Abstract
Objectives:
Recent studies suggest that elevated neutrophil to lymphocyte ratio (NLR) and platelet to lymphocyte ratio (PLR) are poor prognostic factors in sudden sensorineural hearing loss (SSNHL). We aimed to investigate the accuracy of this hypothesis by taking into account the effect of cardiovascular risk (CVR) factors.
Methods:
Medical records of 122 patients with SSNHL were reviewed retrospectively and grouped into 2 as; patients without CVR (group 1; n = 68) and patients having CVR (group 2; n = 54). Moreover, 60 control cases who did not have SSNHL were also included and grouped into 2 as; group 3 (n = 30) with CVR and group 4 (n = 30) healthy controls without having SSNHL or CVRs. Neutrophil (N), lymphocyte (L), platelet (Plt), NLR, and PLR between the groups and their relationship with the severity of hearing loss, recovery rates, and audiogram configurations were analyzed.
Results:
The highest N and NLR values were in group 1 and were significantly higher than the values of group 4 (P < .05, P < .01). There was no significant relationship between the groups in terms of L, Plt, or PLR values. The highest NLR and PLR values were determined in SSNHL patients with mild hearing loss, complete recovery, and up-sloping audiogram configuration (P > .05).
Conclusions:
Elevated levels of N and NLR may be considered as strong laboratory findings showing an inflammatory response in the diagnosis of SSNHL, but the presence of CVR factors does not seem to increase the inflammatory response in SSNHL as expected. In patients with SSNHL, NLR and PLR elevation may indicate better prognosis.
Keywords
Introduction
Sudden hearing loss (SHL) is a subjective feeling of hearing impairment in one or both ears with a rapid onset, within 72 hours. Sudden sensorineural hearing loss (SSNHL) is a type of SHL, having some objective diagnostic criteria such as hearing loss of 30 dB or more, and affecting at least 3 consecutive frequencies. 1 Its incidence is approximately 5 to 20/100 000. 2 It is often seen in the sixth and seventh decades of life, mostly unilateral and it does not discriminate against sex. It may occur as an isolated disease or may be an initial symptom of a systemic disease. Therefore, it is recommended to investigate systemic diseases in all patients. 3 However, in more than 90% of the patients any reason is not defined through adequate investigations and they are considered as idiopathic. 1 -3 In SSNHL development, vascular, infectious, autoimmune, traumatic, metabolic, hematologic, and neurological mechanisms have been suggested and is still being discussed. 3 -6 Advanced ages, vertigo at the onset, degree of hearing loss, audiometric configuration, duration between onset, and treatment are known to be important for the prognosis. 2 -4 On the other hand, the prevalence of hearing loss is reported to be high in males, Caucasians, smokers, and patients with older age, low educational level, diabetes mellitus (DM), and hypertension (HT). 4
In recent years, hypotheses related to the formation of SSNHL have focused on increased risk of ischemia, such as atherosclerosis and microvascular damage caused by chronic inflammation. 5 -7 Nowadays, inflammatory biomarkers such as white blood cell (WBC) count, WBC subtypes counts, neutrophil to lymphocyte ratio (NLR), and platelet to lymphocyte ratio (PLR) have been significantly associated with SSNHL many times and have been suggested to have prognostic values. Among these, NLR has received great attention. 6-7
In this study, we investigated the effects of inflammatory biomarkers including neutrophil (N), lymphocyte (L), NLR, platelet (Plt), and PLR, which were obtained easily and inexpensively with complete blood count, on the severity of hearing loss, recovery rates, and audiogram configurations of patients with SSNHL. In our study, we also tried to find answers to the question of how these inflammatory markers are affected in the presence of cardiovascular risk (CVR)-producing comorbid diseases, such as DM, HT, hyperlipidemia (HL), which frequently accompany SSNHL patients. To our knowledge, this is the first study to investigate the relationship between these parameters and systemic diseases in patients with SSNHL.
Materials and Methods
Ethical Consideration
Our study was started after the TUEK-14 numbered 33646832 / 702.99 of the approval of Medical Education Training Board of our institution.
Study Population
Medical records of patients treated with SSNHL in our clinic between 2016 and 2018 were retrospectively reviewed. Patients who have passed the onset of the disease for more than 3 days; who have taken steroids or other anti-inflammatory drugs before admission; patients with a history of otitis media during the last 4 weeks; those who have active viral infection at the time of diagnosis; patients with a history of otologic surgery, trauma, or barotrauma; patients with retrocochlear pathology determined in magnetic resonance imaging; and those who received treatment other than standard treatment (prednisone in the dose of 1 mg/kg per day, with a progressive dose reduction, maintained for at least 2 weeks); and patients who did not have complete blood count before treatment were excluded.
A total of 122 (n = 122) patients with SSNHL who met the admission criteria were included in the study. In this patient population, patients with only SSNHL were grouped in the group 1 (n = 68), and patients with one or more CVR factors in addition to the diagnosis of SSNHL were grouped in the group 2 (n = 54). Cardiovascular risk factors were determined based on European Guidelines on Cardiovascular Disease Prevention in Clinical Practice (version 2012). 8 Patients were considered to have HT, DM (type I or II), and HL based on their self-reports and if they were already being treated for those diseases. Obesity was defined by a body mass index greater than 30 kg/m2.
As control groups, among age and sex matched patients, who had one or more of the CVR factors defined in group 2 and had no diagnosis of SSNHL were included in group 3 (n = 30); while group 4 (n = 30) was formed from patients who did not have SSNHL and did not have any CVR factors (Table 1). Informed consent was obtained from all patients.
Definitions of Groups.

Abbreviations: CVR, cardiovascular risk; DM, diabetes mellitus, HT, hypertension, HL, hyperlipidemia; SSNFL, sudden sensorineural hearing loss.
Audiometric Assessment
The results of pure-tone audiometry (Interacustic AC 40, Assens, Denmark) of the patients with SSNHL were evaluated by assessing the results before and 1 month after the end of treatment. Audiological evaluation was performed at 250, 500, 1000, 2000, 4000, and 8000 Hertz (Hz) frequencies, and the pure-tone average, speech detection threshold, speech discrimination scores were examined. Hearing loss was classified as mild (< 40 dB loss for any frequency), moderate (up to 80 dB), and severe (profound, > 80 dB).
Hearing recovery was defined as complete, partial, slight, and no recovery in the Siegel’s criteria resembling the previous studies. Partial and slight recoveries were pooled as partial recovery.
According to audiogram configurations, hearing loss was evaluated in 4 groups including up-sloping (hearing loss involving 250-500 Hz), down-sloping (hearing loss involving 4000-8000 Hz), flat (hearing loss with less than 20 dB difference between the best and worst hearing thresholds), and anacousia (complete loss).
Laboratory Assessment
Complete blood count results (Sysmex XE-2100; Kobe, Japan) of patients with SSNHL (groups 1-2) were obtained from routine pretreatment tests, and among those results N, L, and Plt values were recorded. The values of group 3 were determined from the blood samples taken in the routine controls of the patients, and the data of group 4 were determined from the blood samples taken for preoperative anesthesia. The NLR was calculated as the ratio of the absolute N count to the absolute L count, and the PLR was calculated as the ratio of the absolute Plt count to the absolute L count.
Statistical Analysis
The study data were analyzed with the SPSS Statistics version 23 program. Descriptive statistics (mean ± standard deviation) were given for numerical variables. Kolmogorov Smirnov normality test was applied to numerical variables in order to decide on the tests to be applied. As a result of the test, it was seen that all numerical variables provide the assumption of normality and therefore parametric tests were performed in their comparisons. The difference between the 2 independent groups regarding their means was examined by the independent sample t test. The differences between more than 2 independent groups according to the mean values were examined by one-way analysis of variance (ANOVA) and Tukey test was used to determine the groups with significant differences.
One-way analysis of variance was applied to examine whether there are differences between the groups according to the average of the measurements. The power of the study conducted over 182 samples was calculated to be 94.7% with .05 significance level and 0.347 effect width. G * Power 3.1.9.2 package program was used for the power of the study.
Results
Of the patients with SSNHL (groups 1 and 2), 52 (42.6%) were female and 70 (57.4%) were male and the mean age was 62.25 years. In the control groups (groups 3 and 4), 28 (46.6%) were female and 32 (53.4%) were male and the mean age was 50.75 years (Table 2).
Age and Gender Distribution of Groups.

Abbreviations: n, number of patients; SD, standard deviation.
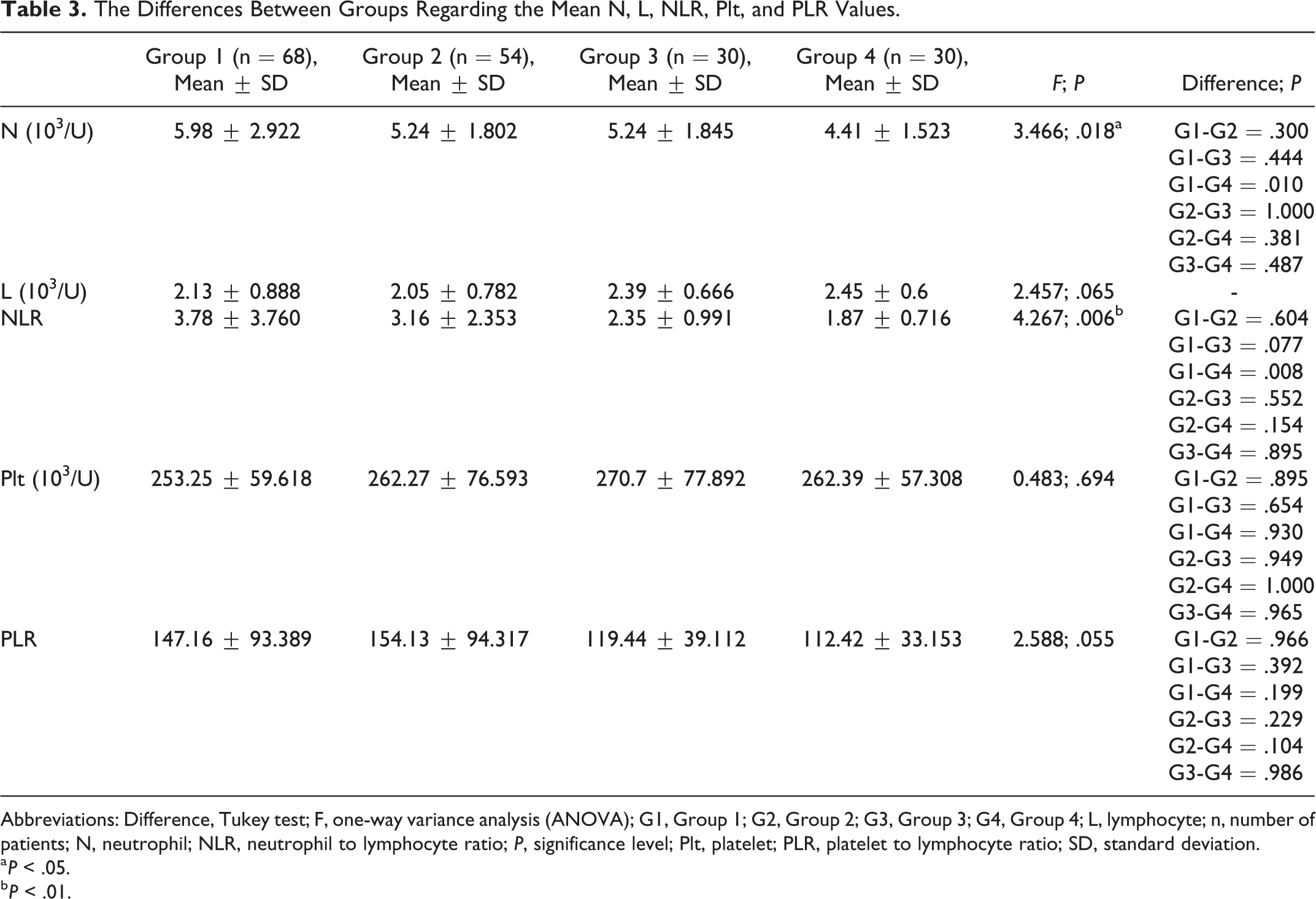
The mean values of N, L, Plt, NLR, and PLR in all groups are shown in Table 3. The highest mean N value was determined in group 1 and the lowest mean N value was found in group 4 (P = .018).
The Differences Between Groups Regarding the Mean N, L, NLR, Plt, and PLR Values.
Abbreviations: Difference, Tukey test; F, one-way variance analysis (ANOVA); G1, Group 1; G2, Group 2; G3, Group 3; G4, Group 4; L, lymphocyte; n, number of patients; N, neutrophil; NLR, neutrophil to lymphocyte ratio; P, significance level; Plt, platelet; PLR, platelet to lymphocyte ratio; SD, standard deviation.
a P < .05.
b P < .01.
As a result of the ANOVA; there was no statistically significant difference between the groups in terms of mean Plt, L, and PLR values (P > .05). The mean NLR value in group 1 (3.78) was significantly higher than that of group 4 (1.87; P = .006). Although not statistically significant, the mean N value of the patients in group 1 was higher than the mean N values of group 2 and group 3 (P = .300, P = .444). The mean N values were similar in groups 2 and 3. Patients with only SSNHL (group 1) had higher NLR and PLR values than patients with only CVR (group 3; P = .077, P = .392; Table 3).
There was no significant relationship between the mean NLR and PLR values and the severity of hearing loss in patients with SSNHL (P = .679, P = 736). However, both NLR and PLR values decreased as the severity of hearing loss increased. Accordingly, the highest NLR and PLR values were observed in the mild and the lowest NLR and PLR values were observed in the patients with severe hearing loss (Table 4).
The Relationship Between Hearing Loss Severity and the Mean NLR and PLR Values.

Abbreviations: Difference, Tukey test; F, one-way variance analysis (ANOVA); n, number of patients; NLR, neutrophil to lymphocyte ratio; P, significance level; PLR, platelet to lymphocyte ratio; SD, standard deviation.
The mean NLR and PLR values in SSNHL patients did not have a significant effect on recovery rates (P = .687, P = .968). But the patients with complete recovery had the highest NLR and PLR values when compared with those with partial and no recovery (Table 5).
The Relationship Between Recovery Rates and the Mean NLR and PLR Values.

Abbreviations: Difference, Tukey test; F, one-way variance analysis (ANOVA); n, number of patients; NLR, neutrophil to lymphocyte ratio; P, significance level; PLR, platelet to lymphocyte ratio; SD, standard deviation.
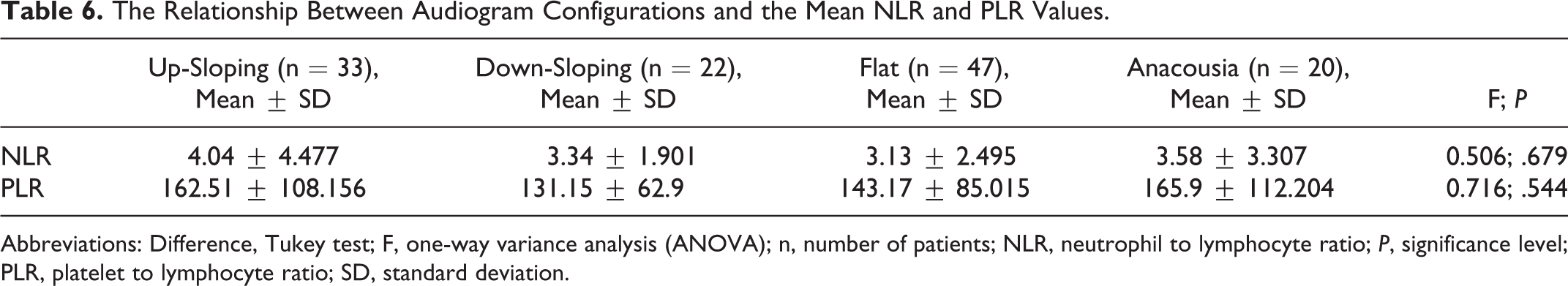
No significant correlation was found between pretreatment audiogram configurations and mean NLR and PLR values of patients with SSNHL (P = .679, P = .544). However, the highest NLR and PLR values were observed in patients with up-sloping configuration (Table 6).
The Relationship Between Audiogram Configurations and the Mean NLR and PLR Values.
Abbreviations: Difference, Tukey test; F, one-way variance analysis (ANOVA); n, number of patients; NLR, neutrophil to lymphocyte ratio; P, significance level; PLR, platelet to lymphocyte ratio; SD, standard deviation.
Discussion
Various hypotheses have been proposed for the formation of SSNHL based on infectious, vascular, traumatic, autoimmune, and metabolic disorders. 6,7 The inner ear is an “end organ” without collateral circulation and its blood supply is mainly provided by the labyrinthine artery. Hair cells in the cochlea consume large amounts of oxygen and are hypersensitive to hypoxia, so a decrease in blood flow is thought to result in SSNHL. 6,7 Due to some clinical features such as acute onset and being mostly unilateral, SSNHL is simulated ischemic vascular diseases such as myocardial infarction, transient ischemic attacks, or amaurosis fugax. 9 Chronic inflammation disrupts blood flow in the inner ear with endothelial damage, triggers the formation of atherosclerosis and subsequent microvascular damage to the striae vascularis, and increases the risk of ischemia. 7 The widespread use of anti-inflammatory steroids in the treatment of SSNHL suggests that inflammation plays an important role in the pathogenesis of this disease.
Many researchers have tried to explain the relationship of SSNHL with diseases carrying CVR-factor such as HT, DM, and HL by vascular-inflammatory hypotheses. 5,9 -12 Haremza et al 5 suggested that SSNHL should be considered as an early vascular symptom that elicits CVR factors such as HL and HT, which are unknown before. Lin et al 10 reported that DM increased the risk of SSNHL and Ryu et al 11 suggested that hyperglycemia could be a potential negative prognostic factor in SSNHL. Many researchers think that the risk factors for ischemic vascular diseases such as HT, HL, DM, and smoking are also risk factors for SSNHL development and that inflammation-induced cochlear ischemia is a common pathway in the development of SSNHL. 12 -15
Neutrophil, L, and Plt, which are the circulating cells in blood, have an important role in the control of inflammation. Nowadays, these inflammatory markers and the values obtained from the ratios of these markers such as NLR and PLR have been the subject of many studies as indicators of systemic inflammation. 6,7,16,17 Among these inflammatory biomarkers, especially NLR achieved large attention since it represents a combination of circulating neutrophil and lymphocyte counts. Chen et al 7 reported that NLR or PLR acted as a comprehensive indicator of 2 complementary immune pathways and NLR was significantly higher in patients with SSNHL than healthy controls. In this meta-analysis, they concluded that NLR is higher in patients who do not recover, and that NLR can be used as both diagnostic and prognostic biomarker for SSNHL. In the meta-analysis of Cao et al, 16 they reported that NLR, PLR, N, and L values were strongly associated with SSNHL, similar to Chen et al. 7 In most of the studies included in both meta-analyzes, 18 -22 patients with SSNHL carrying CVR factors such as HT, DM, and HL were accepted as an exclusion criterion, and the effects of inflammatory conditions caused by these diseases, which have been discussed to have similar pathogenesis, on NLR, PLR, N, and L values have not been investigated in patients with SSNHL. In our study, similar to the literature, we found that patients with SSNHL (group 1) had significantly higher N and NLR values than healthy controls (group 4; P = .018, P = .006), but those carrying CVR in addition to SSNHL (group 2) had lower N and NLR values than patients with isolated SSNHL (group 1; P = .300, P = .604), and we found that N values were very close to each other, especially in groups 2 and 3 (5.24 ± 1.802, 5.24 ± 1.845). Considering that N and NLR values in both groups 2 and 3 were higher than group 4; it can be thought that N and NLR values increase more in acute conditions such as SSNHL and do not create an additional inflammatory condition in the presence of chronic inflammatory conditions carrying CVR factors such as HT, DM, and HL. We could not establish a similar relationship in our findings for PLR values. This abnormal N and NLR elevation in group 1 may be due to psychological and physical stress in patients with SSNHL, as mentioned by Masuda et al. 23 Many authors have shown that comorbidities such as HT, DM, or HL have a negative effect on the prognosis of SSNHL, 10 -14 but according to our findings, N and NLR values were not higher in group 2 than that of group 1 or 3, which was unexpected. However, if NLR elevation was strongly associated with poor prognosis in SSNHL as stated by many investigators, 7,16,18 -22 the highest NLR values would be expected to be in group 2 in our study. Özler 19 found that NLR values were higher in SSNHL patients than in the control group, though the author emphasized that the highest values were determined in patients with the most severe hearing loss the author did not show the same correlation of NLR with moderate or mild hearing loss. Seo et al 20 and Durmus et al 21 found that NLR and PLR values were higher in SSNHL patients than in the control group and in patients who did not recover and therefore they suggested the prognostic values of NLR and PLR. Kum et al 22 detected high NLR levels in patients with SSNHL and those who did not recover but they did not show the same correlation for mean platelet volume, which is an indicator of high platelet activity. Ikinciogullari et al 24 found that NLR and PLR values were higher in SSNHL, but could not correlate with both in terms of recovery, and argued that patients with high NLR values responded better to anti-inflammatory (steroid) therapy. In our results, we observed that as the severity of hearing loss increases NLR and PLR decrease, the highest NLR and PLR values are in patients with complete recovery (P = .687, P = .968) and up-sloping configuration (P = .679, P = .544). Although it is not statistically significant, our results that support each other obviously made us think that high NLR and PLR, like some other authors, may not have a poor prognostic effect. 25,26
One of the most important limitations of our study is the retrospective design and the low number of cases. Another limitation is that, in our study, as in many other similar studies, a cutoff value related to diagnosis and prognosis for NLR and PLR values could not be specified. Lastly, since all SSNHL patients were hospitalized and under aggravated stress, the increased inflammatory response could affect the N, L, and NLR values.
Conclusions
High N and NLR values seem to be a strong laboratory finding supporting inflammatory hypotheses in the diagnosis and pathogenesis of SSNHL. Platelet to lymphocyte ratio values did not show an association as strong as the NLR values with SSNHL. The presence of CVR factors seems to be unable to aggravate the N and NLR levels further in patients with SSNHL. According to our results, high NLR and PLR values tend to indicate better prognosis in terms of hearing loss severity, recovery rate, and audiogram configuration.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
