Abstract
Coal gasification fine slag (CGFS) is a by-product of gasification process of gasification beds that pollutes the environment. To make the utilization of CGF more efficient, poly (vinyl chloride) (PVC)/CGFS composites were prepared via a melt blending process, and their mechanical and thermal properties were investigated. In order to dig deeper into the decomposition mechanism, the thermal degradation kinetics were systematically analyzed by drawing non-isothermal heat maps based on the Flynn-Wall-Ozawa, Friedman, and Kissinger model-free methods. According to the results, the properties of all tests vary significantly with CGFS content. In particular, at a the CGFS content of 20 phr, the tensile strength (tensile modulus) and flexural strength (flexural modulus) of CGFS-20 were 13.5% (47.8%) and 13.9% (41.6%) higher, respectively, than those of conventional CGFS-0 composites. However, the impact strength decreases with the increase of CGFS content. At the same time, the addition of CGFS suppresses the mass conversion rate and improves the thermal stability of PVC composites at high temperature, and the final residual mass reaches 31%, which is 12.7% higher than that of pure PVC. The kinetic analysis of thermal decomposition shows that CGFS promotes the initial degradation of the composites reduces the activation energy of the reaction, and at the same time endows them with higher thermal stability in the high-temperature phase.
Introduction
Poly (vinyl chloride) (PVC) is a widely used, inexpensive thermoplastic, ranking second only to polyethylene (PE) and polypropylene (PP) in global production. Thank you to its high electrical insulation, outstanding corrosion resistance, and superior abrasion resistance, 1 PVC is extensively used in construction materials, imitation leather coatings, packaging materials, wires and cables, medical composites, and other fields.2–4 However, regardless of its competitive mechanical properties compared to traditional resins, PVC suffers from its low tensile strength and poor thermal properties during processing and use. Consequently, reinforcing the PVC matrix by adding a suitable filler is essential to improve the thermal stability and mechanical properties of PVC to meet the requirements for practical applications. The most common fillers for PVC are glass beads, calcium carbonate, talc, montmorillonite, glass fibers, and wood fibers.5–7 However, these reinforcing additives may have some drawbacks, such as excessive consumption of mineral resources, pollution of ecological environment, high production cost, etc. Therefore, the development of low-cost filler products with high added value has become an urgent task.
Coal gasification slag is currently the most promising candidate. It is a by-product of the expansion and rupture of raw coal particles and the gradual consumption of the carbon matrix in a fluidized bed gasification process. Depending on the particle size, it can be classified as coal gasification fine slag (CGFS) and coal gasification coarse slag (CGCS). 8 CGFS is mainly composed of minerals (SiO2, Al2O3, CaO, or Fe2O3) and flocculated porous carbon. 9 Because of its complex chemical composition compared to single-component fillers, CGFS gives more possibilities for the performance of the filling material. For instance, Ai et al.10,11 embedded CGFS in various resins, thereby improving the strength, stiffness and elongation of the materials to a large extent. Zhang et al. 12 prepared a multifunctional and low-cost mesoporous powder (CGSF3) was prepared from coal gasification fine slag. It was found that the tensile, impact and flexural properties of PP -CGSF3-30 composites were 49.84%, 70.81%, and 139.63% higher than those of PP-CC -30, respectively. In addition, CGFS3 exhibited excellent performance for resin deodorization properties. Xu et al. 13 CGFS mesoporous silica was prepared by acid leaching, calcination and pH adjustment, and the filled styrene-butadiene rubber composites showed better mechanical properties and resistance to wetting compared with commercial precipitated silica.
During the last decade or so, many scholars have only considered the influence of filler particles on the mechanical properties alone.14,15 However, the type of filler in the composite formulation exerts a direct impact on the thermal degradation behavior of PVC. 16 In particular, filters play a crucial role in the processing temperature conditions of the composite through extrusion, injection, and compression, which may deteriorate or improve the thermal stability of the final material. The presence of fillers can improve or reduce its thermal stability.17,18 CGFS is composed of various metal oxides and is similar in chemical composition to fly ash from power plant combustion, so a similar effect on the mechanism of PVC thermal degradation is expected.
In order to optimize the utilization of CGFS, the present work aims at studying composites based on PVC with CGFS fillers. It is noteworthy that a suitable filler preparation is a prerequisite for obtaining composites for processing and forming. For this purpose, the composites with different filler contents were prepared via the particle functionalization of CGFS by mechanical ball milling and chemical modification. Particular attention was paid to the influence of the modified CGFS content on the thermal stability and mechanical properties of the PVC composites. In addition, given that thermal degradation greatly impacts the temperature of PVC processing, a kinetic model has been used to elucidate the effects of CGFS addition on the degradation mechanism of the composites.
Experimental
Materials
Chemical composition of CGFS (wt.%).

Surface modification of CGFS
CGFS was ball-milled for 60 min using a planetary ball mill and then sieved through in a 100 mesh sieve to obtained a fine powder. The powder was then treated with a (3-aminopropyl)triethoxysilane solution. Deionized water and anhydrous ethanol were mixed in a ratio of 1:3 (v/v) to obtain 100 mL of the solution, and a small amount of hydrochloric acid was added dropwise to obtain a pH of 3–4. In the obtained solution, 10% silane was dissolved and mixed thoroughly for 30 min so that all components were homogeneously dispersed. Then, 100 g of CGFS were introduced into the silane solution and thoroughly stirred for 15 min. The resulting slurry was dried in an electric fan dryer at 105°C for 12 h and lightly ground to obtain the modified CGFS.
Preparation of PVC/CGFS
Formulation of PVC/CGFS composites.

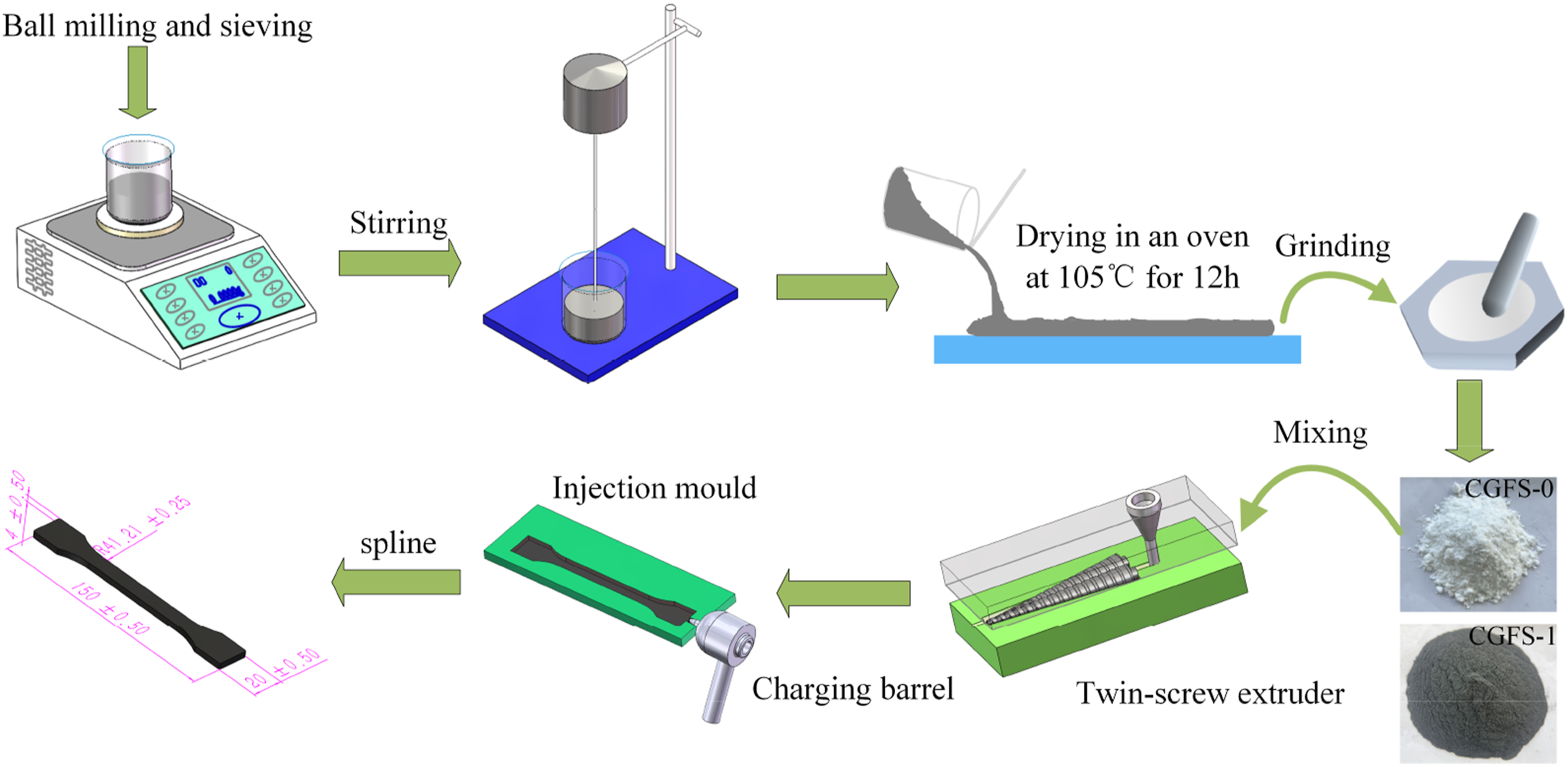
Flow chart of CGFS preparation.
Characterizations
XRF studies
The chemical composition of the CGFS samples was determined using an X-ray fluorescence spectrometer (ARL-980, ARL, Switzerland) with boric acid as the carrier, and the power and current during the measurements were set at 40 kV and 66 mA, respectively, and the temperature was controlled at (25 ± 2)°C.
XRD studies
An X-ray diffractometer (XD-3, Pu-Analysis General Instrument Co., Ltd. (Beijing, China)) with a Cu target was used. The working tube voltage and the current were 36 kV and 30 mA, respectively. The diffractograms were recorded within a 2θ scan range from 12° to 65° at a step size of 2 (°)/min.
Particle size studies
The particle sizes of the samples were determined using a laser particle size analyzer (BT-2003, Danyang Baxter Instruments Co., Ltd. (Jiangsu, China)). Prior to the experiments, 0.05 g of the sample to be measured was added to 2 mL of anhydrous ethanol. The blend was then stirred thoroughly and slowly added to the automatic recirculating injection system for subsequent analysis. The refractive index was controlled to be between 5% and 10%, and the measurements were repeated 3 times to obtain the average value.
Scanning electron microscopy
A scanning electron microscope (ESCAN VEGA3 SBH, TESCAN, Germany) was used to observe the morphology of CGFS samples. Before the experiments, powders were glued to a copper support with a conductive paste and covered with a gold coating via spraying at an accelerating voltage of 20 kV. The appropriate magnification at an accelerating voltage of 30 kV was chosen in the course of measurements.
Mechanical property
A universal testing machine (WDW-50, Shenzhen KQL Co., Ltd., (Guangzhou, China)) was used to determine the tensile strength and flexural strength, as well as to calculate the tensile modulus, flexural modulus and elongation at break from the corresponding values. The tensile strength and tensile modulus were established conforming to GB/T1040-1992 (“Test Methods for Tensile Properties of Plastics”) standard on the dumbbell type specimens (type I) with the dimensions of 150 mm × 10 mm × 4 mm; the test speed was 10 mm/min. The flexural strength and flexural modulus were calculated according to GB/T1048-2008 (“Determination of Flexural Properties of Plastics”) standard. A three-point flexural method was used for testing, and the standard specimen size was 80 mm × 10 mm × 4 mm. The test span was 64 mm, the deflection was 6 mm, and the speed was 2 mm/min. The impact strength was calculated according to GB/1048-2008 (“Test Method for Tensile Impact Properties of Plastics”) standard using a simple beam impact tester (TGG-25, Taihe Testing Machine Co., Ltd., (jilin, China) The “V” notched rectangular strips with the sizes of 80 mm × 10 mm × 4 mm served as the samples, and the notch width was 1/3 of the sample strip width. The average value was calculated from the five trials.
Thermal property
The thermal stability of PVC composites with different CGFS contents was analyzed using a STA 449 F3 simultaneous thermal analyzer (NETZSCH, Germany) under N2 atmosphere at a flow rate of 50 mL/min from room temperature to 1073 K. The temperature was increased at the rates of 5, 10, 15, and 20 K/min. The CGFS sample was taken from room temperature to 1600K at 10 K/min. The TGA and DTG curves were obtained, and the relevant thermodynamic parameters were analyzed.
Results and discussion
Basic analysis of CGFS
Figure 2(a) displays the particle size distribution and accumulation curves of CGFS. The particle size of microfraction has curve exhibits a single peak, covering the range between 0.039 and 39.87 μm. The average diameter (D50) of 50% of the particles was measured to be about 4.479 μm.
19
The XRD analysis (Figure 2(b)) revealed that the CGFS consisted of extensive amorphous silicate glass phase.
20
According to the TGA curves (Figure 2(c)), the CGFS contained moisture, volatile fractions, and a large amount of unburned carbon, of which the porous charcoal was highly compatible with the substrate.
10
The SEM image of CGFS (Figure 2(d)) disclosed spherical particles with a large surface roughness that contained numerous irregular smaller particles on their surface.
21
Basic analysis of CGFS: (a) Particle size distribution; (b) XRD pattern; (c) TGA and DTG curves; (d) SEM image.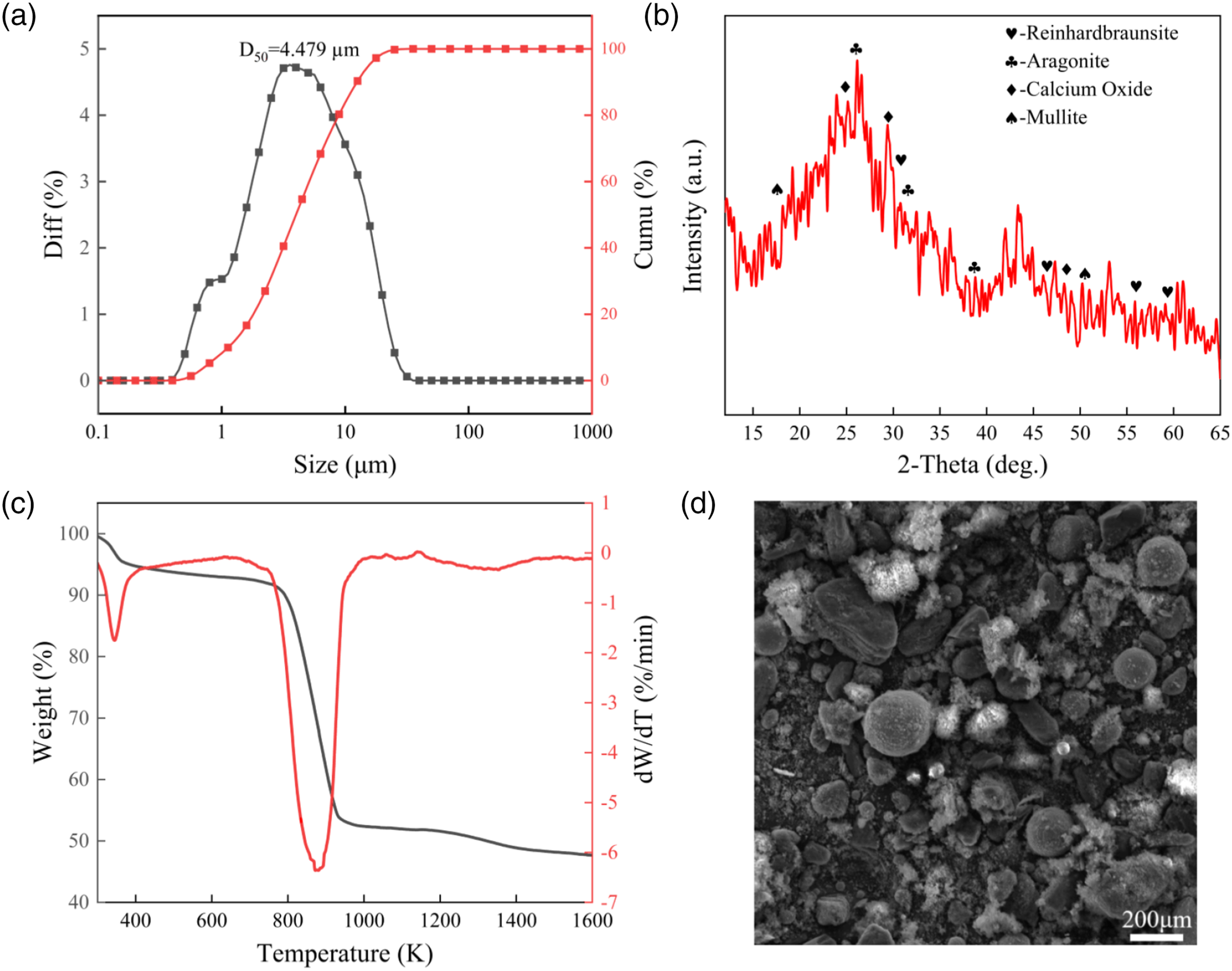
Mechanical properties
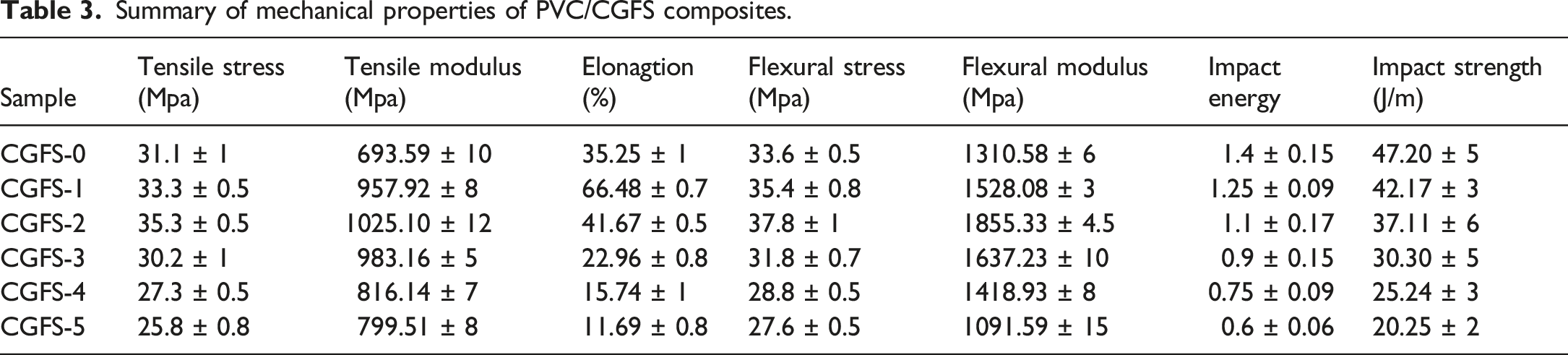
The mechanical response to quasi-static load is one of the fundamental properties of plastic parts. The mechanical parameters of the PVC/CGFS composites with different CGFS additions are illustrated in Figure 3. Summary of mechanical properties of PVC/CGFS composites in Table 3. As shown in Figure 3(a), the tensile strength and tensile modulus with increasing CGFS loading first increased, reaching their maxima (35.3 MPa and 1025.10 MPa, respectively) in CGFS-2, and then decreased. The increase in both values could be due to a small amount of CGFS uniformly dispersed in the polymer matrix and the strong interaction between PVC and CGFS.
22
In turn, excessive CGFS particles could have agglomerated and accumulated in the PVC matrix, causing them to be unevenly dispersed, resulting in a decrease in tensile strength and tensile modulus. Figure 3(b) shows that a small amount of CGFS filler (10 phr) can increase the elongation at break of the composites by 31% compared to the pure PVC material.
23
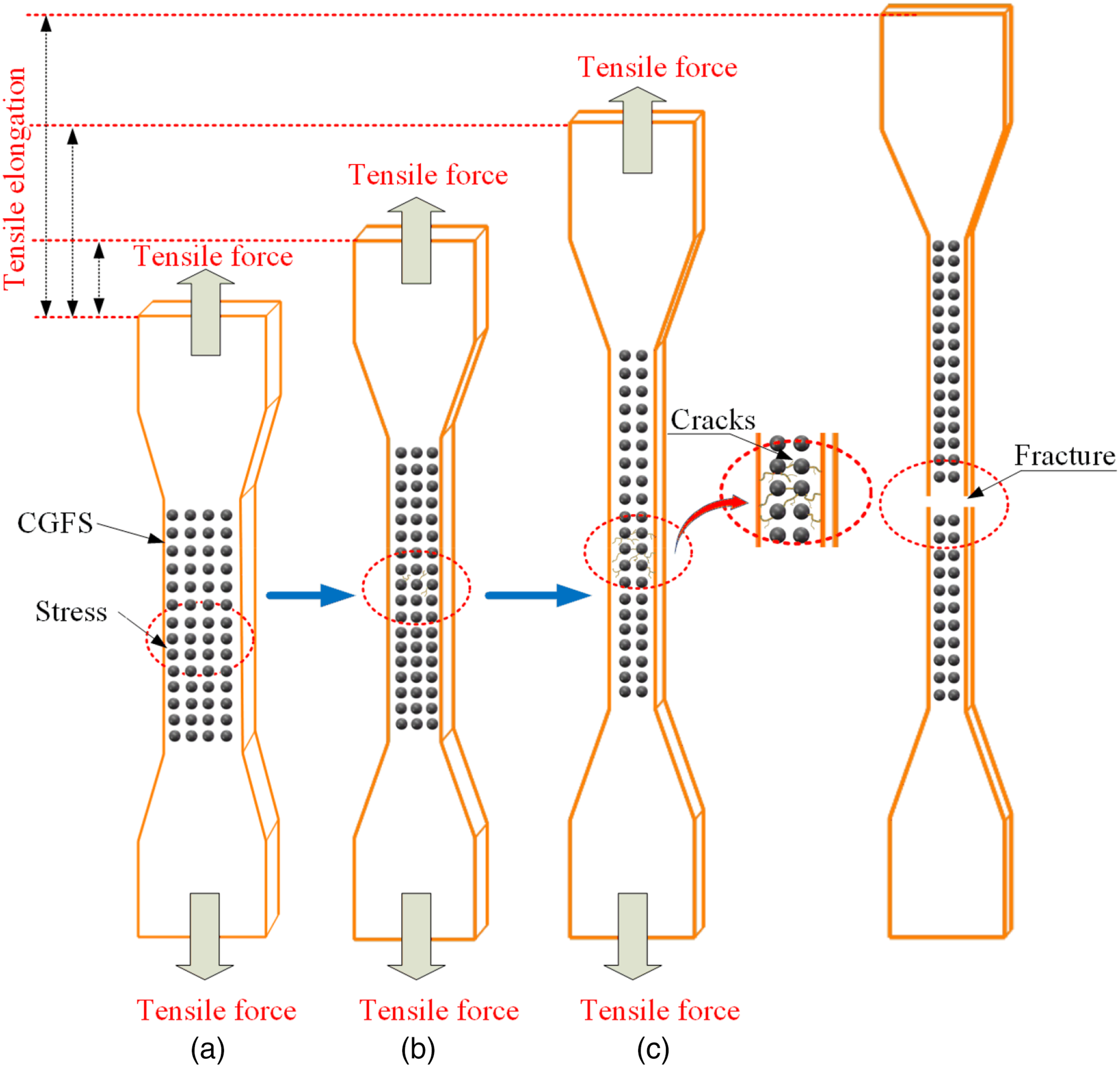
The possible mechanisms affecting this parameter are displayed in Figure 4. When the composite is subjected to tension loading, the force (tensile force) initially acts on the CGFS particles in the polymer matrix, so that the modified CGFS particles can interact strongly with the polar PVC matrix to improve the composite’s overall performance.
24
This force is then transferred to the polymer matrix until the section is deformed or undergoes transition from elastic to plastic state. Figure 3(c) depicts the flexural strength and flexural modulus of the PVC/CGFS composites with different CGFS content. The tensile modulus of the PVC/CGFS composites increase and then decrease with increasing CGFS addition. Compared to the flexural strength and flexural modulus of the pure PVC material (33.6 MPa and 1310.58 MPa, respectively), the flexural strength and flexural modulus increased by 10.4% and 41.6%, respectively, followed by a decrease.
25
This trend is similar to that described above. The lack of impact strength is one of the main drawbacks of PVC materials. The introduction of CGFS filler into the PVC matrix generally resulted in a decrease in impact strength (see Figure 3(d)), which is likely due to the fact that the immobilization of the polymer chains by the CGFS particles may have inhibited plastic deformation, and thereby absorbed the fracture energy.
13
As a result, the tendency for crack expansion increased and the impact work decreased once the composite was subjected to an external force, thus enhancing the brittleness of the material.
26
Mechanical properties of PVC composites: (a) Tensile strength; (b) Elongation; (c) Flexural strength; (d) Impact strength. Summary of mechanical properties of PVC/CGFS composites. Simulation of the tensile mechanism of the composite under tensile stress.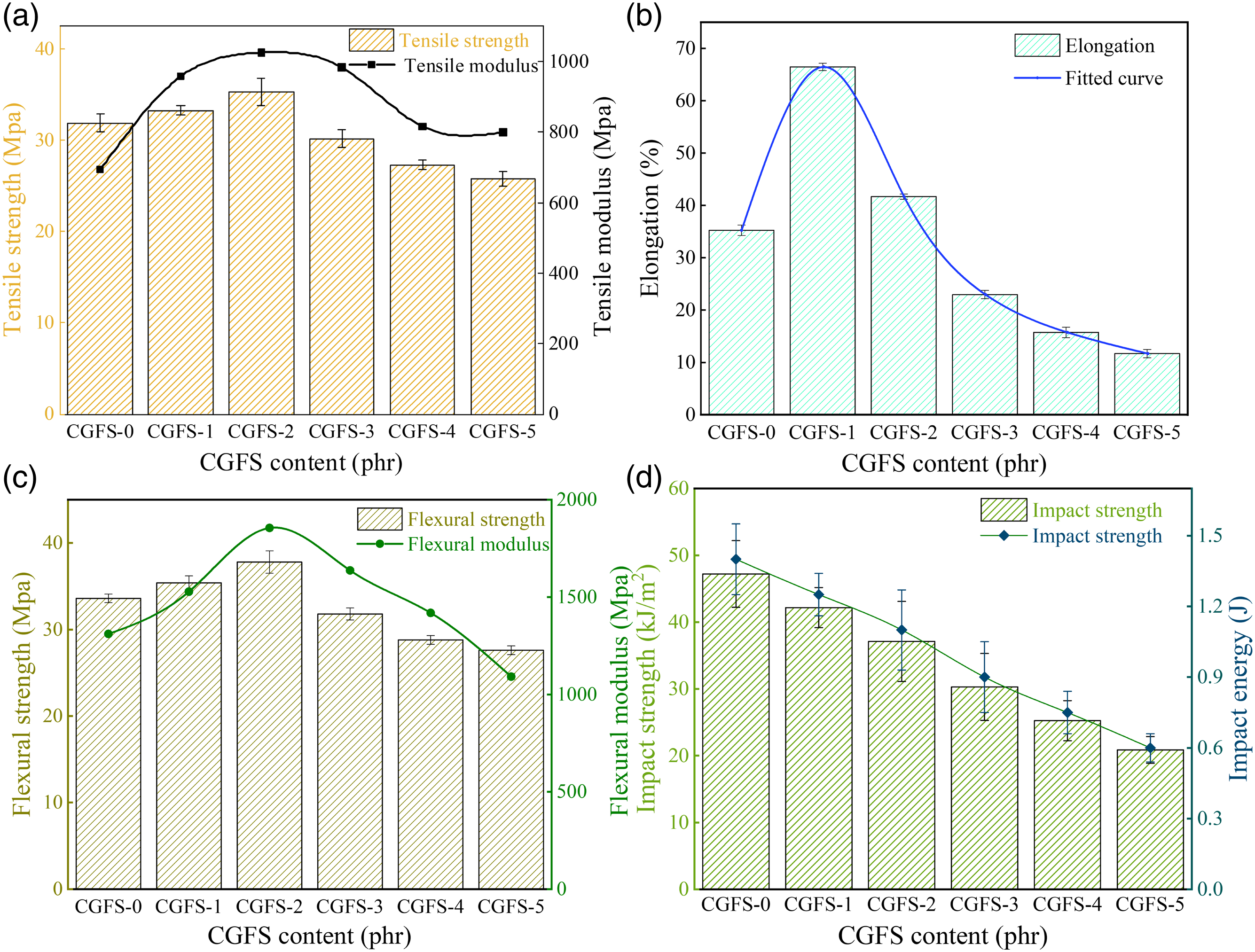
Thermal stability
The thermal stability of a polymer is an important parameter in evaluating the thermal resistance of a material and determining of the appropriate extrusion and injection molding temperature. Figure 5 displays the TGA and DTG curves, the temperature-dependent mass conversion rate (α), and the first-order derivative dα/dT for a PVC/CGFS composite at a heating rate of 10 K min−1. Here, α = W0 - Wt/Wt - W∞ (W0 is the initial weight, Wt is the actual weight, and W∞ is the final weight of the sample). The corresponding data are summarized in Table 4. As for other thermoplastic materials, the pyrolytic weight loss of PVC/CGFS composites can be broadly divided into two stages. The first stage mainly refers to the formation of chlorine radicals and the degradation of other additives such as DOP; this temperature is called the temperature of primary degradation (SDT). The second stage entails degradation of the PVC molecular chains (-(-CH = CH-)n-), resulting in excessive coloration of the polymer and deterioration of its physical properties.
1
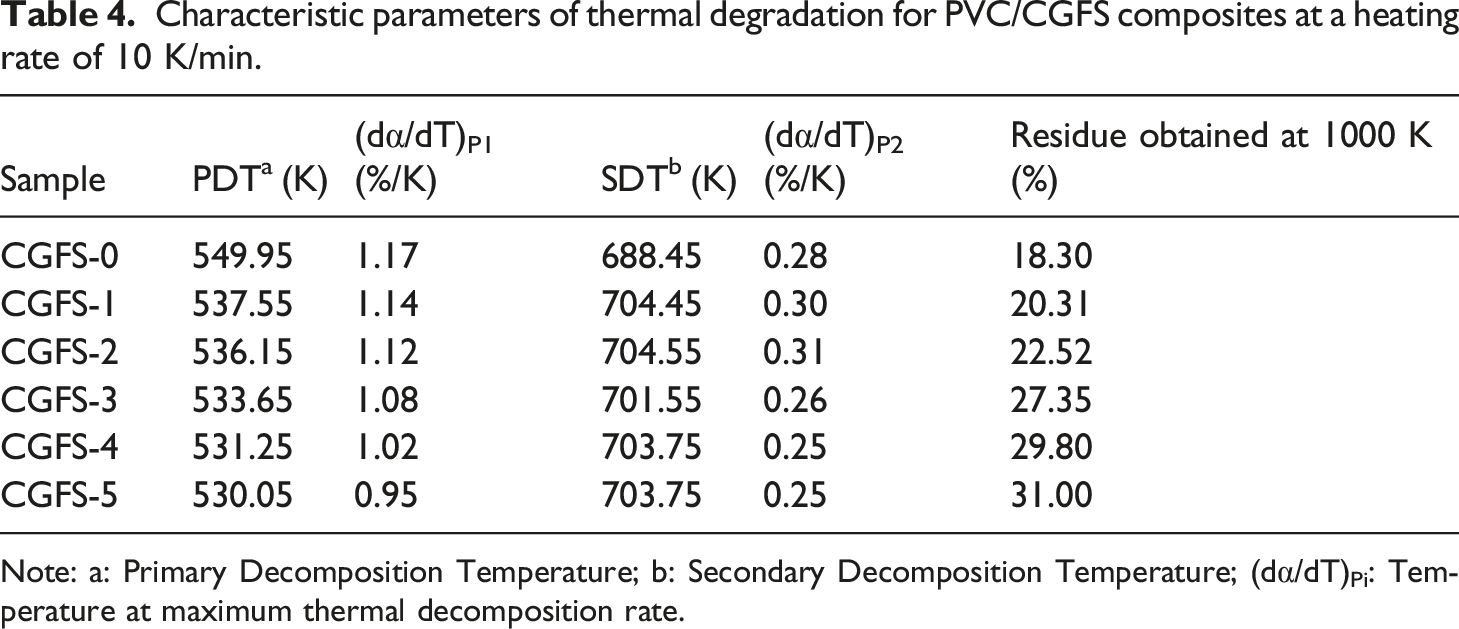
This temperature is referred to as the secondary degradation temperature (PDT). As shown in Figure 5(a), the thermal degradation curve of PVC/CGFS composites are roughly similar, as the filler does not affect the thermal stability of the PVC matrix. However, the thermal stability of CGFS is higher than that of PVC, and the increase of CGFS addition resulted in a slight decrease in the first degradation temperature (PDT) of the PVC/CGFS composite compared to that of the pure PVC. Moreover, the analysis of the dependence of (dα/dT)P1 (%/K) showed that the maximum degradation temperatures were all significantly lower than that of CGFS-1. The same result was also reported by other groups.27,28 The main reason is that the metal oxides (SiO2, Al2O3), which are part of CGFS, have high thermal conductivity and provide the diffusion pathway to reduce the initial temperature of thermal degradation of the composites. However, as the temperature increased, STD moved toward higher temperatures, with the highest second decomposition temperature of the composite occurring at a CGFS addition of 10 phr, indicating that the addition of CGFS retarded the cleavage of the PVC hydrocarbon backbone. It is possible that the chlorine radicals generated during the first degradation reacted with the metal oxides (CaO, Fe2O3) in the CGFS and replaced the oxygen to form metal chlorides without generating HCl gas. On the contrary, according to some reports, SiO2 and Al2O3 from CGFS do not participate in the reaction and therefore still release HCl gas during the degradation process.13,29 At 1000 K, the amount of unburned carbon increased due to the increase in the content of inorganic filler in the matrix, so the residual mass also increased with the addition of CGFS. Since the inorganic filler was stable enough at the measured temperature, it was difficult to remove by combustion. As a result, the thermal stability of the PVC composite could be improved to a certain extent. (a) TGA and (b) DTG curves, (c) α and(d) dα/dT plots of PVC/CGFS composites at a heating rate of 10 K min−1. Characteristic parameters of thermal degradation for PVC/CGFS composites at a heating rate of 10 K/min. Note: a: Primary Decomposition Temperature; b: Secondary Decomposition Temperature; (dα/dT)Pi: Temperature at maximum thermal decomposition rate.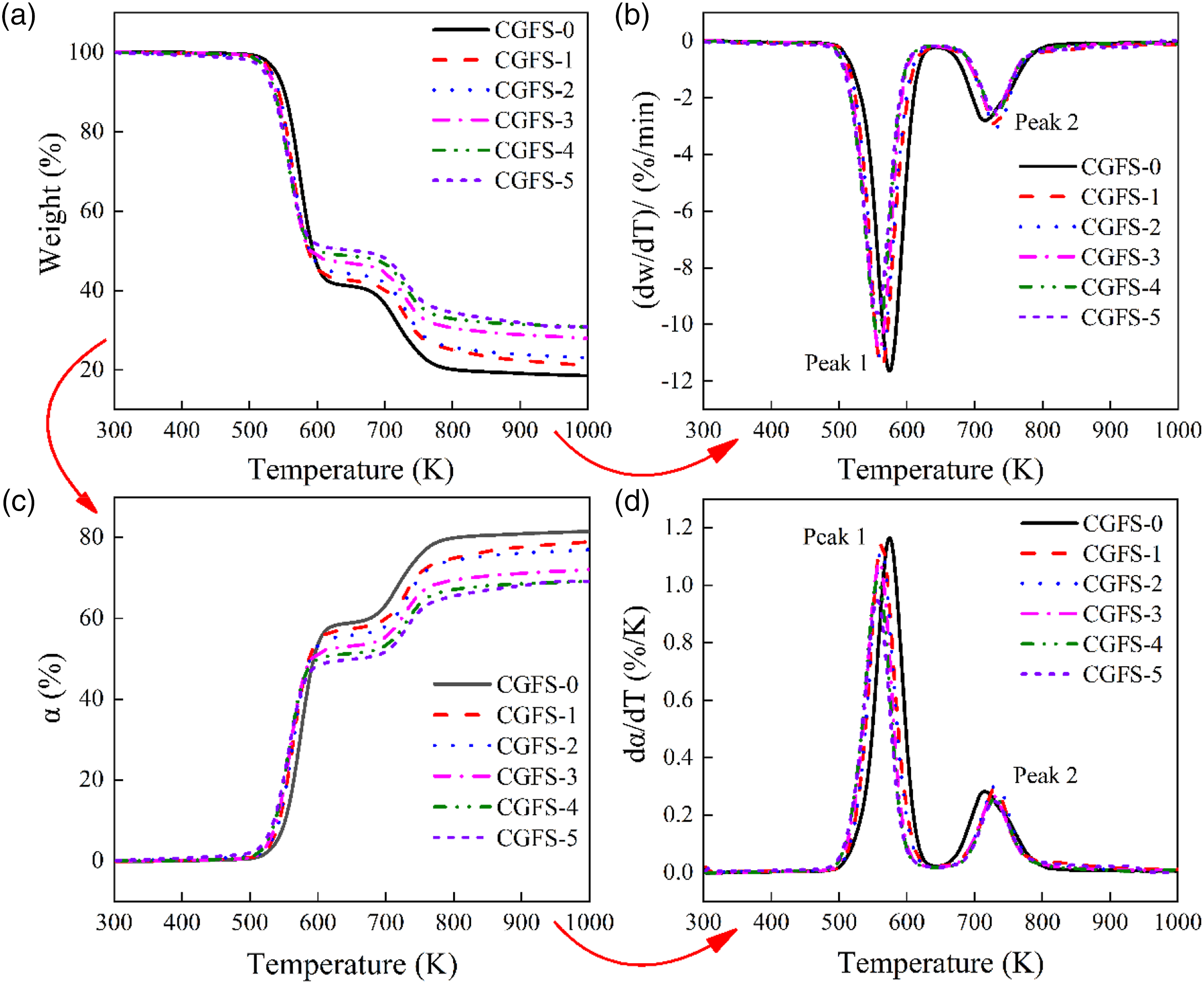
Pyrolysis kinetic analysis of composite materials
In order to gain insight into the macroscopic phenomena and reaction mechanisms accompanying the thermal degradation reactions of CGFS-filled PVC composites, as well as explore the inter-reactant interactions during thermal decomposition a kinetic model based on non-isothermal pyrolysis at a linear heating rate was used.30,31 This can be achieved by using a kinetic model based on non-isothermal pyrolysis at a linear heating rate.
32
Figure 6 depicts the TGA and DTG curves of pure PVC, CGFS-2, and CGFS-4. As the heating rate increased, both the TGA and DTG curves shifted to higher temperatures, indicating that the trend of thermal decomposition is related to the heating rate. The main reason for this is that the higher the heating rate, the shorter the time required for the composite to reach the target temperature, resulting in the incomplete reactions, thermal hysteresis phenomena, and heat transfer limitations,
33
which leads to a shift of the curve toward the high-temperature region. However, the remaining residual weights were approximately the same at 1000 K, indicating that the increased heating rate had a weaker effect on the conversion rate α. Representative TGA and DTG curves of PVC/CGFS composites at heating rates of 5, 10, 15, and 20 K/min: (a) CGFS-0 (b) CGFS-2, and (c) CGFS-4.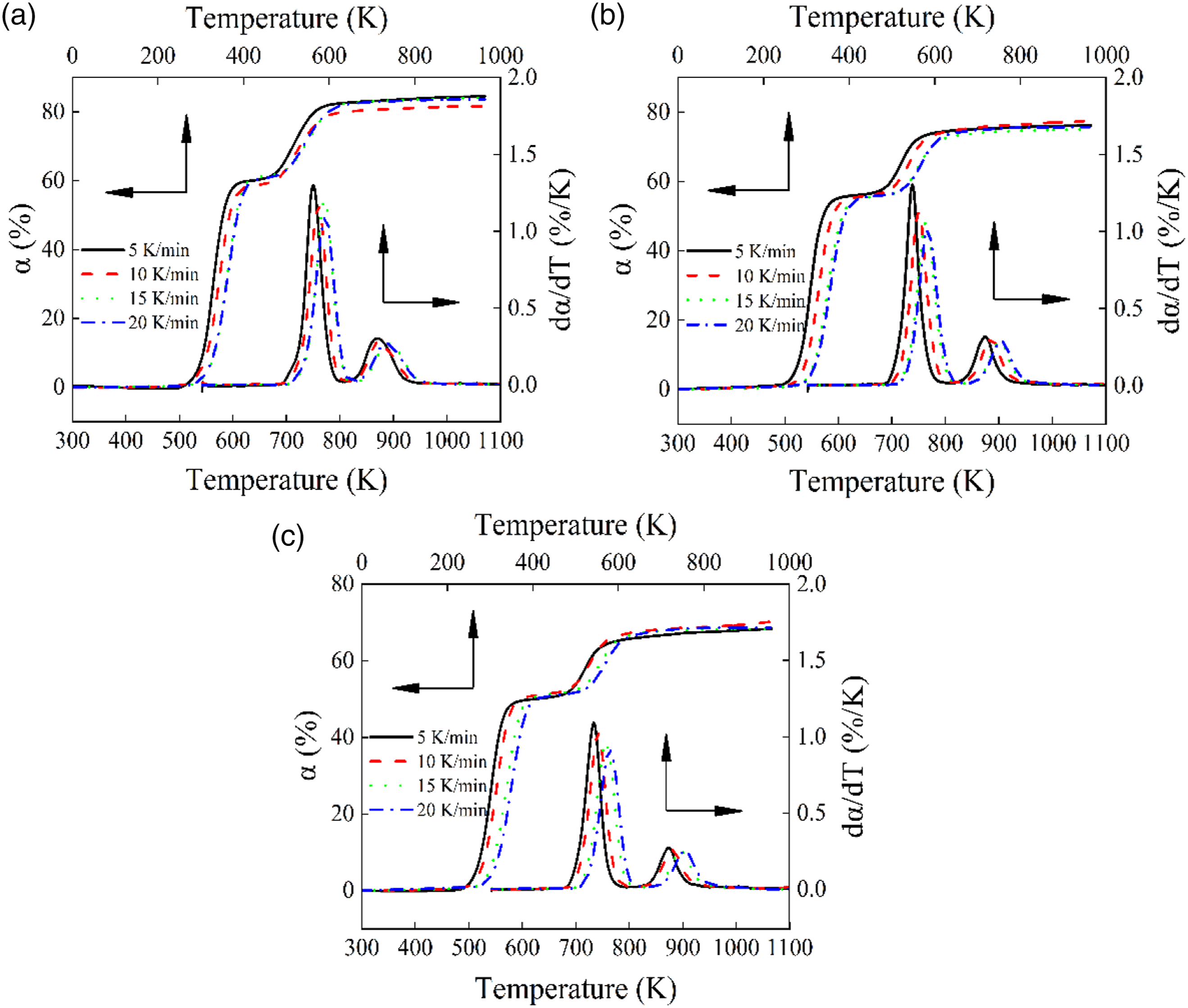
Determination of activation energy
Analysis of thermal degradation kinetics provides insight into the decomposition behavior of polymers. In turn, the kinetic parameters can be obtained from the non-isothermal TGA data. The thermal decomposition reaction process which usually conforms to the Arrhenius equation
34
involves a reaction model mechanism (f(a)), a mass conversion (α), and a change in reaction rate with respect to the temperature (K(T)). Then the decomposition rate (dα/dt) can be expressed as follows:
Based on the Arrhenius equation, the relationship between K(T) and temperature can be expressed as:
Given equations (1) and (2), the reaction rate can be expressed as:
Substituting the rate of heating β = dT/dt into equation (3) allows one to describe the rate of decomposition as a function of temperature rather than time as follows: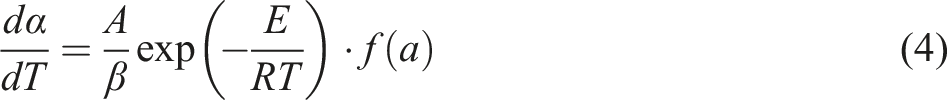
There are two types of mathematical kinetic models widely used in pyrolysis kinetics, which are the model-free method and the model-fitting approach.33,35 The model-free method does not require to consider the reaction mechanism and is well-adapted and effective. 36 Among them, when finding the E of the reaction, preference is given to the equal conversion rate method. 37 At the fixed conversion rate α, the reaction rate is a variable that depends only on the temperature T. The relationship between the apparent reaction E and the conversion rate α is again determined by calculation based on data from multiple TGA curves at different linear heating rates. Three model-free methods - Flynn-Wall-Ozawa (F-W-O),38,39 Friedman (FR), 40 and the Kissinger-Akahira Sunose (KAS) 41 - were used in this study.
The FWO method is derived from the integral equivalence transformation method. The temperature integral is solved using Doyle’s approximation
42
(p (E/RT) = −5.331-1.052 E/RT) as follows: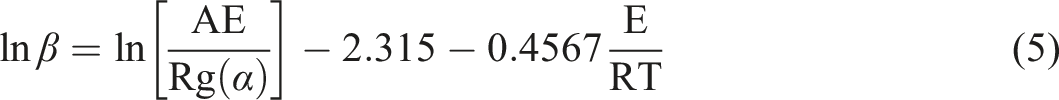
Here, the inversed temperature (1/T) is the independent variable and the logarithm of the heating rate (lnβ) is the response variable. The Friedman method is based on the differential transformation method and considers the logarithm of the heating rate and mass conversion rate (ln (β·dα/dt)) as a function of inversed temperature (1/T):
The KAS method is presented by the expression below: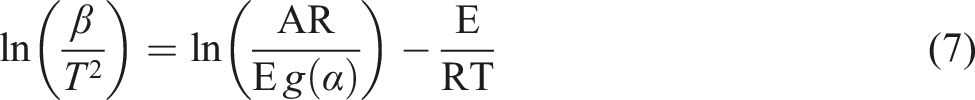
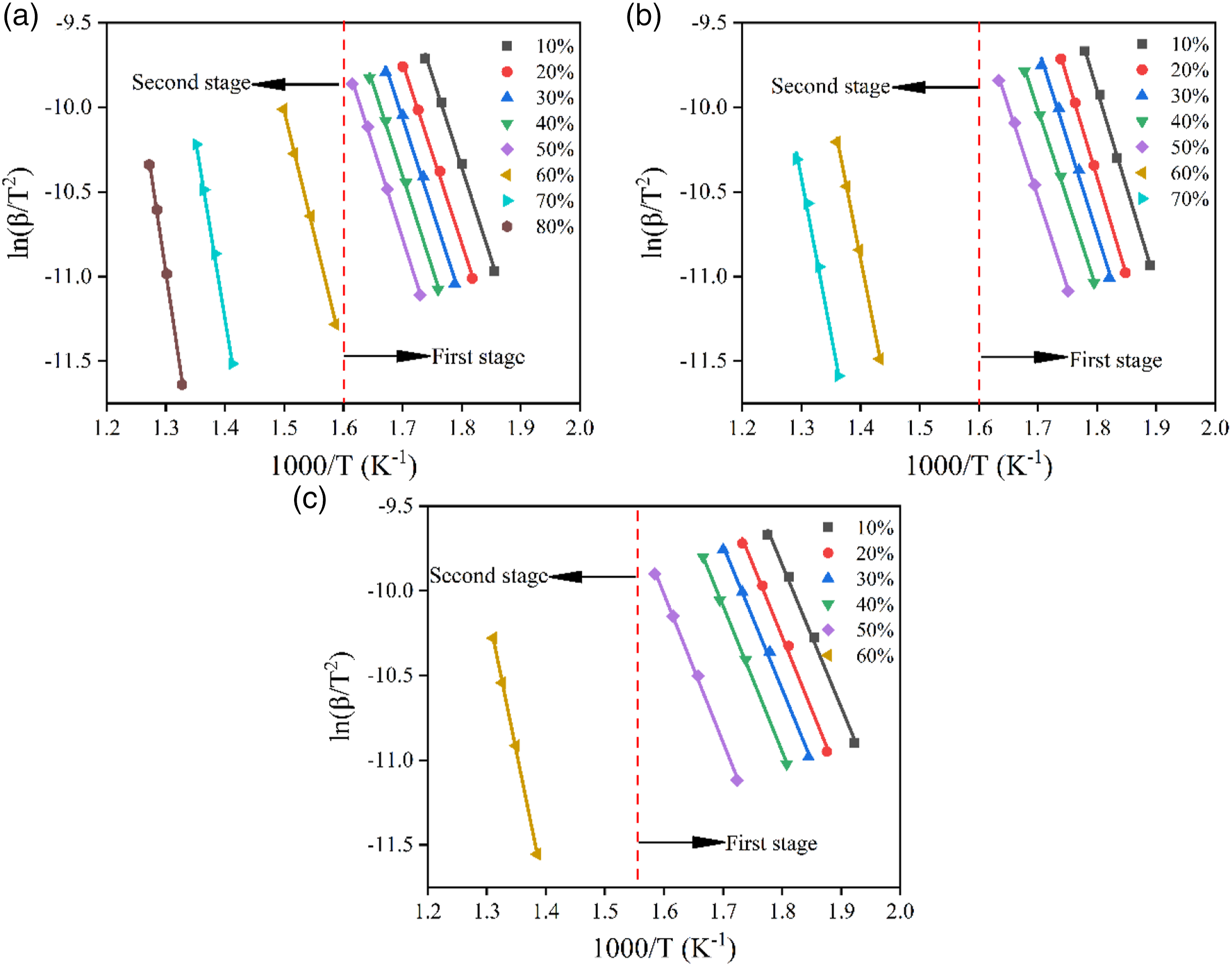
From this equation, the value of ln (β/T2) is a function of 1/T at different heating rates, which enables one to calculate the E.
These three methods can be used to investigate the relationship between the instantaneous E and mass conversion rate α. In particular, E is the energy barrier that allows one to define the chemical reaction to be overcome as well as its feasibility.43,44 It has been shown that the reaction proceeds more efficiently at lower activation energies. Figures 7–9 display the iso-conversion diagrams obtained at different heating rates, from which the instantaneous value of E at different conversion rates can be calculated from the individual slopes according to the linear regression equations. The E values determined based on FWO, Friedman, and KAS methods are listed in Table 5. From Figures 7–9 and Table 5, the range of mass conversion for pure PVC is 10–80%, while it is 10–70% and 10–60% for the composites with 10 phr and 20 phr CGFS, respectively. Since the PVC pyrolysis process occurs in two stages, the E values are not constant for all samples but vary with the α-value, indicating that the reaction mechanism for instantaneous conversion is not the same throughout the pyrolysis. In the first stage of thermal degradation (0.1 < α < 0.5), the E decreases with increasing α-value. Conforming to the FWO, Friedman, and KAS methods, the E of the pure PVC is 97.70 kJ/mol, 91.74 kJ/mol, and 92.11 kJ/mol, respectively. In the second stage (0.6 < α < 0.8), the E of pyrolysis is relatively high, reaching values of 197.67 kJ/mol, 164.15 kJ/mol, and 194.90 kJ/mol, respectively. This is due to the fact that stable chemical bonds are gradually formed in PVC as the increasing pyrolysis temperature increases. At the same time, there is an interaction between the free radical chains generated during the pyrolysis of PVC or other reactions at higher temperatures.
33
The dependence of the E on the conversion rate exhibits almost the same trend in all three models. However, the independence of the E of the composites for mass conversion is completely different from the control samples. Specifically, the E values vary relatively smooth manner in the Flynn-Wall-Ozawa and KAS models. Flynn-Wall-Ozawa plots of samples at different mass conversions: (a) CGFS-0, (b) CGFS-2, and (c) CGFS-4. Friedman plots of samples at different mass conversions: (a) CGFS-0, (b) CGFS-2, and (c) CGFS-4. KAS plots of samples at different mass conversions: (a) CGFS-0, (b) CGFS-2, and (c) CGFS-4. Activation energies of samples based on Flynn-Wall-Ozawa, Friedman, and KAS methods.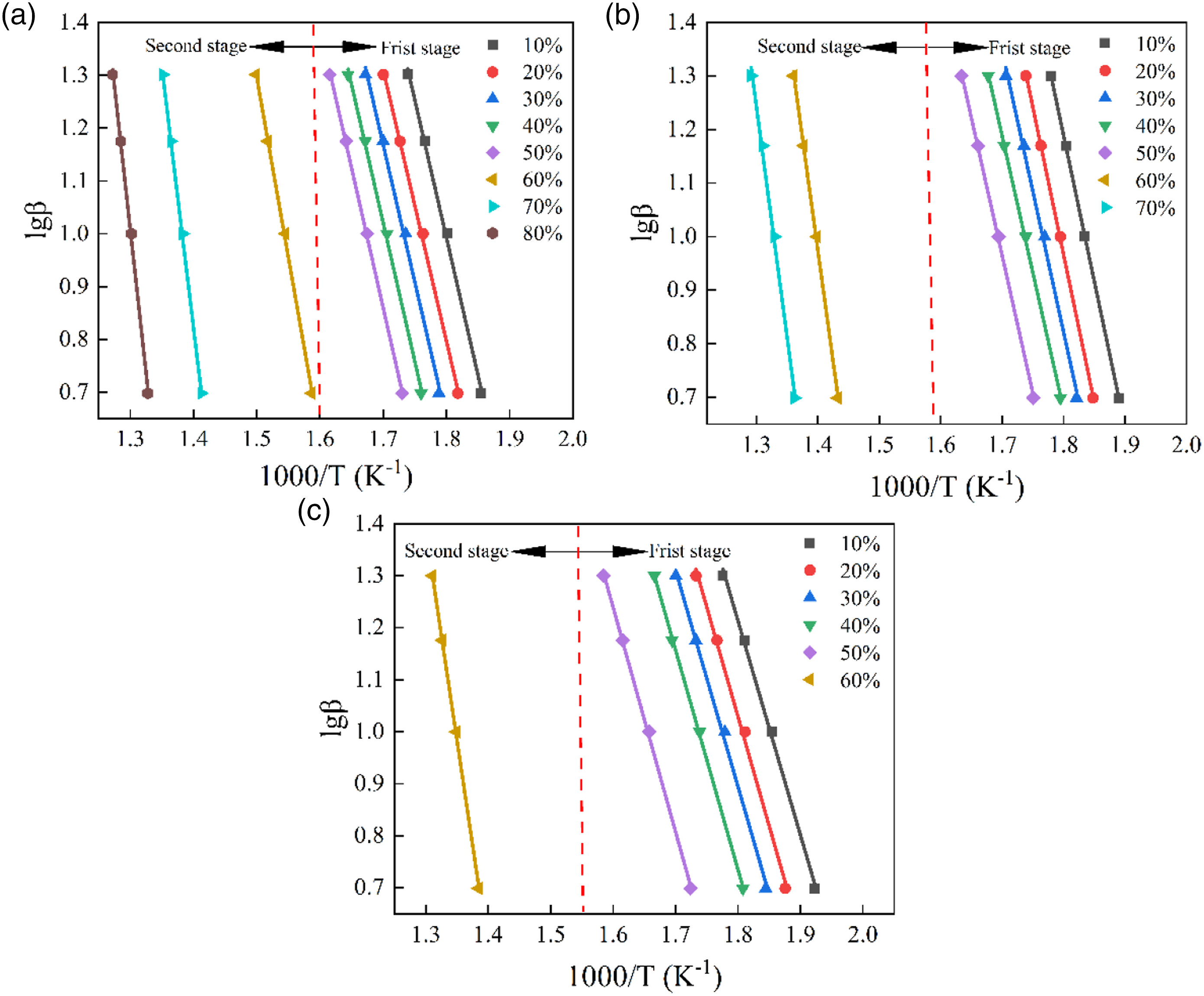
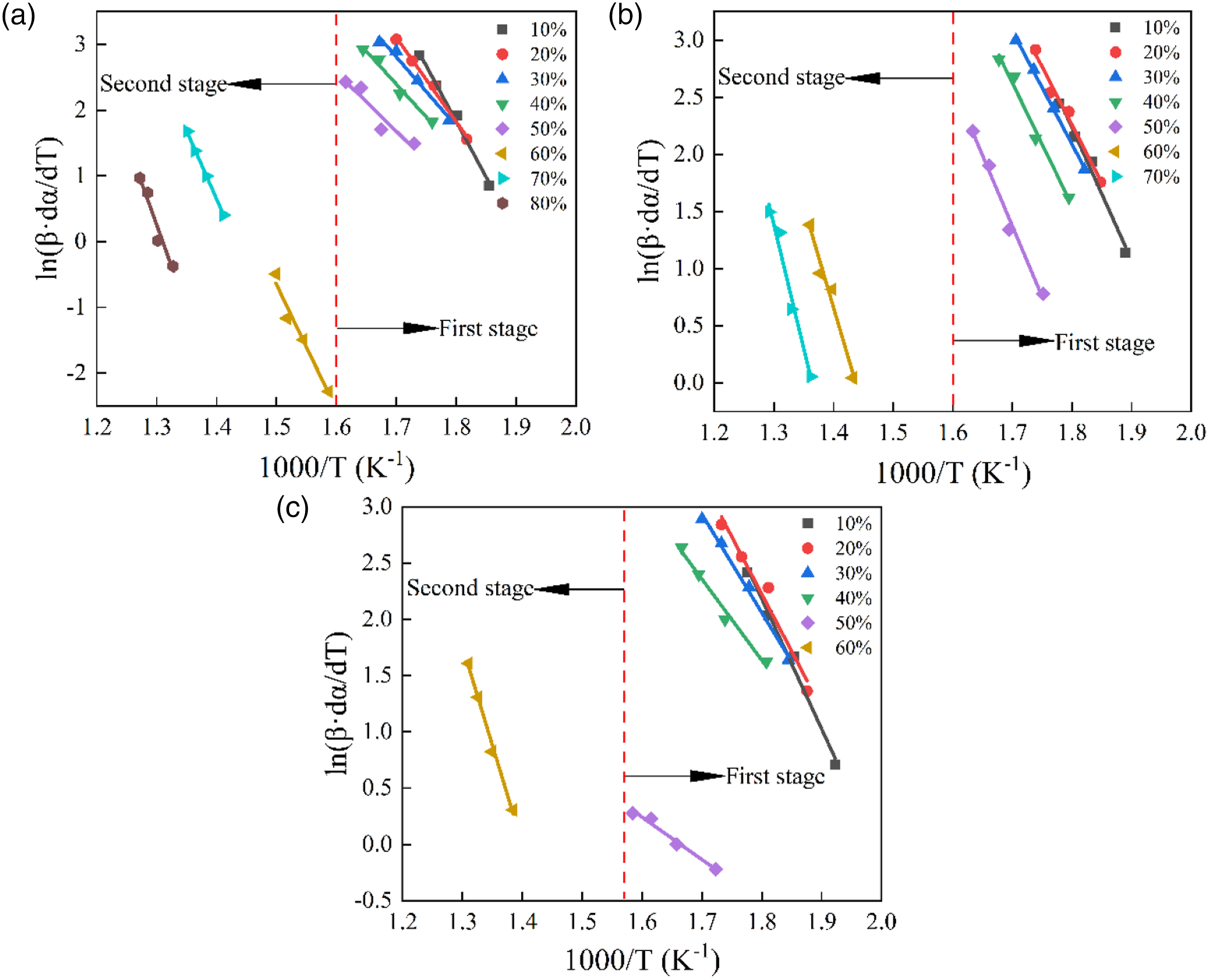
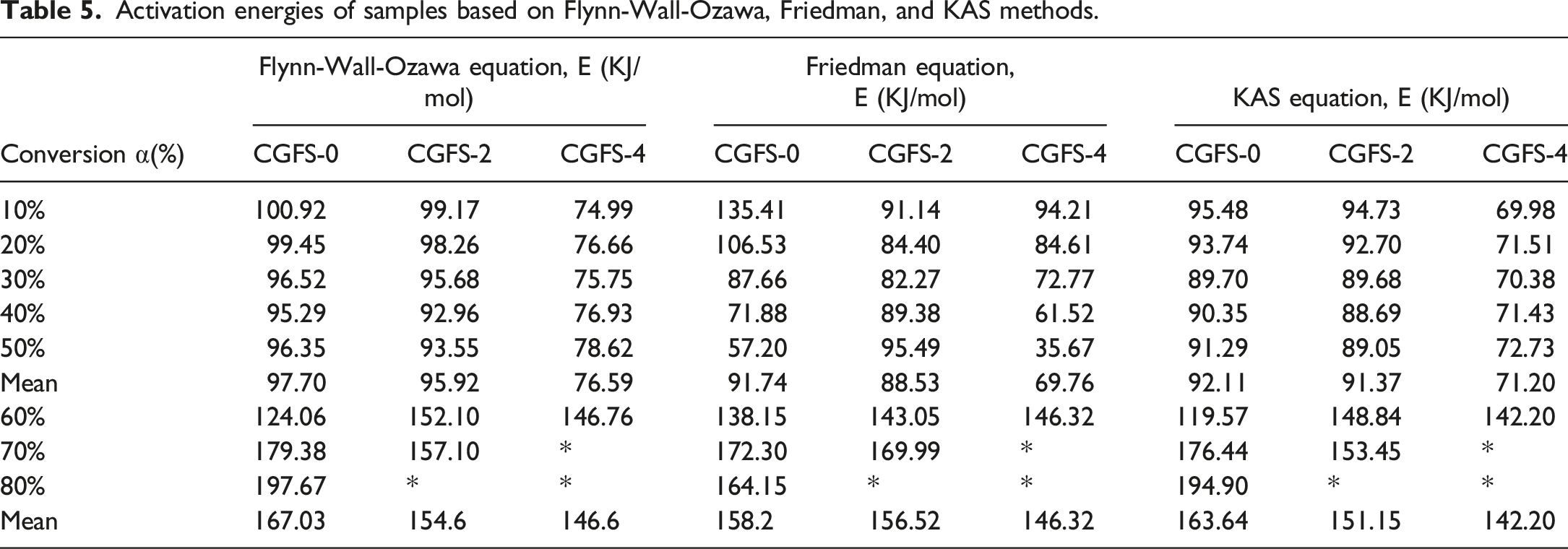
In contrast, the Friedman model has a large variation in E, which may be due to the use of instantaneous conversion rates. However, regardless of the calculation method used, the activation energies of the composites followed the same trend with increasing CGFS concentration. According to the Flynn-Wall-Ozawa model, the average E in the first stage were 95.92 kJ/mol and 76.59 kJ/mol, respectively. Those obtained from the Friedman equation for the composites with CGFS content of 10% and 20% were 89.94 kJ/mol and 69.76 kJ/mol, respectively. The first-stage activation energies provided by the KAS model were 91.37 kJ/mol and 71.20 kJ/mol, respectively. These results suggest that the incorporation of CGFS in the first stage could have a catalytic effect on the composite, which might be related to the chemical composition of the CGFS itself. However, in the second stage, the mass conversion rate decreased because of a large amount of unburned char in the CGFS, with the CGFS-2 conversion rate reaching 70%. However, the conversion rate of CGFS-4 was only 60% of that of the pure CGFS-0 sample. So there is no comparable view.45,46 Based on the three model calculations, the instantaneous activation energies of CGFS-2 and CGFS-4 composites tended to increase at the conversion rate α = 0.6 of the comparative sample compared with the pure PVC. This means that CGFS reacted with the chlorine radicals, preventing the formation of HCl during degradation, so the higher E was required for thermal degradation. Meanwhile, it was also suggested that various metal oxides made their own contribution to inhibit PVC degradation.47,48
Conclusions
In this study, the basic physicochemical properties of CGFS were analyzed for the first time. To improve the surface activity of CGFS, its surface was modified by a mechanochemical treatment. The PVC/CGFS composites were then prepared by using a micro twin-screw extruder and an injection molding machine. The mechanical properties, thermal stability and kinetic analysis of thermal degradation were studied, and the conclusions are as follows. (1) The particle size curve of CGFS exhibited a single-peak distribution with an average value of D50 at about 4.479 μm. According to the XRD data, CGFS was mainly composed of an amorphous-silicate glassy phase with oxides such as SiO2 and Al2O3. The TGA curves revealed that the CGFS contained moisture, volatile fractions, and a large amount of unburned carbon. Finally, spherical and irregular particles were identified by SEM in the filler structure. (2) The tensile strength (tensile modulus) and flexural strength (flexural modulus) of the PVC/CGFS composites increased significantly with increasing CGFS content at loads below 20 phr, while they decreased with further increasing CGFS filler content. The impact strength of the CGFS-5 composite reached 47.2MPa, which was 57.1% lower than that of CGFS-0. (3) The addition of CGFS suppressed the mass conversion and improved the thermal stability of the PVC phase at high temperatures, and the final residual mass reached 31%, corresponding an increase of 12.7% compared with that of pure PVC. The kinetic analysis the thermal decomposition showed that CGFS promoted the initial degradation of the composites, while improving their thermal stability in the high-temperature phase. At the same time, the E decreased with increasing CGFS loading in the early degradation phase, indicating the evolution of degradation and its slight inhibition in the later phase.
Supplemental Material
Supplemental Material - Kinetic study of mechanical and thermal properties and thermal degradation of PVC composites based on coal gasification fine slag
Supplemental Material for Kinetic study of mechanical and thermal properties and thermal degradation of PVC composites based on coal gasification fine slag by Yanhua Teng, Kangli Li, Long Chen, Kerui Wang and Changguo Xue in Journal of Thermoplastic Composite Materials.
Footnotes
Acknowledgements
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by Supported by National Natural Science Foundation of China (Grant Nos.12172002), the Key Research and Development Program Projects in Anhui Province (Grant Nos. 202004h07020026), and Anhui Province Innovation Foundation for Returness.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
