Abstract
Keywords
Introduction
Sudden sensorineural hearing loss (SSNHL) is specifically characterized by a sudden reduction in hearing ability of at least 30 decibels (30 dB), affecting a minimum of 3 consecutive pure-tone frequencies on the audiogram within a 72-hour timeframe. 1 Typically, SSNHL as unilateral hearing loss, often accompanied by concurrent symptoms like vertigo and tinnitus, necessitating a prompt diagnosis and treatment to mitigate associated risks. 2 Recent surveys in Mainland China estimated an annual incidence of 19 cases per 100,000. 3 Audiogram patterns are categorized into low-frequency, high-frequency, and flat-frequency SSNHL, and known to correlate with the underlying pathological mechanisms and severity of SSNHL. This classification holds clinical significance for analyzing the characteristics of SSHNL patients. 5 Hence, understanding the pathological mechanisms, seeking effective treatments, and evaluating treatment options for patients with different audiometric subtypes are of paramount importance.
While the exact cause and progression of SSNHL remain elusive, there is a broad consensus linking viral infections, autoimmune diseases, and vascular diseases to this condition. 6 An emerging concept suggesting the aberrant activation of cellular stress pathways sheds light on the role of chronic inflammation in the pathophysiology of SSNHL. 7 Inflammation likely plays a crucial role in SSNHL development by elevating the risk of microvascular injury and ischemia. 8 C-reactive protein (CRP), an acute inflammatory protein, has shown a significant association with the pathogenesis of SSNHL.9,10 Furthermore, a study conducted by Zhou et al 11 has highlighted CRP’s role in amplifying the risk of SSNHL and acting as a contributing factor for this condition. Conversely, white blood cells, particularly neutrophils and lymphocytes serve as common inflammatory markers. During inflammation, activated neutrophils lead to increased lymphocyte consumption and apoptosis, resulting in decreased lymphocyte counts in peripheral blood. 12 The neutrophil-to-lymphocyte ratio (NLR) serves as a marker for systemic inflammation and a reliable indicator for SSNHL prognosis and diagnosis. 13 Additionally, the platelet-to-lymphocyte ratio (PLR) is considered an inflammation marker, reflecting changes induced by thrombotic states and acute inflammation. 14
Currently, the treatment of SSNHL lacks consensus primarily due to unknown etiology. Corticosteroids are the most commonly used treatment, 15 despite the potential serious complication of systemic corticosteroid therapy, such as immunosuppression. 16 Fortunately, previous studies have explored batroxobin (Bat) as a potential treatment for SSNHL. Bat addresses potential inner ear thrombosis and enhances cochlear blood flow. 17 Derived from Bothrops moojeni, Bat serves as a defibrinogenic agent in clinical treatment for conditions like SSNHL and vibration disease. 18 The combination of intratympanic corticosteroid and intravenous Bat therapy has shown promising results in the treatment of SSNHL, particularly in cases associated with type-2 diabetes, resulting in a high recovery rate. 19 Notably, SSNHL demonstrates anti-inflammatory and antioxidant properties, making systemic glucocorticoid therapy a recognized and effective treatment for various SSNHL types, despite notable potential adverse effects. 20 Recent studies suggest that localized steroid hormone therapy effectively manages SSNHL, with application of intratympanic injection (ITI) and postauricular injection as the 2 common local treatment approaches. 21 However, the efficacy of a combination treatment of Bat with intratympanic dexamethasone injection (Bat and ITDI) for different audiometric curve types in SSNHL remains unclear, as does its impact on peripheral blood inflammatory cytokines. Our research aims to investigate the effect of Bat and ITDI on the efficacy of SSNHL in patients with different audiometric curve types and the alterations of peripheral blood inflammatory cytokines.
Materials and Methods
Ethics Statement
This study obtained approval from the Academic Ethics Committee of Affiliated Dongyang People’s Hospital of Wenzhou Medical University. All patients and their families were duly informed about the study’s objective and willingly signed the informed consent. It is essential to highlight that this study strictly adhered to the guidelines outlined in the Declaration of Helsinki.
Research Subjects
This retrospective study encompassed a total of 161 patients with SSNHL admitted to the Ear-Nose-Throat Department at Affiliated Dongyang People’s Hospital of Wenzhou Medical University between 2020 and October 2022. These individuals were classified into 2 groups based on their treatment modalities. The control group consisted of 86 patients, while the observation group comprised of 75 patients. The control group underwent Bat therapy, whereas the observation group received Bat and ITDI treatment.
Inclusion and Exclusion Criteria
Inclusion criteria were as follows: (1) meeting the diagnostic criteria for SSNHL as established by the Otolaryngology–Head and Neck Surgery of the Chinese Medical Association; (2) being aged between 18 and 75 years; (3) sudden onset of symptoms occurring within minutes, hours, or over a period of 3 days; (4) the absence of systemic autoimmune disease; (5) patients providing informed consent for participation in the study; and (6) having complete clinical data and the ability to cooperate with relevant treatment and examinations.
Exclusion criteria included (1) the presence of middle ear or inner ear disease or a history of middle and inner ear surgery; (2) the identification of other otologic or intracranial conditions through cranial and temporal bone imaging; (3) patients who had previously received systematic SSNHL treatment at another hospital; (4) patients in pregnancy or lactation; (5) individuals who were prescribed any lipid-lowering drugs, anticoagulants, antiplatelet, or fibrinolytic drugs; and (6) those with allergies to or contraindications to the drugs used in this study.
SSNHL Classification Criteria
The classification criteria for SSNHL among patients were based on the diagnostic criteria for SSNHL, which included (1) low-frequency type: hearing loss at frequencies less than 1000 Hz (included) and hearing loss at least 250, 500 Hz ≥ 20 dBHL; (2) high-frequency type: hearing loss at frequencies above 2000 Hz (included) and hearing loss at least 4000, 8000 Hz ≥ 20 dBHL; (3) flat-frequency type: hearing loss across all frequencies, with an average hearing threshold of ≤80 dBHL at 250 to 8000 Hz (250, 500, 1000, 2000, 3000, 4000, and 8000 Hz); and (4) total deafness type: hearing loss across all frequencies, with an average hearing threshold of ≥81 dBHL at 250 to 8000 Hz (250, 500, 1000, 2000, 3000, 4000, and 8000 Hz).
Data and Sample Collection
Clinical data of all enrolled SSNHL patients were compiled for screening, statistical analysis, and examination. Within 24 hours of admission, fasting elbow venous blood (2 mL) were collected from patients both before and after treatment to assess alterations in peripheral blood inflammatory indicators. This involved assessing neutrophils, lymphocytes, and platelets, facilitating the calculation of NLR (ratio of absolute neutrophil count to absolute lymphocyte count) and PLR (ratio of platelet count to lymphocyte count). The remaining peripheral blood samples were utilized for subsequent enzyme-linked immunosorbent assays (ELISAs).
Treatment Methods
In the control group, patients were treated with Bat (H20030295, Tobishi Pharmaceutical, Beijing, China) in combination with conventional drug treatment, including administration of corticosteroid hormones, neurotrophic, and vasodilator drugs. Bat was administered intravenously via a slow drip every other day, starting with an initial dose consisting of 100 mL of 0.9% sodium chloride combined with 10 BU Bat, followed by subsequent doses of 100 mL 0.9% sodium chloride mixed with 5 BU Bat. Prior to each administration, the fibrinogen concentration was assessed, and treatment efficacy was evaluated through pure-tone audiometry after 4 consecutive doses. In contrast, patients in the observation group underwent ITDI treatments following a similar regimen to the control group. The procedure involved assuming a supine position with the healthy ear facing upward. The skin of the external auditory canal was cleansed and disinfected, followed by the instillation of 4 drops of 2% lidocaine (H20057816, Kelun Pharmaceutical, Hunan, China) in the affected ear to induce local surface anesthesia. After a 5-minute interval, the lidocaine was aspirated using a negative pressure aspirator. Using a tympanic membrane puncture needle, 1 mL (10 mg) of dexamethasone (H41021255, Tianyao Pharmaceutical, Tianjin, China) was injected into the tympanic cavity through the posterior inferior quadrant of the tense part of the affected ear’s tympanic membrane. Subsequently, patients maintained a supine head position toward the healthy ear for 30 minutes, minimizing swallowing movements to ensure optimal drug absorption within the tympanic cavity. After the puncture, patients were instructed on ear hygiene and dryness, and the medication was administered 4 times every other day. Treatment efficacy was assessed through subsequent pure-tone audiometry.
Enzyme-Linked Immunosorbent Assay
The level of the peripheral blood inflammatory factor CRP (ml057570) in patients was assessed using ELISA kit from Enzyme-linked Biotechnology, Shanghai, China, following the provided instructions. 23
Evaluation Criteria
After treatment, the assessment of efficacy aligned with the classification of therapeutic effect for SSNHL in China. Briefly, the efficacy was categorized as follows: (1) cured: restoration of hearing threshold to normal at all frequencies, reaching the level of the healthy ear, or returning to the level before the onset of the disease; (2) markedly effective: an improvement exceeding 30 dB in the average hearing loss frequency; (3) effective: enhancement ranging 15 to 30 dB in the average hearing loss frequency; and (4) ineffective: an improvement of less than 15 dB in the average hearing loss frequency. The total effective rate was calculated as (number of cured cases + number of markedly effective cases + number of effective cases)/total number of cases × 100%.
Statistical Analysis
Data analysis and graphing were conducted using SPSS 21.0 (IBM, Armonk, NY, USA) and GraphPad Prism 8 (GraphPad Software, San Diego, CA, USA). The normal distribution of data was assessed using the Shapiro–Wilk test, and outcomes were reported as mean ± standard deviation. Unpaired t-tests were utilized for comparisons between 2 groups, while categorical variables were analyzed employing Fisher’s exact test. Statistical significance was defined at P < .05.
Results
Comparison of Clinical Baseline Data Between the Two Groups of SSNHL Patients
The control group, comprising of 86 patients (48 males and 38 females), exhibited an average age of 50.44 ± 15.46 years, while the observation group, comprising 75 patients (37 males and 38 females), had an average age of 49.19 ± 13.02 years. The clinical baseline data of both groups were compared and analyzed. There were no significant differences between the 2 groups in terms of age, sex, disease course, concomitant symptoms, hypertension, diabetes mellitus, ear side, sudden hearing loss type, and hearing loss degree (all P > .05), signifying their comparability. The data are provided in Table 1.
Comparison of Clinical Baseline Data Between 2 Groups of SSNHL Patients.
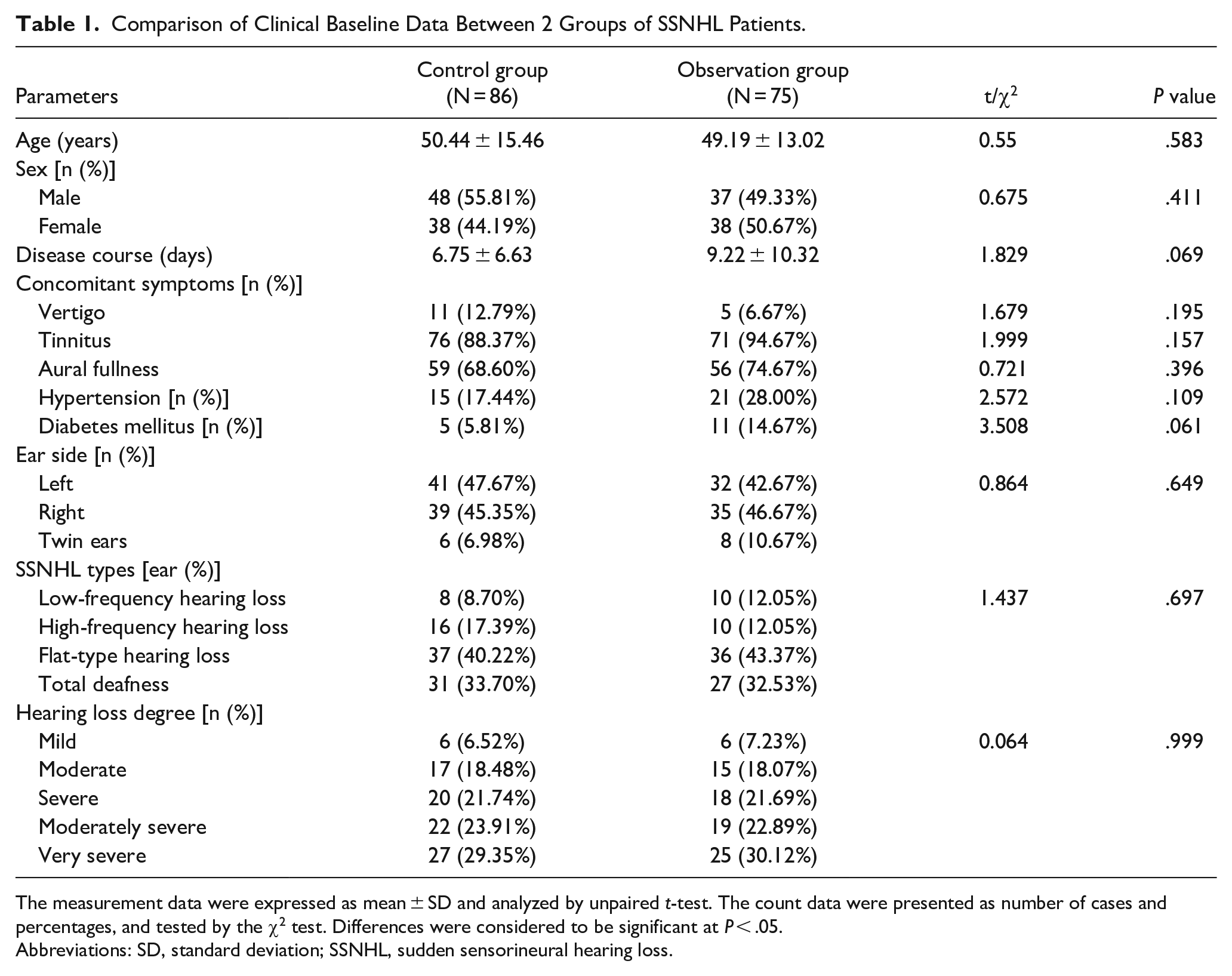
The measurement data were expressed as mean ± SD and analyzed by unpaired t-test. The count data were presented as number of cases and percentages, and tested by the χ2 test. Differences were considered to be significant at P < .05.
Abbreviations: SD, standard deviation; SSNHL, sudden sensorineural hearing loss.
Comparison of Clinical Efficacy Between the Two Groups of SSNHL Patients
In the assessment of clinical efficacy posttreatment, both groups exhibited improvements in hearing. Within the observation group, 14, 16, 33, and 20 cases demonstrated cured, markedly effective, and ineffective levels of efficacy after treatment, culminating in a total effective rate of 75.90%. Contrastingly, the control group showcased 10, 20, 25, and 37 cases exhibiting these corresponding levels of efficacy, resulting in a total effective rate of 59.78% (χ2 = 5.163, P < .05). The data are shown in Table 2. The results suggest that Bat and ITDI treatments deliver superior clinical efficacy for SSNHL patients.
Comparison of Clinical Efficacy Between 2 Groups of Patients [Ear (%)].
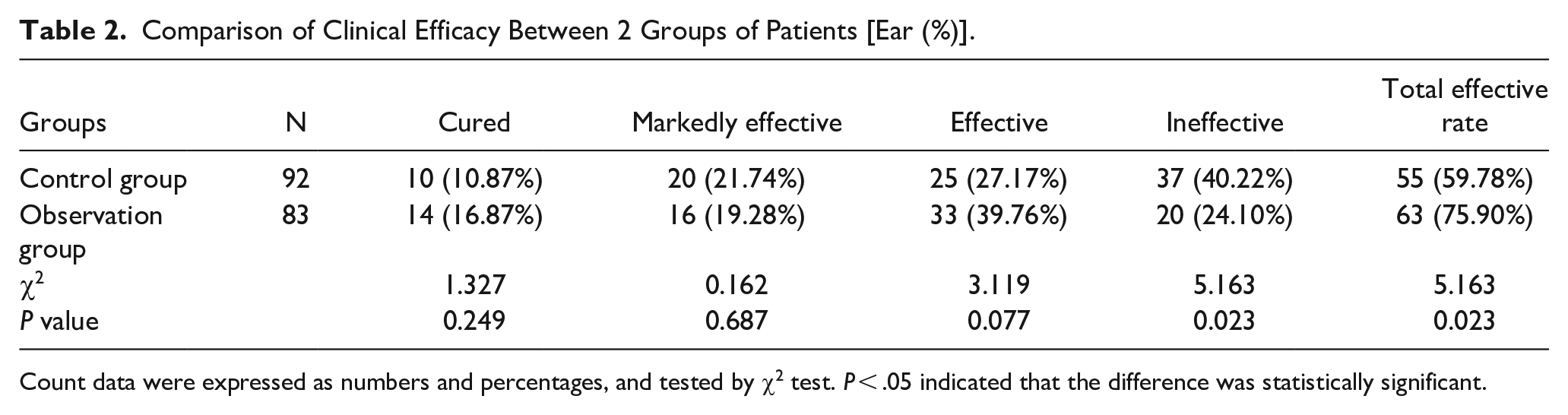
Count data were expressed as numbers and percentages, and tested by χ2 test. P < .05 indicated that the difference was statistically significant.
Comparison of the Total Effective Rate of SSNHL Patients With 4 Audiometric Curve Types After Treatment
Upon categorizing all patients based on their audiometric curves, it became evident for individuals with low-frequency hearing loss, the total effective rate in the observation group was 80.00%, compared to 87.50% in the control group, with no statistically significant difference between the 2 groups (P > .05). In the case of patients with high-frequency hearing loss, the total effective rate in the observation group (80.00%) was higher than in the control group (50.00%), however the difference was not statistically significant (P > .05). Regarding cases with flat-type hearing loss, the 2 groups did not significantly differ in the total effective rate, with the observation group (72.22%) exceeding the control group (56.76%) (P > .05). In instances of total deafness, the total effective rates in both groups did not exhibit a statistically significant difference, yet the rate in the observation group (77.78%) was higher than that in the control group (61.29%) (P > .05). Further details are available in Table 3.
Comparison of the Total Effective Rate of SSNHL Patients With 4 Audiometric Curve Types After Treatment [Ear (%)].
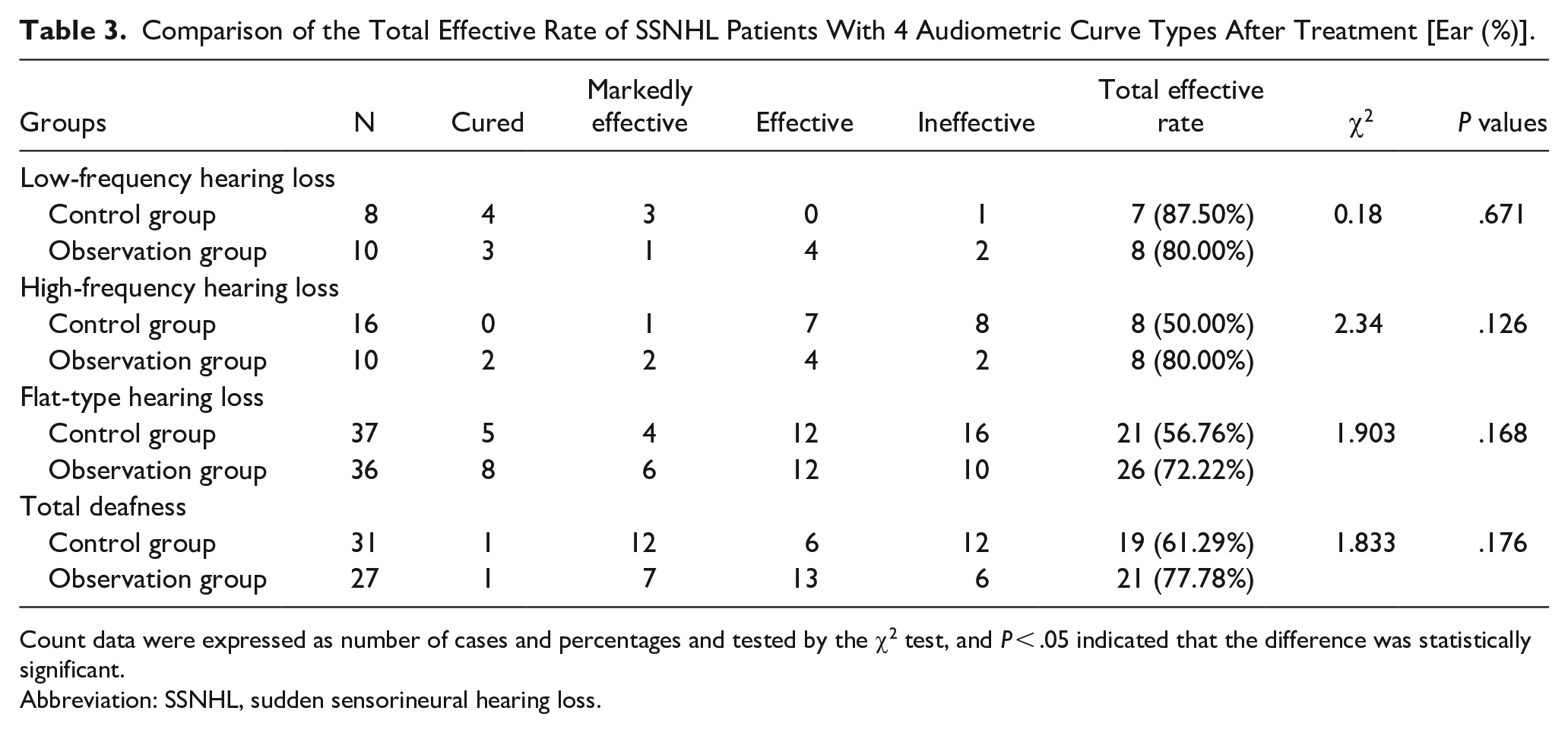
Count data were expressed as number of cases and percentages and tested by the χ2 test, and P < .05 indicated that the difference was statistically significant.
Abbreviation: SSNHL, sudden sensorineural hearing loss.
Comparison of Peripheral Blood Inflammation Levels Between the 2 Groups of SSNHL Patients Before and After Treatment
The comparison of peripheral blood inflammation levels between the 2 groups of SSNHL patients before and after treatment highlighted no noticeable differences in peripheral blood inflammation indicators CRP, NLR, and PLR prior to treatment (all P > .05). However, following treatment, the levels of CRP in peripheral blood and the ratios of NLR and PLR in both groups significantly decreased (all P < .05). Notably, the observation group exhibited lower levels of peripheral blood CRP level and the ratios of NLR and PLR compared to the control group (all P < 0.05). The data are provided in Table 4. These results imply that Bat and ITDI treatments effectively reduce peripheral blood inflammation levels in SSNHL patients.
Comparison of CRP, NLR, and PLR Values Between the 2 Groups Before and After Treatment [Mean ± SD (%)].
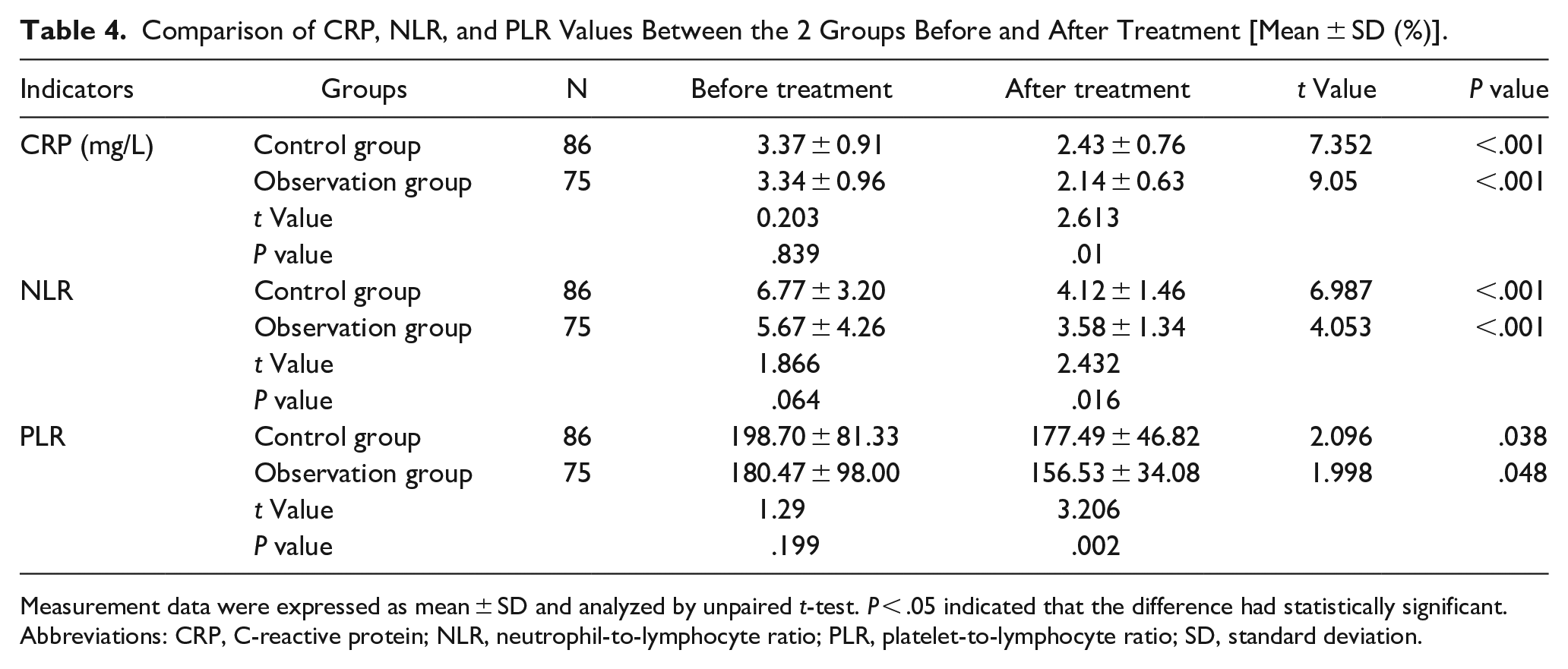
Measurement data were expressed as mean ± SD and analyzed by unpaired t-test. P < .05 indicated that the difference had statistically significant.
Abbreviations: CRP, C-reactive protein; NLR, neutrophil-to-lymphocyte ratio; PLR, platelet-to-lymphocyte ratio; SD, standard deviation.
Comparisons of Peripheral Blood Inflammation Levels in SSNHL Patients With 4 Audiometric Subtypes After Treatment
In the observation group of SSNHL patients, significant reductions were evident in peripheral blood CRP, NLR, and PLR levels among individuals experiencing low-frequency hearing loss, high-frequency hearing loss, flat-frequency hearing loss, and total deafness relative to the control group. Statistically significant differences were particularly identified between the 2 groups, specifically in the peripheral blood NLR level of SSNHL patients with low-frequency hearing loss, the peripheral blood CRP level in cases of high-frequency hearing loss, and the peripheral blood PLR levels in SSNHL patients with flat-frequency hearing loss and total deafness (all P < .05) (Table 5).
Comparisons of Peripheral Blood Inflammation Levels in SSNHL Patients With the 4 Audiometric Curve Subtypes After Treatment.
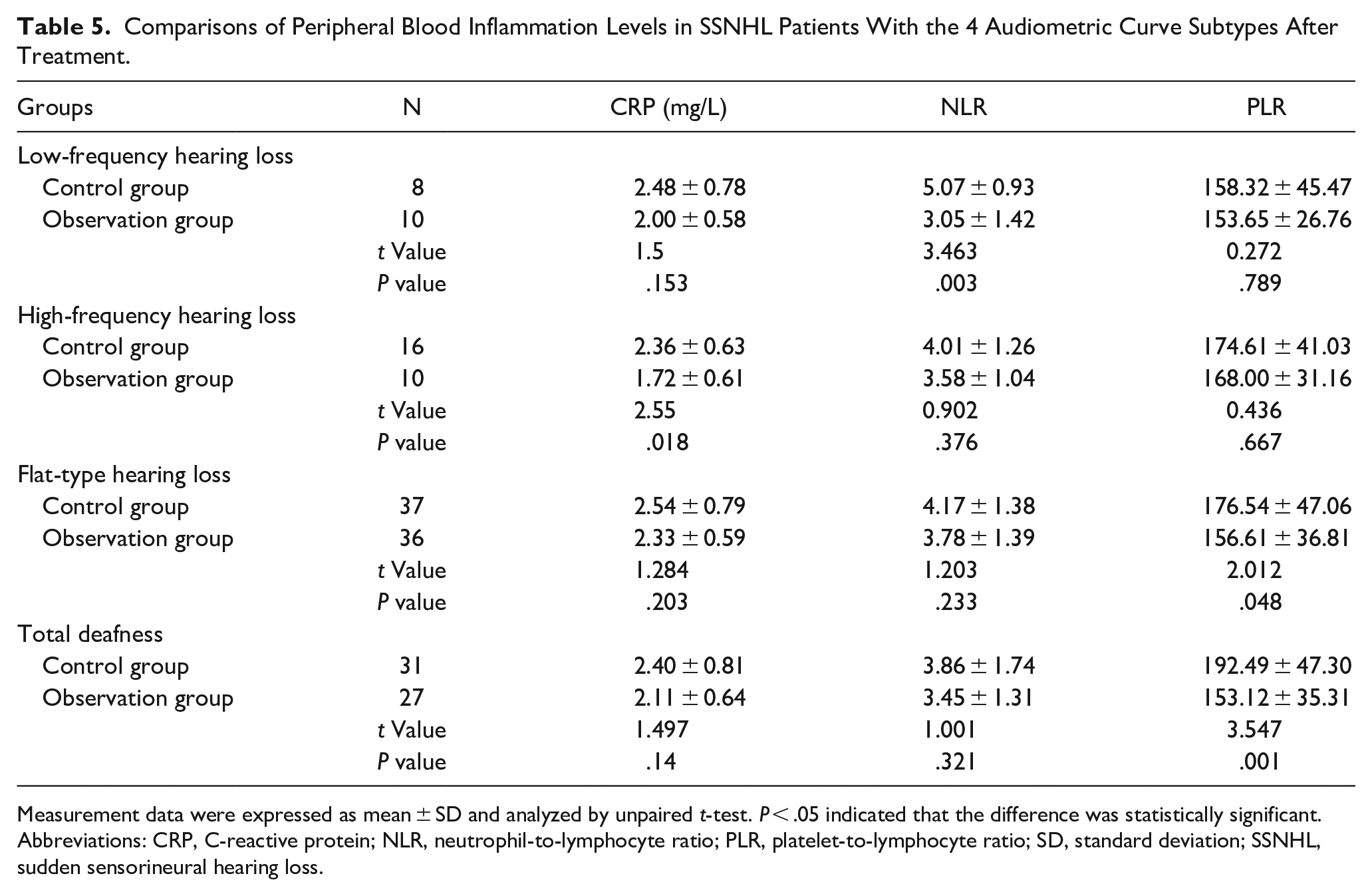
Measurement data were expressed as mean ± SD and analyzed by unpaired t-test. P < .05 indicated that the difference was statistically significant.
Abbreviations: CRP, C-reactive protein; NLR, neutrophil-to-lymphocyte ratio; PLR, platelet-to-lymphocyte ratio; SD, standard deviation; SSNHL, sudden sensorineural hearing loss.
Comparison of Adverse Reactions and Complications Between the 2 Groups of SSNHL Patients After Treatment
Throughout the treatment course, adverse reactions and complications were closely monitored in both groups of SSNHL patients. The results revealed the occurrence of various adverse events and complications, including pain, dizziness, tympanic membrane perforation, tympanic membrane infection, tinnitus aggravation, hearing loss, elevated blood pressure, and raised blood glucose levels in both groups. The incidence of these adverse reactions and complications in the observation group (14.67%) was slightly lower than that in the control group (18.60%). Nevertheless, its vital to note that this difference did not reach statistical significance (P > .05). Detailed data are presented in Table 6.
Comparison of Adverse Reactions and Complications Between the 2 Groups [n (%)].
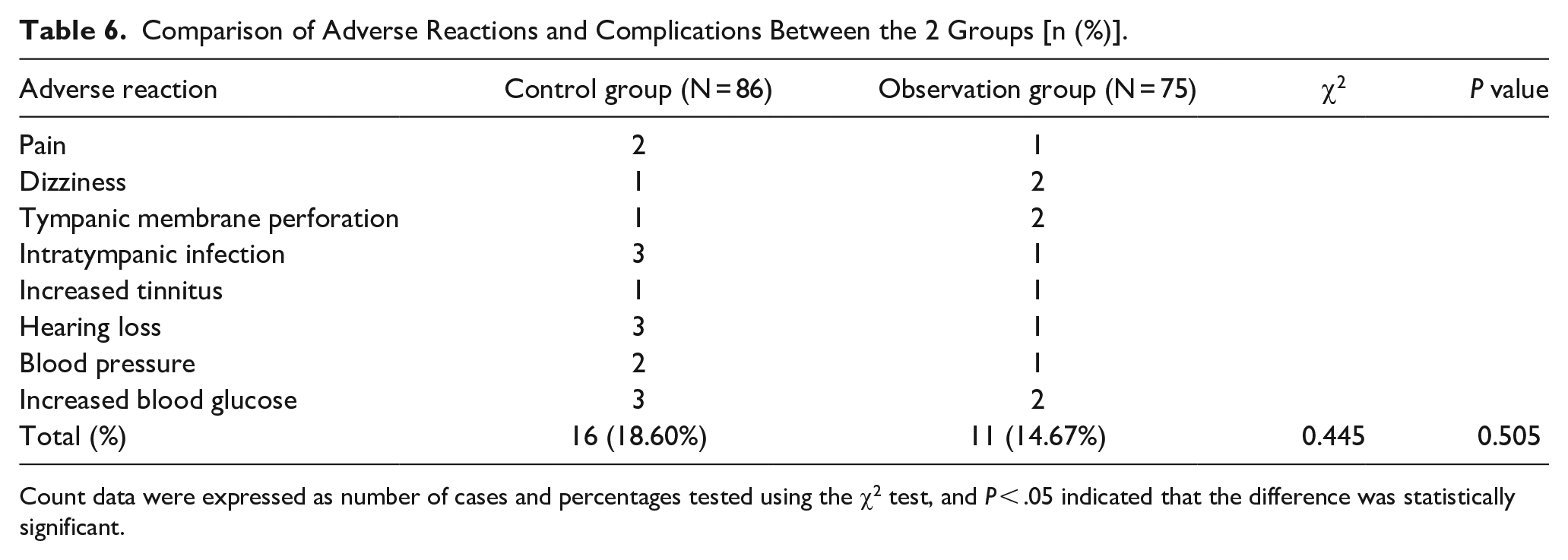
Count data were expressed as number of cases and percentages tested using the χ2 test, and P < .05 indicated that the difference was statistically significant.
Discussion
SSHNL presents a significant challenge within otolaryngology. 24 Individuals affected by SSNHL may often endure distressing symptoms such as tinnitus, deafness, vertigo, and related manifestations. 25 The involvement of factors such as inflammation, microthrombosis, atherosclerosis, and immune dysfunction have been suggested in the onset of SSNHL. 26 Timely identification and treatment of this condition can substantially improve the quality of life for SSNHL patients. 15 Moreover, combination therapy stands as a primary approach in managing total deafness in SSNHL cases. 27 Consequently, there is an urgent need to explore combination therapies and deepen our comprehension of SSNHL’s etiology, which could significantly impact its treatment. Our study aimed to assess the clinical significance of Bat and ITDI treatment in SSNHL patients. The experiment results revealed that Bat and ITDI exhibited superior efficacy in SSNHL patients, showcasing the potential to reduce peripheral blood inflammatory indicators and the incidence of adverse effects or complications.
Intratympanic corticosteroid injections facilitate drug diffusion through the round window membrane into the inner ear, 28 thereby intensifying drug concentration and reducing the occurrence of side effects compared to systemic applications. 19 Previous studies have suggested that combining Bat with significant intratympanic steroid injection effectively treats SSNHL. 29 Aligning with prior research, our study demonstrated that Bat and ITDI improved clinical efficacy in treating SSNHL patients. However, following combined therapy, no significant difference was identified in the total effective rate among SSNHL patients with 4 audiometric curve types. Additionally, both groups of SSNHL patients, whether receiving Bat treatment alone or Bat and ITDI, experienced complications or adverse reactions during treatment, with a slightly lower incidence observed in the combination-treated patients. Nevertheless, this disparity lacked statistical significance. The dedicated efforts of our colleagues have indeed demonstrated that, owing to fewer adverse reactions and high efficacy, ITI of drugs stand as a preferable treatment option for refractory SSNHL. 30 However, limited reports exist regarding the complications or adverse reactions from Bat and ITDI treatment in SSNHL patients, presenting an innovative aspect of our study.
Vascular occlusion, viral infection, cochlear inflammation, as well as immune-mediated mechanisms stand as prominent factors contributing to SSNHL. 31 Inflammation and vascular risk factors are believed to play a role in the occurrence of SSNHL. 32 Bat assists in the depolymerization of fibrinogen polymers, thereby reducing oxidative stress and the inflammatory response. 33 In the context of SSNHL development, inflammatory cytokines like NLR and PLR values, CRP level, and the CRP/serum albumin ratio are involved. 34 Remarkably, CRP levels are elevated in SSNHL patients compared to normal subjects. 35 Previous studies have indicated relatively elevated NLR and PLR values in SSNHL patients, foreseeing the incidence of SSNHL. 36 The higher NLR and PLR values in patients with poor prognoses compared to those with good prognoses further reinforce the correlation between ISSNHL and inflammatory response. 37 Our result indicate that Bat and ITDI can reduce peripheral blood CRP levels, NLR, and PLR values in patients with SSNHL. Essentially, Bat and ITDI led to a reduction in inflammation levels in the peripheral blood of SSNHL patients. Specifically, the levels of peripheral blood CRP, NLR, and PLR levels of SSNHL patients with the 4 audiometric subtypes were decreased.
This study introduces an innovative revelation that the overall effectiveness rate of SSNHL patients in the observation group was significantly higher than that of the control group, indicating a positive impact of Bat and ITDI treatment on SSNHL patients. Specifically, in cases of high-frequency hearing loss, the observation group displayed a higher effectiveness rate compared to the control group. Similarly, for flat-frequency hearing loss cases, the observation group exhibited a superior effectiveness rate in contrast to the control group. Moreover, the effectiveness rate for total deafness cases was elevated in the observation group. However, its important to note that the differences between the 2 groups were not statistically significant, potentially due to the limited number of patients included in the study. Notably, within the observation group, lower effectiveness was observed for patients with low-frequency hearing loss. This observation might be due to the limited efficacy of intratympanic injections, given that low-frequency SSNHL often relates to lifestyle factors and viral infections. These findings emphasize the enhanced effectiveness of Bat and ITDI in enhancing the hearing abilities of SSNHL patients, particularly among those experiencing with flat/high-frequency hearing loss, and total deafness. Consequently, there is a necessity for distinctive treatment strategies. For patients with the 3 types of sudden hearing loss (high-frequency, flat-frequency, total deafness), prioritizing this combined treatment, alongside careful monitoring of fibrinogen levels, could result in positive clinical outcomes. Overall, this strategy holds clinical significance by establishing a standard for treating SSNHL patients and offering crucial guidance to otolaryngologists in selecting precise treatment protocols.
In summary, our study highlights the superior clinical efficacy of Bat and ITDI in the treatment of SSNHL patients with varying audiometric curve types. Furthermore, this treatment approach effectively reduces the levels of inflammatory cytokines in the peripheral blood of SSNHL patients, thereby reducing the incidence of adverse reactions and complications. However, our study is subject to certain limitations. Firstly, the relatively sample size prompts the need for further investigations with larger sample sizes to comprehensively assess the effects of Bat and ITDI on clinical efficacy and alterations in peripheral blood inflammatory indicators in SSNHL patients. Additionally, our present study offers initial insights into changes in inflammatory indicators in peripheral blood, warranting further-in-depth exploration of the relationship between these indicator and SSNHL. Moving forward, our plans induced multicenter studies and expanding the sample size to strengthen the reliability of our results. Concurrently, we are committed to ongoing efforts to verify the role of the inflammatory response in the development of SSNHL.
Footnotes
Author Contributions
CH is the guarantors of integrity of the entire study and contributed to the study concepts; CH and QJ contributed to the study design and definition of intellectual content; QJ and JZ contributed to the literature research, data analysis, and article preparation; QJ contributed to the statistical analysis, article editing, and review; QJ, JZ, DW, and TC contributed to the data acquisition and clinical studies; All authors read and approved the final article.
Availability of Data and Materials
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partially supported by grants Jinhua Science and Technology Bureau public welfare project, 2023-4-218.
Ethics Statement
This study was approved by the Academic Ethics Committee of Affiliated Dongyang People’s Hospital of Wenzhou Medical University. All patients and their families were informed of the purpose of this study and were willing to sign the informed consent. This study was conformed with the Declaration of Helsinki.
