Abstract
The main influencing factors of fouling in coal gasification black water systems were revealed by analyzing coal samples, scale samples, and black water gray water samples. In terms of coal quality, the ash in coal is the main source of scale, and the use of high-ash coal increases the tendency of fouling. The black water gray water is featured by low water quality, high calcium and magnesium ion contents, turbidity, and dissolved solids, which easily precipitate and crystallize when the temperature and pressure of the system change drastically, leading to fouling. On this basis, a magnetic flocculant combined with ferric oxide and polyacrylamide was prepared. Experiments showed the turbidity of black water was effectively reduced under a certain dosage, and the removal rate of turbidity in black water reached 79.89%, but the turbidity was increased by adding excessive magnetic flocculant.
Introduction
Energy is the basis of human production and development. At present, humans mainly use fossil energy, including coal, oil, and natural gas. The coal, oil, and gas industries play a pivotal role in meeting global energy demands, providing the lifeblood for various sectors (Banan and Asadi, 2024). Fossil energy will remain the main energy used by humans for a long time (Li et al., 2021). Coal plays an extremely important role in China's energy use. The basic situation of China's energy distribution is “short of oil, short of gas, and extremely rich in coal.” Therefore, we should fully utilize the advantages of China's rich coal resources and actively make diverse use of coal. Under the current situation, accelerating the clean and efficient utilization of coal is an inevitable choice and a strong cornerstone supporting energy transformation, ensuring national energy security, and achieving the goal of “double carbon” (Li, 2021). China's coal-rich oil-poor gas-poor energy resource endowment characteristics determine the main energy status of coal in the short term and will not be fundamentally changed. In 2021, China's coal consumption of more than 4 billion tons was still as high as 56% of the primary energy consumption, and the use of coal carbon emissions accounted for about 70% of carbon emissions from fossil energy consumption (Xie, 2021). If we do not quickly adjust the current utilization and consumption structure of coal, it will exacerbate the problems of carbon emissions and environmental pollution. At the present stage and for a long time, we should achieve clean utilization of coal resources and ensure the sustainable development of the national economy (Wang et al., 2018a).
Coal gasification projects are one of the ways to promote clean, efficient, and comprehensive utilization of coal. In recent years, coal gasification projects have been carried out in many areas of China (Zhang and Ponomarenko, 2023). In coal gasification engineering, coal is converted via a chemical reaction with a gasification agent (oxygen, water vapor, etc.) using coal as raw material and under specific equipment and conditions into syngas, which is mainly composed of carbon monoxide, hydrogen, and methane (Tawanda et al., 2022). Syngas is used in many applications; for instance, syngas is an important raw material for the production of synthetic ammonia and methanol. Syngas obtained through coal gasification can be used to produce synthetic ammonia and methanol after purification, conversion, and other processes to adjust the ratio of its composition, which is an important basis for the fertilizer and chemical industry. Syngas can be converted into liquid fuels (gasoline, diesel, etc.) through Fischer-Tropsch synthesis and other processes, realizing the indirect liquefaction of coal. At the same time, syngas can also be used to produce chemical products such as olefins, providing an important basic raw material for the chemical industry and reducing dependence on petroleum resources (Adnan et al., 2021). Coal gasification combined cycle power generation is a highly efficient and clean way to generate electricity. The syngas produced by coal gasification is purified and then enters the gas turbine to generate electricity. Then the high-temperature exhaust gas discharged from the gas turbine is used to generate steam to drive the steam turbine to generate electricity. This way achieves the gradual utilization of energy, improves the efficiency of power generation, and reduces the emission of pollutants. Coal gasification is a clean way of utilizing coal, and the sulfur, nitrogen, and other impurities in the coal can be effectively removed through gasification and subsequent purification and treatment, which reduces the emissions of sulfur dioxide, nitrogen oxides, and other pollutants. Compared with the direct combustion of coal, coal gasification can convert coal into more easily utilized gas fuel or chemical raw materials, improving the utilization efficiency of coal and expanding its application field (Dai et al., 2021a; Xu et al., 2022).
In coal gasification, entrained-flow gasification is characterized by high gasification temperature, wide application range of coal, large operating load, and high coal conversion rate. This process has been widely used in recent years and is the first choice technology for large-scale gasification. However, the air bed gasification process has some disadvantages. For instance, the air bed in the gasification process will produce black water with high temperatures and a large flow rate. The process of treatment will produce a certain amount of low-pressure steam, and consume a large amount of circulating cooling water, making the black water treatment system easy to scale (Ammendola et al., 2011). The coal gasification system fouls the main part of the black water pipeline, gasifier, washing tower, and evaporation of the hot water tower black water pipeline. The coal gasification system formed by scale pieces is generally wrapped in part of the hard ash and the formation of the scale piece is composed of magnesium ions and calcium ions. At a higher black water pipeline pressure, the formation of the scale piece of the process of the density is greater, so the texture is very hard (Guo et al., 2022). The main control points of the gasification technology are coal quality, water quality, and system water balance. These control points are influenced by many factors and are hard-to-control, which restrict the long-term stable operation of gasification devices. Hence, it is particularly important to control water quality (Sun et al., 2023). In the coal chemical industry, the black water and gray water system plays an important role. Poor water quality can easily lead to problems such as deposition and fouling in the gray water system. When the pipelines and systems are seriously blocked, the production of the whole gasification device can be completely stopped. Therefore, it is significant to control the water quality index of the gasification device for its long-term, safe, clean, and efficient operation (Cao et al., 2014).
There are mainly electro-flocculation, membrane absorption, and acid gas hard removal technologies for the gasification black water system. With the electro-flocculation technology, a soluble electrode plate is placed in a reaction tank and electrified. Then Fe3+ and Al3+ combine with aqueous OH− to generate polymers (e.g. Fe (OH)3 and Al (OH)3), which can adsorb and settle colloidal particles, suspended solids and other impurities in black water, effectively reducing the total hardness of gasified black water. In the membrane absorption hardening technology, black water first passes through a multi-media filter to remove suspended solids and most solid substances, and then enters a nano-filter to harden and become softened water. The softened water then enters a reverse osmosis membrane recovery device to separate Na+ from water. The permeated water of the reverse osmosis membrane enters the black water system for recycling, and the non-permeated water with high Na+ concentration is recycled to prepare a regenerated solution of the nano-filter. Acid gas hardening technology is improved from the lime-soda method. Acid gases such as CO2 and SO2 react with aqueous Ca2+ and Mg2+ to form CaSO4 and CaCO3 crystals. After that, the solubility of CaSO4 will decrease with the increase in pH, and some CaSO4 crystals will precipitate to form solids. Then the solids will be separated from the black water in a solid–liquid way to reduce the total hardness of the black water (Hu et al., 2019; Hu et al., 2021).
In addition to the above methods, black water flocculation and settling with flocculants are commonly used to reduce the hardness of black water. The most common flocculant is polyacrylamide (PAM). The long-chain molecular structure of PAM contains a large number of active groups, such as −COOH and −CONH2. These polar groups easily adsorb and aggregate suspended solids and colloidal particles in water, and are widely used as flocculation treatment agents for sewage (Manafi et al., 2017). Among them, cationic PAM (CPAM) containing positively charged groups can generate electrostatic attraction with negatively charged suspended particles in black water. This reaction compresses the radius of the hydration layer, strengthens the mutual attraction with colloidal particles, and thus completes adsorption and aggregation. Hence, CPAM is suitable for flocculation treatment of black water (Wang et al., 2014).
However, in recent years, more and more researchers have found that ordinary flocculants rely on gravity settling for solid–liquid separation. The flocculation and settlement time is long when using ordinary flocculants to treat black water. The flocculation process produces a large amount of sludge, which is large in size and high in water content, making it difficult to treat and dispose of ordinary flocculant dosage is large and the treatment cost is high. Therefore, a new type of flocculants is needed to solve these problems.
Magnetic materials have received more attention due to the magnetic properties in magnetic fields. It is of great significance to prepare magnetic composite flocculants to realize solid–liquid separation under the joint action of gravity and external magnetic field. The magnetic particles in magnetic flocculants have large specific surface area and surface activity, which can quickly adsorb suspended particles, colloids, and other pollutants in the water, prompting them to rapidly aggregate to form larger flocs, greatly shortening the flocculation and settling time and improving the efficiency of water treatment. In the treatment of some wastewater with high turbidity or containing a large number of fine particles, the advantages of rapid flocculation are more obvious. In flocculation, the floc structure formed by magnetic flocculant is more compact, the water content is relatively low, and the amount of sludge produced is less. Because the sludge contains magnetic particles, the magnetic field can further thicken and dewater the sludge, reduce the difficulty and cost of sludge treatment, and reduce the secondary pollution of sludge to the environment. The separated magnetic flocculant can be reused after proper treatment (e.g. cleaning and regeneration), which reduces the use cost of the agent. This reusable feature not only saves resources, but also helps to reduce the potential impact of chemicals on the environment (Hu et al., 2024; Ma et al., 2018).
In this study, X-ray diffraction (XRD), scanning electron microscopy (SEM), and energy dispersal spectrum (SEM–EDS) analysis were used to comprehensively and thoroughly analyze the mineralogical, structural, and elemental compositions of the coal and scale flakes. This multi-technology approach, compared with single-technology analyses, reveals the intrinsic mechanisms of fouling in the black water system of coal gasification in a more comprehensive and accurate manner, and provides richer and more detailed information on the microscopic level of the study. The method is more comprehensive and accurate than single-technology analysis. A magnetic flocculant combining non-toxic and environmentally friendly ferric tetraoxide with PAM commonly used in industry was prepared to overcome the shortcomings of traditional flocculants and single materials. Its application effect in the coal gasification black water system was investigated, which is innovative in the study of flocculants applied in this field and expands the research boundary of flocculants in optimizing the water quality of coal gasification black water.
Although previous studies have paid attention to the fouling problem of coal gasification black water systems, this study further clarifies the specific role mechanisms of coal quality, black water and gray water water quality, temperature, and pressure in the fouling process through multi-dimensional analysis. This clarification complements and improves the understanding of the causes of fouling, and provides a more systematic theoretical basis for subsequent studies. Based on the existing research on hardness removal technology and flocculants, a new type of magnetic flocculants was developed and its application effect was studied. This research provides new technical means and practical references for optimizing the water quality of the black water system in the gasification plant, enriches the research results in this field, and provides more choices for practical engineering applications (Song et al., 2016; Zhu et al., 2019).
Materials and methods
Materials
Coal samples, scale samples, and black water/gray water samples were all collected from Huaibei Tanxin Technology Co., Ltd, Anhui Province. Black water and gray water samples were collected at three time points: 16 October, 27 October, and 15 November 2023. Black water was sampled from the mixed wastewater from the cooling chamber and the scrubber tower. Gray water was collected from the gray water tank and was a product from the black water multistage flash steam process. Scale samples were taken from scale deposits in the flash tank. Measurements of water quality conducted within 3 days of sample collection provided a comprehensive assessment.
Digestion and determination of coal samples and scale samples
A coal sample and a scale sample (each 0.2 g) were screened with a 200 mesh sieve, and put into a glass beaker. Then the beaker was added with 15 mL of hydrochloric acid and 5 mL of nitric acid, shaken well, slowly heated, and boiled on an electric heating plate for 20 min. Next, the beaker was taken off and cooled. After 10 mL of perchloric acid was added, the beaker was heated until it started to emit thick white smoke, and then was slowly heated for 15–20 min. The beaker was taken off, and added with 10 mL of a nitric acid solution while it was hot. The beaker was fully stirred to dissolve the salts on the cup wall. After the sample was completely digested, the solution was transferred to a capacity of 100 mL. Then the contents of calcium and magnesium in the sample were determined by atomic absorption spectrometry (Xin et al., 2012).
X-ray diffractometry
A proper amount of samples was screened by a 200-mesh sieve, and tableted by a powder tableting method. The tableted samples were tested on an XRD device under Cu target Ket radiation, smooth tube 40 kV, 15 mA, scanning range 0–90, and scanning rate 8°/min. The XRD patterns of the samples were analyzed using JADE. Determine the major minerals that constitute the composition of coal and scale flakes (Yan et al., 2020).
SEM and EDS
A conductive tape was stuck on the copper sample table. Then a proper amount of samples was screened by a 200-mesh sieve, and evenly stuck on the conductive tape. Next, the excessive samples were blown off, and spray-painted to increase the conductivity of the samples. Then a sample was put into the SEM device and scanned by an electron beam. When the electron beam interacts with the sample, it will excite electrons and X-ray radiation on the surface. The energy characteristics of these radiations were detected and analyzed via EDS. Specifically, an energy spectrometer was used to record the energy spectra of excitation radiation. These energy spectra show the peaks of different elements in the sample, and each peak corresponds to the emission energy of a specific element (Ji et al., 2022).
Proximate analysis of coal
Moisture content of coal: A coal sample was passed through a 200-mesh sieve. Then 10 g of a sample was weighed and put in a glass dish. The total weight was recorded. The temperature of the oven was raised to 105 °C–110 °C. Then the glass dish with the sample was put in the oven and baked for 60 min. After that, the glass dish and the sample were cooled to room temperature before weighing and recording (Zhang et al., 2022).
Ash content of coal: An ash dish was taken out and put into a muffle furnace at 900 °C for combustion until its weight was stable. The organic matter that may remain in the ash dish was removed. After the ash dish was cooled to room temperature, it was weighed and recorded. Then 10 g of a coal sample screened by a 200-mesh sieve was weighed, laid flat on the ash dish, and then put together in a muffle furnace. Next, the muffle furnace was heated to about 500 °C in no < 30 min, and kept for 30 min after reaching the specified temperature. Then the temperature was further raised to about 815 °C, and the sample was burned for 1 h. The gray dishes were taken out of the muffle furnace, put on an asbestos board, and cooled to room temperature. Then they were weighed and recorded as well. The weighed sample was put into a self-sealed bag for storage (Haider et al., 2013).
Volatile matter of coal: A crucible with a cover was put into a muffle furnace at 900 °C for combustion until its weight stabilized, so as to remove the residual substances in the crucible. When the crucible was cooled to room temperature, it was weighed and recorded. Then 1 g of a sample screened by a 200-mesh sieve was weighed and laid flat in the crucible. After the muffle furnace was heated to 920 °C, the crucible was quickly put within 7 min to the muffle furnace for 7 min. Next, the crucible was cooled to room temperature in a clean environment, weighed, and recorded as well (Riley and Marsh, 2022).
Fixed carbon of coal: The difference after deducting water, ash, and volatile matter from 100 was the fixed carbon content of coal.
Coal elemental analysis
The contents of carbon and hydrogen in coal were determined using the methods stipulated in the national standard GB/T476-2008 “Method for Determining Carbon and Hydrogen in Coal.” The content of nitrogen in coal was determined using the method specified in GB/T 19227-2008 “Method for the Determination of Nitrogen in Coal.” To determine the content of the sulfur element in coal using the Echocard method, a coal sample was mixed with the Echocard reagent (a mixture of anhydrous sodium carbonate and magnesium oxide) and scorched at high temperature to convert the sulfur in coal into sulfate. The product of the cauterization was then dissolved, and a barium chloride solution was added to produce a barium sulfate precipitate from the sulfate ions. The sulfur content was calculated based on the mass of the barium sulfate. Due to the complexity of the oxygen element in coal, the indirect method is usually used. In other words, the contents of elements other than oxygen (carbon, hydrogen, nitrogen, sulfur, etc.) in coal are determined first, and then the sum of the mass fractions of these elements is subtracted from 100% to obtain the oxygen content.
Water quality measurement methods
Atomic absorption spectrometry was used to determine the calcium and magnesium contents of black water and gray water. A multi-parameter digital water quality meter was used to determine turbidity, chemical oxygen demand (COD), total organic carbon (TOC), electrical conductivity (EC), salinity (SAL), and total dissolved solids (TDSs). Chemical titration was used to determine the total alkalinity and hardness of water samples.
Preparation of magnetic flocculant
Firstly, Fe3O4 was put in an oven for drying. Then 1 g of Fe3O4 powder was put into a flask, added with 10 mL of 3-aminopropyltriethoxysilane, 20 mL of absolute ethanol, and 20 mL of pure water in turn. The flask was heated in a water bath at 70 °C for 10 h. After that, the products were separated and collected by magnets, washed several times with anhydrous ethanol and pure water, and then dried. A certain amount of acrylamide was weighed into a beaker and dissolved by adding 20 mL of pure water. Then a certain amount of ammonium persulfate was weighed to dissolve it. In a nitrogen atmosphere, the product was stirred in a water bath at 65 °C for 5 min, immediately added into the dissolved acrylamide monomer, stirred for 5 min, and then added with the dissolved ammonium persulfate. After the reaction, the product was collected by a magnet, washed with anhydrous ethanol and pure water several times, and dried at 65 °C for 1 h. Finally, a magnetic flocculant with a core–shell structure was prepared (Wang et al., 2018b).
Results and discussion
Proximate analysis of coal
Table 1 shows the results of the proximate analysis of the coal samples. The contents of moisture, volatile matter, fixed carbon, and ash in coal are determined via industrial analysis. The moisture in coal mainly includes crystal water and free water. Crystal water is generally not determined in the industrial analysis, because it is closely combined with coal and cannot be easily separated. Free water in coal can be further divided into external moisture and internal moisture. External moisture refers to the moisture attached to the coal surface and coal cracks during coal mining, transportation, and storage, but is unstable and easily affected by the environment. Intrinsic moisture refers to the moisture absorbed or condensed in the pores of coal particles and will be lost when heated to a certain temperature (Arisoy et al., 2017). Moisture absorbs heat when coal is burned. Additional heat may be consumed for moisture evaporation during gasification and may lead to problems such as coal deterioration and caking during transportation and storage, affecting coal usability and economy. Overall, No. 4 and No. 6 coals contain lower intrinsic moisture and have an advantage in this regard.
Results of coal proximate analysis.
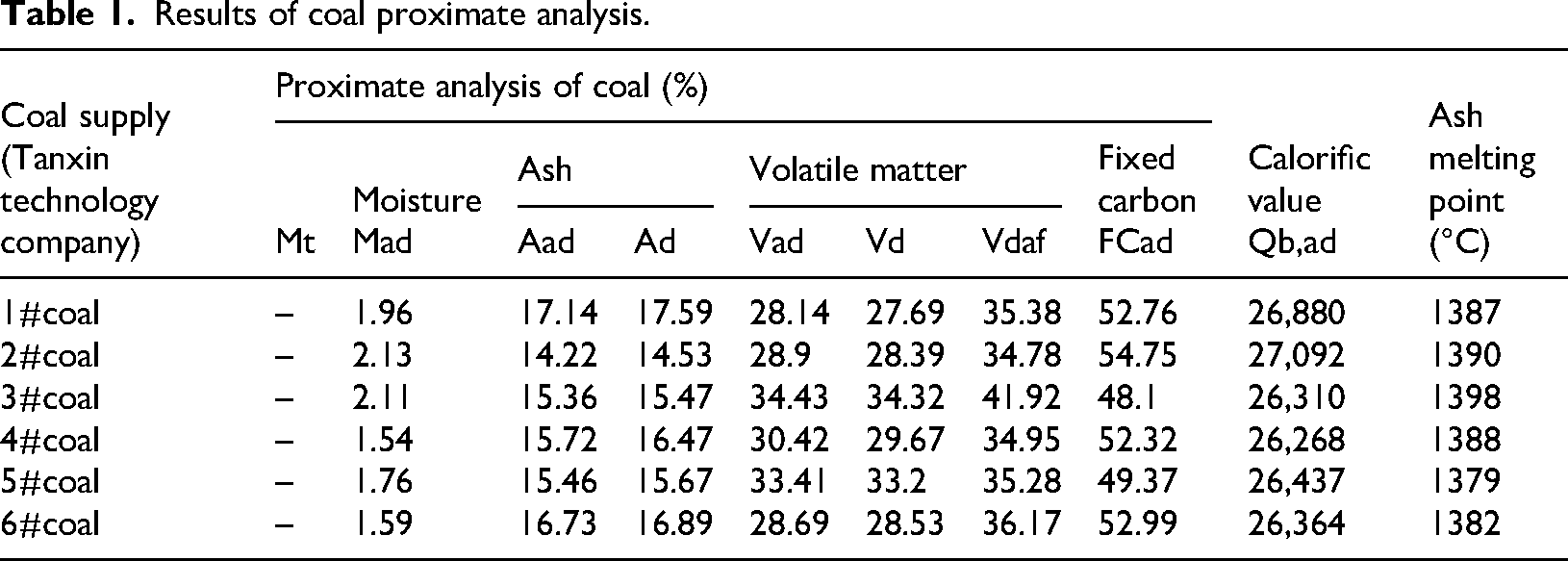
Volatile matter is a measure of the gaseous and liquid products from coal pyrolysis. Coal with a higher volatile matter content can be more easily ignited and burnt completely during combustion and has a longer flame, but may also generate more pollutants. Higher volatile matter in coal gasification facilitates rapid coal ignition and reaction in the gasification process. No. 3 coal has the highest volatile matter and may be more active in the initial stage of the gasification reaction, making it more easily react with the gasifier. Fixed carbon is a non-volatile solid combustible in coal. A higher content of fixed carbon means a higher calorific value and a longer combustion duration. Fixed carbon is one important index to measure the quality of coal. A higher fixed carbon content in coal gasification indicates more energy can be released from the coal in the gasification, which can improve the production and quality of gas. No. 2 coal has the highest fixed carbon content, meaning it can produce more heat and gas in the gasification (Su et al., 2016).
The ash in coal refers to the residues left after the complete combustion of coal and is mainly composed of oxides, silicates, and aluminates. Ash in coal can also be hardly utilized in coal gasification. In coal gasification, ash in coal will become liquid slag under high temperatures and high pressure. Then the liquid slag together with hot synthesis gas will enter the quenching chamber of the gasifier to mix with quenching water. The liquid molten slag will quickly become solid slag to be removed by slag removal, and the quenching water will become black water and go to the black water treatment system (Chai et al., 2023). Due to contact with liquid slag and hot synthesis gas, black water will contain abundant calcium and magnesium ions, and the temperature is very high. After that, the temperature of black water in the black water treatment system will gradually decrease after passing through the flash tank. During this process, calcium and magnesium ions in black water will react to form precipitates, which will attach to the black water system reactor. Eventually, the black water system reactor will scale. From the perspective of coal gasification, the main component of scale in the black water system comes from ash in coal, which is the most important factor affecting the scale in the black water system. Therefore, to reduce the fouling of the black water system, we can reduce the ash content in coal. Enterprises can use low-ash coal or pretreated coal to reduce the ash content in coal, which may increase the operating cost of enterprises (Cao et al., 2023; Kobayashi et al., 2015). No. 2 coal has the lowest ash content, indicating that No. 2 coal is relatively more favorable for coal gasification. This sample can reduce the fouling tendency of the system, and decrease the amount of ash handling, wear and tear on the gasification equipment.
The calorific value directly reflects the amount of energy that can be released during coal combustion or gasification. The calorific value of No. 2 coal is the highest among all the coal samples. Hence, No. 2 coal can provide more energy for the gasification reaction during gasification, which is conducive to the gasification reaction. It also can increase the gasification efficiency and the output of coal gas. The higher ash melting point can avoid premature slagging in the gasifier, which affects the normal progress of gasification. The ash melting points of the coal samples are all between 1379 °C–1398 °C, without significant difference. Comparatively, No. 3 coal has the highest ash melting point (1398 °C) and thus may have some advantages in preventing slagging. On the whole, each coal has its own characteristics and advantages in coal gasification. No. 2 coal has excellent performance in terms of lower ash, higher fixed carbon content, and highest heat generation. No. 3 coal has some advantages in terms of volatile matter and ash melting point. No. 2 and No. 3 coals are more suitable for coal gasification.
Coal elemental analysis
Coal is mainly composed of inorganic substances and organic matter. Inorganic substances include minerals and water, and ash mainly comes from minerals in coal. Organic matter is mainly composed of carbon, hydrogen, oxygen, nitrogen, sulfur, and other elements. Organic matter is the main component of coal, and the sum of carbon, hydrogen, and oxygen accounts for more than 95% of organic matter. The content of each element in coal can indicate the quality of coal to a certain extent. The higher contents of carbon, hydrogen, and oxygen in coal gasification mean it is more favorable for coal gasification, because this process is to gasify coal into a mixture rich in carbon monoxide and hydrogen in an oxygen atmosphere (Ujala et al., 2023). In coal gasification, sulfur in coal may be oxidized into sulfur dioxide to escape into the air, and finally, form acid rain through a series of physical and chemical reactions, which will affect human production and life. Therefore, a lower sulfur element in coal means the quality of coal is better. The mineral content in coal also affects coal gasification. In coal gasification, the minerals in coal cannot be gasified and utilized, and will eventually become residues and be discharged. The process of turning minerals in coal into residues will cause many problems, such as excessive calcium and magnesium ions in black water, which will lead to fouling in the black water system and affect the efficiency of coal gasification.
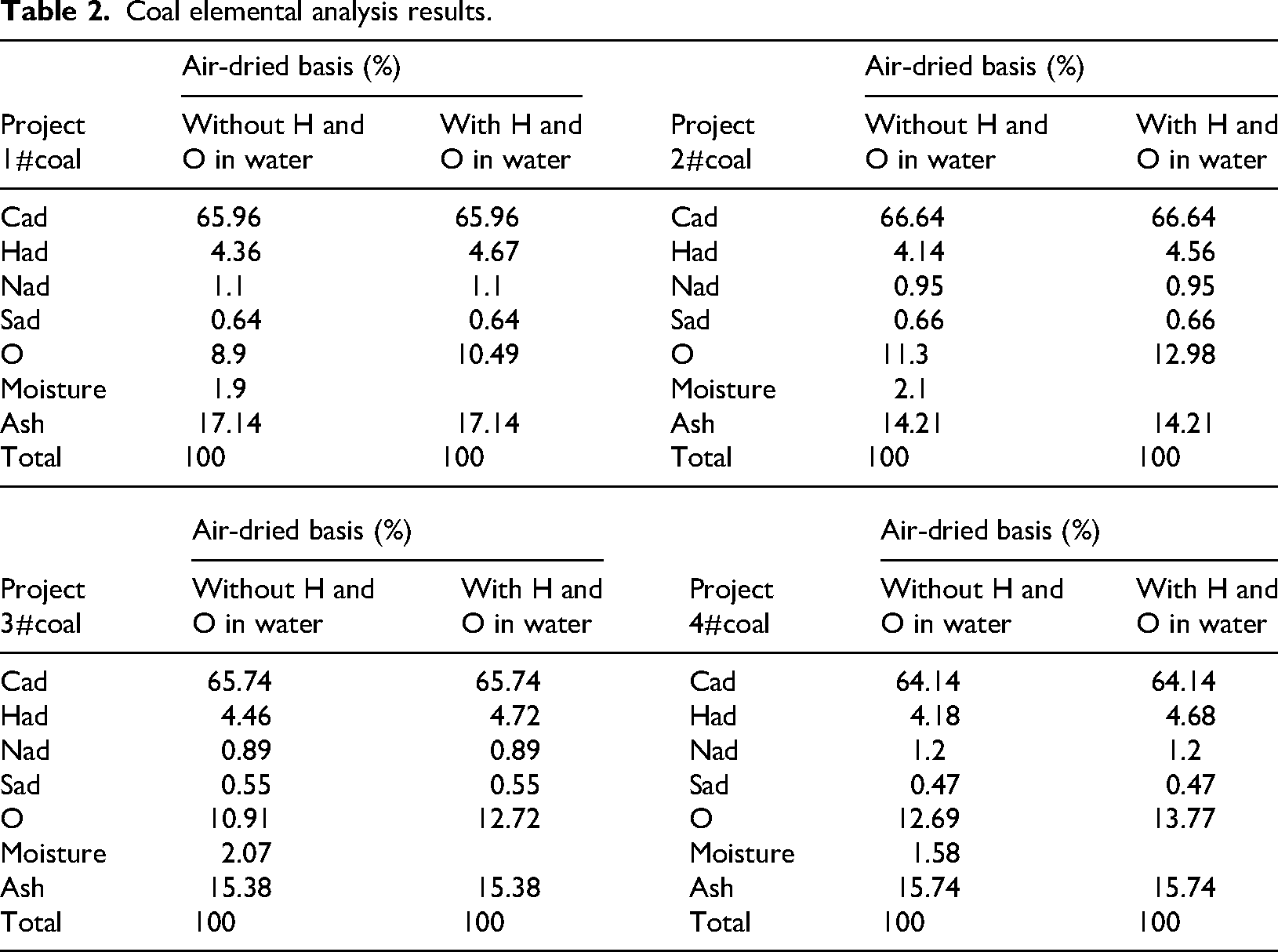
Table 2 shows the results of elemental analysis of coal samples. The carbon contents of the four coal samples were close to each other, ranging from 64.14% to 66.64%. The carbon content is the highest at 66.64% in No. 2 coal and is relatively low at 64.14% in No. 4 coal. Carbon is the main element that produces heat when coal is burned, and a higher carbon content usually means a higher potential for heat generation. Hydrogen content fluctuates from 4.14% to 4.72%. No. 3 coal has the highest hydrogen content of 4.72% when hydrogen and oxygen are included in the water. No. 2 coal has a relatively low hydrogen content of 4.14% when hydrogen and oxygen are not included in the water. Coal with high hydrogen content may produce more heat during combustion, and the combination of hydrogen and oxygen to form water may also affect processes such as combustion products and heat transfer. Differential oxygen reduction data show that the oxygen content ranges from 8.9% to 13.77%. Coal #4 has the highest oxygen content of 13.77% when it contains hydrogen and oxygen in water. Coal #1 has the lowest oxygen content of 8.9% when it does not contain hydrogen and oxygen in water. Excessive oxygen content reduces the heat content of the coal because oxygen does not produce heat and combines with other combustible elements to reduce the proportion of combustible components. Nitrogen content ranges from 0.89% to 1.2%. No. 3 coal has a minimum nitrogen content of 0.89% and No. 4 coal has a maximum of 1.2%. Nitrogen may generate pollutants such as nitrogen oxides during combustion, so a relatively low nitrogen content is better for the environment. Sulfur content ranges from 0.47% to 0.66%, maximizing to 0.66% in No. 2 coal and minimizing to 0.47% in No. 4 coal. Sulfur combustion produces harmful gases (e.g. sulfur dioxide), which are a major factor in causing environmental problems such as acid rain. In addition, elemental sulfur may form sulfate ions after gasification, which combine with metal ions such as calcium and magnesium in black water to generate insoluble salts such as calcium sulfate and magnesium sulfate, which is one of the important factors leading to fouling of black water system. So a lower sulfur content is better (Dai et al., 2020; Liang et al., 2021).
Coal elemental analysis results.
XRD
XRD is a non-destructive technology widely used in coal quality evaluation. The principle is that when X-rays pass through coal, each coal type has a unique structure and chemical composition and a unique diffraction form. These characteristics can be observed by the corresponding distance d between diffraction surfaces and the relative intensity of diffraction lines. The mineral composition of coal can be obtained by analyzing the coal structure with XRD. XRD can also be used to analyze the scale structure and thus the mineral composition of the scale (Yang et al., 2021).
Figure 1 shows the XRD diagrams of four types of coal samples and scales. The coal samples are mainly composed of kaolinite, quartz, calcite, and muscovite, and some coal samples also contain albite and pyrite. The scales are mainly composed of calcite, gypsum, and dolomite.
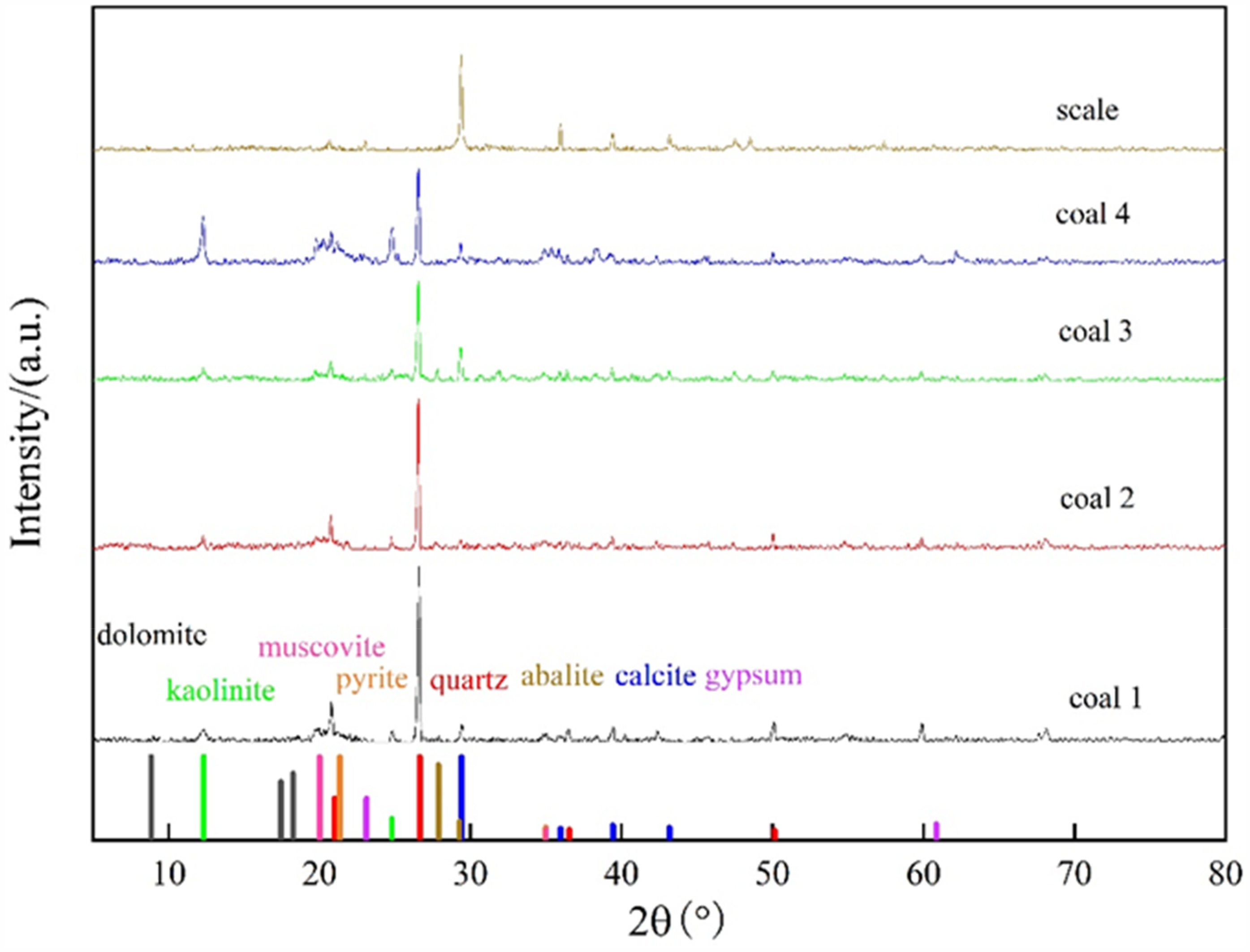
X-ray diffraction (XRD) results of coal and scale samples.
Figure 2(a) and (b) shows the weight ratio of each mineral in coal and in scale, respectively. Combined with Figure 2 (XRD quantitative analysis diagram), the content of mineral quartz in coal accounts for the highest proportion, with a weight ratio of 43.3%. Quartz is the most widely distributed mineral in the quartz group of minerals and is mainly composed of SiO2. Moreover, the weight ratio of kaolinite is 37.3%. Kaolinite mainly results from natural alteration of feldspar and other silicate minerals and is a water-containing aluminosilicate. Kaolinite is dense or loose, generally white, and is chemically composed of Al4[Si4O10](OH)8. The oxide form of kaolinite is 2Al2O3·4SiO2.4H2O, and the theoretical composition (wB%) is 39.5% Al2O3, 46.5% SiO2, and 14.0% H2O. There is also muscovite, with a weight ratio of 16.0%. Muscovite belongs to silicate, and its chemical formula is KAl2(AlSi3O10)(OH)2, including 45.2% SiO2, 38.5% Al2O3, 11.8% K2O, and 4.5% H2O. In addition, it contains a small amount of Na, Ca, Mg, Ti, Cr, Mn, etc. Finally, the coal samples also contain a small amount of calcite (3.3%). Calcite is a calcium carbonate mineral, and its chemical composition is mainly CaCO3. In addition, there is a small amount of other minerals in coal (about 0.1% by weight) (Jiang et al., 2021).
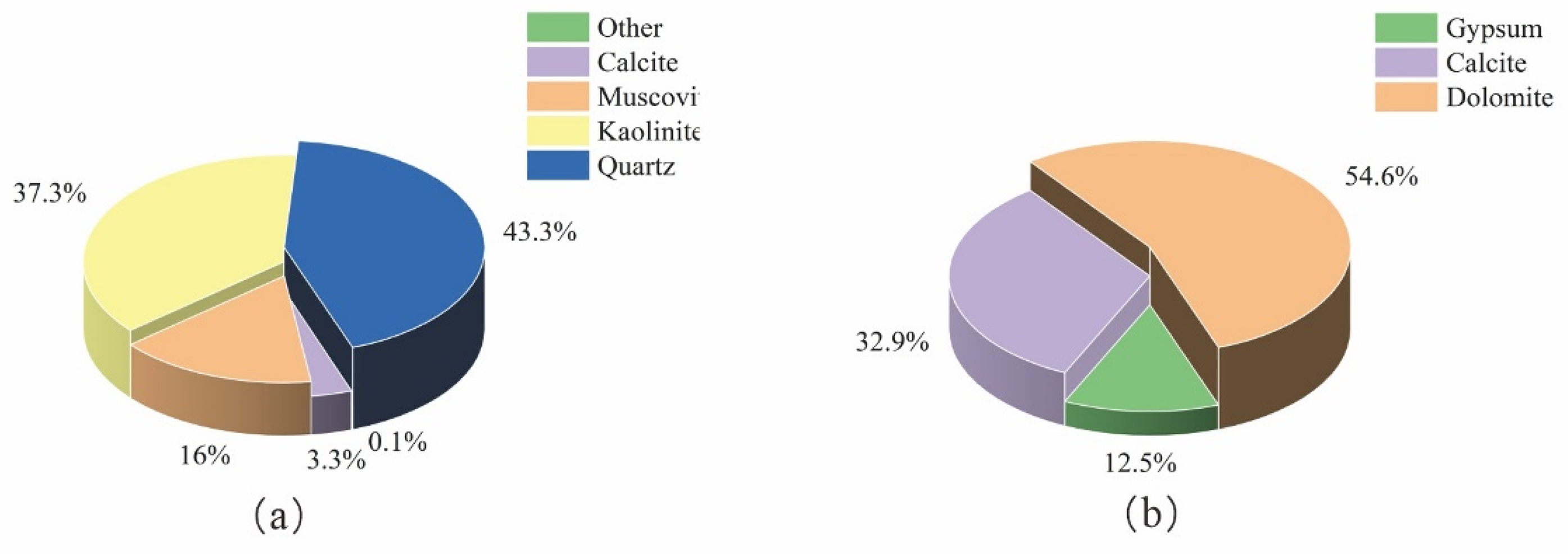
(a and b) Quantitative analysis results of coal and scale samples.
Dolomite accounts for the highest proportion on the scale, with a weight ratio of 54.6%. The chemical composition of dolomite is a compound of calcium carbonate and magnesium carbonate, and its molecular formula is CaCO3·MgCO3, or CaMg(CO3)2. Dolomite is mainly composed of calcium carbonate and magnesium carbonate (CaCO3/MgCO3 ratio ∼ 1:1). The theoretical composition is 30.41% CaO, 21.87% MgO, and 47.72% CO2, and the CaO/MgO ratio is 1.39. The weight ratio of calcite is 32.9%. Calcite is a calcium carbonate mineral, with the chemical composition of CaCO3. The chemical composition is 56.03% CaO and 43.97% CO2 as well as Mn and Fe. Gypsum accounts for the smallest weight ratio of 12.5%. Gypsum is mainly composed of calcium sulfate dihydrate (CaSO4·2H2O) and contains a small amount of silicic acid, aluminum hydroxide, sulfide, and trace amounts (e.g. iron and magnesium). The theoretical components are 32.6% CaO, 46.5% SO3, and 20.9% H2O.
Combining Figures 1 and 2, we conclude that the mineral composition of the scale flakes (mainly calcite and gypsum) is very different from the coal samples (mainly kaolinite and quartz). This difference indicates that the formation of fouling does not simply result from ash accumulation in the coal, but involves complex physicochemical changes. Raw coal contains a variety of minerals. Under the high temperature and pressure environment of coal gasification, most of the minerals will become ash and be removed from the coal gasification system. However, some minerals will undergo physico-chemical reactions, and dissolve and enter the coal gasification system. Afterward, the minerals enter the black water and follow the black water into the black water flash system. In this system, the temperature and pressure drop rapidly, which causes some salts in the black water to re-precipitate. For example, calcium carbonate dissolves at high temperatures and precipitates at low temperatures. As the gasification process proceeds, the aqueous concentrations of various ions increase. When the concentrations of these ions exceed their saturated solubility in water, precipitation begins. For example, calcium ions combine with carbonate ions and sulfate ions to form insoluble salts, such as calcium carbonate and calcium sulfate (Suyatno et al., 2024).
SEM and EDS
SEM–EDS, a combination of SEM and EDS, is often used to analyze the surface morphology and elemental composition of coal. This combined analysis method provides details about the microstructure and element distribution of coal and is significant for studying coal combustion and gasification characteristics (Kwiecińska et al., 2019).
Porosity in coal is a very important factor in coal gasification. The presence of more pores in coal is beneficial for coal gasification because more pores are beneficial for full contact between oxygen and coal samples and for complete reaction (Xu et al., 2023). Figure 3(a) shows the SEM image of sample No. 1. Sample No. 1 has an uneven surface and consists of many fine grains that are tightly combined together (Figure 3(a)). This sample has many surface pores and presents a porous structure, indicating it is loose. Sample No. 1 has a large number of loose pores on the surface, which are conducive to coal gasification. Figure 3(b) shows the SEM image of sample No. 2. Clearly, the surface of sample No. 2 is relatively flat, and mainly consists of large lumps and small lumps of coal. The lumps are not tightly intercombined but are simply stacked on top of each other. The gaps between the lumps of coal are more and larger, and the whole coal sample has more pore spaces. Sample No. 2 has more pores, which are good for coal gasification. However, sample No. 2 has many large lumps of coal, which are not good for coal gasification. Figure 3(c) shows the SEM image of sample No. 3. The structure of sample No. 3 is basically similar to that of sample No. 2, and also consists of many large lumps and small lumps. Nevertheless, there are more large lumps and fewer small lumps in sample No. 3 compared with sample No. 2. Therefore, sample No. 3 coal is not conducive to coal gasification compared with sample No. 2 (Gonzalez et al., 2022).

(a) to (f) Scanning electron microscopy (SEM) results of coal and scale samples.
Figure 3(d) shows the SEM image of sample No. 4. The surface of sample No. 4 is smooth and consists of many laminar structures that are simply stacked on top of each other. Moreover, sample No. 4 is more porous and has a larger specific surface area, which are favorable for coal gasification. Figure 3(e) and (f) shows the SEM images of the scale sheets. The structure of the scale sheets is similar to that of coal sample No. 1, as the surface is rough and uneven with many fine particles. The inside of the scale sheets has many pores and crevices, and the whole scale sheet is a loose and porous structure. The reason is that the main components of the flake are CaCO3 and MgCO3, which are both loose and porous, leading to a loose and porous structure of the flake (Zhou et al., 2015).
Analysis of the SEM images of scale flakes indicates scale flakes are mainly composed of fine particles. This result suggests that the coal gasification process produces many tiny sediment particles that will constantly collide with each other in the water. When a collision happens, the tiny particles will coalesce through physical or chemical action to form larger particles. The original suspended solids, ash particles, and residual flocculants in the black water may also coalesce or flocculate these particles together. As the particles continue to grow, they under gravity will gradually settle to the inner wall surface of the pipeline, and equipment. At the same time, due to the solid-liquid interface with a dipole layer, coal gasification wastewater with positively charged mineral solute ions (e.g. Ca2+ and Mg2+) will be attracted to the negatively charged surface of the wall of the device under the electrochemical field formed by the potential difference of the dipole layer. These changes accelerate particle deposition on the wall surface, and these deposits of particles accumulate, and gradually foul (Jiao et al., 2024).
Figure 4(a) shows the EDS result of sample No. 1. Clearly, the elemental distribution of sample No. 1 is dominated by C and O, with a certain amount of Al and Si and a small amount of K, Ca, and Fe. This result indicates sample No. 1 contains a certain amount of kaolinite minerals (aluminum silicate minerals marked by Al) and calcite minerals (carbonate minerals marked by Ca), which further validates the results of XRD. Figure 4(b) shows the EDS result of sample No. 2. The elemental composition of sample No. 2 is also dominated by C and O, with a certain amount of N, Al, and Si, and a small amount of K, Ca, Fe, and Mg (Rompalski et al., 2019). Unlike sample No. 1, sample No. 2 has more N and Mg, which may be because sample No. 2 contains a certain amount of Mg-containing dolomite minerals. Figure 4(c) shows the EDS result of sample No. 3. Sample No. 3 contains up to 90% C and O, and a certain amount of Al, Si, Ca, and a small amount of K, Fe, Mg, and S. Compared with the previous two coal samples, sample No. 3 has more S, which is a harmful element in coal gasification. In coal gasification, S in coal will finally enter the atmosphere and cause pollution to the environment. Figure 4(d) shows the EDS result of sample No. 4. Like the previous coal sample, sample No. 4 is also dominated by C and O. However, compared with the other coal samples, sample No. 4 contains very few other elements, and only contains a certain amount of Al, Si, N, and a small amount of S, but no Ca, Mg, Fe, or K. This result indicates the mineral species contained in sample No. 4 are fewer mineral species and mainly kaolinite minerals (Hower et al., 2022; Kingdom et al., 2017).
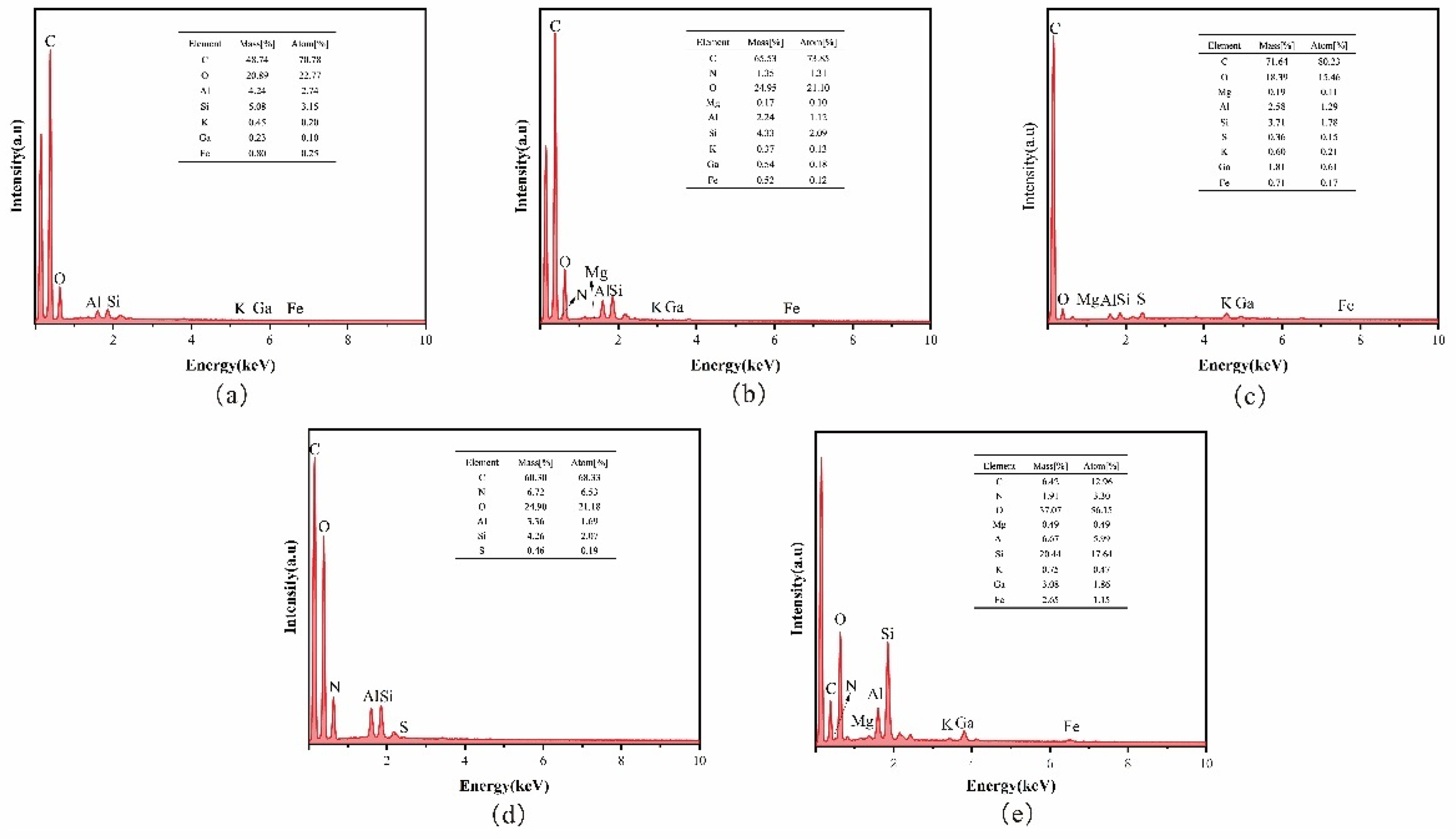
(a) to (e) Plot of energy spectrum analysis results of coal and scale samples.
Figure 4(e) shows the EDS result of the scale flake. The scale flake is mainly composed of C, O, and Si, and a certain amount of Ca, Al, Fe, N, and a small amount of K and Mg. This result indicates the scale flake is mainly composed of dolomite and gypsum, and a certain amount of Al and Si, suggesting the flake may also contain kaolinite minerals (kaolinite is an aluminosilicate mineral). Various elements in the scale flakes are found in coal, which can further verify that the fouling in the coal gasification system comes from coal and that the ash content in coal is an important influence on the fouling of the coal gasification system (Karami et al., 2021).
Gasification system process
Figure 5 is a process flow diagram of the coal gasification system. The coal gasification process is mainly divided into three parts: a gasifier gasification system, a syngas preliminary purification system, and a black water treatment system. The gasifier gasification system includes a gasifier, a cooling chamber, a slag breaker, a lock hopper, and a slag pool. The syngas preliminary purification system includes a venturi scrubber and a scrubber tower. The black water treatment system includes a medium-pressure flash evaporation tank, a low-pressure flash evaporation tank, a vacuum flash evaporation tank, a cooler, a separator tank, a settling tank, and a gray water pool (Singh et al., 2014).
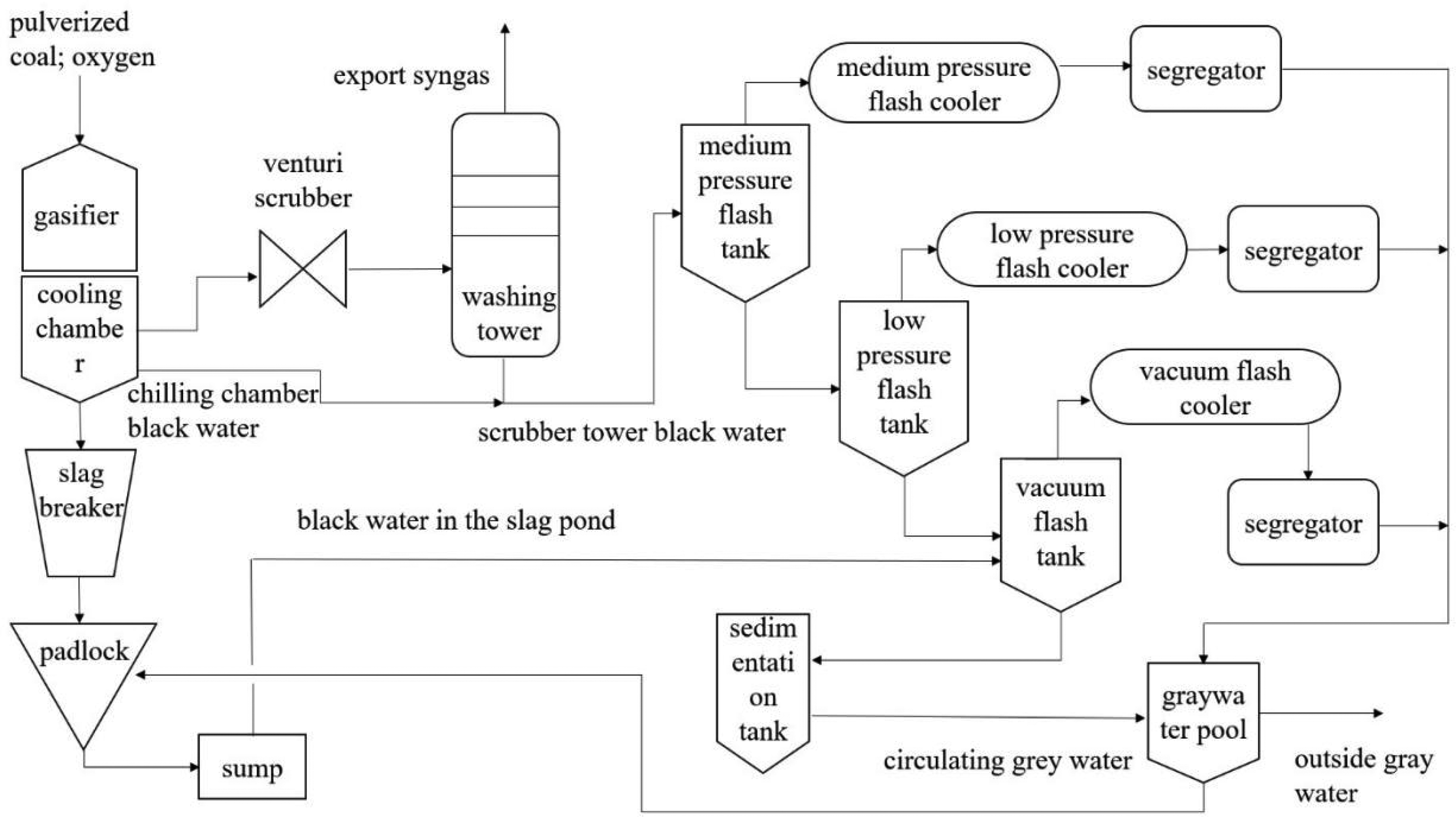
Flow diagram of gasification system process.
Firstly, through the dense-phase pneumatic conveying system, the pulverized coal will be pressurized into a fluidized state. Then the fluidized pulverized coal will be transported through the pipeline to the reaction chamber of the gasifier and will be gasified under the high temperature and high pressure conditions in the reaction chamber of the gasifier. Oxygen needs to be added when the pulverized coal is gasified, and the amount of oxygen added for the complete gasification of pulverized coal is much larger than the amount of oxygen needed for pulverized coal gasification. Thus, the pulverized coal undergoes a partial oxidation reaction in the reaction chamber to produce syngas rich in H2 and CO (Wang et al., 2024).
During the reaction process, the ash in the coal will become liquid slag, and other components in the coal will become hot syngas. The resulting hot syngas and liquid slag will leave the gasifier reaction chamber through the drop tube into the gasifier cooling chamber. The hot syngas and liquid slag will be mixed with the gasifier cooling chamber in the cooling water reaction. Specifically, the liquid slag will be rapidly solidified into solid slag, and most of the coarse slag first goes to the slag breaker in the downstream of the cooling chamber, then to the lock hopper, and finally to the slag pool. A small part of the fine ash will go with the syngas to the downstream process. The hot syngas will pass through the gasifier cooling chamber, and the cooling water will undergo heat exchange. The temperature of the hot syngas will decrease while the temperature of the cooling water will increase (Zhan et al., 2022). When the cooling water is mixed with the liquid slag and the hot syngas, the liquid slag will form some fine slag, which cannot be filtered and will remain in the cooling water. At the same time, some ions such as calcium and magnesium in the liquid slag and hot syngas will also enter the chilled water. Eventually, the chilled water will become a high-pressure, high-temperature, high-hardness, high-alkalinity, and high-suspended matter content of black water (He et al., 2022).
The crude syngas enters the venturi scrubber and is atomized, scrubbed, dusted, and wetted in the venturi throat. The dusted and wetted crude syngas enters the scrubber tower. In the scrubber tower, the crude syngas is further scrubbed and the fine ash in the crude syngas remains in the scrubber water. The syngas goes out through the outlet at the top of the scrubber tower, and the scrubber water is scrubbed into black water, which is finally sent to the black water treatment system together with the black water from the cooling chamber (Li et al., 2022).
Black water first goes into the medium-pressure flash tank for flash evaporation, and then flashes out of steam and non-condensable gas into the medium-pressure flash cooler. The cooled gas enters the separation tank, and the acidic gas is separated to the recycling device, while the condensate enters the gray water pool. After the first level of flash evaporation, the black water enters the low-pressure flash tank for the second level of flash evaporation. The gas is flashed out into the low-pressure flash condenser for cooling. The cooled gas enters the separation tank for separation. Acidic gases are separated and go to the recovery device, while the condensate goes to the gray water pool. The secondary flash black water and slag pool black water enter the vacuum flash tank for vacuum flash evaporation. The gas is flashed out into the vacuum flash condenser for cooling. The cooled gas enters the separation tank. Acidic gases are separated and go to the recycling device, while the condensate goes to the gray water pool. The black water after flash vaporization enters the settling tank for flocculation and settling, and the settled black water enters the gray water tank. Most of the gray water in the gray pool is recycled, and a small portion is sent as wastewater to the wastewater treatment unit for further treatment (Wang et al., 2021).
Water quality analysis of black water and gray water
Black water is discharged from the gasifier, the washing tower, and the slag pond, while gray water is the water formed after multi-stage flashing, flocculation, and clarification of black water. Most of the gray water is then sent to the gasification unit for recycling through the low-pressure gray water pump, and a small portion of the gray water is sent as wastewater to the wastewater treatment unit for further treatment.
The tables below show the measurements of black water quality and gray water quality. In conclusion, the black water has a total hardness of ∼ 1100 mg/L, calcium hardness of ∼ 400 mg/L, and magnesium hardness of ∼ 30 mg/L. This result indicates the hardness of black water mainly comes from calcium and magnesium ions. The high hardness makes calcium and magnesium ions easily form insoluble salts in the coal gasification system, increasing the possibility of fouling. The hardness of gray water (∼ 760 mg/L) is lower than that of black water, and the theoretical risk of fouling is smaller. The COD and TOC of black water and gray water are not high and change very slightly. COD and TOC mainly reflect the organic matter content in the water, and a lower organic matter content generally has a small direct effect on the fouling. However, the organic matter and metal ions form a complex, which will indirectly affect the fouling. The total alkalinity of black water and gray water is higher, indicating the water contains more carbonate, bicarbonate, and other alkaline ions. These materials coexisting with calcium and magnesium ions will promote the formation of calcium carbonate, magnesium carbonate, and other precipitates, increasing fouling tendency.
The TDS content of black water is 3160–3360 ppm, and the EC content is 6.42–6.78 Ms/cm. Both TDS and EC content are higher, indicating more dissolved salts exist in the black water. A large amount of salts may precipitate out due to over-saturation during the system operation, which in turn promotes fouling. Compared with black water, gray water has a TDS content of 2303–2610 ppm and an EC content of 4.84–5.16 Ms/cm. The lower TDS and EC content indicate it contains fewer salts, and the potential risk of fouling is lower than that of black water. The turbidity of the black water is 106.6–111.2 NTU, indicating certain amounts of suspended particles and other substances exist in the water. These suspended particles may become the core of fouling, and provide attachment points for scale formation, accelerating the fouling process. The gray water has lower turbidity (45.7–55.2 NTU) than that of black water and contains relatively few suspended particles, indicating it may have a certain but weaker effect on fouling (Cui et al., 2024).
The black water is mainly characterized by high hardness, alkalinity, and turbidity, which is because coal contains many minerals, such as calcium carbonate and magnesium carbonate. During the gasification, high temperature and chemical reactions cause these minerals to dissolve and enter the black water in the form of ions. As a result, the concentrations of calcium and magnesium ions, and the hardness of the black water all increase. In addition, coal contains alkaline minerals such as calcium oxide and magnesium oxide. In the gasification, they will react with water to generate corresponding hydroxides, which will ionize abundant hydroxide ions, thus increasing the alkalinity of black water. The turbidity of black water is high because numerous solid particles will be generated in coal gasification, including incompletely reacted cinders, and ash. These particles will enter the black water along with the reacted products, making the black water become turbid. Compared with black water, the turbidity and total hardness of gray water are significantly reduced. This is because gray water is formed from the multi-stage flash, flocculation, and clarification of black water. In the black water flash flocculation, calcium, and magnesium ions and suspended solids are removed, and the turbidity and total hardness of gray water are significantly reduced (Xu et al., 2020; Yin et al., 2023).
Analysis of the causes of scaling in coal gasification black water and gray water systems
Through XRD, SEM, and energy dispersal analysis of coal samples and scale samples, industrial analysis, and elemental analysis of coal samples, the process flow of coal gasification system operation, black water, and gray water quality. In conclusion, the main influencing factors affecting the fouling of coal gasification in black water and gray water systems are coal quality, black water, and gray water quality, temperature, and pressure during system operation.
Coal quality is an important factor affecting the fouling of coal gasification black water and gray water systems. The scale samples are mainly composed of CaCO3 and MgCO3, and a certain amount of CaSO4, which comes from the ash in coal. The main components of coal ash are CaO, MgO, SiO2, Al2O3, and Fe3O4, which generally account for more than 95% of the total ash. In the coal gasification system operation, ash is difficult to be gasified, and most of the ash will become residue. Large particles of residue will be removed, while some small particles of residue are difficult to be removed. With the operation of the system in the various equipment, the small particles in the appropriate conditions will be continuously attached to the equipment to form fouling (Deng et al., 2025).
The black water is mainly generated in the cooling chamber, while forms gray water after multi-stage flashing, flocculation, and clarification. Most of the gray water is then sent to the gasification unit for recycling through the low-pressure gray water pump, while only a small portion of the gray water is sent to the wastewater treatment unit as wastewater for further treatment. Tables 3 and 4 show the results of black water and gray water quality measurements, respectively. In conclusion, the total hardness, total alkalinity, turbidity, dissolved solids, and other indicators of gray water compared to black water are reduced, but the contents are still high. The black water will experience many temperature and pressure changes in the flash tank, and most of the gray water used as recycled water in the coal gasification system will also experience temperature and pressure changes. With the continuous change of temperature and pressure in the coal gasification system, a large number of calcium and magnesium ions in the black water and gray water will combine with CO32− and OH− in the surrounding environment to form crystals, which will then precipitate and attach to the equipment and pipelines to form scale.
Black water quality measurements.
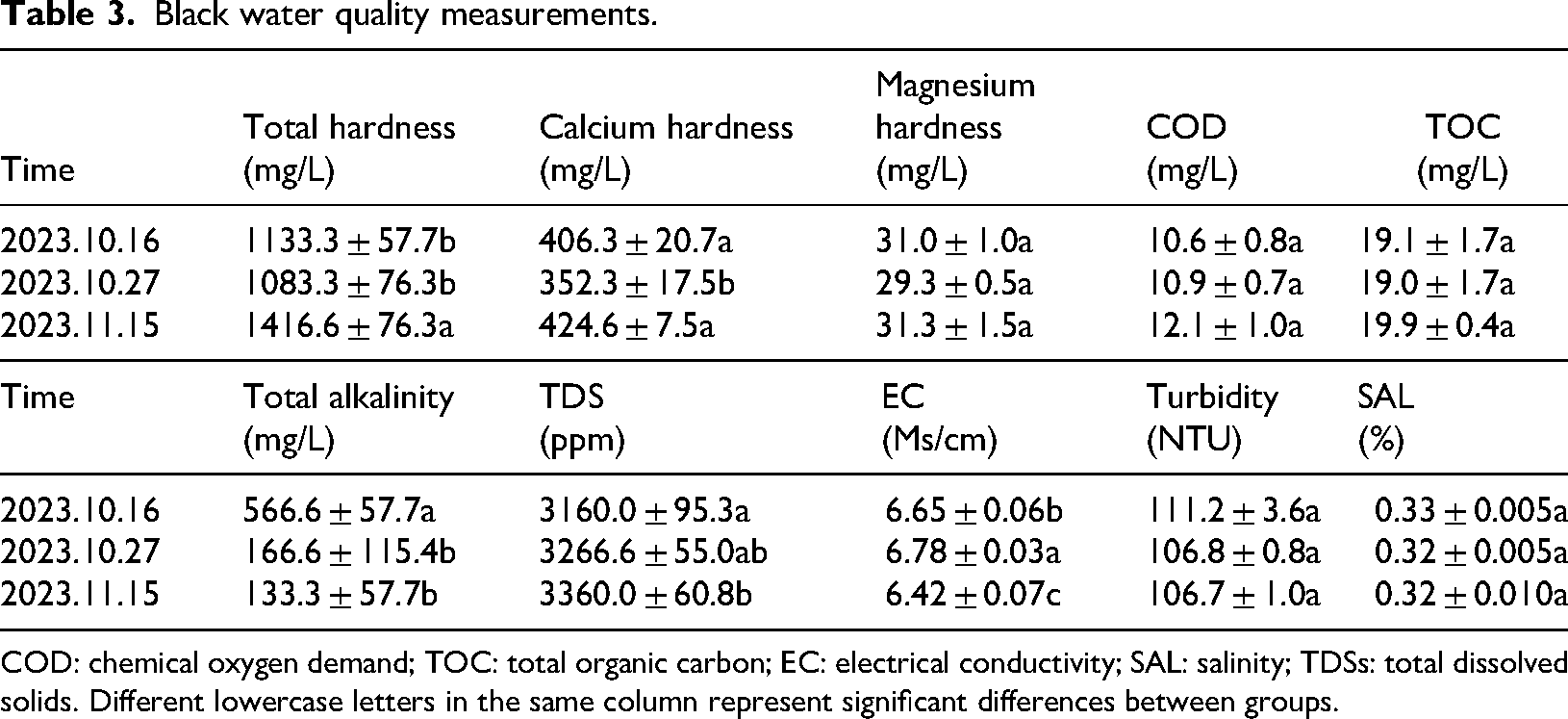
COD: chemical oxygen demand; TOC: total organic carbon; EC: electrical conductivity; SAL: salinity; TDSs: total dissolved solids. Different lowercase letters in the same column represent significant differences between groups.
Gray water quality measurements.
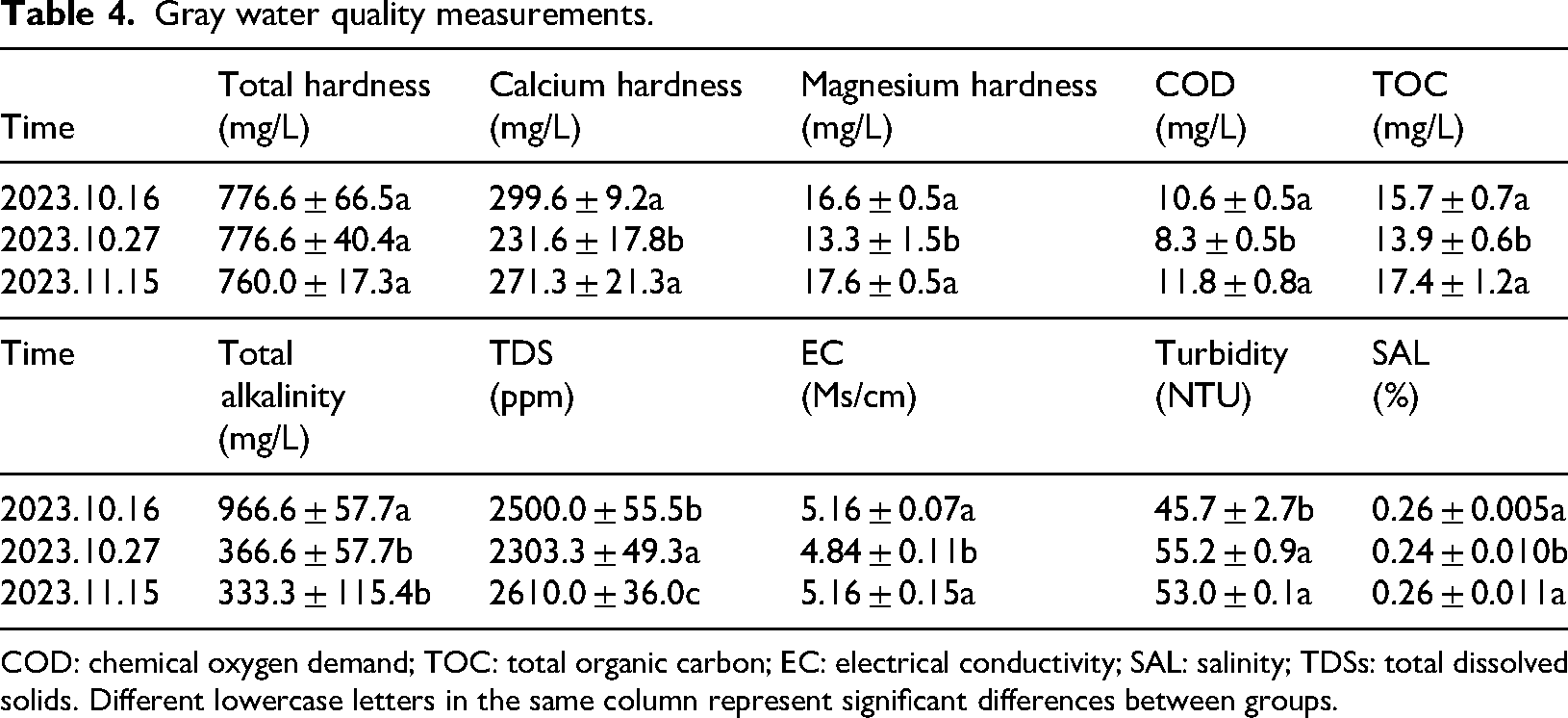
COD: chemical oxygen demand; TOC: total organic carbon; EC: electrical conductivity; SAL: salinity; TDSs: total dissolved solids. Different lowercase letters in the same column represent significant differences between groups.
Temperature and pressure are also the main influencing factors of coal gasification system fouling, and the temperature and pressure changes are particularly large during the operation of a coal gasification system. The temperature of gray water in the upper part of the cooling chamber in contact with syngas can reach up to about 300 °C, and the temperature of gray water after flashing drops to about 50 °C. The recycled gray water is flashed and heat-exchanged to about 150 °C, and the temperature of the water varies greatly between 300 °C and 50 °C, which will increase the repeated fouling of the system. The pressure of the cooling water in the cooling room and of the washing water in the carbon washing tower is basically equal to the gasifier operating pressure. The pressure is gradually reduced by the step-by-step flash evaporation, and becomes atmospheric when it finally enters the settling tank. The gray water is pressurized again to return to the system for recycling, and the probability of system fouling is also obviously increased when the pressure amplitude is changed greatly and the pressure is gradually reduced in the process of the black water and gray water treatment (Sharma et al., 2024; Zhao et al., 2024).
Flocculation and sedimentation experiments with magnetic flocculants
A new type of magnetic flocculant was prepared from non-toxic and environmentally friendly ferric oxide combined with PAM, which is commonly used in the industry, to overcome their respective shortcomings and apply them to black water. Shenning furnace gasification black water was taken on-site, and added with a magnetic flocculant. The dosage of the magnetic flocculant was adjusted to investigate the flocculation effect.
Figure 6 shows a graph of the experimental results of the magnetic flocculant. The turbidity of the black water was highest at 69 NTU when the amount of magnetic flocculant was 0, but decreased as the amount of magnetic flocculant increased. The turbidity of black water minimized to 22 NTU when the dosage of magnetic flocculant was 2 × 10−6, but increased to 38 NTU when the dosage of magnetic flocculant further rose to 3 × 10−6. These results indicate the turbidity of the black water can be reduced by increasing the dosage of magnetic flocculant, but may increase when the dosage is above a certain amount.
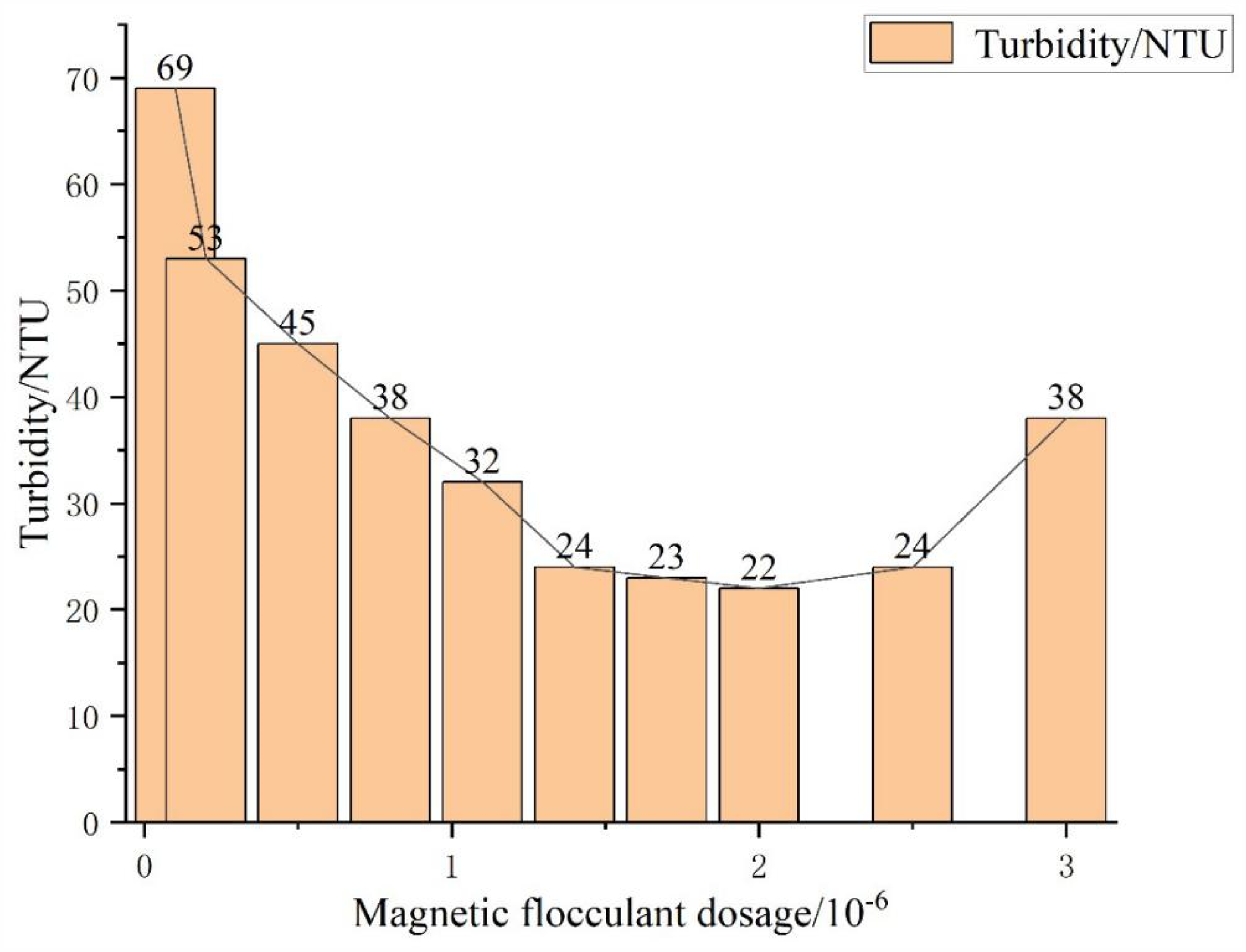
Experimental results of flocculation and sedimentation.
In conclusion, with the gradual increase of magnetic flocculant dosage, the turbidity of coal gasification black water gradually decreases. However, when the turbidity of black water decreases to a certain extent with a further increase in the magnetic flocculant, the turbidity of black water rises. The possible reason is that when the dosage of the magnetic flocculant exceeds a certain range, the spatial potential resistance in the solution increases. As a result, the macromolecules on the surface of the magnetic flocculant cannot spread out completely in the solution, which in turn affects its bridging adsorption and worsens the flocculation effect (Zhang and Mao, 2023). When the dosage is too large, the magnetic flocculant will form a “polymer adsorption film” layer on its surface, which will hinder its adsorption of the polymer. Moreover, due to the huge spatial resistance of the adsorption film, the polymer particles can easily exclude each other, which will be re-stabilized and dispersed into solution. Consequently, the turbidity of the solution will be increased (Dai et al., 2021b).
Discussion of water quality optimization
Water quality optimization of the coal gasification black water system is a key link to ensure efficient and stable operation, which is directly related to the efficiency of clean coal utilization. Currently, the system is facing more serious water quality problems. The high hardness, high turbidity, and high dissolved solids content of black water and gray water not only increase the risk of system fouling, but also cause difficulties for subsequent treatment and reuse.
The magnetic flocculant prepared in this study demonstrated significant advantages in water quality optimization. Compared with traditional polyacrylamide and other common flocculants, the magnetic flocculant can achieve rapid solid–liquid separation under the combined effect of gravity and external magnetic field, which greatly shortens the flocculation and settling time. In the treatment of coal gasification black water, when the magnetic flocculant dosage was in the appropriate range (1.4–2.0 × 10−6), the black water turbidity can be effectively reduced to 22–24 NTU, indicating it has a strong adsorption and aggregation capacity for suspended and colloidal particles. At the same time, the floc formed by the magnetic flocculant has a compact structure, low water content, and produces less sludge. The magnetic particles in the sludge can be easily thickened and dewatered through the magnetic field, which reduces the difficulty and cost of sludge treatment, and reduces the secondary pollution to the environment. In addition, the magnetic flocculant also is reusable, and can be used again after appropriate treatment, which reduces the use cost of pharmaceuticals and improves the utilization rate of resources.
However, magnetic flocculant also has certain limitations in practical application. Its treatment effect is greatly affected by the dosage. When the dosage exceeds a certain range (e.g. when it reaches 3.0 × 10−6), solution turbidity is increased. This is because the increase of spatial site resistance affects the bridging and adsorption of the flocculant, and leads to the formation of “polymer adsorption film” hindering the adsorption of the polymers, which makes the redispersion of the particles. In addition, the stability and durability of magnetic flocculants during long-term operation are still relatively under-studied. Whether the performance will be degraded after repeated recycling and how to maintain its stable treatment effect need to be further explored.
In order to achieve more efficient water quality optimization, magnetic flocculants can be considered for joint use with other treatment technologies in the future. For example, combined with membrane absorption and hardness removal technology, magnetic flocculants are first used to remove suspended solids and colloidal particles in the water to reduce the turbidity of the black water. Then the membrane absorption and hardness removal technology is used to remove calcium, magnesium ions, and other hardness components in the water, so as to achieve the depth of the black water purification. Alternatively, it can be used in conjunction with electroflocculation and hardness removal technology, which can produce polymers with adsorption effect through electrode reaction, and the adsorption effect of magnetic flocculant complements each other to improve the effect of removing pollutants.
From a longer-term perspective, water quality optimization also requires comprehensive consideration of the operating parameters and process characteristics of the entire coal gasification system. On the one hand, the coal gasification process shall be further optimized, the coal ash and other impurities in the black water system be reduced, and the degree of water quality pollution from the source be reduced. On the other hand, the real-time monitoring and control of black water and gray water quality shall be strengthened according to the water quality changes in a timely manner to adjust the treatment process and the amount of chemicals added. This process ensures the water quality always meets the requirements of system operation. In addition, the development of new environmentally friendly water treatment chemicals and technologies can be explored to improve the efficiency and effectiveness of water quality treatment, while reducing the impact on the environment.
In summary, water quality optimization is crucial for coal gasification systems. Magnetic flocculants, as a new type of water treatment chemicals, provide new ideas and methods for water quality optimization but still need to be continuously improved and refined. Through comprehensive use of various treatment technologies and optimization of system operating parameters, it is expected to effectively optimize water quality in coal gasification systems and promote the development of clean and efficient utilization of coal (Tian et al., 2024; Wang et al., 2019).
Conclusions
Through industrial analysis, elemental analysis, XRD analysis, and SEM–EDS analysis of the coal samples, it is clear that the ash in coal is the most important factor affecting the scaling of the black water system. Ash in coal mainly consists of CaO, MgO, SiO₂, Al₂O₃, Fe₃O₄, and so on. During coal gasification, most of the ash turns into residue, and the small particles of residue enter into the equipment, and scale is formed by adhering to suitable conditions. Scale flakes are mainly composed of calcite, gypsum, and dolomite, and the main components are CaCO₃ and MgCO₃, and also contain a certain amount of CaSO₄. The calcium and magnesium ions in the scale flakes mainly come from dolomite and calcite in coal, which enter the black water system during coal gasification, and as the temperature of the black water decreases, they combine with carbonate and sulfate ions to form precipitates, which attach to the equipment and affect the operation of the system. The magnetic flocculant prepared combines non-toxic and environmentally friendly ferric oxide and PAM to overcome the shortcomings of traditional flocculants. When the dosage is 1.4–2.0 × 10−6, the flocculation effect is good and the turbidity of black water can be reduced to 22–24 NTU. However, when the dosage exceeds a certain range, such as 3.0 × 10−6, the turbidity of the solution rises, and the flocculation effect deteriorates (Fan et al., 2020).
After a series of experiments, this project studies the main influencing factors of coal gasification system fouling, and puts forward some improvement measures. We prepare a magnetic flocculant that can effectively alleviate the fouling of the coal gasification system. This flocculant can provide an idea for the coal gasification black water system in the Huaibei area, and a method for alleviating the fouling of the black water system. It helps to develop the coal gasification industry, strengthen the utilization of coal raw materials, and accelerate the construction of strategic bases for coal-to-oil and coal-to-gas. It also strengthens the production capacity and technology reserve of coal-to-oil and coal-to-gas. It promotes the coupling development of the coal chemical industry with green power, green hydrogen, energy storage, carbon dioxide capture and utilization and storage, etc. Moreover, it creates a low-carbon recycling industry chain for the efficient conversion of coal, promotes the high-end, diversified, and low-carbon development of the coal chemical industry, and is conducive to the clean production and utilization of coal (Tan et al., 2022).
However, this study still has some limitations. For instance, coal gasification system fouling is caused by a variety of influences. In addition to these influencing factors, there may be some other influencing factors. The study of magnetic flocculants is relatively simple, and the stability and durability of magnetic flocculants in long-term operation are still relatively insufficient in terms of application. Whether its performance will be degraded after many cycles of use, and how to maintain its stable treatment effect need to be further explored. Future research can be carried out in a number of areas. In terms of preparation process optimization, simpler, more efficient, and low-cost preparation methods need to be further explored to reduce the consumption of raw materials and the generation of waste, and to improve the production efficiency and quality stability of magnetic flocculants. New raw materials or preparation techniques can be studied to reduce the preparation cost and improve its environmental friendliness. For the improvement of application performance, the adaptability of magnetic flocculants in different water quality and working conditions should be thoroughly studied. Through a large number of experiments and simulation studies, the optimal conditions of use and scope of application should be determined. At the same time, the research on the joint application of magnetic flocculants and other treatment methods (e.g., combining with membrane separation technology, chemical precipitation method) in order to improve the overall treatment effect and adaptability to complex water quality (Xuan et al., 2022).
Footnotes
Author contributions
Laiyang Zhu: investigation, methodology, and conceptualization; Dongpeng Zhang: project administration; Tiantian Ma: funding acquisition; Nie Sun: software; Fei Liu: resources and supervision.
Conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Huaibei Major Science and Technology Project (grant number HK2021012); the Process Research on Water Quality Optimization of the Shenning Furnace Black Water System (Enterprise-commissioned Projects, 2024-32 and 2021-13).
