ×25). B, Mature hyaline cartilage containing benign chondrocytes without cellular atypia, mitotic activity, or necrosis (hematoxylin & eosin stain, magnification ×200). AT indicates adipose tissue; C, hyaline cartilage; DG, dilated glands.
Discussion
Chondromesenchymal hamartoma was included in the World Health Organization classification of head and neck tumors for the first time in 2016, in the fourth revision. 2 However, it was recognized as a distinct tumor subtype 2 decades ago by McDermott et al. 9 The exact origin of this tumor is not known, but it is hypothesized to derive from embryologic rests. 3 It occurs predominantly in young children and infants as a lesion arising from the nasal septum and vestibule, paranasal sinuses, nasal turbinates, and skull base. The tumor is benign, but local invasiveness with intracranial involvement through the cribriform plate has been observed.2-4 However, few cases have been reported in adult patients.3,4 According to the systematic literature review by Mason et al, 4 the mean age of presentation is 9.6 years with a range from birth to 69 years. The male to female ratio is 2.2:1. 4 Recently, an association has been found between CHM and the predisposition for the pleuropulmonary blastoma tumor disorder in children, which is due to germline or somatic mutations of DICER1 gene.6,10 However, it is difficult to explain the origin of CMH in adult population.
Nasopharyngeal involvement of CMH takes place through the protrusion of this lesion from the nasal cavity, paranasal sinuses, and skull base to the nasopharyngeal space, and only 4 such cases have been reported in the world literature.5-8 We report the first 2 cases of CHM completely situated in the nasopharynx. In the first patient, the origin of the tumor was the posterior edge of the nasal septum and, in the second one, the posterolateral wall of the nasopharynx, near the pharyngeal part of the Eustachian tube. Besides the nasal obstruction and postnasal mucopurulent discharge in both patients, the first one had mild postnasal hemorrhage, and the second had several episodes of otitis media with effusion with conductive hearing loss, probably due to the mechanical obstruction of the Eustachian tube. Histologically, in both patients’ lesions, we found islands of hyaline cartilage and spindle mesenchymal cells in a myxoid background, without the presence of hyperchromasia, mitotic activity, cell atypia, or necrosis. In the second patient, however, we found the presence of seromucinous glands and mature adipose tissue components in the excised mass.
In the first patient, due to episodes of mild postnasal hemorrhage and the localization of the lesion, we first expected a diagnosis of angiofibroma or vascular malformation. Those lesions have been eliminated after the arteriography and the histopathological analysis. Through arteriography, we found no signs of pathological vascularization, and through histopathological examination, we found regular blood vessels, ranging from capillaries and sinusoids to large vessels, that were lined with one layer of endothelial cells lying in a fibrous stroma. 11
Regarding the second patient, the main differential diagnostic problems were nasopharyngeal dermoid (choristoma) and lymphoid hamartoma. Dermoid is a bigeminal lesion derived from ectodermal and mesodermal germ layers. It is a pedunculated or sessile polypoid mass, with the grayish-white external appearance of skin, also known as “hairy polyp.” It is present at birth and is usually discovered can be found during the neonatal period or in infancy, although, in exceptional cases, “hairy polyp” may be found in adulthood. 12 Nasopharyngeal dermoids are commonly elongated finger-like masses that hang down from the nasopharynx, causing respiratory obstruction. Most (about 60%) of them arise from the lateral wall of the nasopharynx and the nasopharyngeal surface of the soft palate. 12 Another frequent primary localization is the pharyngeal portion of the Eustachian tube. Microscopically, choristoma is lined with normal-appearing skin with hair follicles, arrector pili muscles, and sebaceous glands. The fatty/fibrofatty stroma commonly contains cartilage, muscles, and nerves. 12 In our patient, the excised mass was covered by respiratory epithelium and the stroma-contained fibromyxoid tissue with islands of mature hyaline cartilage, seromucinous glands, and adipose tissue. Park et al 13 reported a case of a lesion originating from the posterolateral wall of the nasopharynx, between the orifice of the right Eustachian tube and the adenoid, diagnosed as lymphoid hamartoma. Histopathology revealed that the mass consisted of surface respiratory epithelium and fibrous stroma containing islands of lymphoid tissue and dilated vascular channels but without the cartilage islands, seromucinous glands, and adipose tissue.
Although CMH is typically a benign lesion, in 2013, the first and only reported case of malignant transformation of CMH was described. 14 The authors described a 40-year-old female patient with nasal obstruction and epistaxis. A CT scan of the nose and paranasal sinuses confirmed a heterogeneous polypoid soft-tissue mass filling the nasal cavity and extending into the maxillary and ethmoid sinus. The patient underwent a complete radical resection. Histological and immunohistochemical analyses showed a portion of the mass was consistent with typical nasal CMH. However, some areas of mass exhibited cytological atypia, marked mitotic activity, and foci of necrosis. The final diagnosis was that of nasal CMH with malignant transformation. 14
Footnotes
Authors’ Note
This investigation was conducted as a part of scientific project of the Military Medical Academy Faculty of Medicine, Belgrade, Serbia (MFVMA02/19-21/). Ethical approval not required. The patient’s permission was obtained.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
