Abstract
Coal pyrolysis is the basis of coal gasification, as it is the necessary reaction step in the coal conversion process. To study the pyrolysis characteristics of different coal grades under temperature, thermal simulation tests of three different ranks of coal were conducted, and the pore structure and gas products were analyzed by using liquid nitrogen adsorption and gas chromatographic, and perform fractal fitting for pores of different size. The results show that the pyrolysis gas output of Inner Mongolia lignite is higher than that of Xinjiang long flame coal and Hancheng bituminous coal. With the increase of coal rank, the gas output of each gas component also shows a decreasing trend and the initial temperature and peak temperature of various gases gradually shift to high temperature. With the increase of temperature, the average pore size, volume, specific surface area, and fractal dimension of the three kinds of coal char show an increasing trend. The average pore size and pore volume of coal char increase, and the fractal dimension increase with the increase of coal rank. The comprehensive comparative analysis of the gaseous products, average pore size, pore volume, and fractal dimension of the three coal chars shows that Inner Mongolia lignite has high gas production, large average pore size, good connectivity, and simple pore structure, which is conducive to the escape of pyrolysis gas and the transportation of gasification agent and is the best favorable coal for gasification. The research results are expected to provide experimental guidance for the actual production of underground coal gasification.
Introduction
At present, the waste of coal resources and the destruction of the ecological environment caused by traditional methods of coal reclamation, transportation, and use cannot be ignored (Friedmann et al. 2009). To solve these problems, changes in the traditional methods of coal production and consumption must be made without delay. Underground coal gasification has provided an important research topic as the cutting-edge technology for clean coal mining (Jiang et al. 2020, Mandal et al. 2020, Liu and Liu 2021, Feng et al. 2021, Rnm and Pkg 2021, Hu et al. 2021, Huang et al. 2021). Pyrolysis is the basis of other chemical processes, such as coal gasification, and is the first step in the clean utilization of coal. Coal pyrolysis is a complex process in which a series of physical and chemical reactions occur at different temperatures when coal is heated in isolated air or inert atmosphere. Its reaction is very rapid, producing gas(The gases are mainly low molecular compounds such as CH4, H2 and CO), liquid and solid products, generally starting at 300°C and ending at 900°C (Wiatowski et al. 2016, Peng et al. 2016, Li et al. 2021, Chmielniak et al. 2021). The increase of temperature, volatile decomposition and intermolecular fracture during pyrolysis will change the pore structure and permeability of coal (Self et al. 2012, Bhaskaran et al. 2015, Meng et al. 2020, Yu et al. 2021, Wang et al. 2021), it will not only affect the escape of gas in the pyrolysis process, but also affect the transportation of gasifying agent in the subsequent gasification process. Therefore, it is of great theoretical and practical significance to study the gasification products and pore size changes of coal char in the process of coal pyrolysis to realize safe, efficient, and clean utilization of coal.
The internal structural characteristics of coal, the peripheral functional groups and side chains of structural units, surface properties and spatial structural properties all change with the change of coal metamorphic degree, and their elemental compositions such as carbon, hydrogen, oxygen, nitrogen, and sulfur are also different, resulting in different characteristics of different coals in the pyrolysis process. Han et al. (2021) took the lignite in Erlian Basin as an example to study the evolution law of pores in lignite during pyrolysis. Hong and Guo (2017) studied the effect of heating rate on the pyrolysis characteristics of Zhundong coal. Huang et al. (2019) studied the changes in pore structure and combustion characteristics of Zhundong coal during slow and fast pyrolysis. Xue et al. (2013) discussed the production characteristics of products of Taiyuan Formation coal measure source rock in Ordos Basin at different temperatures. Zhao et al. (2018) took Shanxi Formation coal in Ordos Basin as an example to discuss the influence of heating rate on the pyrolysis process. In the past decades, considerable efforts have been made to study the coal pyrolysis characteristics and microstructure changes. Some scholars found that the pore structure of coal char becomes more complex during the pyrolysis, and the pore types are diverse and disorderly distributed. In this case, it is difficult to describe it with the traditional geometric model, some scholars studied the pore size change of coal char based on fractal theory, and the fractal theory can well study the pore size change of coal char during pyrolysis(Xu et al. 2010, Wang et al. 2008).
However, most of these studies focus on a single coal type and the main research is the change of fractal dimension of a single aperture, there are few studies on the changes of pore structure of different coal grades and the comprehensive fractal dimension of full pore size, Accordingly, the main purpose of this work is to analysis the aperture changes and products characteristics during the pyrolysis of three kinds of coals with different metamorphic degrees. The evolution behavior of microstructure for three coals and their pyrolysis characteristics were studied using low temperature liquid nitrogen experiment and gas chromatograph, respectively. and it is expected to provide useful information for simulation study or optimization of the operational condition for underground coal gasification.
Samples and experimental
Coal
The lignite, long flame coal and bituminous coal were collected for comparison. The lignite samples (NM) came from Saihantala Formation in Shenhua Coal Mine in the northeast of Erlian Basin. The long flame coal samples (XJ) came from Badaowan Formation in Fukang located in the southeast of Junggar Basin, and the bituminous coal samples (HC) came from Taiyuan Formation in Hancheng mining area, Ordos Basin, and the coal seams are widely distributed. The specific sampling location is shown in Figure 1.
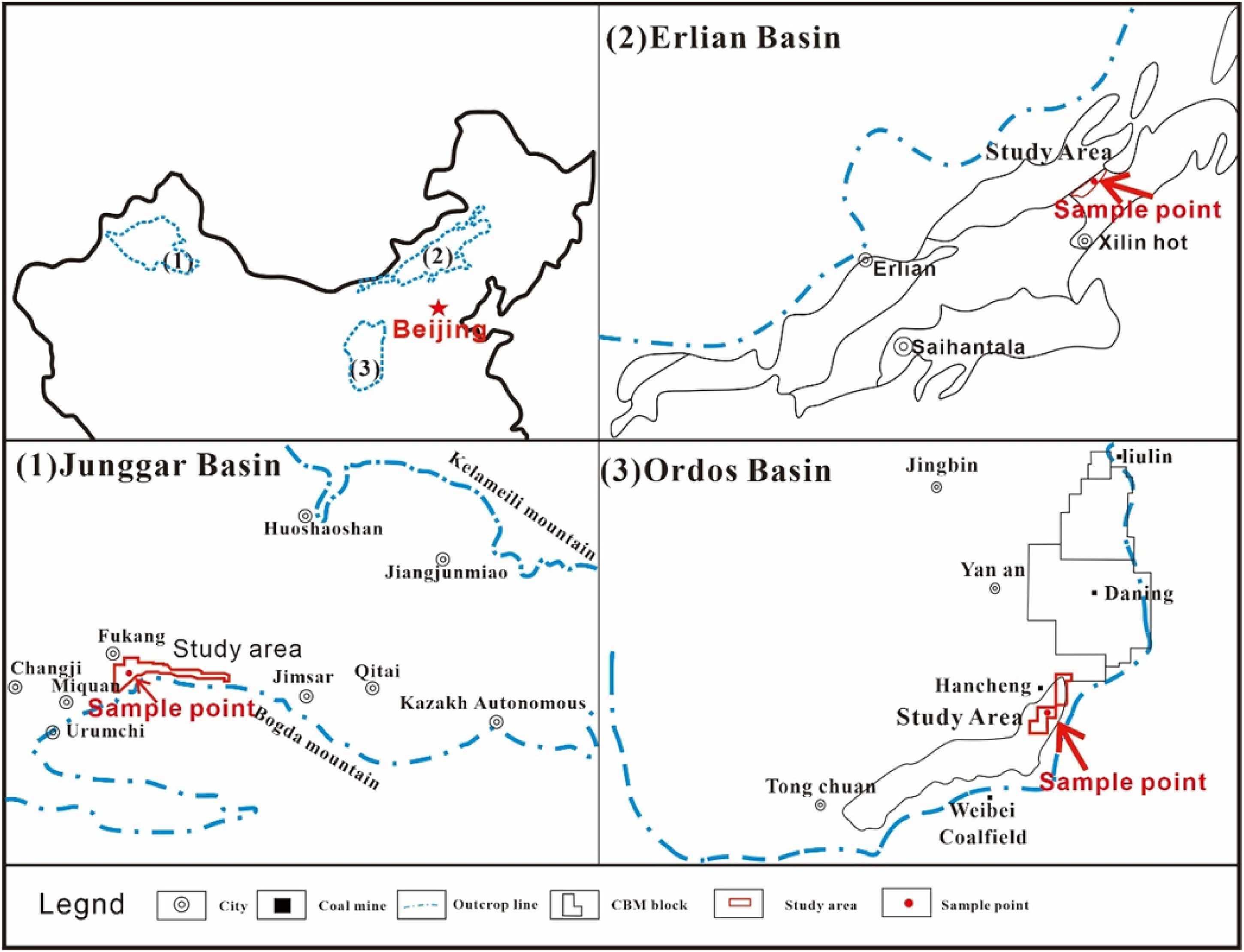
Distribution map of sampling points(Yin et al. 2020).
Experimental procedure
First, the vitrinite reflectance, elemental analysis and macerals were measured and analyzed according to the national standard GB/T212-2008. The ash, moisture, volatile and C, H, O, N content of the three coal samples were measured and analyzed (Donahue and Rais 2009). Then, the samples from each area were divided into three groups, the mass of each test sample is 10g. After crushing and sieving to a predetermined size of 3 mm, the coal samples were filled in the gasifier. Before the experiment, the original air in the device was replaced with nitrogen for 30min, and then the air was replaced with nitrogen in nitrogen atmosphere. After the start of the experiment, the three groups of raw coal are heated to 600, 750, and 900°C respectively at a heating rate of 10 °C / min and kept constant for 30min to obtain coal coke, in which the nitrogen flow is 10ml/min (Figure 2). The gas products are collected with a gas collecting bag, and the solid products are left in the quartz tube. After the temperature in the furnace is cooled to room temperature, they are weighed and sampled for analysis.
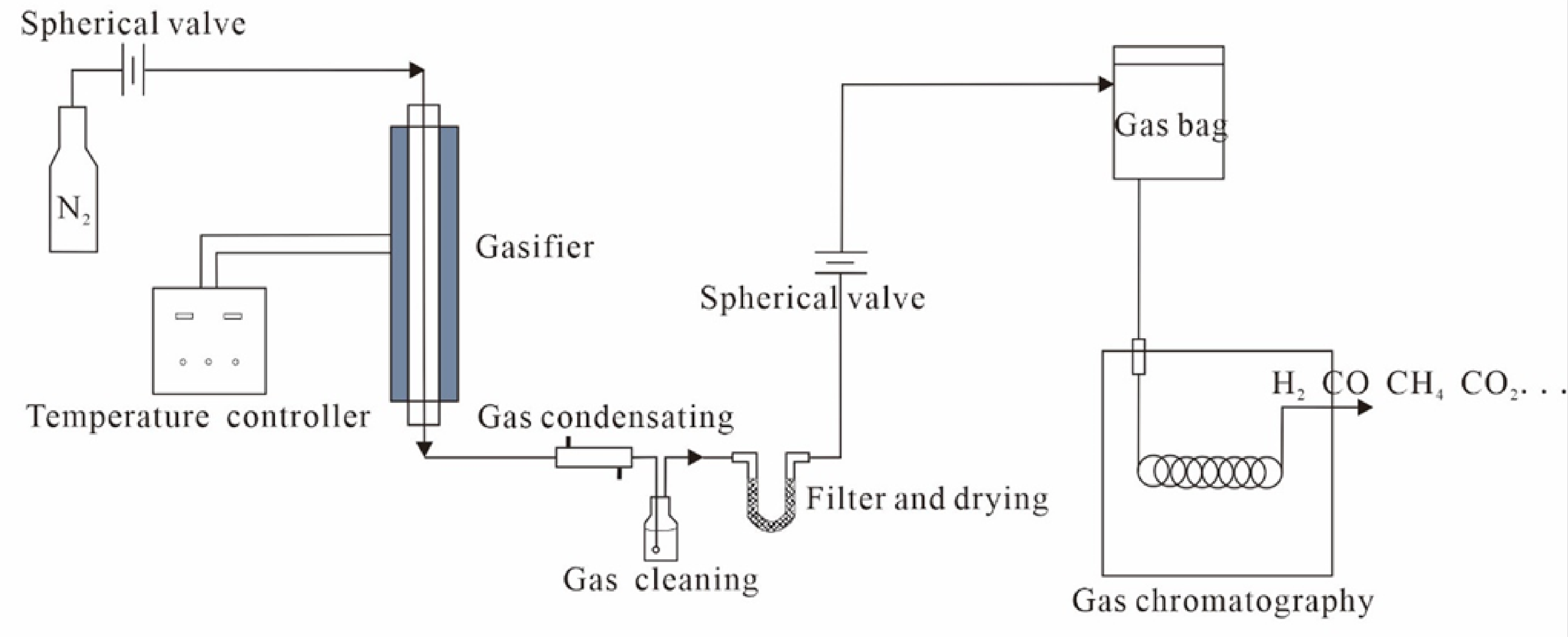
Pyrolysis experimental device diagram.
Analysis of gas composition from coal samples
The pyrolysis gas products of coal samples with pyrolysis temperature of 900 °C are cooled, washed and dried, and then collected with gas collection bags. During the heating process, extract the pyrolysis gas products at the inlet of the air bag every five minutes, the content of pyrolysis gas of the three coal chars were analyzed by gas chromatograph (GC-4000A, China). After the experiment, the gas in the gas bag was fully mixed, and the total volume of pyrolysis gaseous products and the percentage content of each component were measured by gas chromatograph (GC-4000A, China).
Analysis of pore variation in coal
The pyrolysis solid product coal char of different final pyrolysis temperatures (600°C,750°C, and 900°C) was crushed into 60-80 meshes at and dried for 48 h. The pore variation was measured by ASAP2000; this instrument uses the principle of gas adsorption (typically nitrogen) (Li et al. 2020, Yin et al. 2020).
Results and discussion
Gas composition during pyrolysis
Figure 3 shows the changes in main gas composition and content of 3 coal samples with temperature during the pyrolysis process in a nitrogen atmosphere. The diagram shows that the main gases produced during coal pyrolysis include CH4, H2, CO and CO2.
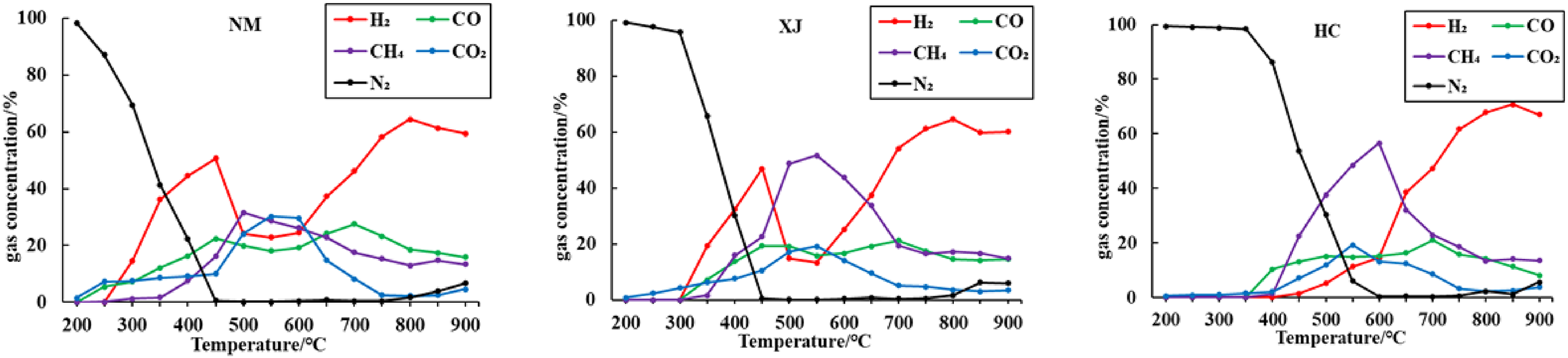
Pyrolysis performed on 3 coal samples at different temperatures.
The release of CH4 during pyrolysis is caused by the breaking of aliphatic chains and aromatic side chains containing methyl functional groups in coal. (Liu et al. 2014, Lin et al. 2017). The precipitation temperature of CH4 in Inner Mongolia lignite is about 300°C and reaches the peak at about 500°C. The precipitation temperature of CH4 in Xinjiang long flame coal is about 350 °C and reaches the peak at about 550°C. The precipitation temperature of CH4 in Hancheng bituminous coal is about 400°C and reaches the peak at about 600°C. The methane emission of the three raw coals increases first and later decreases with an increase in pyrolysis temperature, thereby reaching the maximum at 500−600°C. With the increase of coal metamorphism, the initial precipitation temperature and peak temperature of CH4 gradually shift to high temperature.
It can be seen from Figure 3 that with the increase of temperature, the release of hydrogen generally shows an increasing trend. The process of H2 release from pyrolysis of Inner Mongolia lignite and Xinjiang long flame coal is similar. This process is mainly divided into two steps, which is an obvious bimodal type. (Ying et al. 2013, J. and Patrick 1985). The first stage is 300–500 °C, the Inner Mongolia lignite starts to release from 300°C and the Xinjiang long flame coal starts to release at about 350°C, the first wave peak appears at 450°C, indicating that this stage is a period of severe thermal reaction due to the rupture of organic macromolecular network structure in coal. At this time, the release of H2 is mainly caused by pyrolysis dehydrogenation reaction; The second stage is 550–900°C, starting at about 600°C, H2 begins to release in the second stage, reaching the maximum at about 800°C, and then the release of H2 gradually decreases with the continuous increase of temperature; Hancheng bituminous coal with higher coal rank begins to release H2 at about 450°C, and the H2 peak in the first stage is missing. With the increase of temperature, the hydrogen content gradually increases, and the H2 peak in the second stage appears at about 850°C, which is produced by the polycondensation reaction in the later stage of coal pyrolysis. The second stage H2 release peak appeared in the three coals with different metamorphic degrees, and the temperature reaching the second peak gradually increased with the increase of metamorphic degree. This shows that the binding strength of hydrogen ions in coal molecules increases with the increase of coal metamorphic degree.
The release of CO and CO2 is mainly affected by the oxygen atoms in coal. The oxygen content is different, which means the amount of CO and CO2 release are not the same (Ying et al. 2013, Meng et al. 2019, Daggupati et al. 2010). The formation of CO is different from the formation of CO2 due mostly to the different chemical bond energies of different oxygen-containing functional groups. The thermal decomposition temperatures of these functional groups are different(Yi et al. 2019, Guo et al. 2019). The thermal stability order in coal is hydroxyl > carbonyl > carboxyl > methoxy. During pyrolysis the content of CO fluctuates slightly with temperature; as the temperature increases, the CO content reaches a maximum at approximately 400 and 700°C of Inner Mongolia lignite and Xinjiang long flame coal, the CO content of Hancheng bituminous coal reaches a maximum at 700 °C, which is related to the source of CO. The generation of CO is mainly caused by the breaking and decomposition of ether bonds and carbonyl groups at low temperature section. The carbonyl can generate CO at about 450 °C, and the first peak appears. In addition, the generation of CO is mainly due to the fracture of oxygen-containing heterocycles and the reaction between CO2 and coal char at high temperature section, and the second peak appears. The lower the degree of coal metamorphism, the higher the oxygen content and the greater the amount of CO released, the oxygen content of lignite in Inner Mongolia is as high as 13%, while the oxygen content of Xinjiang long flame coal and Hancheng bituminous coal is between 8% and 6%; therefore, the CO content of lignite in Inner Mongolia is the highest
Meanwhile, the release of CO2 during the pyrolysis of the three coals are similar, with the increase of coal rank, the initial temperature of CO2 release gradually increases. At about 550°C, the CO2 release of the three kinds of coal chars reaches the maximum. In the drying and degassing stage, a small amount of CO2 adsorbed in coal gradually escapes. In the pyrolysis stage, the CO2 released from coal is mainly composed of the removed materials of carboxyl groups and the decomposed of oxygen-containing heterocyclic or oxygen-containing groups. The carboxyl group has poor thermal stability and can generate CO2 at low temperature, the content of CO2 is higher than that of CO in the range of 300−550°C. Comparing the relative escaped amounts of CO2 generated by the pyrolysis of the three coal types, we observe that the differences in the coals’ oxygen contents lead to large differences in the amounts of CO2 produced.
Figure 4 shows the variation in gas product yields with coal type at 300−90°C. The composition of each gas product produced by pyrolysis is different from that of coal at the same heating rate and the same pyrolysis end temperature (Xu et al. 2019). When the final temperature of pyrolysis is 900°C, the combustible gas products are mainly H2, CO, and CH4, with H2 comprising the clear majority. As can be seen from Figure 4, the total pyrolysis gas output of Inner Mongolia lignite is 2.82L/10g coal, the total pyrolysis gas output of Xinjiang long flame coal is 1.86L/10g coal, and the total pyrolysis gas output of Hancheng bituminous coal is 1.54L/10g, the pyrolysis gas output of Inner Mongolia lignite is higher than that of Xinjiang long flame coal and Hancheng bituminous coal. The released H2 yield, CO yield, and CH4 yield t in Inner Mongolia lignite are higher than Xinjiang long flame samples and Hancheng bituminous coals, with the increase of coal rank, the gas output of each gas component also shows a decreasing trend. Table 1 shows that the atomic ratio of H/C is Inner Mongolia>Xinjiang > Hancheng, and O/C atomic ratio is Inner Mongolia > Xinjiang > Hancheng. Due to the high C/O ratio and C/H of Xinjiang long flame coal and Hancheng bituminous coal, the CO and H2 yield is significantly lower than that of Inner Mongolia lignite. Therefore, the results show that the variation of hydrocarbon gases, and CO and CO2 production with coal type is almost consistent with the order of the O/C and H/C atom ratios.
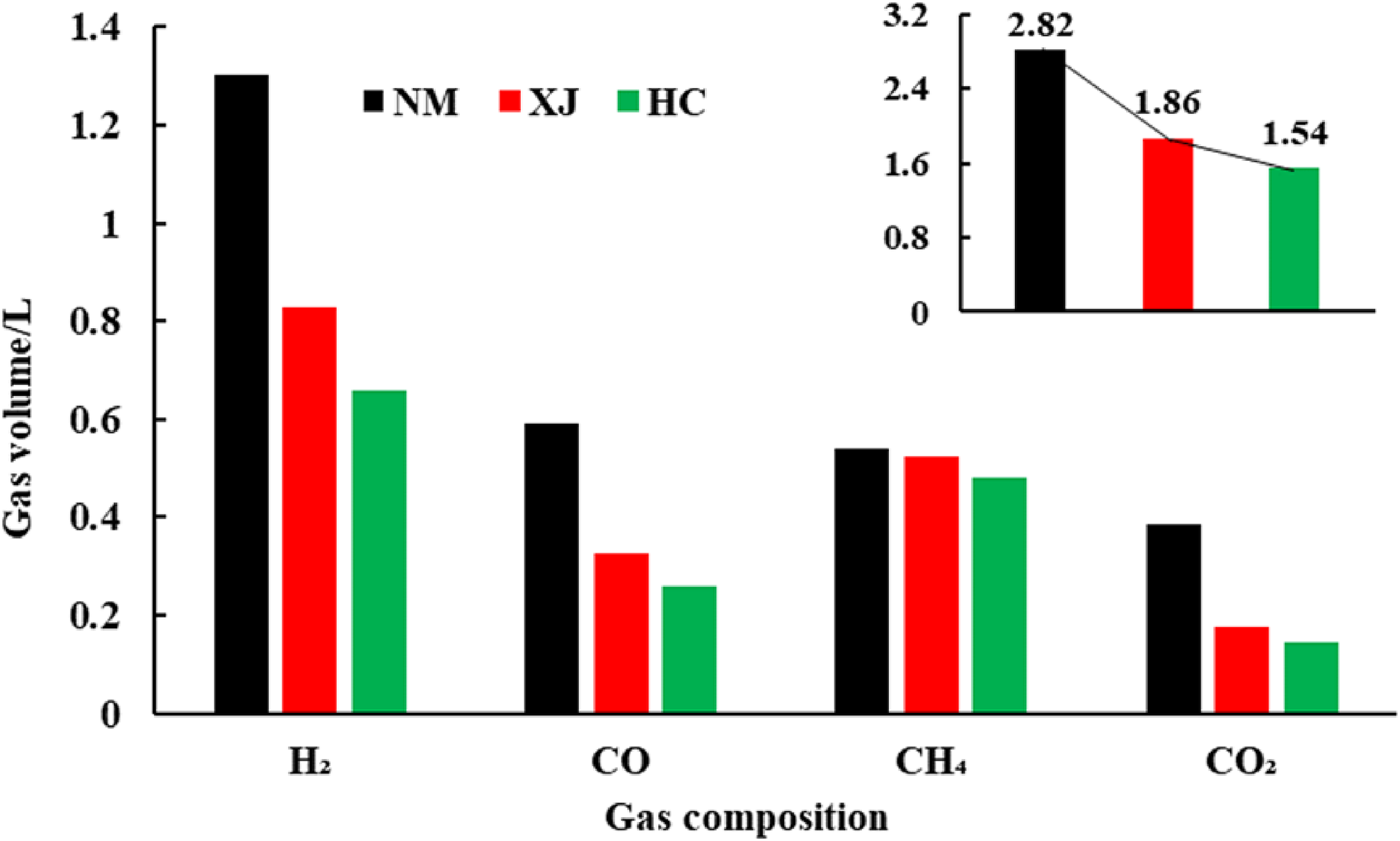
Volume of gas product at final pyrolysis temperature.
Proximate and ultimate analysis of 3 coal samples.
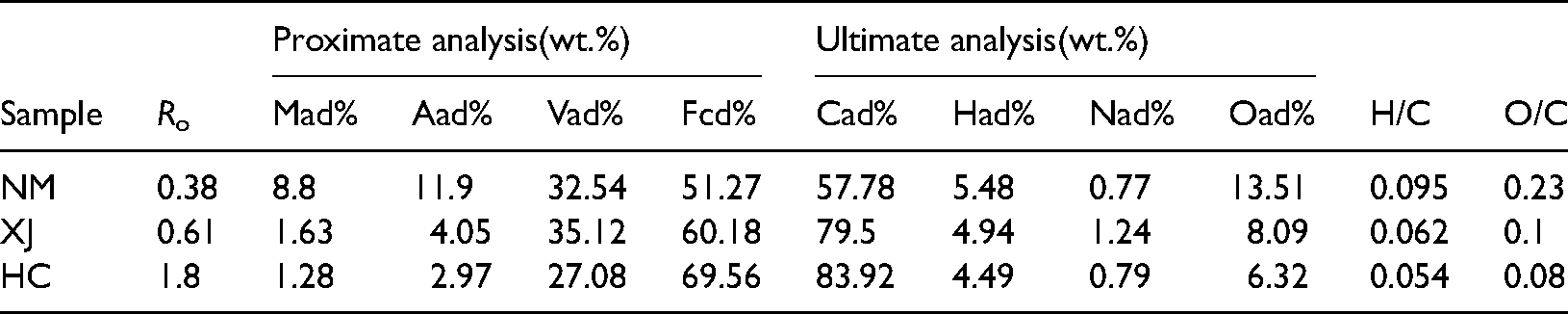
Distinct coal ranks are different in composition, structure, and properties. The basic law is: as the deepening of coal metamorphism, the content of carbon in coal increases gradually, while oxygen content, intrinsic moisture and volatile matter decrease gradually (Table 1). The aromatic condensation degree of low coalification coal is small, but the bridging bonds and functional groups occur more, the low-molecular-weight compounds occur more, the structure is not directional. As the coalification degree deepens, the aromatic rings becoming condensation, the bridging bonds, side chains and functional groups decrease gradually, the arrangement of molecules becomes gradually ordered, and the degree of parallel orientation between molecules increases, thus showing anisotropy. At the anthracite stage, the arrangement of molecules gradually tends to be the graphite structure of highly condensation of aromatic rings.
As shown in Figure 5, coal particle pyrolysis is generally divided into drying stage and pyrolysis stage. With the increase of coal rank, the types of functional groups and fracture temperature in coal are different, in addition, during the pyrolysis process, with the release of volatile matter and the molecular bonds gradually broke, resulting in the change of pore structure, a large number of small holes will be formed on the coal surface, with the continuous increase of pyrolysis temperature, the volatile matter is released more violently, resulting in the expansion of small holes into mesopores and macropores, the collapse and closure of large holes will occur. The pyrolysis process determines the surface activity and structural characteristics of coal char, which is an important factor affecting the subsequent gasification reaction efficiency.
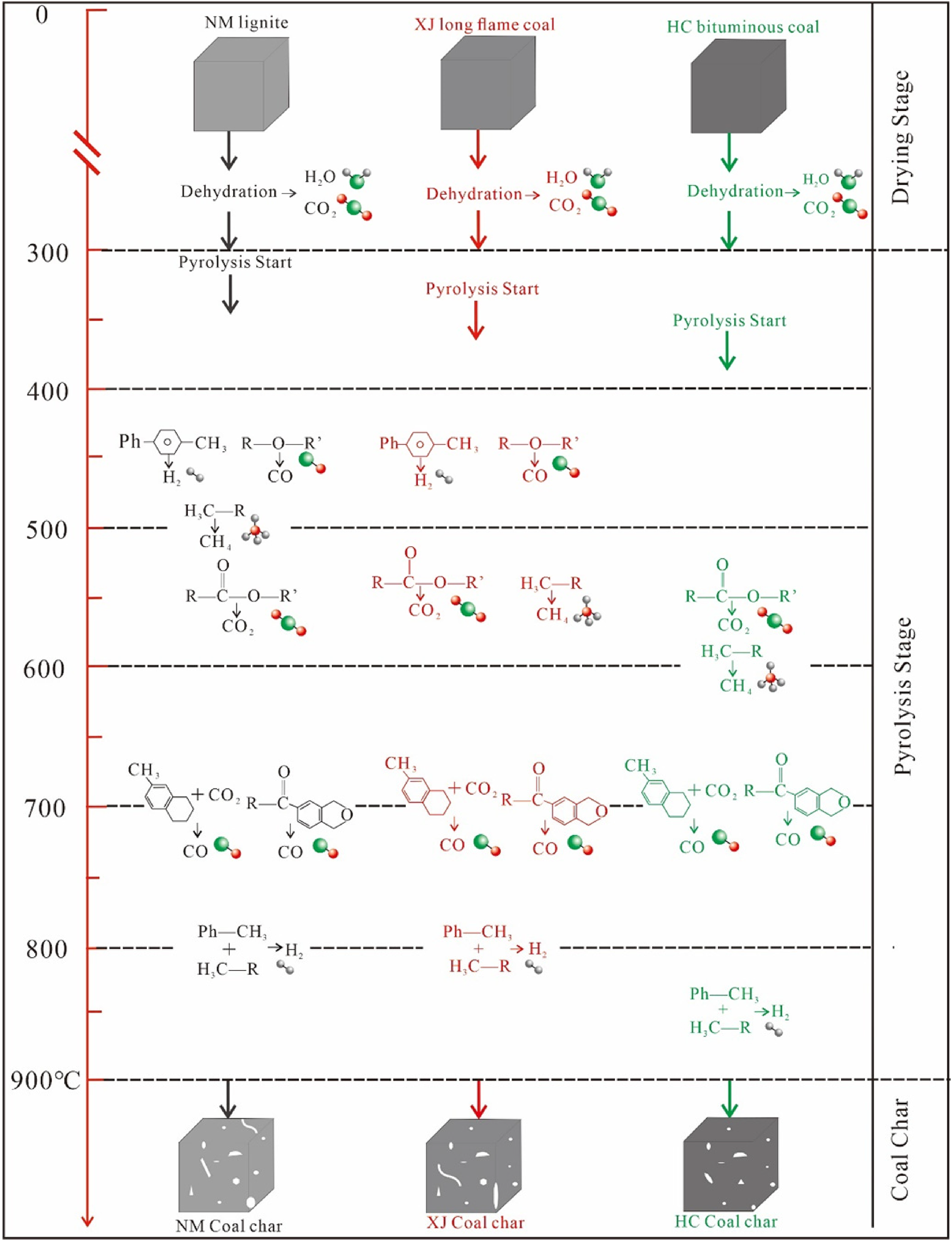
A simple diagram of three coal pyrolysis process.
Measurement of pore structure
Pore structure characteristics
Figure 6 shows that the adsorption curve of this type is parallel to the desorption curve when the relative pressure is less than 0.8, and there is no hysteresis loop between the adsorption and desorption curves, thus indicating that there were larger pores (Yao et al. 2006, Daggupati et al. 2010). The adsorption volumes of samples with different coal grades show discernible differences. The adsorption volume of Inner Mongolia lignite is 8−12mL/g, the Xinjiang long flame coal is 1.2−2 mL/g, and the Hancheng bituminous coal is approximately0.9−1.2 mL/g. The adsorption volume negatively correlates with coal rank. This curve is mainly composed of semi connected, semi open pores, which have the weakest adsorption and accumulation capacities. However, these pores are helpful for the diffusion of pyrolysis gas.
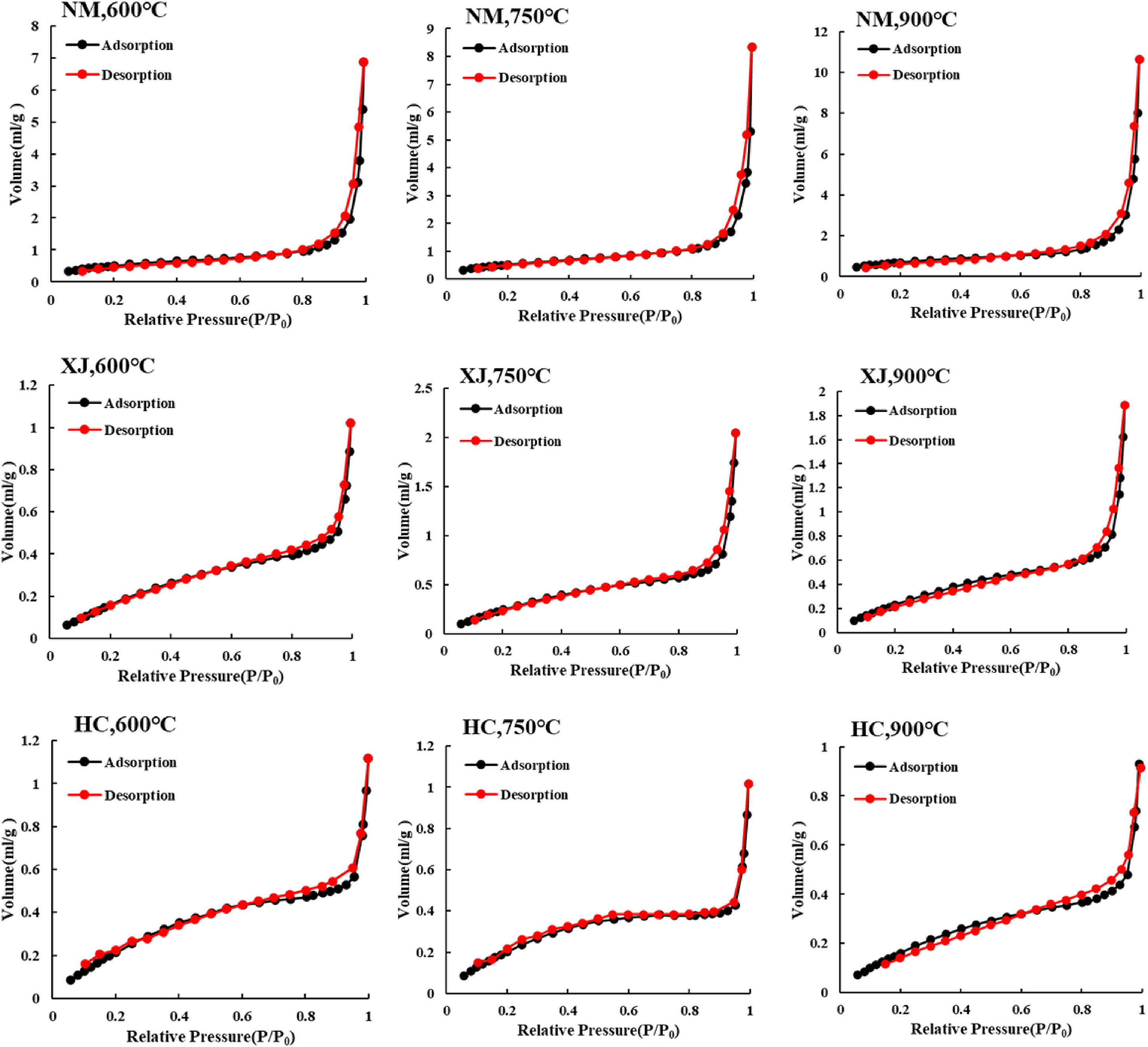
N2 adsorption-desorption isotherms of 3 coal char at different temperatures.
For the Inner Mongolia coal, the adsorption capacity of its coal char positively correlates with temperature, and may be related to its higher ash content. As a lignite, the ash content in Inner Mongolia coal is the highest The presence of more minerals hindered the collapse of the carbon skeleton in the coal char. The nitrogen adsorption amounts of the coal char continuously increased at high temperatures. For Xinjiang coal chars, their adsorption capacity reached a maximum at 750°C. This dynamic is due to the more intense volatile precipitation and more frequent pore openings caused by the increase in temperature. However, when the temperature was too high (> 900°C), the adsorption capacity decreased, which was due to the melting collapse of the coal coke skeleton and the transformation of some micropores into transition pores and mesopores. For the Hancheng coal char, the adsorption capacity of coal char decreased gradually. The analysis shows that the ash content of Hancheng coal char is low, the volatile matter is precipitated, the pore of the coal char is gradually opened, and the carbon skeleton in coal char collapses, which promotes the transformation from micropores to transition pores and mesopores. The amount of gas adsorption decreases gradually at high temperature. In addition, by comparing the ash content of Xinjiang and Hancheng, we observe that a higher temperature collapses the carbon skeleton for coal chars with high ash content; this is also the reason why the temperature of the carbon skeleton collapse for the Xinjiang coal char is 750°C, but the Hancheng coal char is 600°C.
Pore size distribution
Figure 7 and Table 2 shows that the specific surface area of the Inner Mongolia coal char, Xinjiang coal char and Hancheng coal char is 2.2−2.9m2/g, 1.35−2.0 m2/g and 1.26−1.75m2/g,With the increase of coal rank, the specific surface area decreases. The specific surface area of three kinds of coal char come from the contribution of the 2−3 nm hole. The specific surface area of Inner Mongolia coal char reaches the maximum at 900°C, that of Xinjiang coal char reaches the maximum at 750°C, and that of Hancheng coal char reaches the maximum at 600°C.
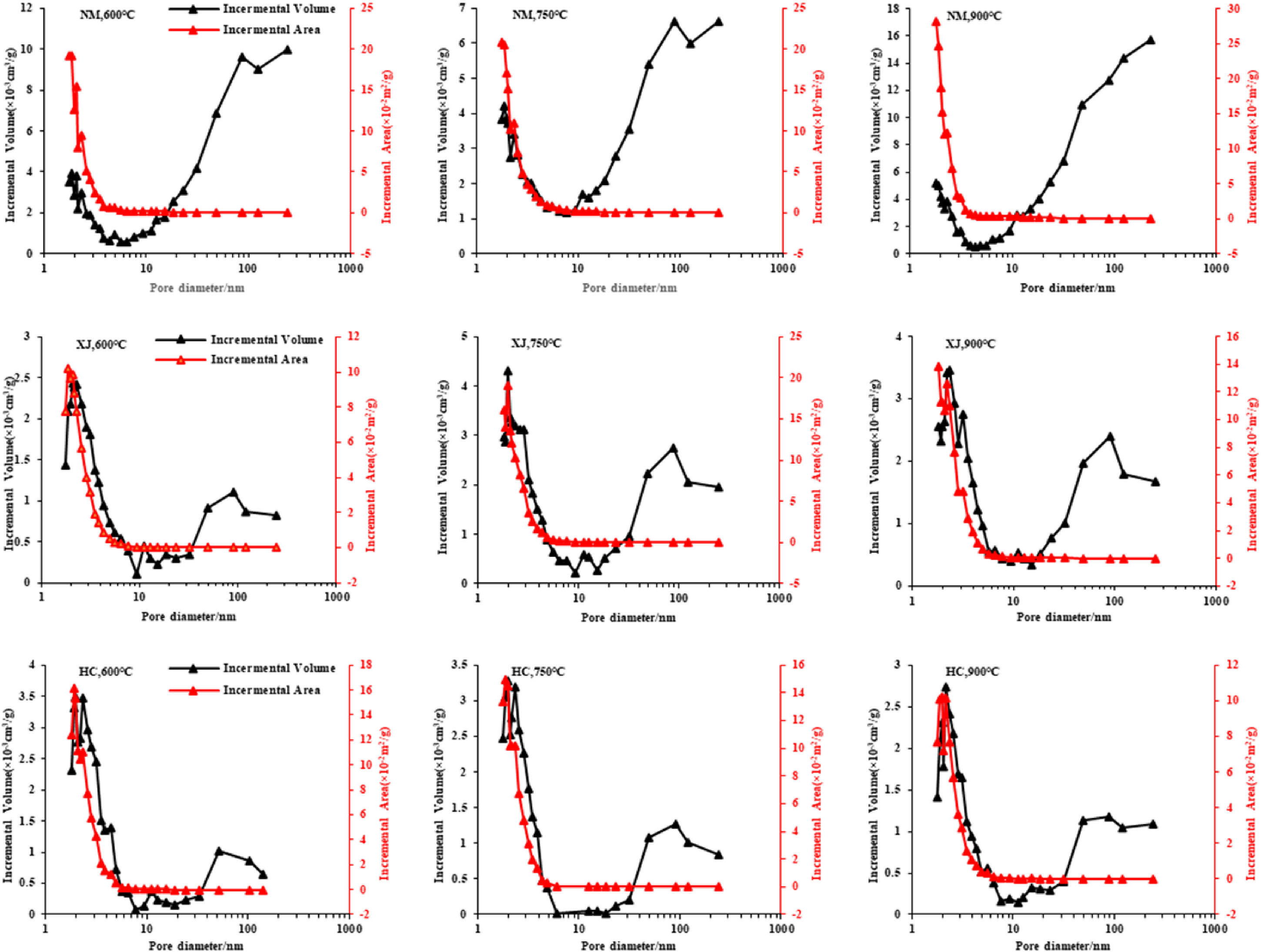
Pore volumes and SSA distributions of 3 coal char at different pore sizes.
Specific surface areas and pore structure parameters of 3 coal char at different temperatures.
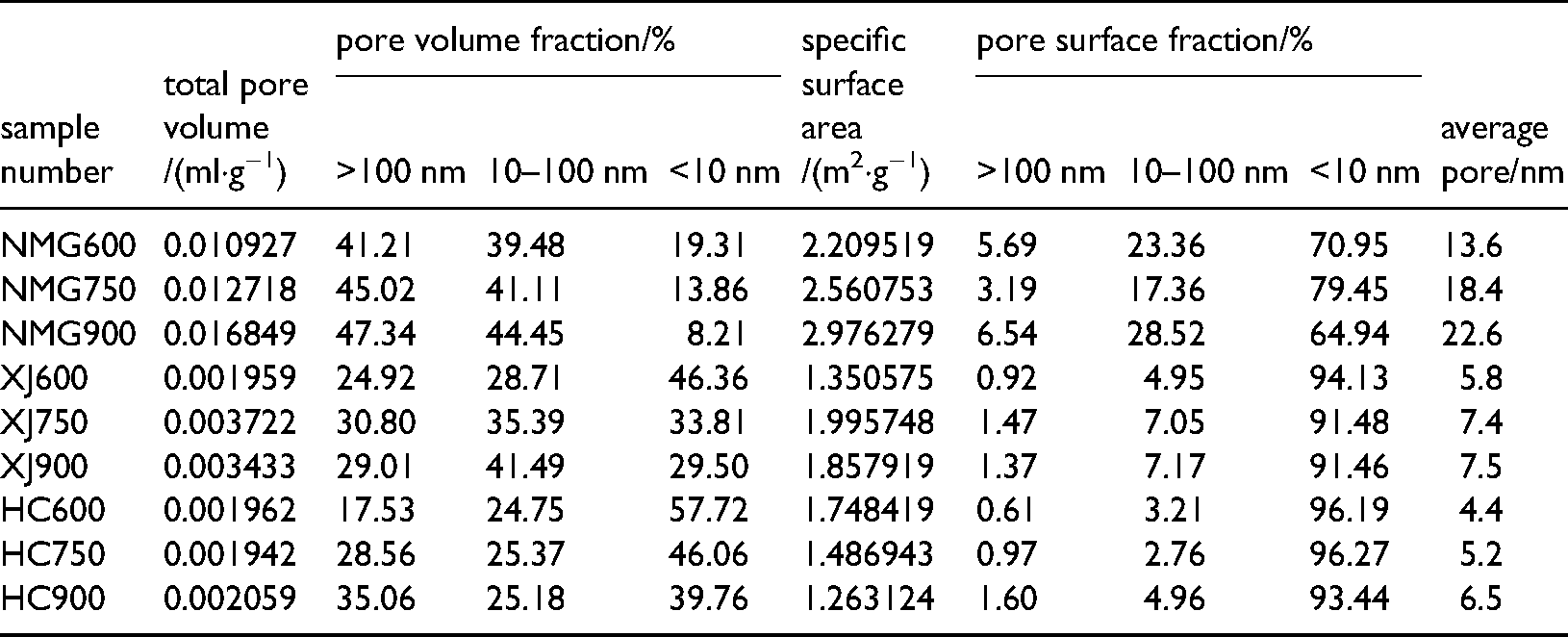
The pore volumes of Inner Mongolia coal char, Xinjiang coal char and Hancheng coal char are respectively is 0.009−0.017mL/g, 0.0019−0.0037mL/g and 0.0019−0.002mL/g (Figure 7, Table 2). With the increase of coal rank, the pore volume decreases gradually. The pore volume of Inner Mongolia coal char mainly comes from the contribution of the 80−100 nm pore, but the pore volume of Xinjiang coal char and Hancheng coal char mainly comes from the contribution of the 2−3 nm pore. It shows that Inner Mongolia coal char mainly develops large and medium pores, while Xinjiang coal char and Hancheng coal char mainly develop small pores. Compared with the average pore size, the average pore size of Inner Mongolia coal char is obviously larger (Table 2).
This is mainly due to the more intense volatilization analysis with the increase of temperature, which makes more pores open. However, the excessive temperature makes the carbon skeleton collapse in the coal char, and some microporous coal chars will transition to mesoporous and macroporous pore types.
Fractal characteristics of pores
As a complex porous organic rock, the pore structure of coal char becomes more complex due to t the thermal action of rock in the pyrolysis process. According to the previous analysis, the traditional geometric method is difficult to describe quantitatively. Fractal geometry is an effective means to analyze the complex pore structure of coal char. Based on the low temperature liquid nitrogen adsorption data. The fractal dimension of coal char is calculated by FHH model (Sun et al. 2015, Fu et al. 2017). The classical FHH equation is as follows: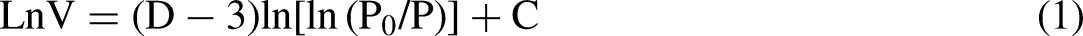
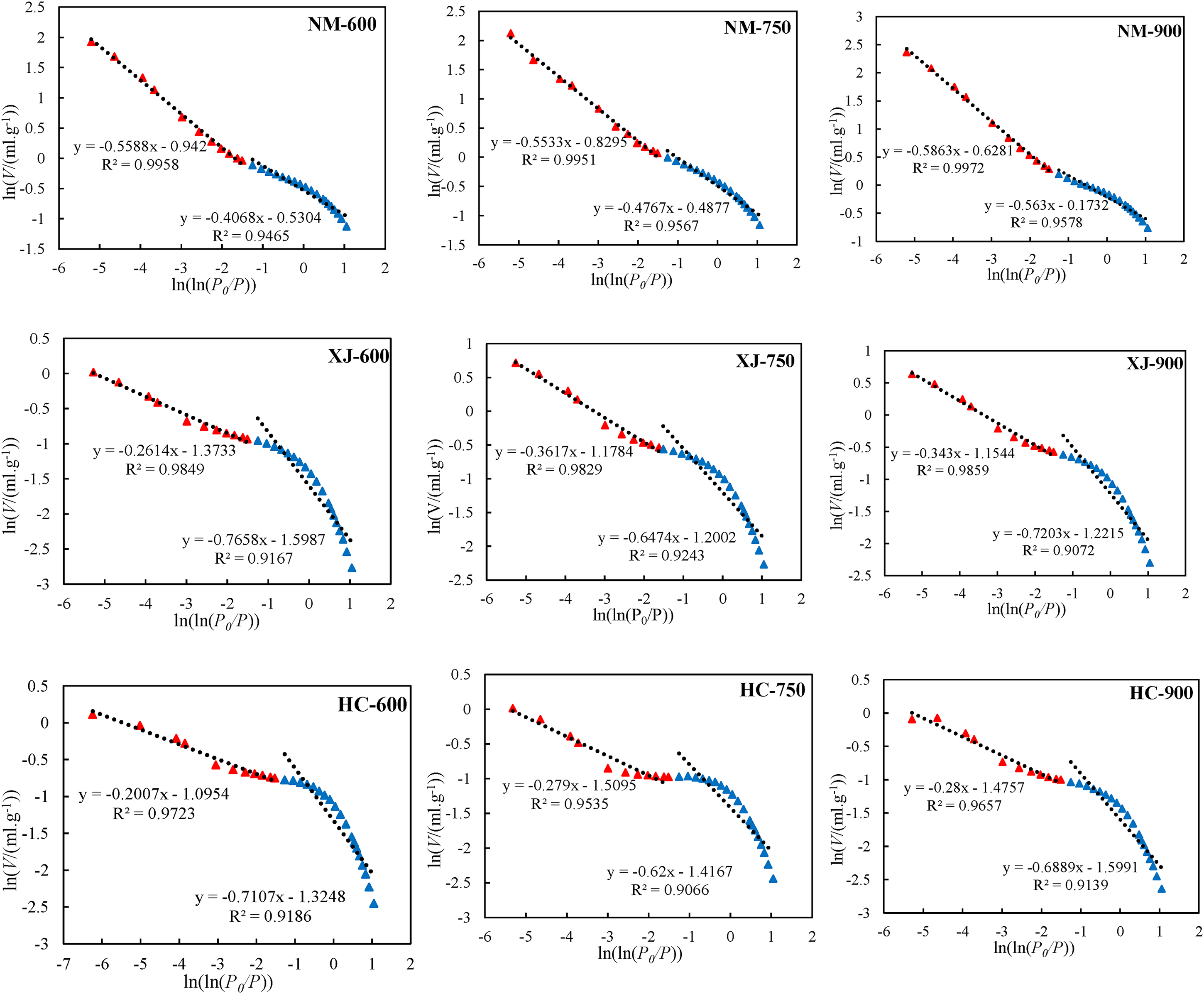
Fractal dimensions of coal nm, xj and hc with different temperature.
The results of fractal dimension.
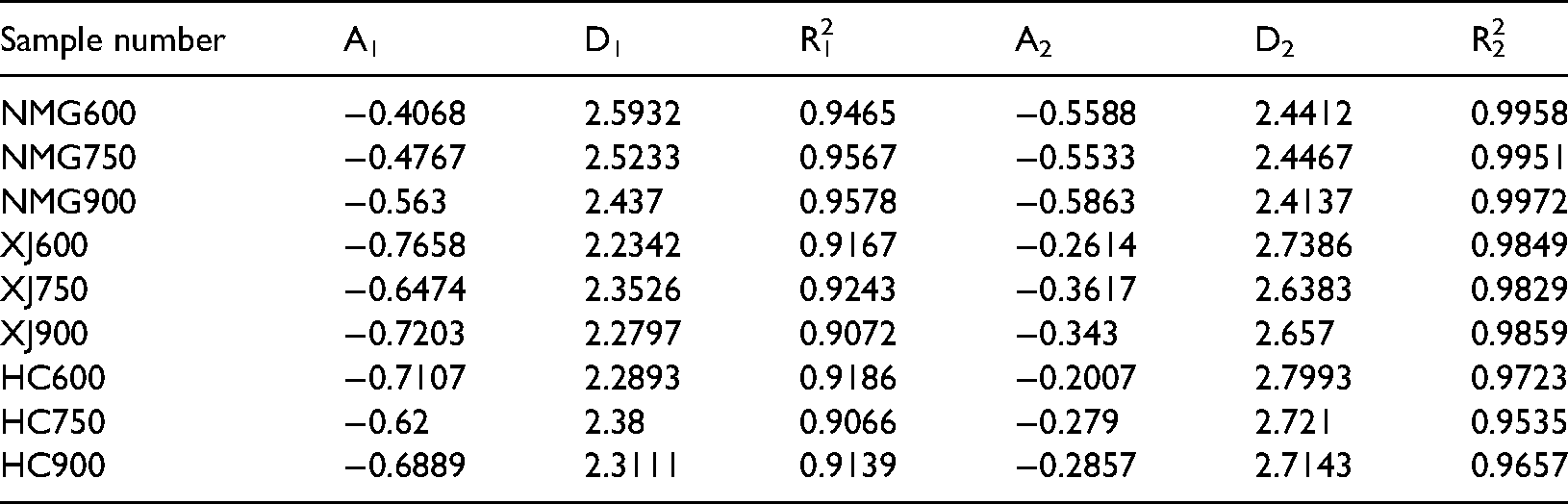
However, the fractal dimension of different aperture segments can't represent the overall complexity and roughness of coal. Therefore, it is necessary to calculate the comprehensive fractal dimension of coal by some method. According to the method for calculating the comprehensive fractal dimension of coal char proposed by Ren et al. (2014), the pore volume ratio of different pore diameter sections is taken as the weight, and the value obtained by weighted summation of the fractal dimensions of different pore diameter sections of coal is called the comprehensive shape dimension of coal, which is recorded as Dt, and its calculation formula is
Integrated fractal dimension of 3 coal char at different temperatures.
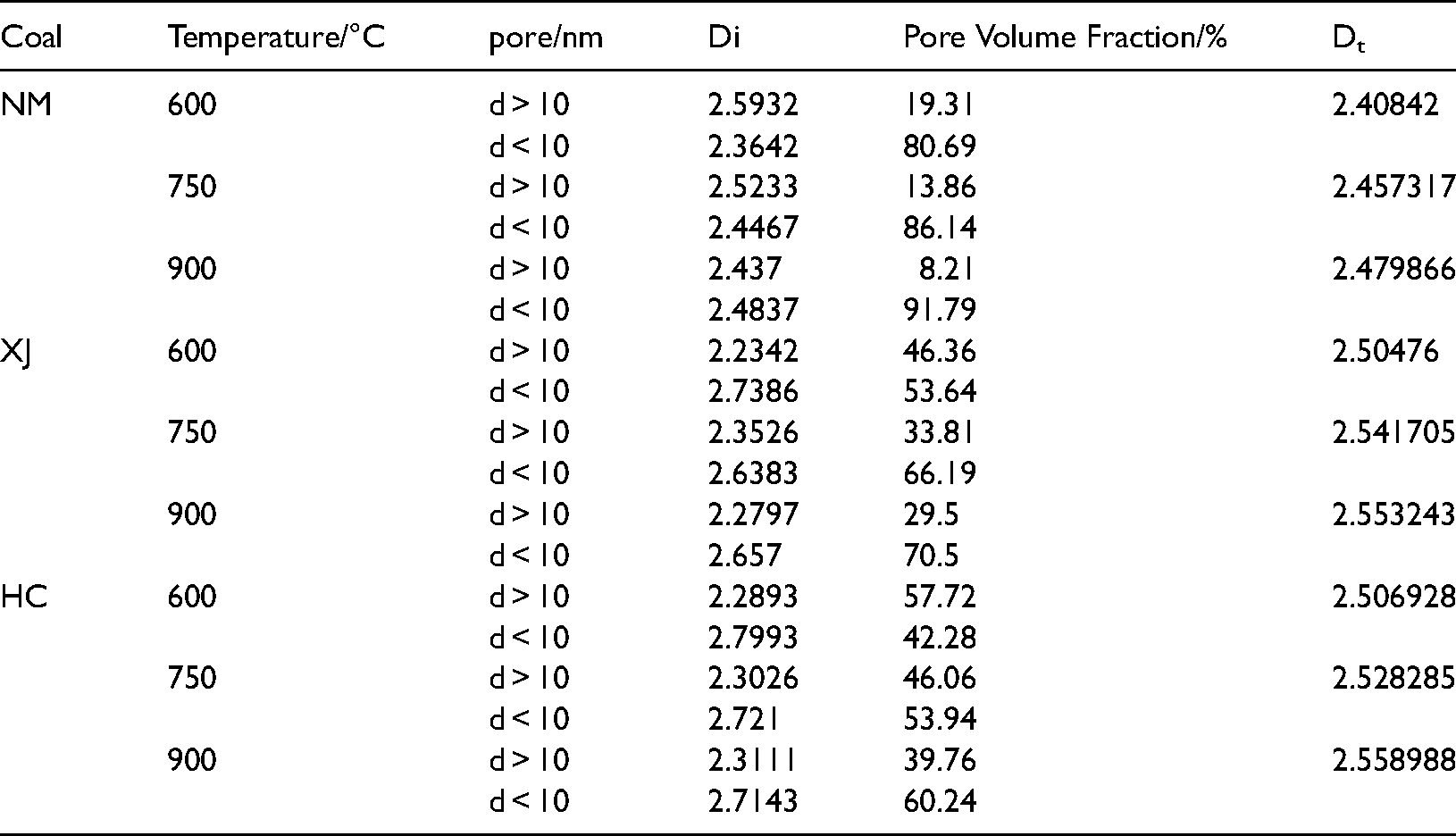
The volume integral dimension represents the distribution and complexity of pore structure in coal body. The higher the value, the more complex the pore structure (Sun et al. 2015, Fu et al. 2017). The relationship between fractal dimension and temperature, coal grade, pore volume and average pore size is shown in the Figure 9. We can know that the fractal dimension of coal of three kinds of coal char increases with the increase of temperature, it indicates that the pore structure of coal char becomes more complex, some micropores are formed with the precipitation of volatile matter in the process of temperature rise, and the number of micropores increases, which makes the pore structure more diverse, so the fractal dimension increases. Because the fractal dimension represents the complexity of pore structure, the larger the fractal dimension, the more complex the pore structure, the greater the diffusion resistance of gas in the pores, and the increase of fractal dimension will reduce the combustion rate of coal char. By comparing the fractal dimensions of three kinds of Coal Chars at the same temperature, it can be seen that the fractal dimensions of Xinjiang long flame coal and Hancheng bituminous coal are relatively high, indicating that its pore surface is rough, the pore system is relatively complex. On the contrary, the fractal dimension of Inner Mongolia lignite is the lowest at 900°C, indicating that its pore surface is relatively smooth, the pore system is relatively simple, and its resistance to gas migration is the smallest.
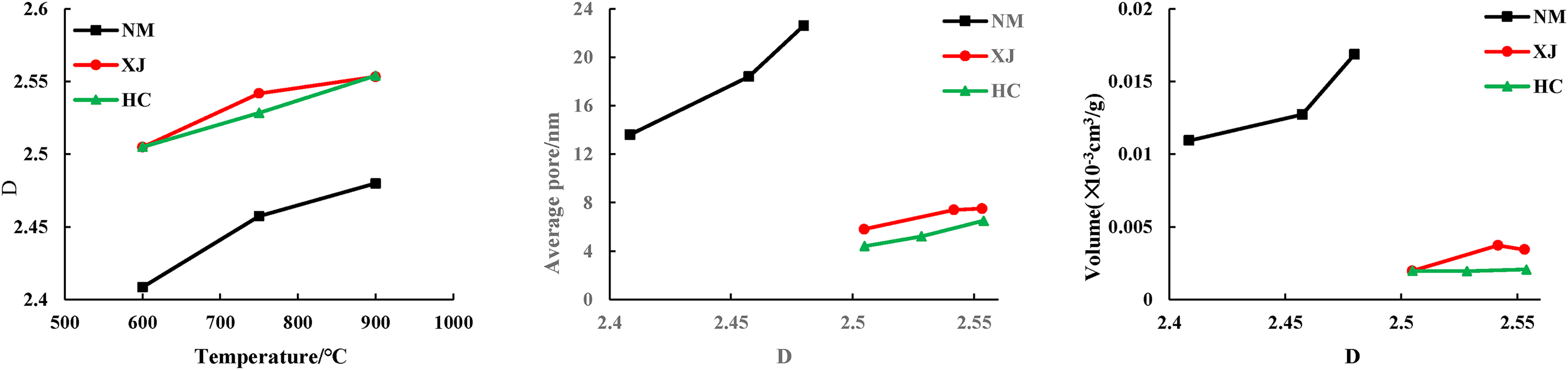
The relationships of temperature, average pore, and volume with fractal dimensions.
Figure 9. Shows that as the temperature rise, the average pore diameter of the three kinds of coal chars gradually increases. The total pore volume and average pore diameter of Inner Mongolia coking coal are the largest among the three kinds of coal char, and the corresponding comprehensive fractal dimension is the smallest, ranging from 2.4084 to 2.4799, indicating that there are many large and medium-sized pores, On the contrary, the total pore volume and average pore diameter of Xinjiang long flame coal and Hancheng bituminous coal are relatively low, but they have large fractal dimension, indicating that there are many micro pores, uneven pore distribution, relatively complex pore system and poor connectivity.
By comprehensively analyzing the combustible gas H2, CO, CH4, and total gas production laws of three coals with different metamorphic degrees, as well as the changes of average pore diameter, pore volume and fractal dimension, and normalize them. It can be seen from Figure 10 that Inner Mongolia lignite has high pyrolysis gas production, large average pore diameter and the lowest fractal dimension, indicating that its pores are developed and simple in structure, which is conducive to the migration of pyrolysis gas and gasification agent in the process of gasification.
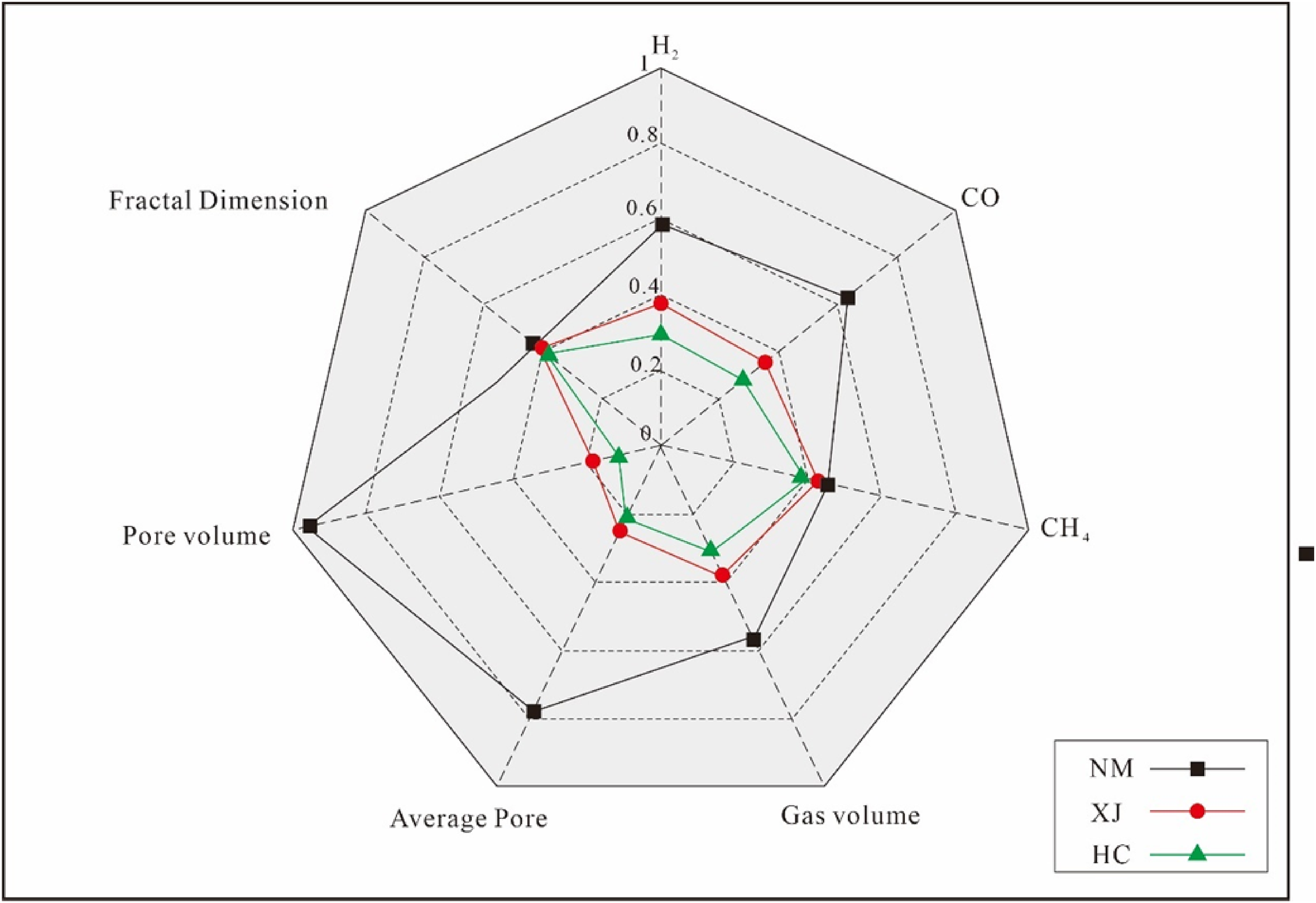
Comprehensive comparative analysis of three coal.
Conclusions
The pyrolysis gas output of Inner Mongolia lignite is higher than that of Xinjiang long flame coal and Hancheng bituminous coal. The released combustible gas H2 yield, CO yield, and CH4 yield in Inner Mongolia lignite are higher than Xinjiang long flame samples and Hancheng bituminous coals, with the increase of coal rank, the gas output of each gas component also shows a decreasing trend and the initial temperature and peak temperature of various gases gradually shift to high temperature. The release order of H2, CO and hydrocarbon gases is related to the stability of functional groups, and the release amount is related to the atomic ratio of O/C, H/C, and the number of functional groups in raw coal.
The pore connectivity of the three coal chars is good, with the increase of temperature, the average pore size, volume, and specific surface area of the three kinds of coal char show an increasing trend. The pore volume of Inner Mongolia coal char is mainly contributed by 80–100nm, the pore volume of Xinjiang coal char is mainly contributed by 80–100nm and 2–3 nm, the pore volume of Hancheng coal char is mainly contributed by 2–3 nm, and the specific surface area of the three kinds of coal char is mainly contributed by pore size of 2–3 nm. Inner Mongolia coal char mainly develops large and medium pores, while Xinjiang coal char and Hancheng coal char mainly develop small pores. The pore volume and average pore diameter decrease with the increase of coal rank.
The comprehensive fractal dimension of Inner Mongolia coal char is 2.41–2.47, that of Xinjiang coal char is 2.50–2.55, and that of Hancheng coal char is 2.51–2.56. With the increase of coal rank, the comprehensive fractal dimension increases gradually. The comprehensive fractal dimension of coal has obvious correlation with temperature, total pore volume and average pore diameter. The comprehensive fractal dimension is negatively correlated with total pore volume and average pore diameter, and positively correlated with temperature.
The comprehensive comparative analysis of the gaseous products, average pore size, pore volume and fractal dimension of the three coal chars shows that Inner Mongolia lignite has high gas production, large average pore size, good connectivity, and simple pore structure, which is conducive to the escape of pyrolysis gas and the transportation of gasification agent, and is the best favorable coal for gasification.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: the National Natural Science Foundation Project, China (Grant No. 42172188, U1703126) and the PetroChina Science and Technology Major Project (2019E-25).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China, PetroChina Science and Technology Major Project, (grant number Grant No. 42172188, U1703126, 2019E-25).
