Abstract
The suitable bed material is important to inhibit ash-related problems including agglomeration and slagging in furnace and fouling on tail heated surfaces during coal gasification in fluidized bed. For newfound high-alkali vast-reserve Zhundong coal in China, the selection of bed material in fluidized bed should be paid more attention because of its more serious ash-related issues. Five bed materials including quartz, kaolin, corundum, sludge ash, and phosphate rock were used to investigate their feasibility to solve the ash-related issues of two typical types of Zhundong coal including Tianchimulei coal and Shaerhu coal. All of five bed materials could effectively impact the release and transformation of alkali and alkaline earth metal species, especially Na species. The capture order of Na by five bed materials from high to low was sludge ash, quartz, kaolin, corundum, and phosphate rock for the two types of coal, and ash fusion temperatures of residual ash added different bed materials were in orders of phosphate rock > corundum > sludge ash >kaolin > quartz for Tianchimulei coal and corundum > phosphate rock > sludge ash > kaolin >quartz for Shaerhu coal. This influence of bed materials was mainly related to their chemical compositions, especially Al-, Si-, and P-based compounds. Due to the strong capture of alkali and alkaline earth metal-based compounds and high ash fusibility temperature, sludge ash was suggested as the appropriate selection of the bed material during Zhundong coal gasification in fluidized bed. Moreover, this suggestion would contribute to the sewage sludge treatment.
Introduction
Coal is still the most important source of energy in China. With the continuous consumption of coal, the discovery and exploitation of Zhundong coalfield, the largest integrated coalfield in China with an estimated reserve of 390 Gt (Zhang et al., 2013), will offer a strong support for China’s coal industry. However, due to the high Na content in Zhundong coal, severe ash-related problems including slagging and fouling greatly limited this coal utilization (Dai et al., 2015; Wang et al., 2015).
When Zhundong coal was used in industrial coal powder boilers, severe slagging usually occurred in the high-temperature radiation zones (1500–1700℃), and the convection zones were mostly covered by fouling (Dai et al., 2013; Wu et al., 2016). The too high content of alkali and alkaline earth metal (AAEM) elements in Zhundong coal has been considered as the main reason for slagging and fouling (Li et al., 2015, 2016a, 2016b). The phase analysis of slags by X-ray diffraction (XRD) collected from boilers with Zhundong as fuel showed lots of AAEM-rich silicates and aluminosilicates, which melted at ∼1100℃ (Li et al., 2015). The severe slagging on water walls near the combustor will decrease the heat exchange efficiency, resulting in an increase of gas temperature in the furnace exit. The too high gas temperature further causes the tube burst of superheater and reheater because of the overheating. Besides, AAEM elements in low-rank coals are volatile (Quann, 1982), which also is suitable for Zhundong coal. It is reported that most of AAEM and Fe in Zhundong coal have been volatilized at 1000℃ (Li et al., 2016). The evaporated AAEM vapors condense and deposit at 600–800℃ in boiler tails (Wang et al., 2015). Fine ash particles are easily coated by AAEM-rich compounds and then become sticky, which is the reason for stronger fouling tendency of Zhundong coal than other coals (Li et al., 2013; Quann and Sarofim, 1982; Zhou et al., 2013a, 2013b, 2009). Unfortunately, the fouling was only swept away by soot blowers (Bilirgen, 2014), but this method could not work well (Du et al., 2015). The flues will be jammed without effective actions, resulting in unscheduled shutdown.
The above introduction shows that the slagging and fouling characteristics of Zhundong coal are greatly influenced by high temperatures. If the operation temperature is controlled at low levels, the temperature difference between the operation temperature and the melting points of AAEM-rich species present in furnace will increase, which conduces to inhibit the molten slagging. Besides, the low temperature could reduce the volatilization of AAEM elements in Zhundong coal (Li et al., 2016), decreasing the sticky of ash particles and further weakening fouling. Therefore, FB with low operation temperatures (850–950℃) will be a better solution to these ash-related problems of Zhundong coal.
For high Na coals, agglomeration is not an negligible issue when they are used in FB. To solve this problem, mineral additives (Kyi and Chadwick, 1999; Wei et al., 2016) and bed materials (Linjewile and Manzoori, 1999; McCullough et al., 2011; Van Eyk et al., 2012; Vuthaluru et al., 1999; Xu et al., 2014) were reported as the main breakthrough. For example, silicon (Si)-rich minerals were found to be good sorbents for retaining Na-based compounds within coal ash, reducing the possibility of fouling in boiler tails (Kyi and Chadwick, 1999). Aluminum (Al)-rich minerals could increase the melting points of ash particles to inhibit the formation of low-temperature eutectics (Linjewile and Manzoori, 1999; McCullough et al., 2011; Van Eyk et al., 2012; Vuthaluru et al., 1999; Wei et al., 2016; Xu et al., 2014). Phosphorus (P)-rich sludge could effectively retain alkali metals in residual ash in the form of high-melting-point phosphates, avoiding defluidization of biomass in FB (Li, 2014). Besides, the content of Fe2O3, CaO, and MgO in coal ash influenced the ash fusibility a lot (Saikia et al., 2016). Therefore, it can be deduced that the ash-related problems of Zhundong coal in FB will be significantly reduced or even avoided if certain effective mineral additive or bed material could retain Na in the furnace in the form of high-melting-point matters, accompanied by periodically refreshing and removing these Na-rich bed materials (Van Eyk et al., 2012). Overall, an effective and low-cost mineral additive or bed material will be the key to solve above problems during this process.
Although Zhundong coal could be used as a good gasification fuel because of its high volatile content, low ash, and medium heat value (Ye et al., 2016), to date, unfortunately, most of studies on Zhundong coal have been restricted to combustion and few studies have been involved in gasification. Moreover, it was reported that the release of Na would be inhibited under the reducing atmosphere (Kosminski et al., 2006b). This indicates fouling will be alleviated during Zhundong coal gasification, compared with Zhundong coal combustion. In our previous studies (Qi et al., 2016; Song et al., 2016a, 2016b, 2016c, 2016d), slagging and fouling characteristics of Zhundong coal during circulating fluidized bed (CFB) gasification with quartz as bed material were initially proved, thus some measures still need to be taken to solve the ash-related problems ensuring smooth operation.
In this paper, the gasification of Zhundong coal in a vertical tube furnace was conducted to simulate the gas–solid reactions in FB and investigate the influence of five minerals on slagging and fouling of Zhundong coal. The experimental minerals were used as bed materials in FB, not only including some common ones such as quartz, kaolin, and corundum, but also including some uncommon ones such as sludge ash and phosphate rock. Two types of Zhundong coal with different properties were used to study the universality of minerals on the inhibition of slagging and fouling of different-property Zhundong coal. The aim of the study is to select one effective and low-cost mineral as slagging and fouling preventative in Zhundong coal gasification in larger scale FBs.
Experimental section
Fuel
Properties of Zhundong coal.
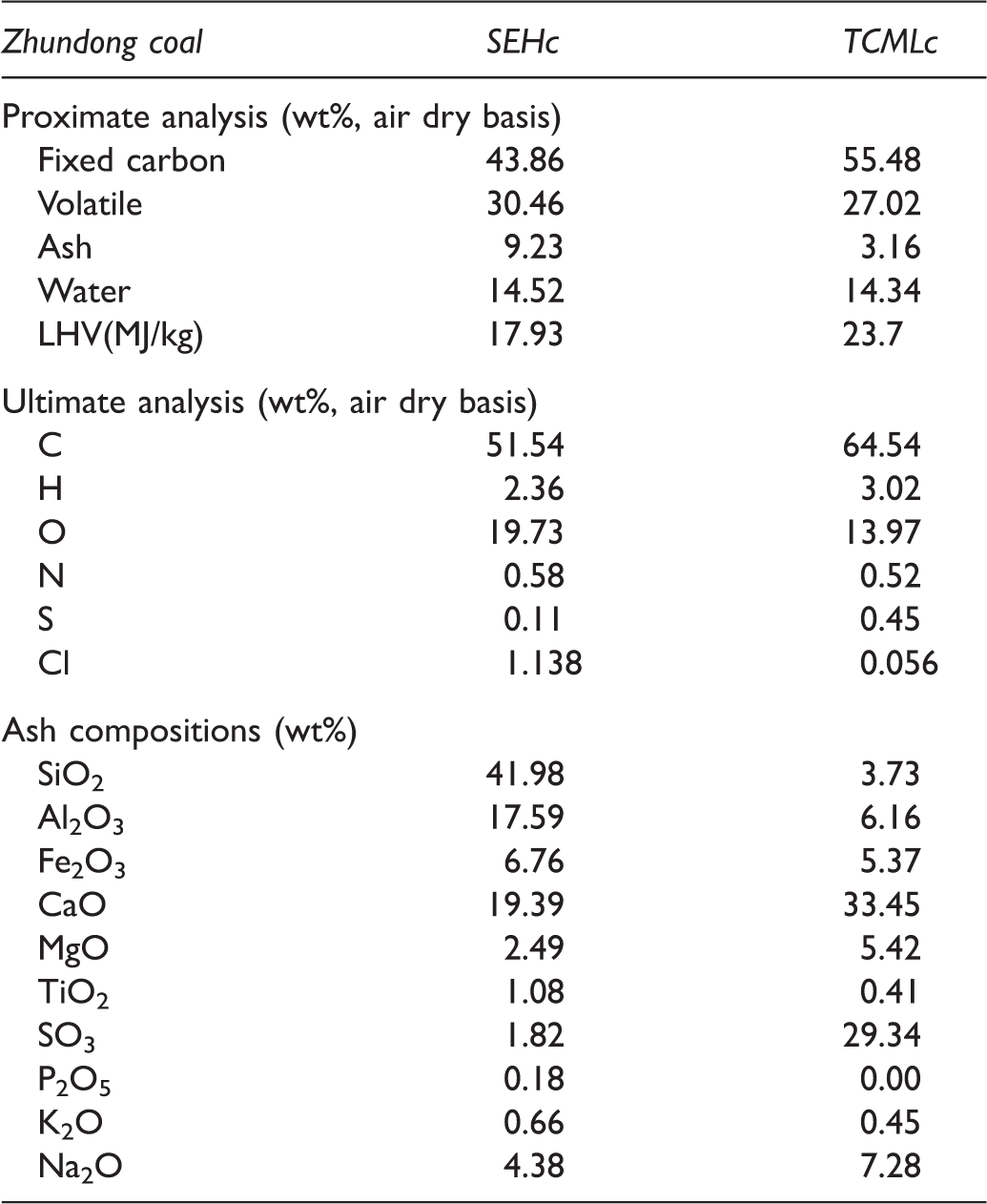
SEHc: Shaerhu coal; TCMLc: Tianchimulei coal.
Bed materials
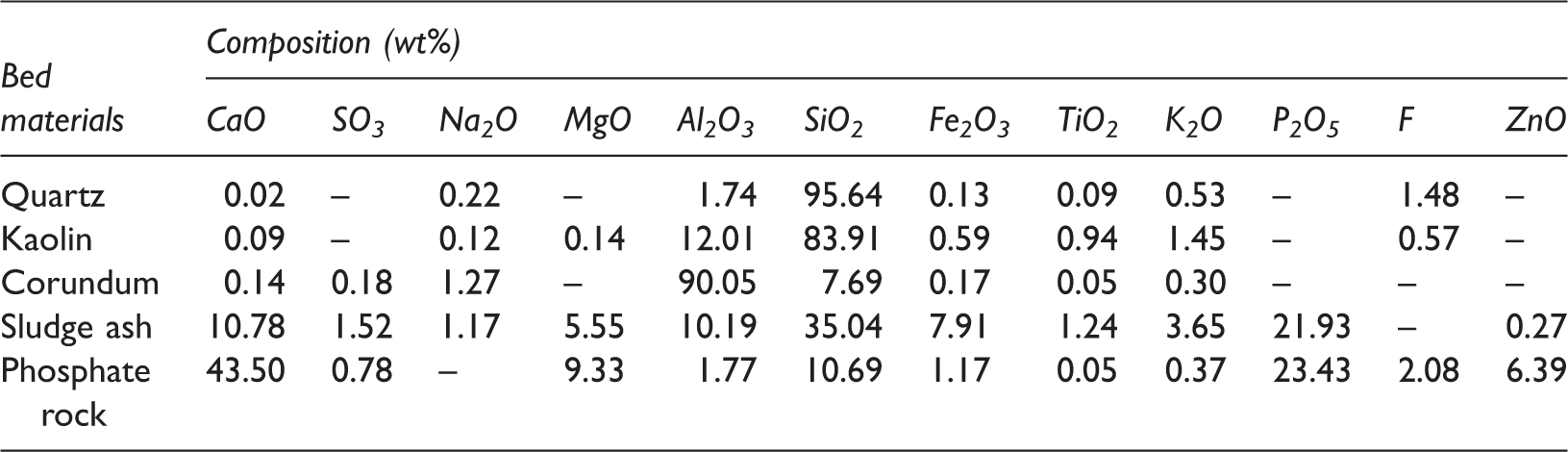
Composition of bed materials.
Mineral phase analysis of bed materials.
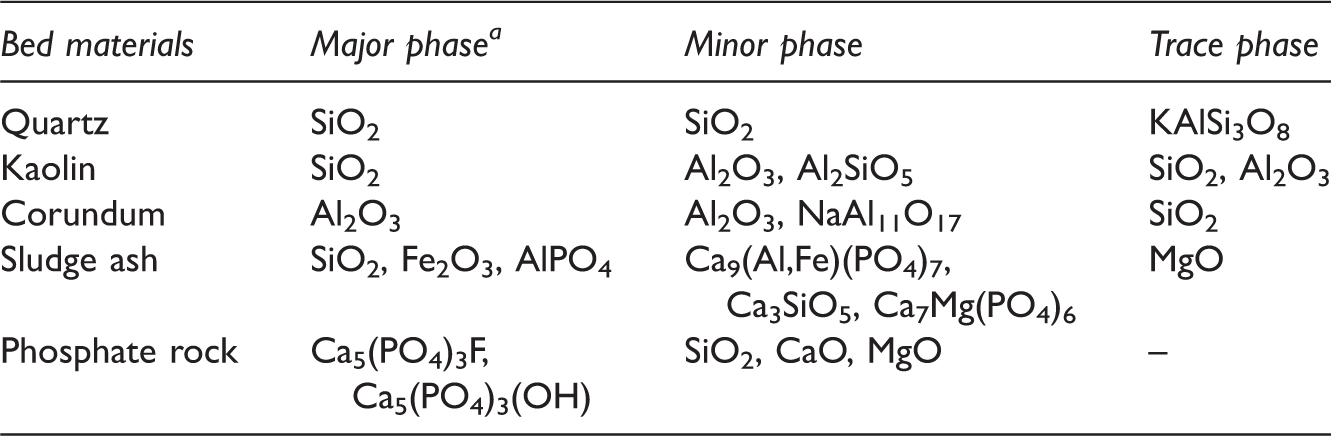
Major (>30%), minor (5–30%), trace (<5%).
The main crystalline phase in quartz is SiO2, accounting for as high as 95.64%. In addition to SiO2, the trace phase of KAlSi3O8 can be detected. For kaolin, SiO2 and Al2O3 are the main components, 83.91 and 12.01%, respectively. Si and Al in the form of Al2SiO5 also occupy certain proportion. Up to 90.05% Al2O3 exists in corundum. In addition, Si in the form of SiO2 and Na in the form of NaAl11O17 are identified. Clearly, the three minerals including quartz, kaolin, and corundum are mainly composed of Si and Al. The influence of Si and Al on ash fusion temperature has been verified (Wei et al., 2016). The three minerals were used here to investigate the influence of Si/Al-based minerals on the slagging and fouling.
Sludge, the by-product of urban sewage treatment, is facing the problem of utilization. As reported, sludge could effectively capture Na forming high-melting-point compounds and then inhibit defluidization during biomass combustion in the FB (Li, 2014). Therefore, sludge ash was selected as a potential bed material. In this paper, sludge was first calcined for 6 h at 500℃ and then the residual ash was selected as the used sludge ash. It can be obtained from Tables 2 and 3 that sludge is a complex mixture containing lots of mineral elements including Si, P, Ca, Fe, and Mg. Main crystalline phases in sludge ash are SiO2, Fe2O3, AlPO4, Ca9(Al,Fe)(PO4)7, Ca3SiO5, and Ca7Mg(PO4)6. The enrichment of Si, Al, and P is a remarkable feature of sludge ash. It is well known that sludge is rich in toxic heavy metals. The volatilization of heavy metals was a troublesome problem during sludge incineration (Werther and Ogada, 1999). Generally, the problem could be reduced or even avoided by adding solid adsorbents into sludge to chemically absorb heavy metals, and further transforming heavy metals into steady metal complexes (Werther and Ogada, 1999; Yao et al., 2004). Complex minerals in Zhundong coal, functioning as clay, offer lots of active sites to react with heavy metal compounds forming stabilized crystal (Liu et al., 2013). Therefore, the problem caused by heavy metals can be neglected during these experiments.
Phosphate rock is a mineral mainly in the form of phosphates, Ca5(PO4)3F and Ca5(PO4)3(OH). Phosphate rock is here for the comparison with sludge ash to investigate the influence of calcium phosphates on inhibiting slagging and fouling of Zhundong coal.
All of the five minerals were crushed and sieved to the size range of 0.063–0.125 mm before each experiment.
Apparatus
The schematic diagram of the vertical tube furnace system is shown in Figure 1. The system mainly consists of a compressed reaction gas source, a temperature-controlled oven, and a reactor. The reaction gas is CO2, which is controlled by a flow meter with a minimum mark of 20 ml/min. The oven with a rated temperature of 1200℃ is divided into two parts, which could be adjusted independently by the temperature controller. The inner dimension of the oven is 66 mm × 50 mm × 420 mm. The height of the reactor from the sintered plate to the outlet is 390 mm, and the inner diameter is 18 mm. The sintered plate plays a role of air distributor in actual FBs. In addition, the air inlet is designed from the top to the bottom of the oven, which provides a long route to reheat the reaction gas.
Schematic diagram of the vertical tube furnace system.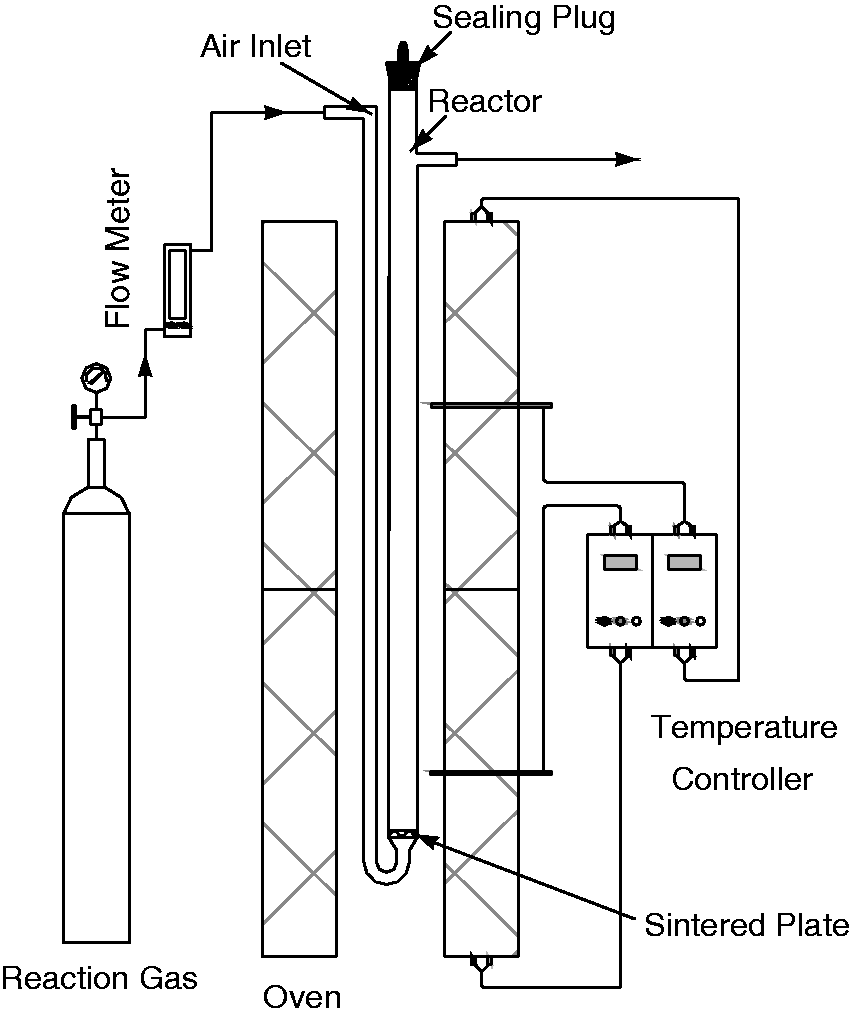
A muffle furnace (CNT, TSX 1400) was also used in this study for ashing samples.
Procedure and analysis
In each experiment, 3 (±0.001) g coal was loaded into the reactor through the sealing plug, and each bed material was mixed with the coal sample as 1% by mass basis. The oven temperature was set at 900℃. The reaction gas of CO2 was controlled at 500 (±20) ml/min. When the temperature was stable at 900℃, the reactor was quickly put into the oven. The gasification time was about 60 min. After that, the reactor was withdrawn from the oven and still cooled by CO2 to room temperature. Each experiment was conducted for a few times in order to obtain enough samples.
The samples collected from the vertical tube furnace were weighed and sealed as the standby. Some operations including digestion, evaporation of acid, and dilution were first done in order to detect Na contents in these samples using an ion chromatograph (IC, ICS-900, DIONEX). About 0.1 (±0.001) g solid sample was dissolved with 11 ml digestion solution (HNO3: HF: H2O2 = 6: 3: 2) at 210℃ for 25 min in a microwave digestion instrument. Then the solution was removed from the microwave digestion instrument and heated on the heating plate at 150℃ for 2 h to completely evaporate the acid. The solid residues were dissolved and diluted by ultrapure water to 50 ml before liquid sample injection into IC. In addition, the gasification residual was analyzed by a field emission scanning electron microscope (FESEM, S-4300, HITACHI), accompanied with an energy dispersive spectroscopy (EDS, HITACHI), to obtain the microstructure and element distribution.
Before further analysis, gasification samples should be calcined at 575℃ for 2 h to eliminate unconverted carbon interference in analysis. Mineral phase analysis was conducted on the calcined samples by an XRD (Empyrean, PANalytical B.V.). The measurement conditions were listed as follows: Anode material was Cu, generator voltage was 40 kV, tube current was 40 mA, scan step size was 0.026°, scan range was from 10° to 90°, and scan time was 4.16 min. Besides, ash fusion temperatures of calcined samples were measured by an ash fusibility analyzer (AFA, AF700, LECO) to investigate the influence of bed materials on the ash fusibility. The test method was referred to the standard GB/T219-2008, and the test was conducted under reducing atmosphere, i.e., 40% CO2 and 60% CO. The results of Table 2 were obtained by an X-ray fluorescence spectrometer (XRF, AXIOS-MAX, PANalytical B.V.).
Results and discussion
Retention of mineral elements
Since Zhundong coal is low rank, most of AAEM elements in coal are easily volatile (Quann, 1982). AAEM-based compounds could be captured by chemical reactions between these compounds and bed materials and stay on the surface of bed material particles. To accurately calculate the AAEM retention because of the addition of bed materials to coal, the mass loss of bed materials was first calculated after heating for 60 min at 900℃, shown in Figure 2.
Mass retention of five minerals after heating for 60 min at 900℃. A: quartz, B: kaolin, C: corundum, D: sludge ash, and E: phosphate rock.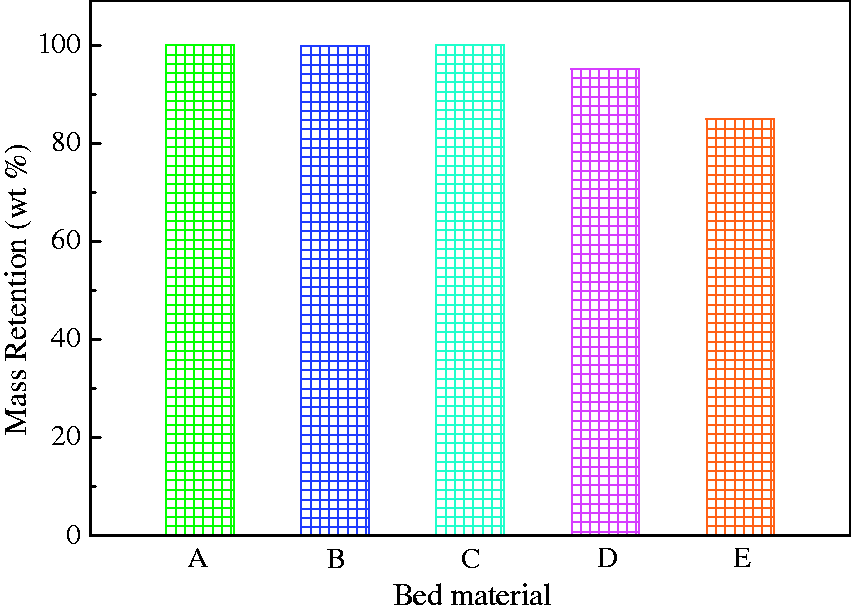
For bed materials of quartz, kaolin, and corundum, little mass loss was observed because the three minerals mainly consist of Al2O3 and SiO2 with a high heat stability. For the used sludge ash, a higher temperature (900℃) enhanced the volatilization or decomposition of mineral salts in the sludge ash prepared at 500℃, resulting in a small amount of mass loss, only 4.8%. However, the decomposition of Ca5(PO4)3(OH) in phosphate rock, listed as follows, caused the highest mass loss among the five bed materials
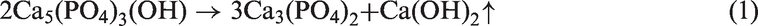
To some degree, the capture capacity of AAEM species by bed materials could be reflected by the mass change of Zhundong coal during the gasification process. The mass retention of Zhundong coal (Rcoal, %) caused by bed materials could be calculated by the following formula (2), supposing that the mass loss of bed materials during this process was the same as the results of Figure 2
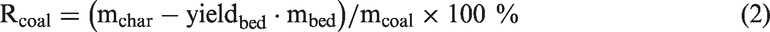
Na is the most important element for slagging and fouling (Song et al., 2016a, 2016b, 2016c, 2016d). Therefore, the capture capacity of Na by bed materials should be focused. The Na retention (RNa, %) could be calculated by formula (3), assuming that the Na content in bed materials remained the same as the results of Table 2
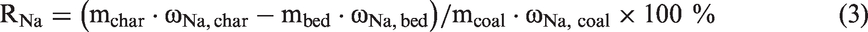
The mass retention of Zhundong coal after gasification at 900℃ for 60 min is shown in Figure 3. The mass retention of the two types of Zhundong coal with bed materials is higher than that without bed materials, which indicates the capture of inorganic elements by five bed materials did exist during the gasification. Although the ash content of TCMLc is lower than that of SEHc as shown in Table 1, the corresponding mass retention of TCMLc is higher than that of SEHc, regardless of with bed materials or not. This could be attributed to the stronger volatility of minerals in SEHc. As shown in Table 1, Cl content in SEHc is as high as 1.279%. Mineral chlorides are usually of lower melting points and strong volatility, compared with other minerals (Niu et al., 2016), finally resulting in a lower mass retention of SEHc. For TCMLc, however, the difference value between the average mass retention with bed materials (40.86%) and one without bed materials (32.41%) is lower than that for SEHc (36.15 and 26.68%, respectively). This can also be contributed to the stronger volatility of SEHc: The mass change will be more obvious for compounds with a stronger volatility once their volatilization was suppressed.
Mass retention of Zhundong coal after gasification at 900℃ for 60 min.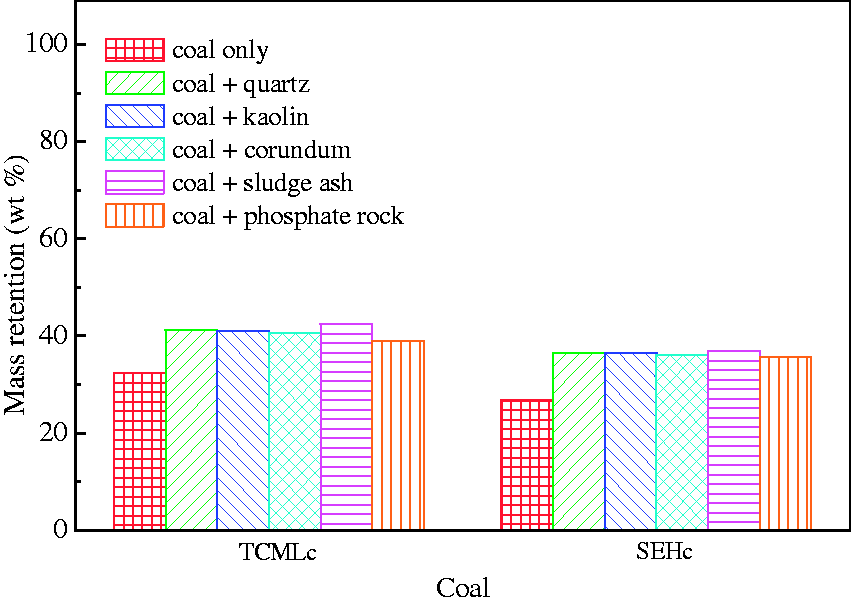
The differences in coal mass retentions among the five bed materials probably indicate the capture of AAEM elements by these minerals was selective. To verify the selective capture, the distribution of mineral elements on the particle surface of bed materials was analyzed by FESEM–EDS. The analysis was only conducted for the cases of SEHc at 1000 amplification, shown in Figures 4 to 8. It should be noted that each element concentration can be reflected by the corresponding color depth.
Mineral element distribution on the surface of quartz particle. Mineral element distribution on the surface of kaolin particle. Mineral element distribution on the surface of corundum particle.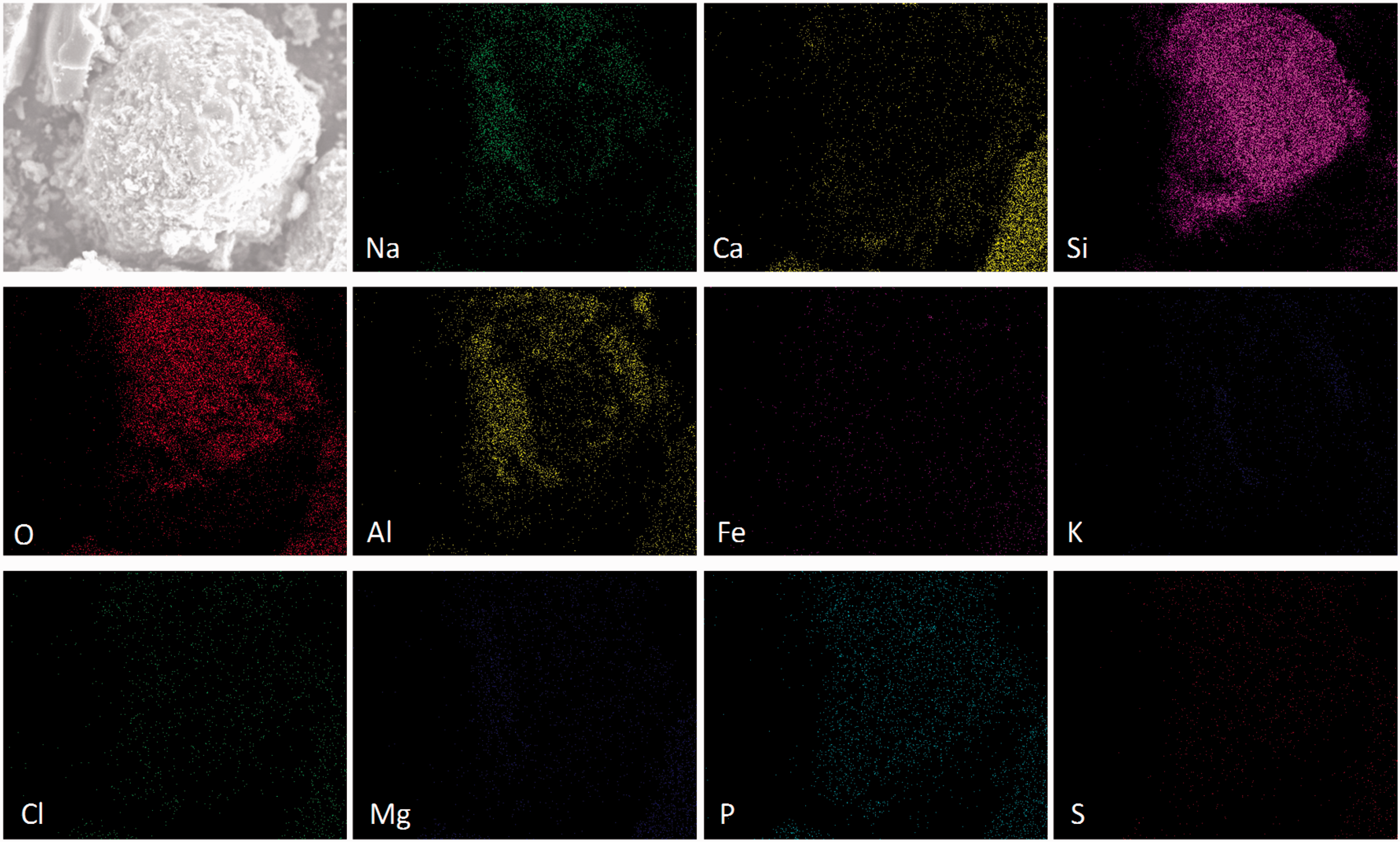
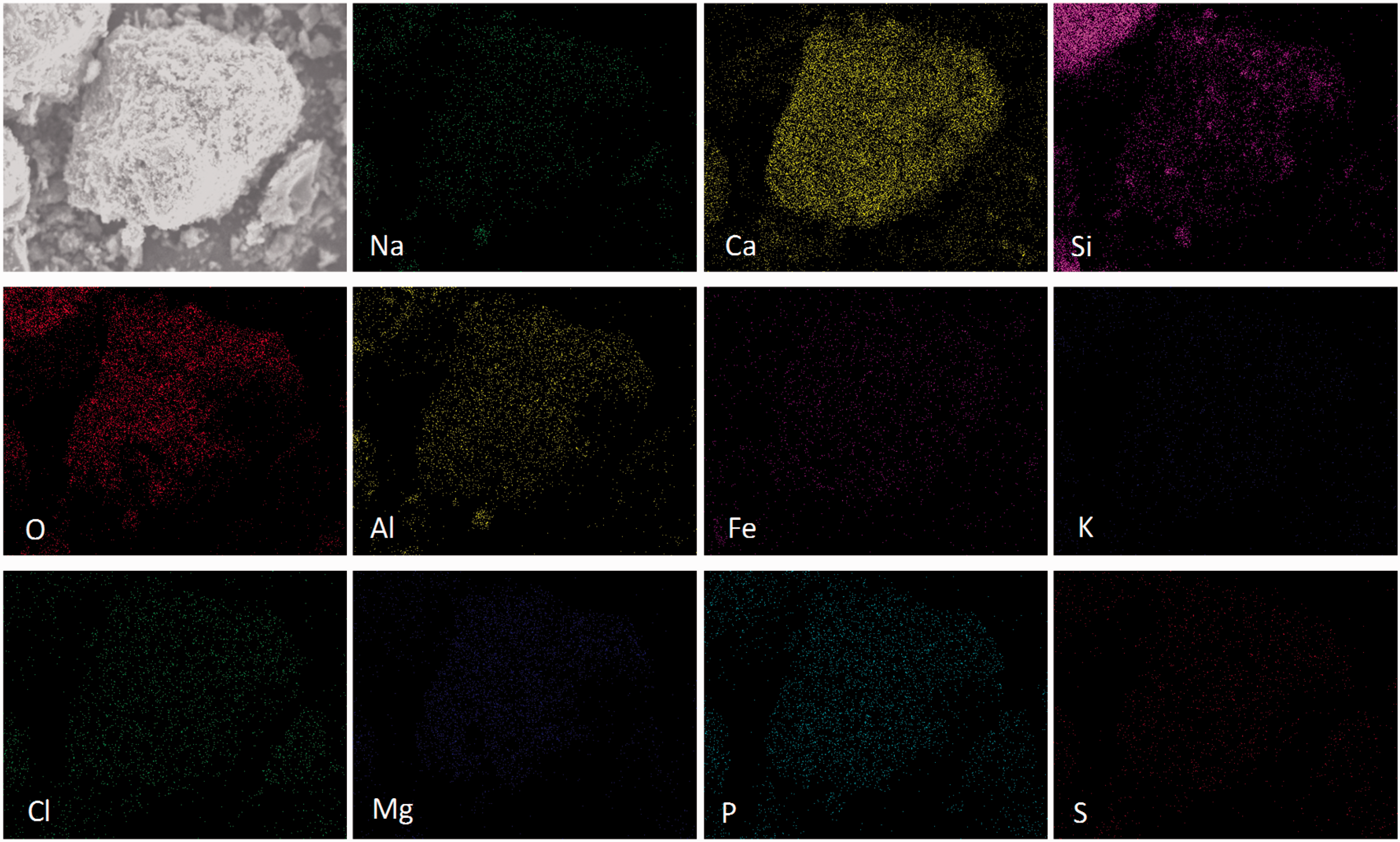

As shown in Figure 4, in addition to Si and O, another three mineral elements including Al, Na, and Ca were enriched on the particle surface of quartz. The quartz particle without any corners might be coated by these captured mineral species. The microstructure and mineral element distributions of the kaolin (Figure 5) are similar to those of the quartz because of their similar compositions shown in Table 2. On the particle surface of kaolin, however, the color of Na was slightly light and those of Ca and Mg were deepened. For corundum mainly composed of Al2O3, the light color of mineral elements other than Al indicates the capture of mineral elements by corundum was not strong, as shown in Figure 6. In addition, the corundum particle was still in a regular shape with corners due to its high thermal inertia.
It could be concluded based on the results of Figures 4 to 6 that the capture capacity of minerals by bed materials was influenced by the ratio of Si/Al in bed materials. Especially, the influence on Na was more obvious: The larger the ratio of Si/Al was, the stronger the capture capacity was. Although no P was detected in quartz, kaolin, and corundum, the presence of P on particle surfaces indicates the capture of P by Si/Al-based minerals existed.
It is observed from Figure 7 that lots of mineral elements including Na, Ca, Si, Al, Mg, and P were enriched on the particle surface of sludge ash. The enrichment of mineral elements was not only related to the high content of mineral elements in sludge ash, but also related to the strong capacity of sludge ash for capturing these mineral elements. The strong capturing capacity also resulted in a high mass retention of Zhundong coal with sludge ash as bed material after gasification (Figure 3). It could be obtained from Table 2 that phosphate rock contains more P than sludge ash. However, phosphate rock with a higher P content failed to cause a stronger capture, indicating that the P content is not the determining factor. Due to lack of interaction with minerals in Zhundong coal, the phosphate particle was still in the original shape. It is worth noting that lots of Si and Al in addition to P were also present in sludge ash, and as reported, the two mineral elements (Si and Al) had a strong ability to capture Na (Kyi and Chadwick, 1999). Therefore, it could be concluded that Si, Al, and P promoted together the high mass retention for the case of sludge ash.
Mineral element distribution on the surface of sludge ash particle. Mineral element distribution on the surface of phosphate rock particle.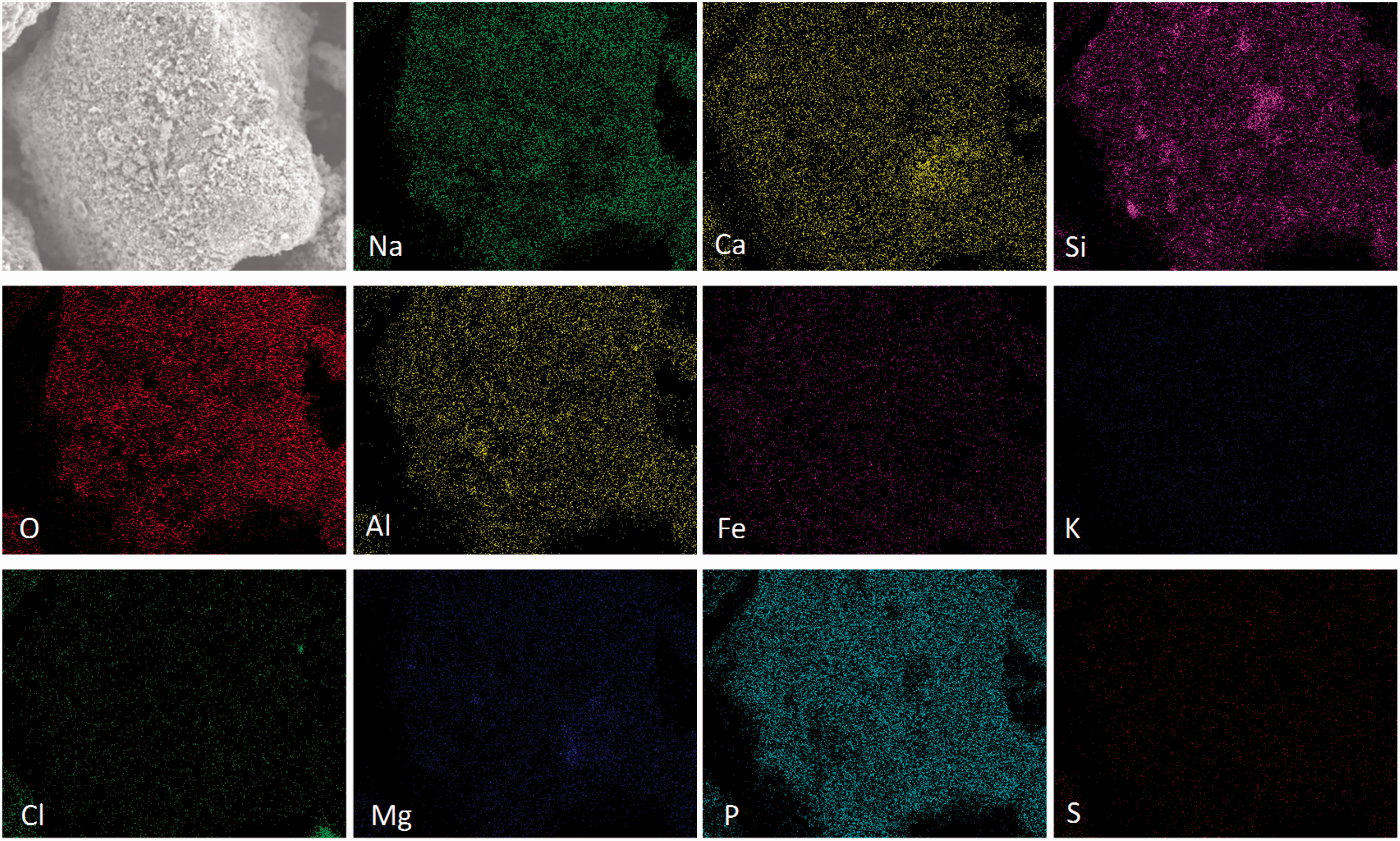
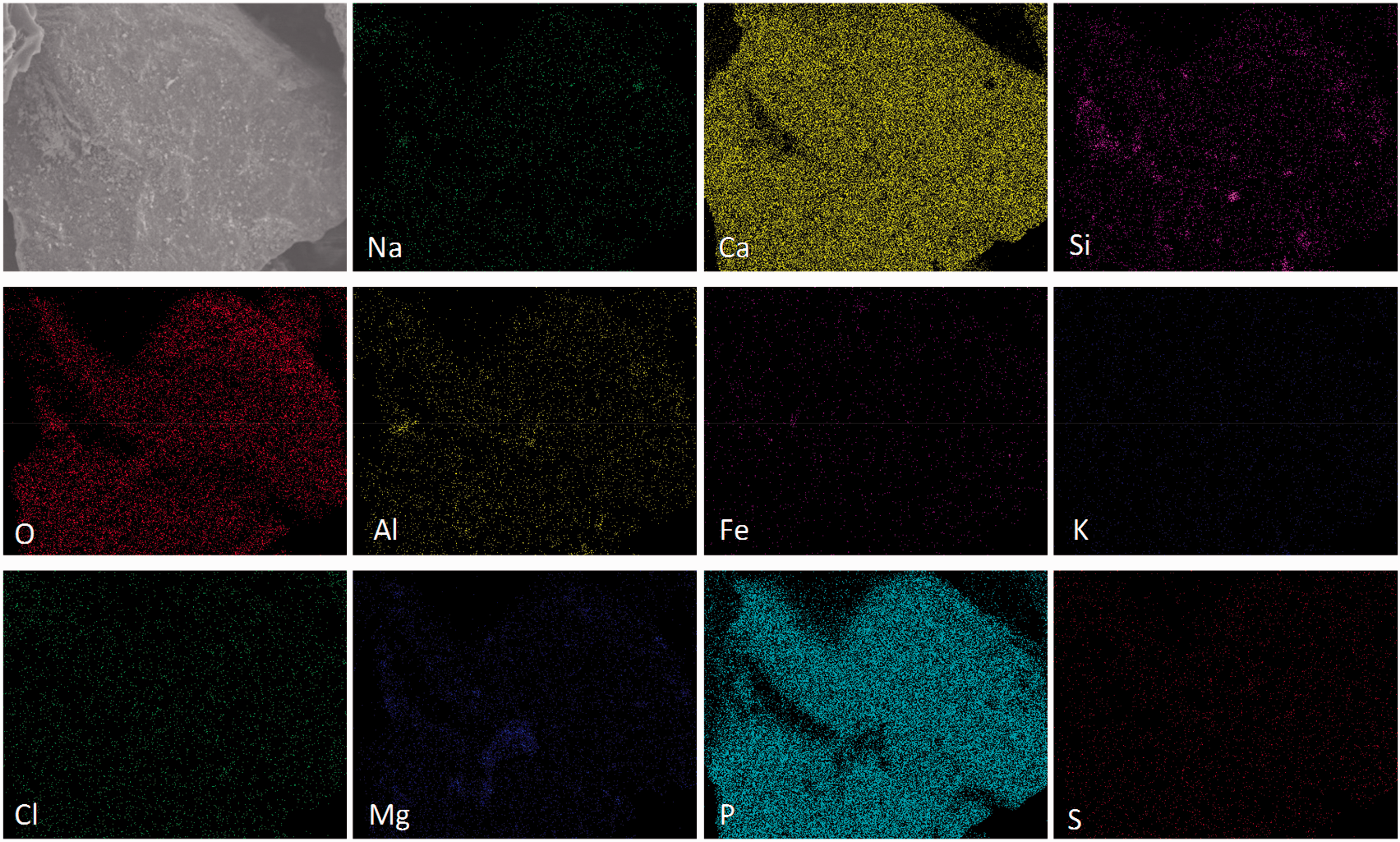
An obvious interaction between minerals and coal corresponds to a high mass retention (Kyi and Chadwick, 1999). Both the results of Figures 3 and 4 to 8 consistently show that all of the five bed materials had a capturing effect on mineral elements. For the high Na coal, the capture of Na by bed materials should be the center of attention, which is given in Figure 9. The results show a more obvious influence of bed materials on Na retention than that on mass retention, compared with the results of Figure 3. For TCMLc, the Na retention increased from 67.03 to 80.48–89.47%. The capture order of Na from high to low is sludge ash, quartz, kaolin, corundum, and phosphate rock. For SEHc, the Na retention increased from 56.46 to 76.42–81.56%, and the order from high to low is also sludge ash, quartz, kaolin, corundum, and phosphate rock. The calculations agree with the results of FESEM-EDS and indicate the five bed materials have a same influence on the two types of Zhundong coal.
Na retention after gasification at 900℃ for 60 min.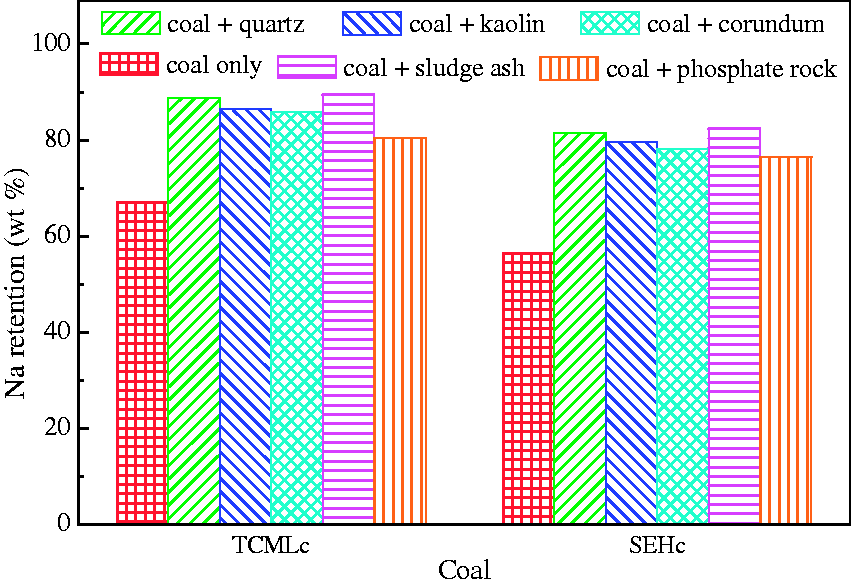
Reactions between minerals and Zhundong coal ash
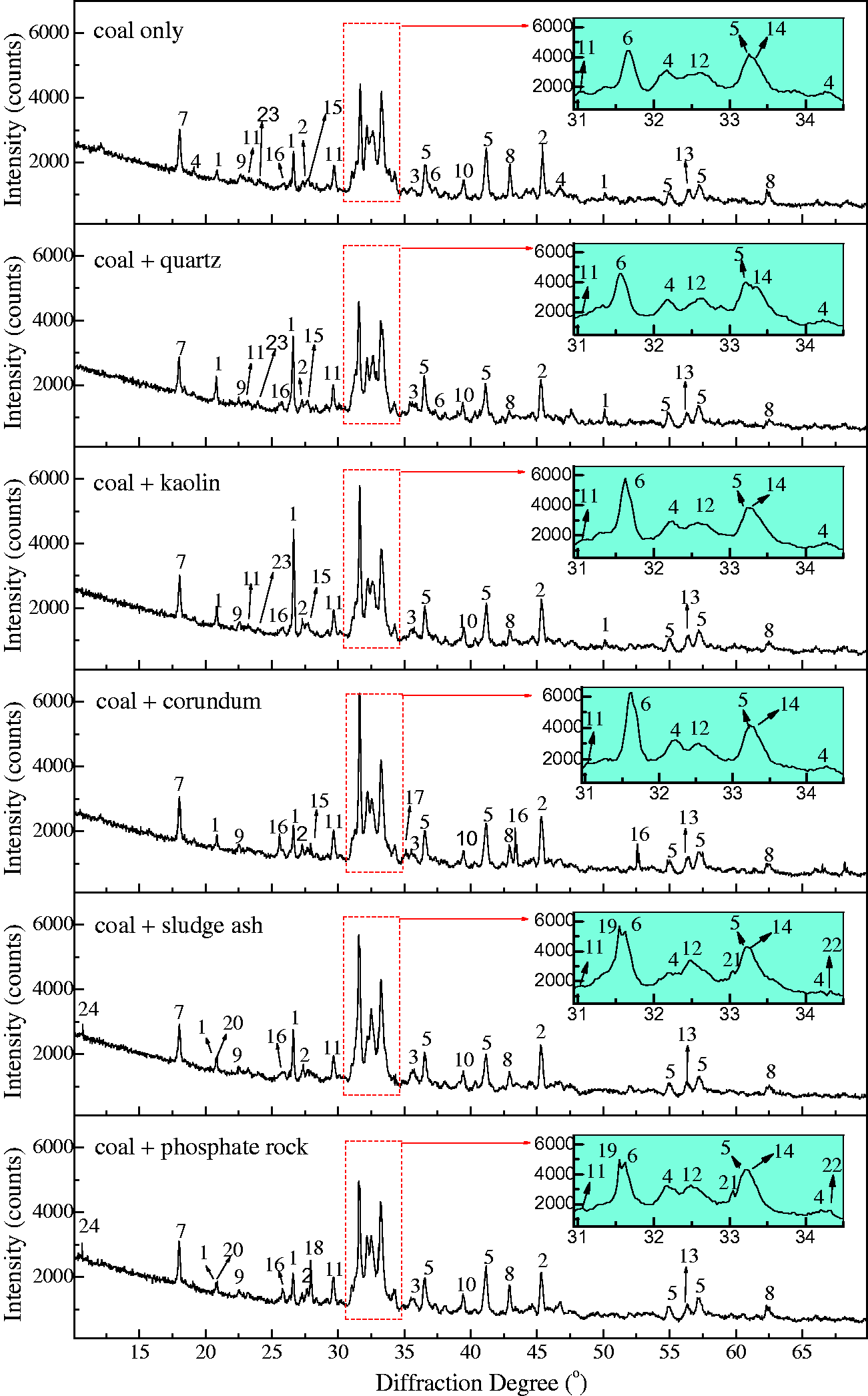
Although the five bed materials have a capturing effect on AAEM species in Zhundong coal, this effect will bring severe ash-related problem (slagging or agglomeration) if the retaining AAEM-based compounds have low melting points (Kosminski et al., 2006b). Therefore, the slagging characteristics of residual ash are greatly influenced by the existing form of these retaining mineral species. Here, the mineralogical phases of residual ash with/without bed materials were analyzed by XRD, shown in Figures 10 and 11.
XRD patterns of TCMLc ash mixed with minerals. 1—CaSO4, 2—CaO3–MgO, 4—Ca2Al2SiO7, 5—(CaO)12(Al2O3)7, 6—NaAlSi2O6, 7—Na2Si2O5, 8—Na2SO4, 9—Al2Ca3(SiO4)3, 10—Al2O3, 11—Ca2SiO4, 12—Ca3SiO5, 13—NaAlSiO4, 14—Ca2Fe2O5, 15—Na3.12Fe2.44(P2O7)2, 16—Na3Al2(PO4)3, 17—Ca5(PO4)3Cl, 18—Na7Ca4.5(P2O7)4, 19—Ca3Fe2(SiO4)3, 20—NaAlSi3O8. XRD: X-ray diffraction. XRD patterns of SEHc ash mixed with minerals. 1—SiO2, 2—NaCl, 3—Fe2O3, 4—Ca2SiO4, 5—Ca3Fe2(SiO4)3, 6—Ca2Al2SiO7, 7—(CaO)12(Al2O3)7, 8—MgO, 9—Al0.5Si0.75O2.25, 10—CaO, 11—KNa3(AlSiO4)4, 12—Na2Ca2Si2O7, 13—MgSiO3, 14—NaFeO2, 15—CaAl2Si2O8, 16—Al2O3, 17—NaAlO2, 18—Ca3(PO4)2, 19—Ca5(PO4)3Cl, 20—Na2Al3(PO4)3, 21—Na2Fe3(PO4)3, 22—Ca5(PO4)2SiO4, 23—Na2Si2O5, 24—Na3.12Fe2.44(P2O7)2. SEHc: Shaerhu coal; XRD: X-ray diffraction.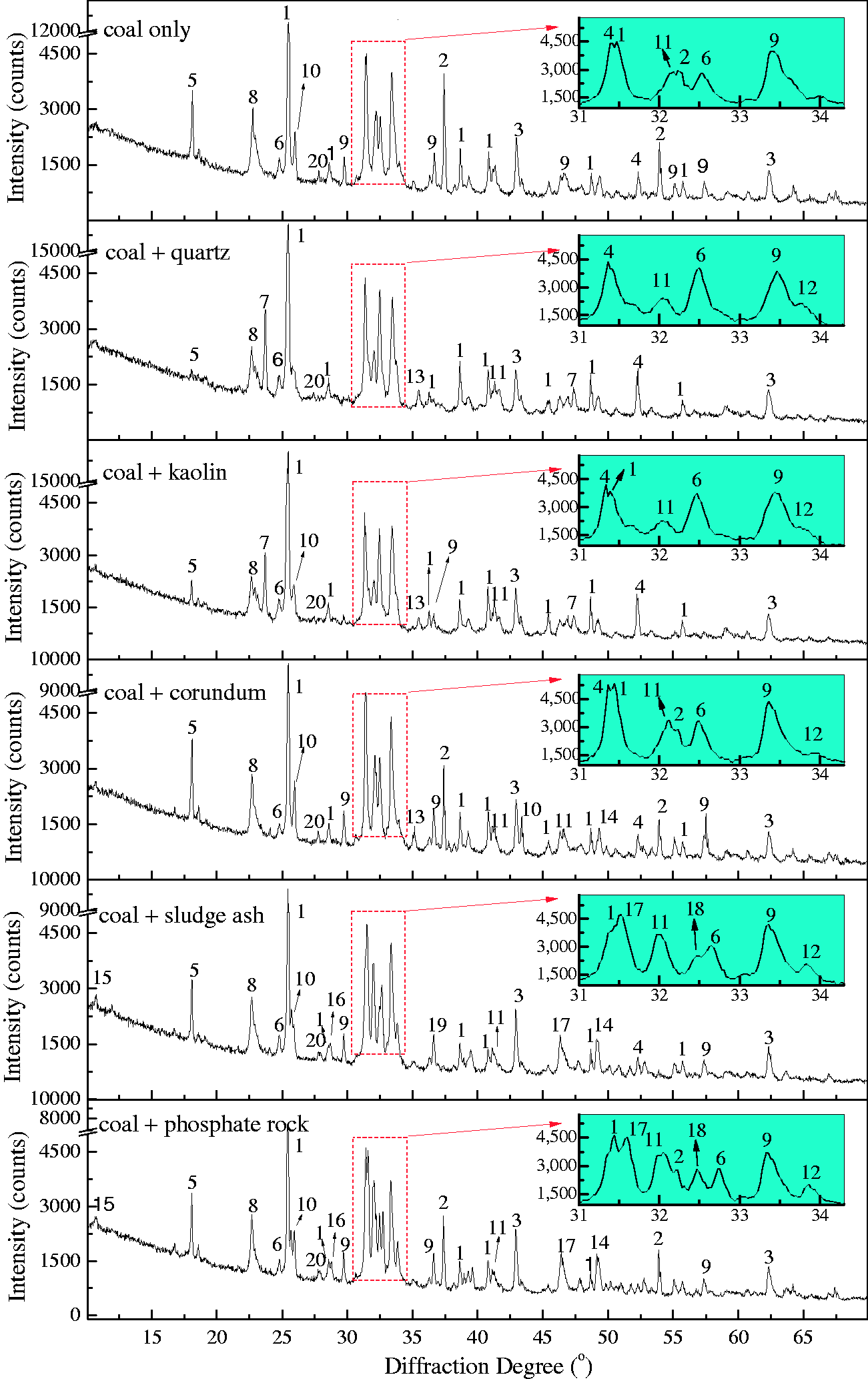
For TCMLc, the top three mineral elements are Ca, S, and Na. At 900℃, the three mineral elements mainly existed in the form of CaSO4, CaO, and Na2SO4. CaO was generated from the decomposition of calcite (CaCO3) in Zhundong coal (reaction (4)). Without bed materials, reactions among mineral species in coal ash occurred, resulting in the formation of (CaO)12(Al2O3)7, Ca2Al2SiO7, Al2Ca3(SiO4)3, Ca2SiO4, NaAlSi2O6, and NaAlSi3O8 (reactions (5) to (10))

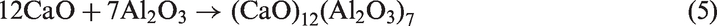
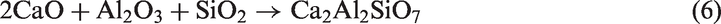
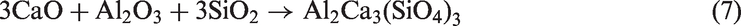
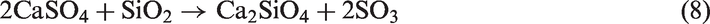
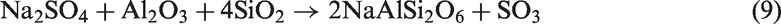
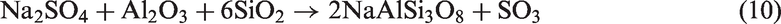
Therefore, the residual Na (67%) in gasified char without bed materials was possibly in the form of Na2SO4 and NaAlSi2O6. The melting point of Na2SO4 is 884℃. NaAlSi3O8 and NaAlSi2O6 melt at 1000–1100℃. Besides, these Na-based compounds are nonvolatile, easily decreasing the ash fusion temperature of Zhundong coal ash.
When SiO2-rich quartz and kaolin were added into TCMLc, no SiO2 were detected by XRD, indicating the two bed materials were easily consumed by TCMLc. The quick consumption of SiO2 might be ascribed to the low SiO2 content in TCMLc ash, only 3.73%. Compared with the case without bed materials, for those cases with quartz and kaolin as bed materials, the CaO peak disappeared, the peak intensity of Na2SO4 was weakened, whereas peaks of Ca3SiO5 and Na-based compounds including Na2Si2O5 and NaAlSiO4 appeared, and the peak intensity of NaAlSi2O6 was increased. Hence, possible reactions consuming SiO2 might occur including those listed below (reactions (11) to (13))

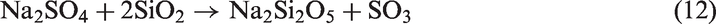
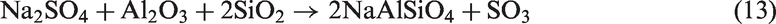
In addition to Na2SO4, the NaCl and organic Na in TCMLc also possibly reacted with SiO2 forming the above Na-based compounds (Kosminski et al., 2006a, 2006b). Since these formed Na silicates or aluminosilicates were nonvolatile, the two retentions including residual char mass and Na increased. However, these formed low-melting-point compounds, especially Na silicates such as Na2Si2O5 with a melting point of as low as 874℃ (Kosminski et al., 2006b), would enhance slagging.
Al2O3 in corundum could steadily exist in the residual ash when corundum was added in the prepared TCMLc as the bed material. The high peak of Na2SO4 indicates the reactions between Na2SO4 and Al2O3 were not obvious. However, the CaSO4 peak intensity was decreased, and the peak intensity of compounds containing Al, such as (CaO)12(Al2O3)7, Ca2Al2SiO7, and Al2Ca3(SiO4)3, was increased. This indicates CaSO4 in TCMLc ash reacted with corundum to form the above compounds (reactions (14) to (16))
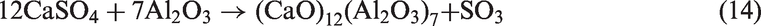
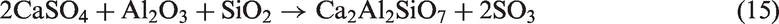
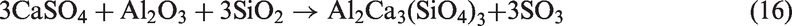
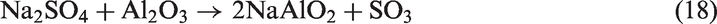
When sludge ash and phosphate rock were used as bed materials, more P-based compounds including phosphates (Na3Al2(PO4)3 and Ca5(PO4)3Cl) and pyrophosphates (Na3.12Fe2.44(P2O7)2 and Na7Ca4.5(P2O7)4) were present in TCMLc ash. From Table 3, sludge ash and phosphate rock are rich in phosphates. It was reported that the affinity between mineral elements including Ca, Na, Mg, Fe, and Al and the phosphate group (PO43−) was strong (Li, 2014). At 900℃, these mineral elements and the phosphate group would easily combine together in the form of phosphates. Moreover, under SiO2- and carbon (C)-rich conditions, phosphates could be transformed into pyrophosphates (Arnout and Nagels, 2016). During this gasification, reactions (19) to (21) are the reason for the presence of pyrophosphates in the residual ash
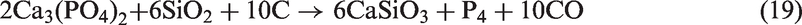

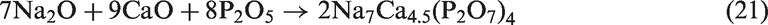
For SEHc, more mineral species including SiO2, NaCl, Ca2SiO4, Ca3Fe2(SiO4)3, Ca2Al2SiO7, and (CaO)12(Al2O3)7 were present in SEHc ash at 900℃, compared with TCMLc ash. These complex mineralogical phases in SEHc ash were partly attributed to the high contents of Si, Ca, Al, Fe, and Na in SEHc. The Cl content in SEHc is up to 1.279% shown in Table 1. This indicates that Na and Cl in SEHc were mainly existed in the form of NaCl (Song et al., 2016a, 2016b, 2016c, 2016d). Under the action of SiO2 and other minerals, Na silicates and aluminosilicates were formed by the following reactions (22) to (24)
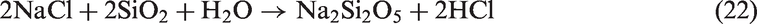
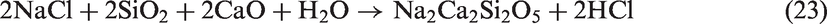
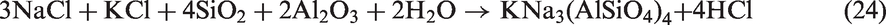
The Na and Fe oxide, NaFeO2, were also detected in SEHc, indicating Na-based compounds easily reacted with Fe2O3 to form eutectics. These eutectics and Na silicates have low melting points, promoting slagging.
Since the SiO2 content in SEHc ash is up to 41.98% (Table 1), the join of SiO2-rich bed materials such as quartz and kaolin failed to promote the interaction between bed materials and SEHc ash. Therefore, the join of quartz and kaolin only caused a stronger SiO2 peak intensity other than the formation of new minerals. On the contrary, the interaction was enhanced when Al2O3-rich corundum was added as the bed material. For example, the SiO2 peak intensity was weakened, aluminosilicates (such as Ca2Al2SiO7 formed by reaction (6)) peak intensity was strengthened, and Na aluminates (NaAlO2 formed by reactions (17) and (18)) were generated.
When sludge ash and phosphate rock were added into SEHc ash, more phosphates and pyrophosphates were formed. Meanwhile, more Na was retained in the residual ash in these two forms. For the high-Cl coal such as SEHc in this paper, Cl-caused corrosion should be focused (Enestam et al., 2013; Miltner et al., 2006). However, the two P-rich bed materials including sludge ash and phosphate rock could reduce the corrosion by inhibiting the release of Cl, as shown by reactions (25) and (26) (Li, 2014). Besides, due to the presence of SiO2 in SEHc, SiO2 also reacted with phosphates to form silicophosphates by reaction (27)
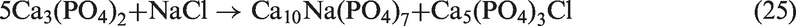

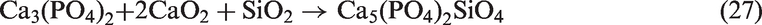
Ash fusibility
Slagging characteristics of Zhundong coal could be reflected by its ash fusion temperature to a certain degree. Ash fusion temperatures of Zhundong coal ash added bed materials are shown in Figure 12. Only in terms of the melting temperature (MT, which is considered to equal the softening temperature in this paper), the two types of Zhundong coal with MTs of 1315 and 1343℃, respectively, are not considered as the serious slagging coal, which failed to match the serious slagging characteristics of Zhundong coal during combustion in actual boilers (Zhou et al., 2014). This could be attributed that the AAEM species (mainly referring to Na) have volatilized completely because of the slow heating rate (about 5℃/min) during measuring MTs. In actual boilers, the heating rate is very high, hundreds of degree per second, resulting in little AAEM elements volatilization within such a short time. Moreover, the volatilized AAEM species would be captured by solid particles under the drastic gas–solid disturbance. Finally, the high AAEM element retention caused serious slagging of Zhundong coal in actual boilers.
Ash fusion temperatures of Zhundong coal ash mixed with minerals: (a) TCMLc and (b) SEHc.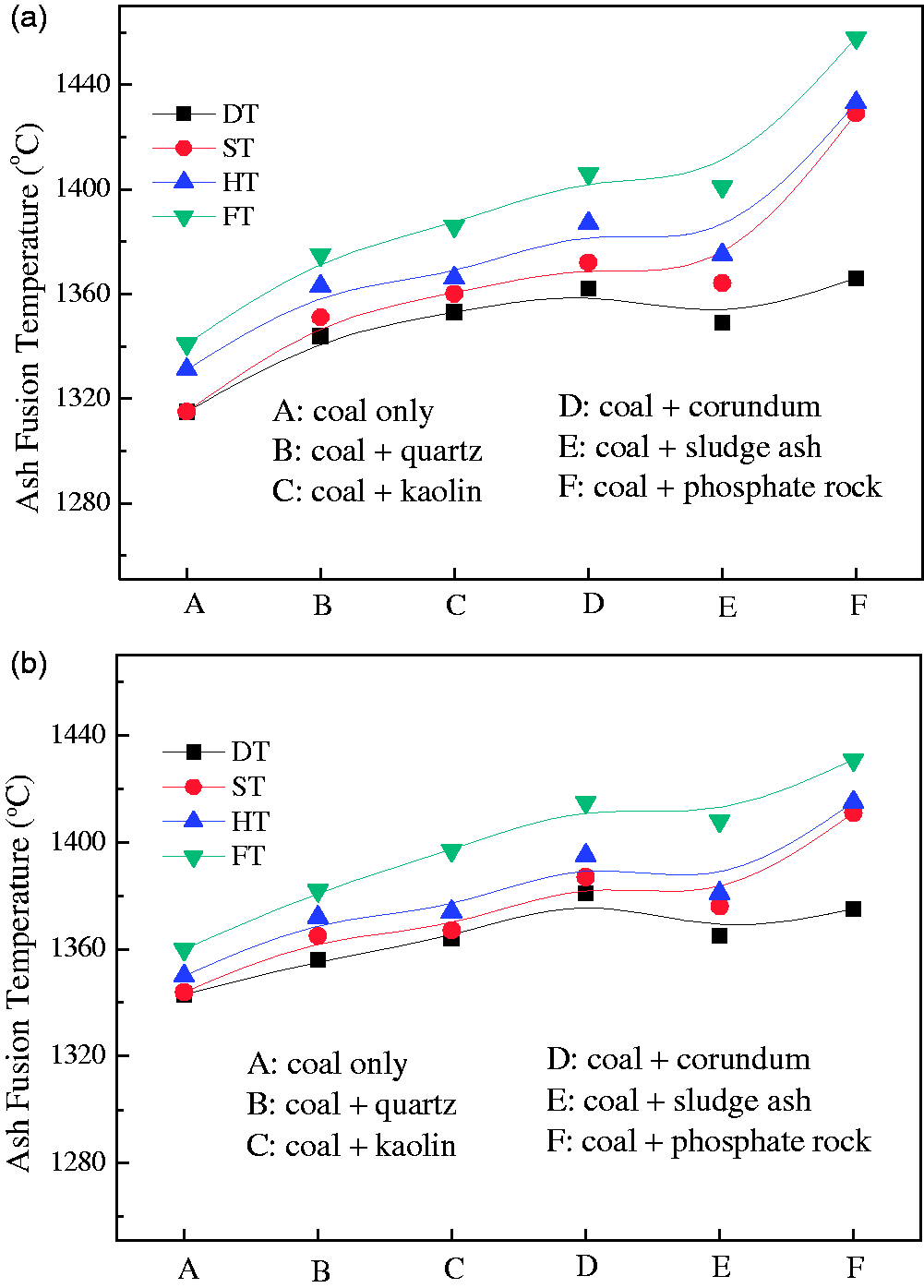
From properties of bed materials shown in Tables 2 and 3, quartz and corundum could be approximately considered as pure SiO2 and Al2O3, respectively, of which MTs are 1650 and 2050℃, respectively. The composition of kaolin is somewhere in between those of quartz and corundum and closer to that of quartz. Thus, the MT of kaolin is considered slightly above 1650℃. The MT of sludge ash measured by AFA is 1336℃. Ca5(PO4)3R (R refers to F or OH) is the main compound in phosphate rock. At high temperature, Ca5(PO4)3R would be changed to Ca3(PO4)2 with the MT of 1391℃ by reaction (1). From Figure 12, it could be concluded that all the five bed materials improved the ash fusion temperature of the two types of Zhundong coal. Although MTs of the five bed materials could be ordered as (from high to low, the same below) corundum, kaolin, quartz, phosphate rock, and sludge ash, MTs of Zhundong coal ash added bed materials were ordered as phosphate rock, corundum, sludge ash, kaolin, and quartz for TCMLc, and corundum, phosphate rock, sludge ash, kaolin, and quartz for SEHc. The order difference between bed materials and Zhundong coal ash added bed materials indicates the join of bed materials greatly influenced the ash fusion temperature of Zhundong coal.
Since the amount of coal used in actual boilers is huge, the amount of AAEM-based compounds in coal, which should be captured for avoiding fouling in boiler tails, is far above that in these experiments. The bed material would face more severe challenge of slagging or agglomeration because of the enrichment of AAEM species. Therefore, in actual boilers, the selected bed materials not only capture AAEM species decreasing the possibility of fouling on the tail surfaces for maximum efficiency, but also improve the ash fusion temperature of materials in furnace as high as possible avoiding the risk of slagging and agglomeration at high temperature. Although the used quartz and kaolin rich in SiO2 effectively increased Na retention in ash, the excessive consumption of SiO2 in a short time caused the formation of low-melting-point compounds easily, thereby resulting in slagging. Hence, the two minerals were not suggested as bed materials during Zhundong coal utilization in industrial FB. Similarly, phosphate rock was also not suggested as bed materials because of its lowest Na retention among the five minerals. By contrast, corundum and sludge ash were suggested as bed materials in industrial FB in terms of their higher Na retentions and better ash fusibility. Moreover, it should be noted that the utilization of sludge ash as the bed material or additive during Zhundong coal gasification in FB could greatly alleviate the challenge of sewage sludge treatment. Therefore, sludge ash is considered as the optimum selection as the bed material, of which the performance should be further verified in larger scale FBs.
Conclusions
Five bed materials including quartz, kaolin, corundum, sludge ash, and phosphate rock were investigated in lab-scale test apparatuses to verify their feasibility as the bed material on preventing slagging and fouling during Zhundong coal gasification in FB. The main conclusions are listed below.
The theoretical calculation and SEM-EDS analysis illustrate that the used bed materials effectively captured AAEM species in Zhundong coal, and their capturing capacities varied little with Zhundong coal properties. The high capturing capacity would inhibit the alkali metal release and then reduce the risk of fouling on tail heated surfaces. The order of capturing capacity of Na (from high to low) was sludge ash, quartz, kaolin, corundum, and phosphate rock. The XRD analysis shows that the mechanisms of chemically capturing AAEM species by different bed materials were different. For Al/Si-rich minerals, the formation of silicates and aluminosilicates was the main reason for their retention, which was influenced by the value of Si/Al in minerals. For P-rich minerals such as sludge ash and phosphate rock, AAEM-based compounds in Zhundong coal would be retained in residual ash in the form of phosphates and pyrophosphates. Moreover, the Si–Al–P system in sludge ash together inhibited the release of AAEM species. The tests of ash fusibility indicate all of five bed materials increased the ash fusibility of Zhundong coal. The orders of minerals which increased the ash fusion temperature of Zhundong coal from high to low were phosphate rock, corundum, sludge ash, kaolin, and quartz for TCMLc, and corundum, phosphate rock, sludge ash, kaolin, and quartz for SEHc. The mineral effectively increasing the ash fusibility would greatly alleviate slagging. Due to the strong capture of AAEM species and high ash fusibility temperature, sludge ash was suggested as the appropriate selection of the bed material during Zhundong coal gasification in FB. Besides, this suggestion would contribute to the sewage sludge treatment.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the Strategic Priority Research Program of the Chinese Academy of Sciences (No. XDA07030100) and the International Science & Technology Cooperation Program of China (No. 2014DFG61680).
