Abstract
A 45-year-old man presented with a history of chronic left nasal congestion. Nasal endoscopy revealed a pedunculated polypoid mass with glandular epithelium surface on the posterior nasal septum. Computed tomography revealed a 25-mm mass-like growth in the left posterior nasal cavity attached to the nasal septum with a stalk. The patient underwent transnasal endoscopic surgery, and the tumor was removed under a block with safety margin. The final pathological diagnosis was sinonasal seromucinous hamartoma (SH). Sinonasal SH is a rare tumor with only 31 reported cases. Transnasal endoscopic surgery is currently the first-line treatment for sinonasal SH. Differential diagnoses of this lesion include inflammatory polyps, respiratory epithelial adenomatoid hamartoma, and adenocarcinoma. Although SH is a benign tumor, its progression to adenocarcinoma has been reported. Therefore, unilateral posterior nasal tumors must be diagnosed precisely.
Introduction
Respiratory epithelial adenomatoid hamartoma (REAH), seromucinous hamartoma (SH), chondro-osseous and respiratory epithelial hamartoma (CORE), and nasal chondromesenchymal hamartoma (NCH) are typical hamartomas that are observed in the sinonasal region. REAH and SH arise from the respiratory epithelium, whereas CORE and NCH comprise mixed mesenchymal elements. The fourth edition of the World Health Organization (WHO) classification of head and neck tumors 1 has categorized SH and REAH as sinonasal respiratory epithelial lesions. SH is a benign proliferation characterized by the overgrowth of the indigenous seromucinous gland; however, its origin has not been identified. To the best of our knowledge, the incidence of sinonasal SH is rare, with only 31 cases reported in the English literature (including 2 cases of the nasopharynx). 2 SH is not associated with any specific etiological factors. Although SH is a benign tumor, its malignant transformation to adenocarcinoma was recently reported. 3 Herein, we report a case of SH of the posterior nasal septum.
Case Report
A 45-year-old man presented with a history of chronic nasal congestion. He was referred to the ENT clinic at our hospital for the evaluation of a mass in the left nasal cavity. Nasal endoscopy revealed a pedunculated polypoid mass with a glandular mucosal surface in the posterior nasal septum causing nasal congestion (Figure 1). Computed tomography (CT) revealed a 25-mm mass in the left posterior nasal cavity extending to the choanae, which was attached to the nasal septum via a fine stalk (Figure 2). A biopsy was performed in the office, leading to the diagnosis of SH. The patient underwent transnasal endoscopic surgery under general anesthesia to remove the lesion. The stalk of the tumor was located in the left posterior and upper nasal septum. The tumor was resected under a block with the safety margins around the stalk (Figure 3). The tumor has not recurred for about 11 months after the surgery in nasal endoscopy. Histopathology revealed polypoid nasal mucosa covered by ciliated respiratory epithelium with respiratory epithelial invaginations (Figure 4a). The tumor contained a variable combination of small and large glands and tubules, growing haphazardly in clusters or lobules. Small glands and tubules were lined by a single layer of cuboidal cells (Figure 4b). Immunoreactivity, the basal cell layer of the respiratory mucosa stained positive for p63 (Figure 5a) and the serous glandular component stained positive for s100 (Figure 5b). Thus, the final pathological diagnosis was SH.
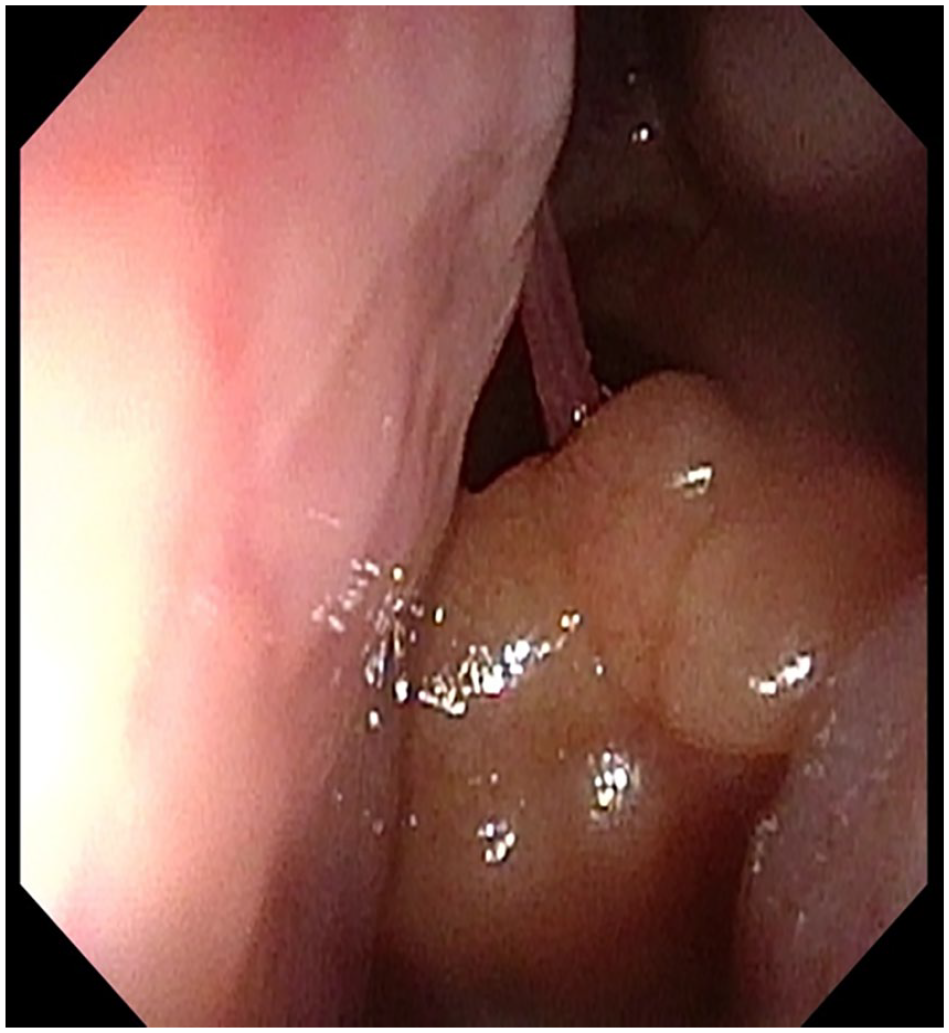
Nasal endoscopy showing a yellowish polypoid mass with a thin stalk on the left posterior septum.
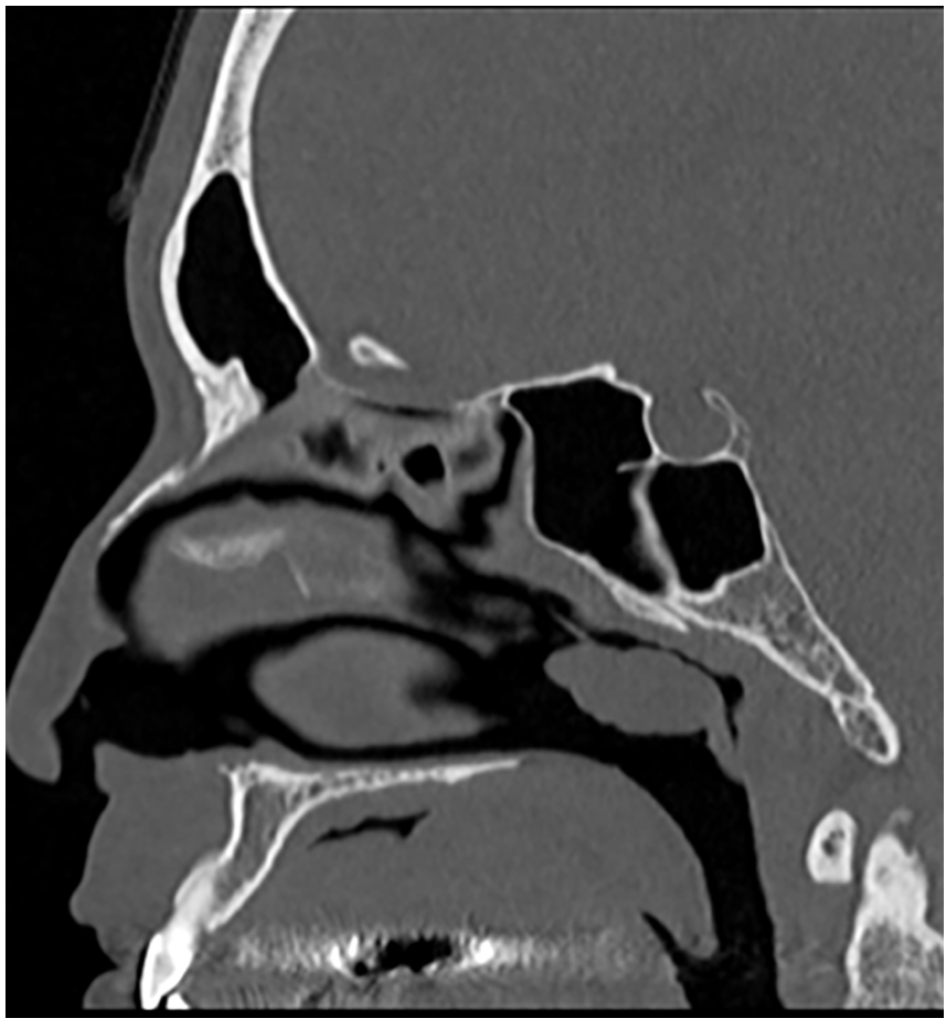
Sagittal view of computed tomographic scan without contrast showing a 25-mm mass with a stalk in the posterior nasal cavity.
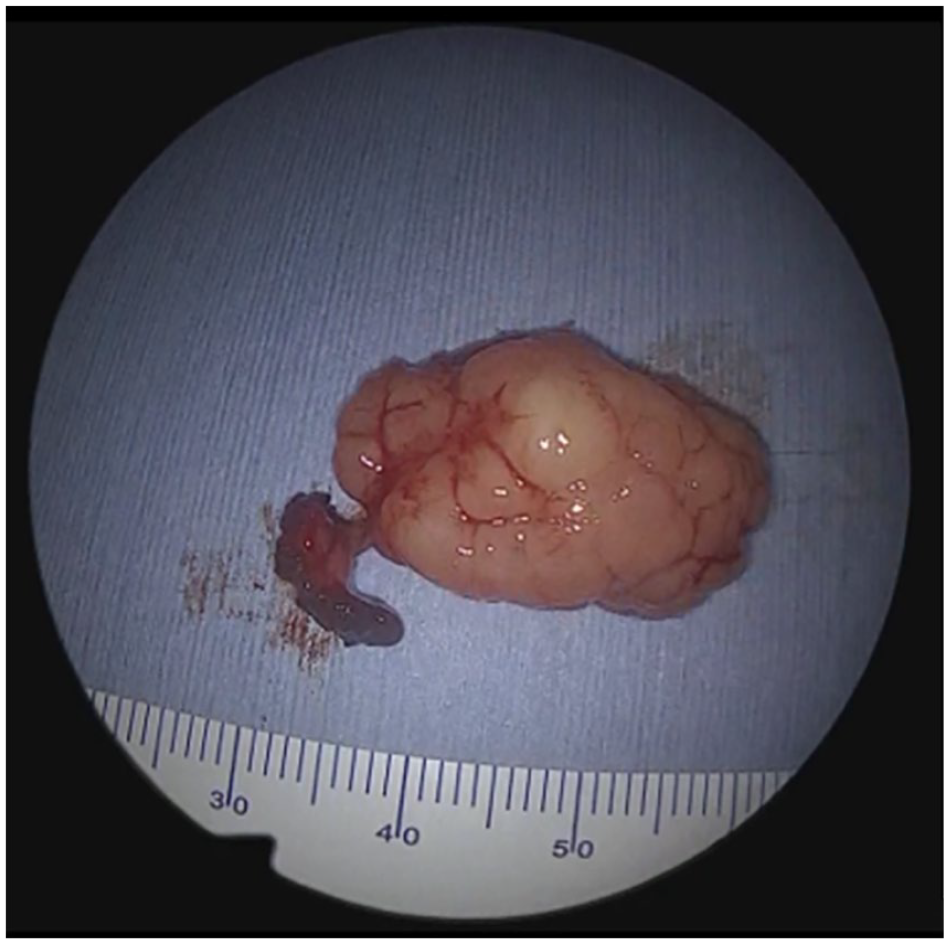
The resected specimen shows a 25-mm polypoid mass with a glandular mucosal surface and a stalk.
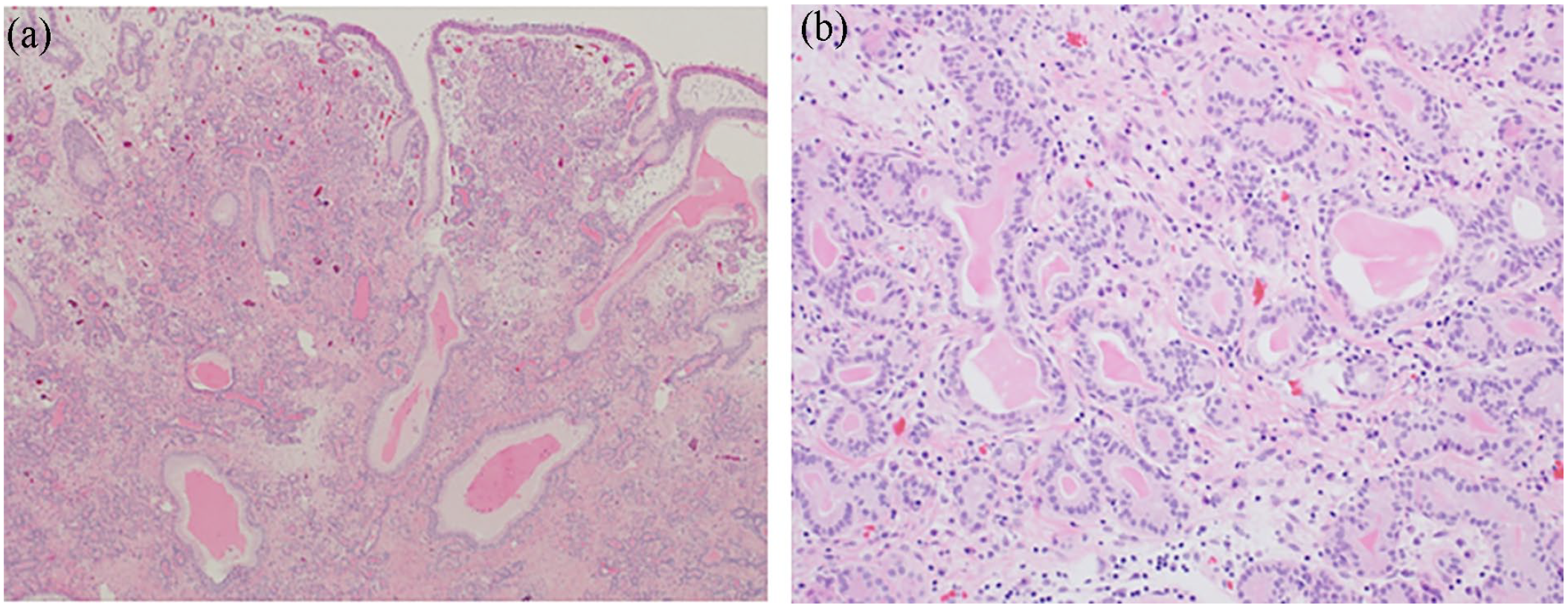
Pathological features. (a) At low power, the mass is covered by ciliated respiratory epithelium with invaginations and (b) at higher power, the tubules comprise a single layer of cuboidal epithelium.
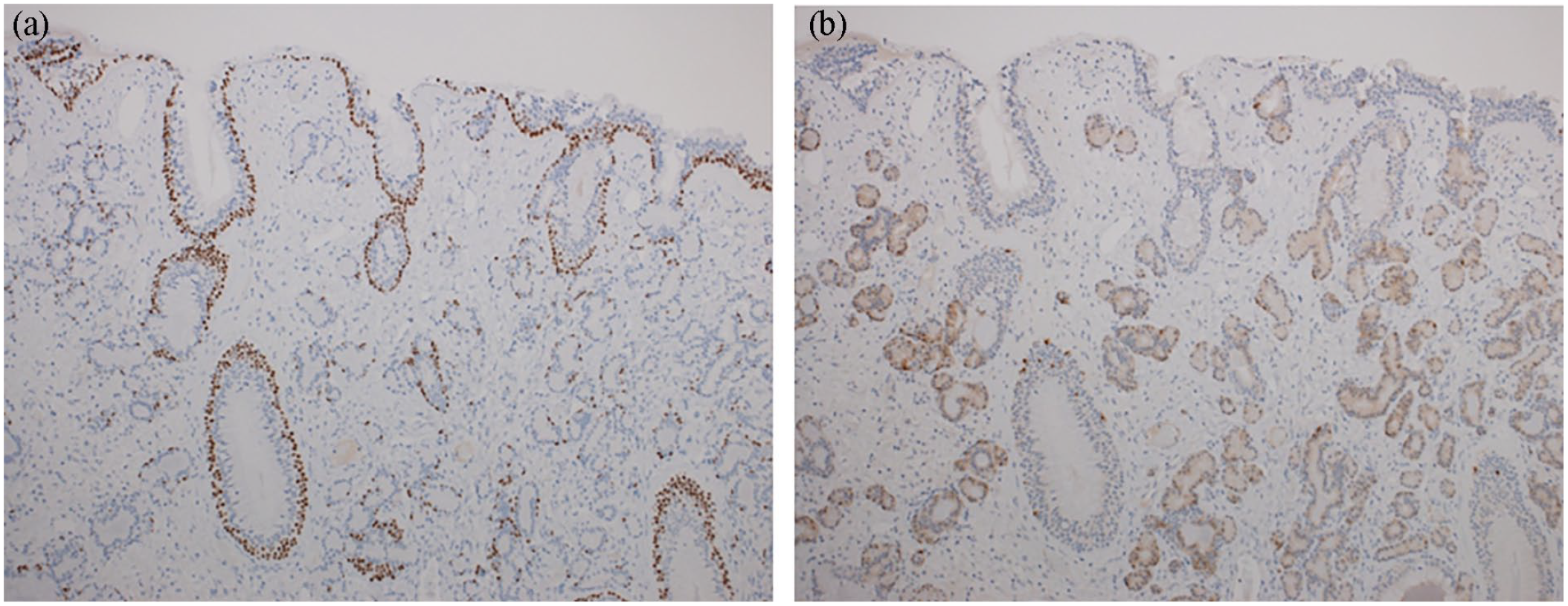
Immunohistochemical features. (a) The basal cell layer around the invaginated respiratory epithelium stained positive for p63; however, staining was absent around proliferating seromucinous tubules and (b) the serous glandular component stained positive for S100.
Discussion
SH was first reported in 1974 by Ballie and Batsakis. 4 The fourth edition of the WHO classification of head and neck tumors 1 has categorized SH as a tumor of the nasal cavity that originates from the respiratory epithelium. Sinonasal SH is a rare tumor, and to date, only 31 cases have been reported in the English literature. 2 Sinonasal SH is commonly located on the nasal cavity or septum. 2 The mean age of the patients is 55.5 years, and the incidence of sinonasal SH is similar among females and males. 2 Unilateral nasal congestion is the most common presenting symptom, and nasal discharge and epistaxis are sometimes recognized incidentally. An association between SH and rheumatoid arthritis, Parkinson’s disease, and chronic sinusitis 1 has been reported; however, the relationship is unclear. Sinonasal SH ranges from 0.6 to 6.0 cm in size and often has a polypoid surface. 5 Sinonasal SHs may have stalks attached to the nasal septum or wide-based attachments. 2 It seldom occurs in the olfactory cleft. SH does not exhibit aggressive characteristics associated with local infiltration and bone destruction. In the present case, a tumor without bone destruction was seen in sinus CT. Histopathologically, the outer layer of the tumor was covered by respiratory epithelium and comprised lobular or haphazard proliferation of small to large glands and ducts lined by a single layer of cuboidal or flattened epithelial cells.5,6 Immunohistochemistry revealed that the serous glands of SH were positive for S100 at least focally, and focal staining for p63 was observed on the outer basal layer. 7 However, diagnosis using only immunohistochemical staining was challenging. Transnasal endoscopic surgery is currently the first-line treatment for sinonasal SH, with only one case of recurrence reported. 7 SH can be cured completely through simple resection. The differential diagnoses of SH include inflammatory polyps, REAH, and adenocarcinoma. SH has a more polypoid surface compared with an inflammatory polyp as revealed by endoscopic findings, and it can be relatively easily distinguished from inflammatory polyps via biopsy. Dis-tinguishing between REAH and SH is challenging as they have many common features; however, the epithelium in REAH is multi-layered, whereas it is single-layered in SH. Immunohistochemistry is not usually required for diagnosis; however, REAHs were negative for CK20, CDX-2 and S100. 8 Therefore, there may be a similar series of pathological changes over time. Weinreb et al. 7 suggested that SH and REAH may be a spectrum of lesions rather than 2 separate entities. Most REAHs often originate from the olfactory cleft, and Hawley et al. 9 reported olfactory cleft widening as a reliable feature that indicates REAH. Davison et al. 10 described that evaluation of the combination of olfactory cleft widening and sagittal discoid mass should be performed to diagnose REAH. However, our patient had a mass on the posterior nasal septum with a stalk and no discoid appearance; therefore, the diagnosis of sinonasal SH, rather than REAH, was made. Kossai et al. 11 reported a third subtype of adult sinonasal hamartoma—olfactory epithelial hamartoma (OEH). OEH resembles sinonasal SH and is characterized by the presence of areas of olfactory epithelium as well as its development from the olfactory cleft. However, further ex-amination is necessary as there are only 6 cases of OHE. Another important differential diagnosis is low-grade non-intestinal-type adenocarcinoma, 12 especially low-grade sinonasal adenocarcinoma (LGSNAC). LGSNAC typically has complex growth patterns, including micropapillary architecture and glands that are back-to-back or fused; in contrast, cellular atypia, nucleoli, and mitosis are frequently absent in SH. Although SH is a benign tumor, Rengifo et al. 3 reported the transition from SH to adenocarcinoma. Thus, although rare, the malignant transformation potential of SH must be considered. In conclusion, SH should be included in the differential diagnosis of unilateral posterior nasal tumors.
Footnotes
Acknowledgements
Data Availability Statement
Data sharing is not applicable to this article as no new data were created or analyzed in this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Statement of Informed Consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
Meeting Presentation
None.
