Abstract

Keywords
Multi-organ system diseases, such as granulomatosis with polyangiitis and sarcoidosis, may manifest in the nasal cavity with crusting, congestion, rhinorrhea, epistaxis, olfactory impairment, and mucosal inflammation. 1 Sjögren syndrome, an autoimmune disorder caused by exocrine gland dysfunction and destruction, has been shown to cause mucosal dryness of the upper respiratory mucosa.2,3 We present a unique case of acquired nasal cavity synechiae and nasopharyngeal stenosis associated with Sjögren syndrome.
A 55-year-old female with no past history of sinonasal surgery presented with 2 years of bilateral nasal obstruction, facial pressure, oral dryness, and nasal drainage. Nasal endoscopy revealed extensive synechiae between the nasal septum and inferior and middle turbinates, and near complete nasopharyngeal stenosis (see Figure 1). Corresponding computed tomography scan demonstrated soft tissue along nasal floor extending superiorly to occupy the right and left nasal passage (Figure 2A) and near complete stenosis of the nasopharynx (Figure 2B). Mucosal biopsy in clinic ruled out lymphoma and repeat biopsy in the operating room revealed nongranulomatous fibrosis and inflammation. A thorough laboratory workup was performed with the following tests: Proteinase 3 , sed rate, C reactive protein, phosphorus 4 , magnesium, complete metabolic panel, cyclic citrullinated peptide, angiotensin converting enzyme, rheumatoid factor, Jo-1 antibody, immunoglobulin G, anti-nuclear antibody, anti-sjogren's syndrome A antibody (anti-SSA), anti-sjogren's syndrome B antibody (anti-SSB), proteinase 3. Immunoglobulin G subclasses.
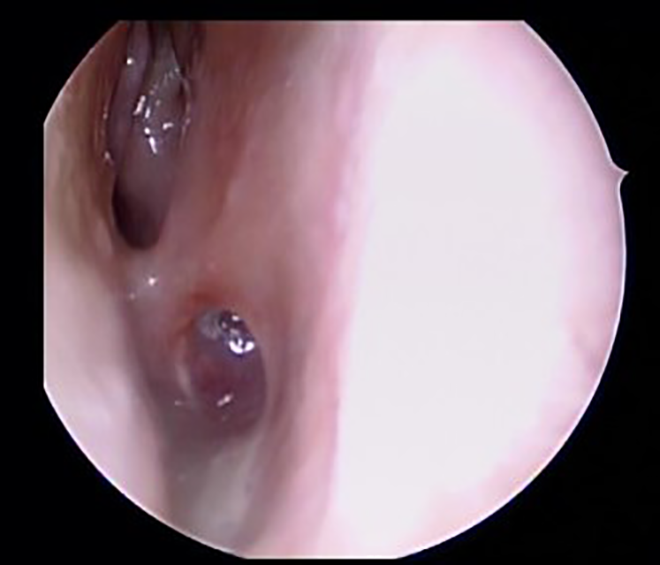
Nasal endoscopy reveals synechiae between the nasal septum and turbinate tissue with near complete nasopharyngeal stenosis.
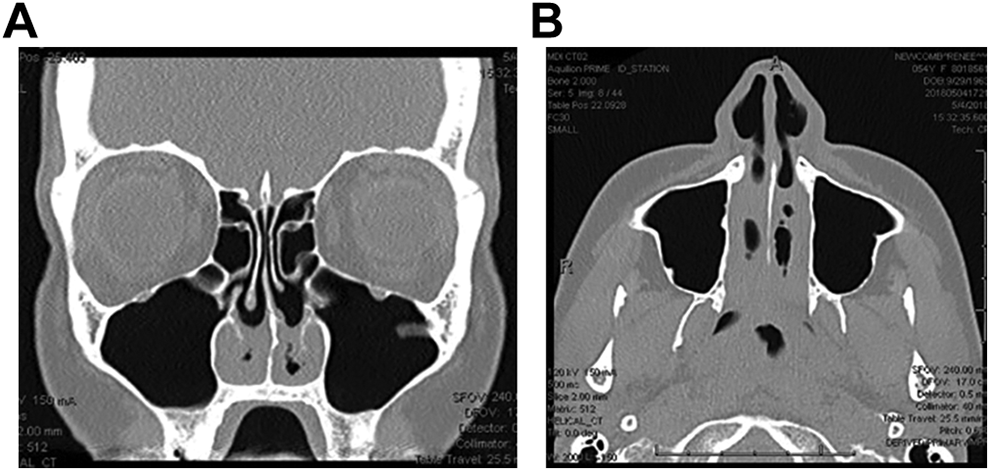
A, Computed tomography (CT) of sinuses, coronal orientation, demonstrates excessive soft tissue along nasal floor. B, Computed tomography (CT) of sinuses, axial orientation, demonstrates excessive soft tissue in bilateral nares and nasopharynx.
Laboratory analysis was positive for anti SS-A/B/Ro autoantibodies, and rheumatology consultation confirmed the diagnosis of Sjögren syndrome and the patient elected to forego any immunomodulating therapies.
The patient underwent endoscopic repair of nasopharyngeal stenosis and lysis/division of nasal synechiae with nasal splinting and postoperative oral steroids. Thorough cutting instrumentation was used to divide and resect the scar tissue while attempting to preserve remaining inferior turbinate tissue. The coblator was used to excise nasopharyngeal scar tissue and recreate a nasopharynx. At the termination of the procedure, silicone nasal splints were secured to the nasal septum and left in place for 2 weeks. A moderate dose steroid taper, as well as saline irrigation was prescribed postoperatively. Twelve-month follow-up demonstrated complete patency of her nares and nasopharynx (Figure 3) with improvement in her Sinonasal Outcome Test—22 score from 46 to 6. The patient was tapered off oral corticosteroids 14 days postoperatively and rheumatologic care was continued with supportive symptom management.
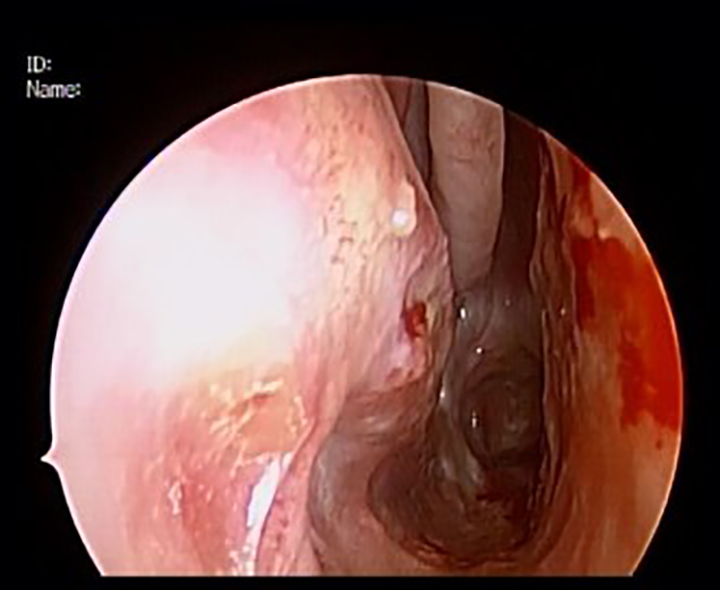
Postoperative endoscopy demonstrating patent nasal airway and nasopharynx.
Sjögren syndrome has important and widespread otolaryngologic considerations, with an estimated global prevalence of 1%-3%. By some calculations, the syndrome is the second most common rheumatic autoimmune disorder. 5 Sjögren syndrome has a strong female predominance and the severity of symptoms increase with age.6,7
Interplay between genetic and environmental factors leads to progressive destruction of exocrine glands in the setting of lymphocytic infiltration and serum autoantibodies. 8 In particular, nasal mucosal biopsies of patients with Sjögren syndrome demonstrate severe periglandular infiltration of lymphocytes and plasma cells. There is also glandular atrophy and periglandular fibrosis, which appear to reflect disease severity. 9 The mechanism underlying the nasal manifestations may be related to (1) autoimmune-mediated damage of the nasal exocrine gland elements, (2) interference with mucosal muscarinic receptors, and (3) interference with gland extracellular matrix via metalloproteinases. 8 Additionally, mucociliary function within the nose appears to be affected in these patients. A thick outer mucus layer with high viscosity does not move effectively with periciliary fluid, prolonging mucociliary transport time. 10 In the tracheobronchial system, this dysfunction results in xerotrachea. 11 The end result of these destructive processes in Sjögren syndrome is decreased mucociliary clearance and nasal mucus production, leading to chronic inflammation.10,12,13
A common manifestation of the underlying pathophysiology of Sjögren syndrome is xerostomia (due to salivary gland involvement) and keratoconjunctival sicca (from lacrimal gland involvement). Furthermore, as nasal mucosa is composed of exocrine glands, some Sjögren patients experience atrophy of this mucosa. 3 Xerorhinia, hyposmia, hypogeusia, and nasal crusting leading to nasal obstruction have also been described. 14 Epistaxis is seen in nearly 1/3 of patients and nasal septum perforation in up to 6.5%. 3 Sjögren is a risk factor for chronic rhinosinusitis, portending 2.51-fold increase in risk compared to matched control patients. 12
Ultimately, in the present case, it can be postulated that significant mucociliary dysfunction resulted in chronic inflammation in the form of chronic rhinitis and crusting along opposing mucosal surfaces, leading to synechiae formation. This case demonstrates that surgical lysis and division of nasal synechiae with splinting and postoperative steroids can offer successful resolution of nasal obstruction induced by Sjögren syndrome. In the setting of nasal congestion, crusting, and synechiae, sinonasal manifestations of Sjögren syndrome should be considered as a part of the differential diagnosis and disease workup.
Footnotes
Author’s Note
Presented at the American Rhinologic Society 65th Annual Meeting in New Orleans, LA, on September 13-14, 2019.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Toby Steele is a consultant for Intersect ENT, and Stryker.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
