Abstract
Sinonasal alveolar soft-part sarcoma (ASPS) is a rare malignant disease that comprises 0.4% to 1.2% of all soft-tissue sarcomas. Alveolar soft-part sarcoma is usually difficult to diagnose because it has many clinical and pathological mimickers. In this case study, ASPS occurred in the olfactory cleft, representing the second case in this location in the literature. This article presents the clinical presentation, radiologic, and histopathological case, and reviews the literature regarding the differentials.
Introduction
Sinonasal sarcomas are relatively rare tumors. An even rarer entity is alveolar soft-part sarcoma (ASPS). Among the English literature, there are only 5 reported cases of sinonasal ASPS and only 1 case report of olfactory cleft involvement. 1 Detecting cases with these tumors and diagnosing them require a high index of suspicion from both the clinician and pathologist. This article presents a case of ASPS of the olfactory cleft. Reporting this case and discussing the diagnosis and treatment approach are important for overcoming the deficits in the literature regarding this type of tumor. This article reviews the pertinent literature with a focus on differential diagnosis of such a rare entity, which makes it challenging to diagnose.
Case Presentation
A healthy 24-year-old female presented with recurrent right-side epistaxis and facial pain for 6 months. The patient denied any hyposmia, facial numbness, or visual changes. The patient underwent septoplasty in another center to correct a septal deviation, during which a tumor in the right nasal cavity was discovered intraoperatively. Multiple biopsies were nonconclusive but suspicious for rhinoscleroma versus inflammatory polyps with histiocytes.
On presentation, there was a right nasal mass filling the olfactory cleft, which bled on touch. A computed tomography (CT) scan showed an expansile soft-tissue mass within the anterosuperior aspect of the right nasal cavity with coarse intimal calcifications and bony remodeling of the adjacent nasal septum, possible extension to the floor of the right frontal sinus, and no orbital or intracranial extension. To delineate between the tumor and the dura, magnetic resonance imaging was done. It showed a soft-tissue mass expanding to the superior anterior aspect of the right nasal cavity, causing remodeling of the bony outline and adjacent parts of the nasal septum without erosion or destruction. The mass had intermediate signal intensity on T2-weighted sequences with mild enhancement in the postcontrast sequences and multiple small areas of punctate calcification/ossification within the mass. It measured about 2.0 × 3.5 cm in the maximum transverse and anteroposterior dimensions, respectively. No orbital or intracranial extension was found. Therefore, a proper biopsy was taken under general anesthesia (Figure 1).
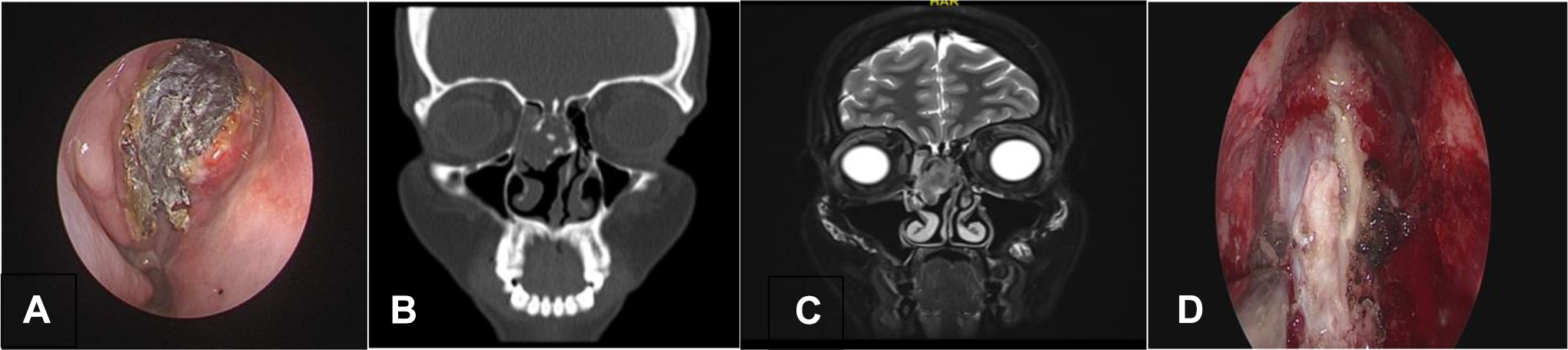
A, Endoscopic view of the mass partially covered with debris. B, Noncontrasted CT coronal section showing the mass originating from the cribriform plate with intimal calcifications. C, MRI T2-weighted image showing intermediate signal intensity of the mass which fills the right nasal cavity. D, Intraoperative and endoscopic view of the mass involving the olfactory nerve. CT indicates computed tomography; MRI, magnetic resonance imaging.
The first biopsy was received in 10% neutral buffered formalin and was routinely processed and paraffin embedded. The hematoxylin and eosin-stained section showed a polypoid piece of mucosal tissue lined by respiratory epithelium, with an underlying proliferation of large polygonal to round cells with abundant granular eosinophilic cytoplasm, prominent nucleoli, focal nuclear pleomorphism, and scarce mitosis. The tumor had a mainly solid pattern of growth, but a focal area could be seen near the mucosa, where the tumor was arranged in small nests with central necrosis (pseudoalveolar pattern). Another growth pattern in the form of trabeculae and single cells within a fibrotic stroma could be seen at the deeper edge of the tumor. The stroma contained delicate vasculature and few inflammatory cells. A small fragment of bone surrounded by tumor cells was noted (Figure 2).
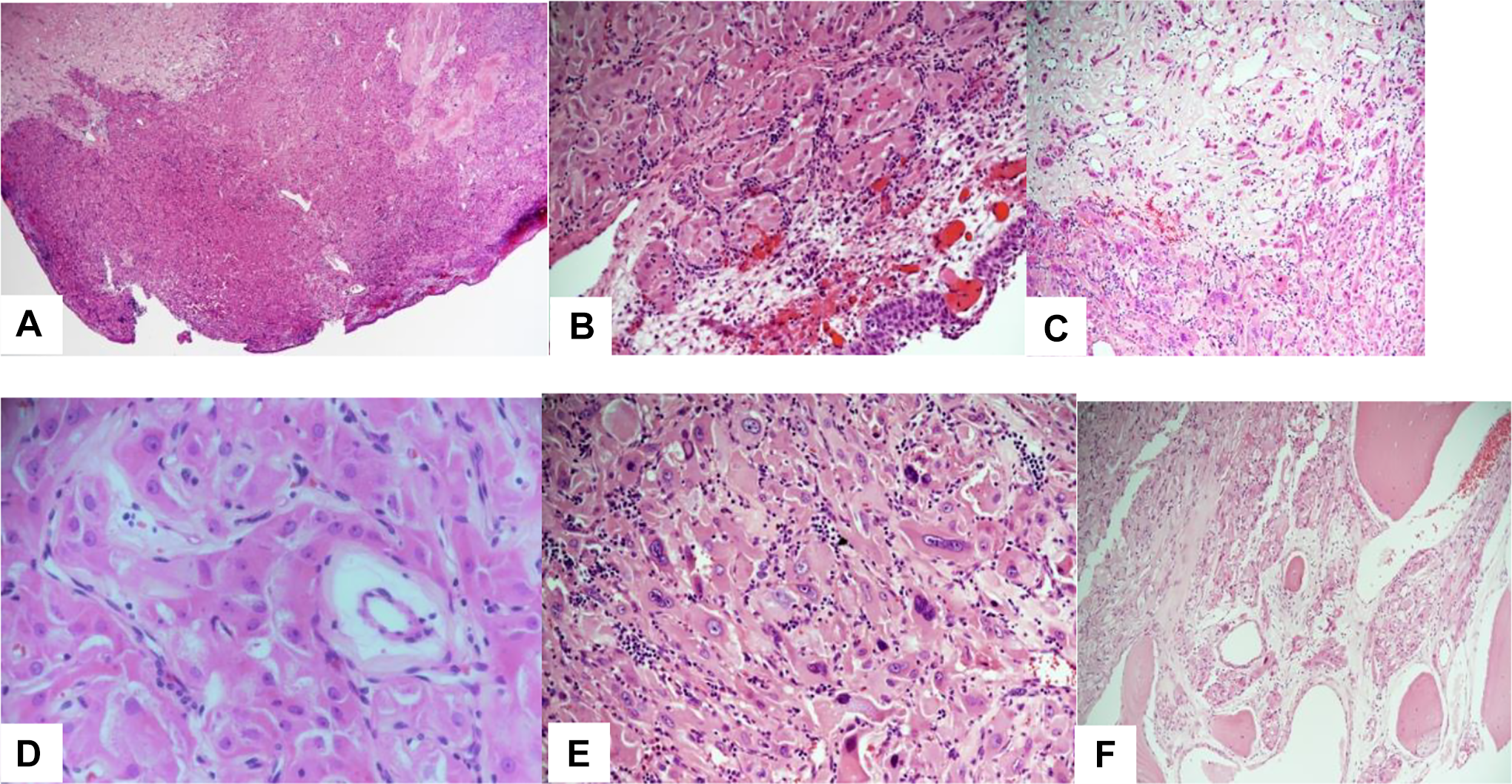
Hematoxylin and eosin-stained sections of the tumor showing a polypoid piece of tissue with solid proliferation of eosinophilic cells (A), nested/alveolar pattern seen near the mucosa (B), while single cells and trabecular arrangement within a fibrotic stroma seen at the periphery (C), the nuclei are monomorphic (D), with few foci showing nuclear atypia (E). Evident bone invasion from the second operation (F).
The immunohistochemical studies showed that the tumor cells were positive for CD68 with retained INI-1 while being negative for CKAE1/AE3, EMA, Cam5.2, LMWCK, HMWCK, CK7, vimentin, S100 protein, HMB45, calretinin, inhibin, desmin, myogenin, CD31, CD34, synaptophysin, chromogranin, GFAP, CD163, PAX8, and HepPar1. Periodic acid-Schiff with diastase showed rod-shaped crystals. Transcription factor for E3 (TFE3) antibody showed diffuse nuclear staining of tumor cells (Figure 3). By this, the diagnosis of ASPS was established. A positron emission tomography (PET)-CT showed minimal fluorodeoxyglucose-avid soft-tissue density at the right nasal cavity superiorly/ethmoidal sinus (standardized uptake values maximum 2.9), with no local or distant metastasis.
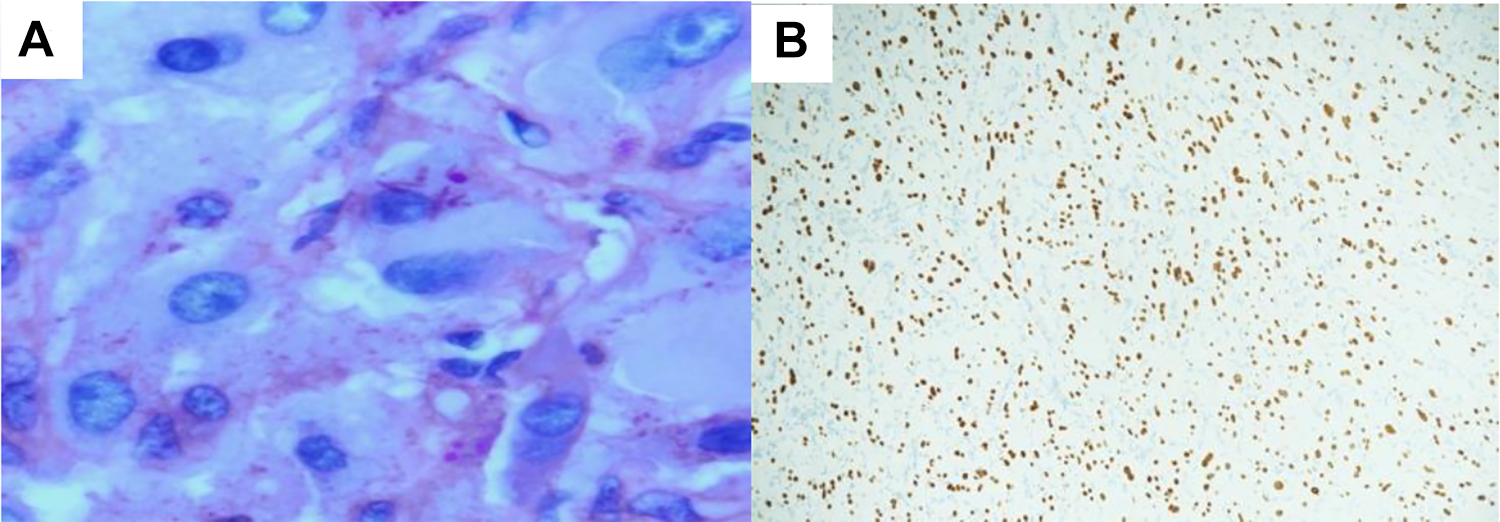
A, Periodic acid-Schiff, diastase-resistant rod-shaped crystals. B, Diffuse and strong nuclear positivity for TFE3 antibody. TFE3 indicates transcription factor for E3.
The patient was then taken for endoscopic resection, where the mass was resected along with the involved olfactory bulb and the surrounding dura and reconstructed with fat, tensor fascia lata, nasoseptal flap, and mucosal graft, as the flap did not reach the most anterior part of the defect (Figure 1). Histologic sections from the second operation showed more evidence of bone invasion. Six weeks later, the patient started on radiotherapy. A 6-month follow-up was negative for residual or recurrence based on endoscopic assessment and PET-CT.
Discussion
Alveolar soft-part sarcoma was first described by Christopherson et al. 2 Alveolar soft-part sarcoma is a relatively rare malignant soft-tissue tumor that represents approximately 0.4% to 1.2% of all soft-tissue sarcomas.3,4 The most common site for this tumor is the soft tissues of the limb, specifically the thigh. It less commonly occurs in other sites, such as the head and neck, mediastinum, retroperitoneum, breasts, and orbits. 5 However, it should be noted that ASPS of the head and neck region generally involves younger patients compared to ASPS of the extremities. 4 Alveolar soft-part sarcoma of the head and neck constitutes approximately 25% of all ASPS and usually occurs in the orbit or the tongue.4,6
The age of onset ranges from 15 to 35 years, and a presentation prior to 5 or after 50 years of age is rare.4,7 It has been reported that women have a higher incidence rate than men. 4 These masses require a proper biopsy, and sometimes multiple biopsies are needed to reach a proper diagnosis, as was the case here. 8 Clinical Characteristics of Sinonasal ASPS are described in Table 1.
Clinical Characteristics of Sinonasal ASPS.
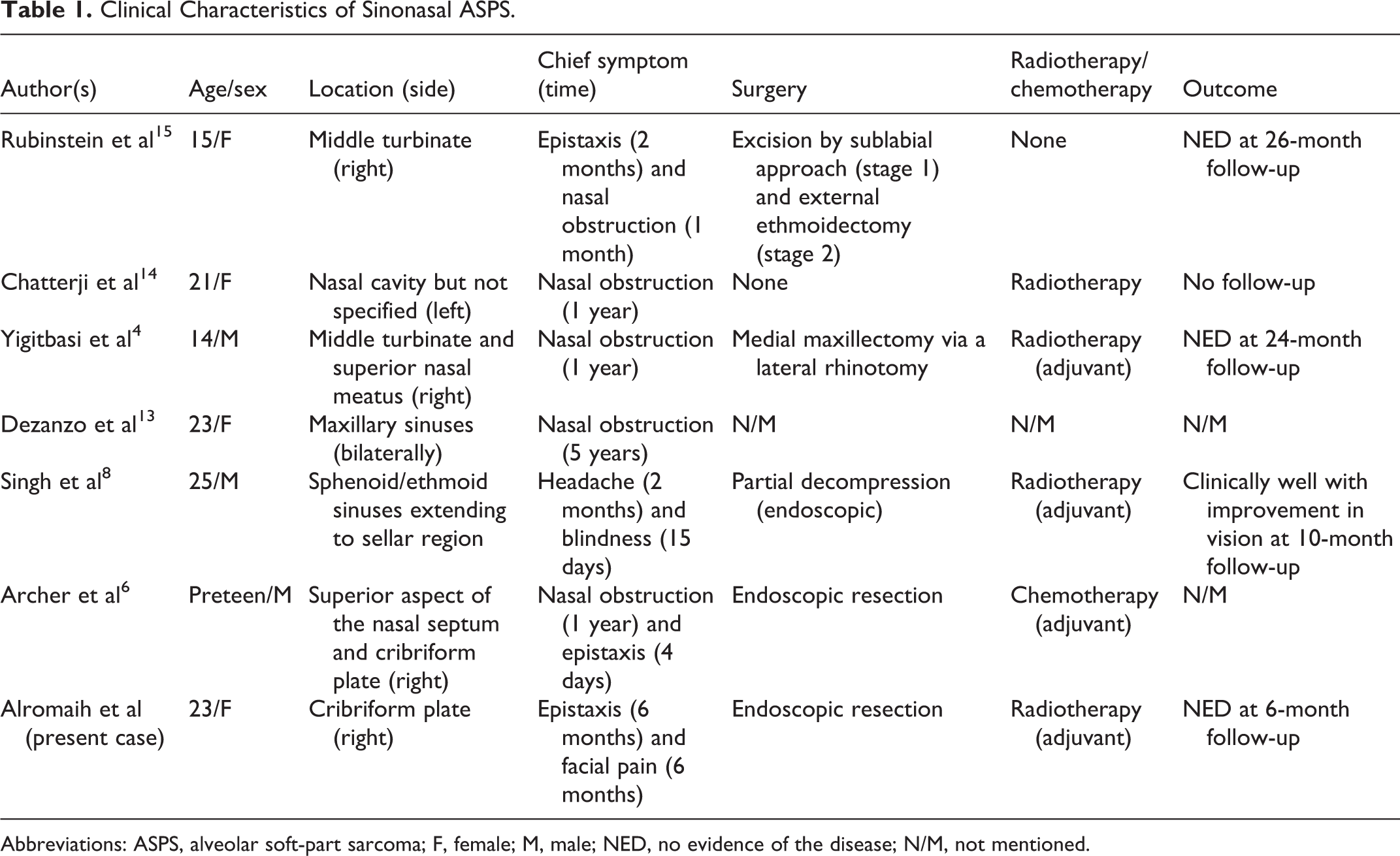
Abbreviations: ASPS, alveolar soft-part sarcoma; F, female; M, male; NED, no evidence of the disease; N/M, not mentioned.
Alveolar soft-part sarcoma is part of a group of malignant tumors with uncertain differentiation. The site of origin remains questionable, with either myogenic or neurogenic origin having been proposed.9,10 Alveolar soft-part sarcoma tends to grow gradually and without obvious clinical symptoms; as a result, identification of the tumor is challenging. 5 Unlike the majority of sarcomas, ASPS frequently metastasizes early, primarily to the lungs (in 42% of cases), bones (19%), brain (15%), and lymph nodes (7%), and it has a high recurrence rate following conservative surgical excision. 11
The histopathological pattern of classic alveolar arrangement is the basis of ASPS diagnosis. Difficulties in diagnosing ASPS can stem from an unusual location or lack of the classical alveolar pattern. Based on morphology, differential diagnoses include granular cell tumor; paraganglioma; perivascular epithelioid cell neoplasm (PEComa); clear cell sarcoma; epithelioid vascular tumors; mucosal melanoma; Rosai-Dorfman disease; and metastatic tumors such as renal cell carcinoma, hepatocellular carcinoma, or adrenocortical carcinoma. Detailed pathological differential diagnoses and their morphological features, ancillary studies, and molecular findings are discussed in Table 2. 8
Pathological Differential Diagnosis and Their Features.
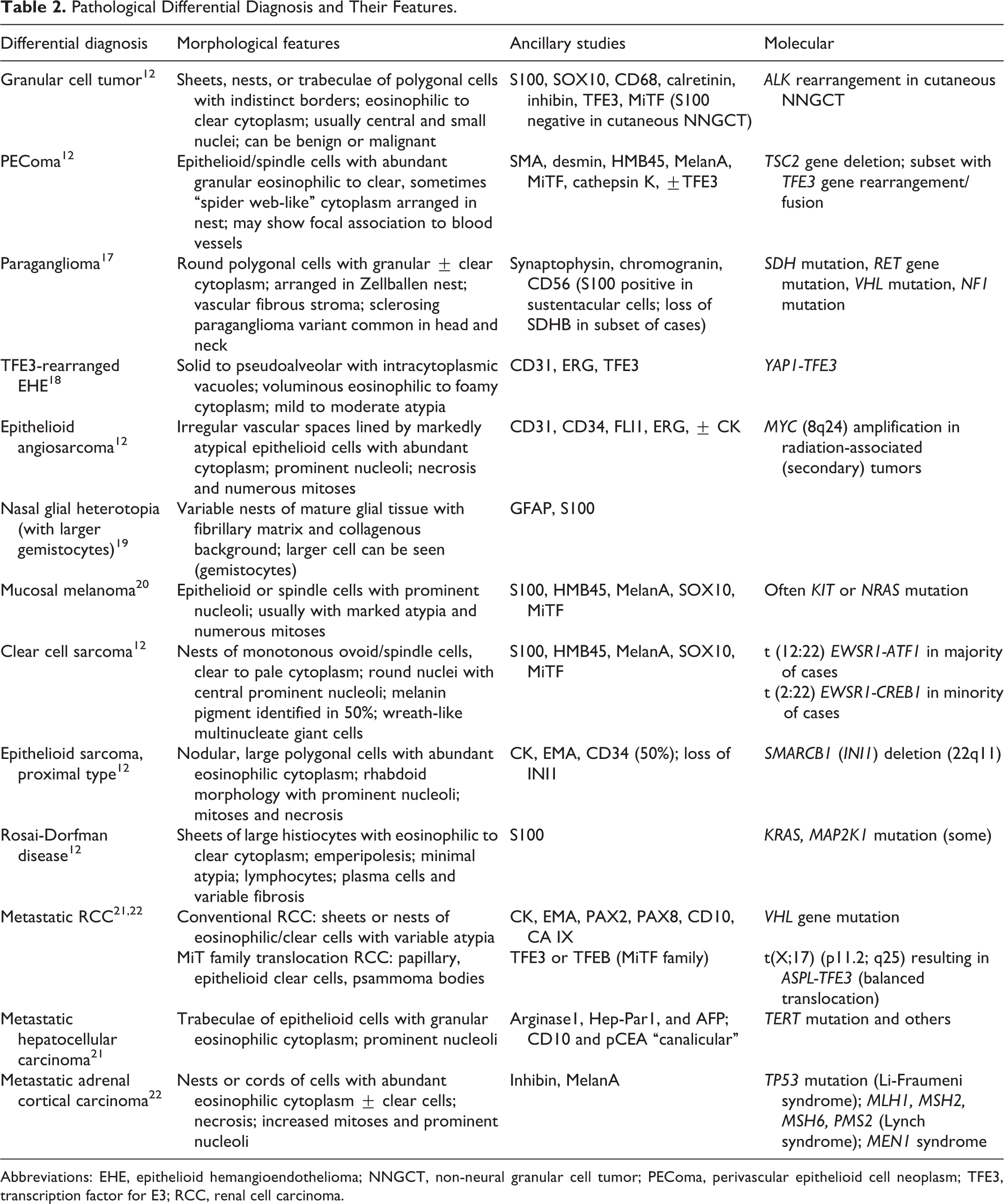
Abbreviations: EHE, epithelioid hemangioendothelioma; NNGCT, non-neural granular cell tumor; PEComa, perivascular epithelioid cell neoplasm; TFE3, transcription factor for E3; RCC, renal cell carcinoma.
Alveolar soft-part sarcoma is composed of a nested/alveolar or solid/compact growth pattern; the latter is especially seen in the head and neck of pediatric patients. The neoplastic cells are usually monomorphic polygonal to round with abundant granular eosinophilic to clear cytoplasm. Rare examples with marked nuclear atypia have been reported. 12 The nuclei are centrally located, with prominent single or multiple nucleoli. Mitosis is rare. Periodic acid-Schiff with diastase highlights intracytoplasmic rhomboid or rod-shaped crystals that are characteristic of ASPS. By immunohistochemical stains, the neoplastic cells show strong nuclear positivity for TFE3 and cytoplasmic staining for cathepsin K. Unfortunately, these 2 markers, while sensitive, are not specific for ASPS, as they can also stain PEComa, renal cell carcinoma, granular cell tumor, and melanoma. 12 Other immunostains that can be positive include desmin, muscle-specific actin, and S100. 1 Moreover, in regard to molecular genetics, ASPS is characterized by unbalanced translocations of der(17)t(X;17)(p11;q25) that result in the ASPL-TFE3 gene fusion. 13
To date, there is no consensus on the best treatment plan for sinonasal ASPS, but radical surgical excision with tumor-free margins is preferred. 4 Radiotherapy, which produces improved local control, is recommended following subtotal surgical removal. The role of chemotherapy is still debatable. 11
Sinonasal involvement has been reported in 7 cases.4,6,8,13-16 Only one of these cases involved the nasal septum and cribriform plate. 6 In this study, we report a case of ASPS presented as a cribriform plate mass. These cases (with the exception of Barbareschi et al, 16 which was inaccessible) are described in Table 1.
It should be noted that all the patients of sinonasal ASPS have been uniformly young. Four of the cases were females and 3 were males. Nasal obstruction was the chief complaint in 5 of the 7 patients, followed by epistaxis in 3 patients. The location of origin of the sinonasal ASPS varied. Regarding metastasis, only 1 case had cervical adenopathy, which was not biopsied. 14 Surgical resection was mentioned as the primary treatment in 5 cases. However, it should be noted that in 1 case, only partial decompression was done. 8 Chatterji et al 14 used radiotherapy as the main and only treatment in their case. They utilized radiotherapy in the form of telecobalt therapy for 5 weeks, which yielded fair response. 14 Regarding the outcome, 3 of the case studies mentioned that there was no evidence of the disease at follow-up.
Conclusion
Although ASPS is a rare entity, it is crucial to differentiate it from its mimickers, as this can change the treatment and prognosis of such cases. In this report, we presented a case of olfactory cleft ASPS and discussed its clinical and pathological perspective.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
