Abstract
Massive dolostones replacing build-ups and containing H2S of thermochemical sulfate reduction (TSR) origin occur in the Upper Permian reef and bank deposits, northeastern Sichuan, China, despite the absence of gypsum or anhydrite deposits, which are usually present in TSR cases. Fluid chemistry from fluid incluions and δ13C, δ18O and 87Sr/86Sr values from different diagenetic phases were measured to determine the dolomitization regime, and to assess the relationships between the occurrence of dolomitization and H2S accumulation. Dolomitization was initiated by seawater with slightly increased salinities (penesaline) at shallow depths prior to chemical compaction. The micro- to fine-crystalline cloudy dolomite formed with relatively high Na and Sr contents, and with δ13C, δ18O and 87Sr/86Sr values (2.2‰∼4.8‰, −3.9‰∼-5.0‰ and 0.70724∼0.70746, respectively) inherited from seawater. The evaporation of Permian seawater in back-reef and inter-reef lagoons during sea-level fall and the subsequent seepage reflux into reef-beach bodies led to greater Mg2+ and SO42− concentrations in higher-salinity pore waters. Further massive dolomitization was promoted by compactional flow of hotter residual seawater at shallow to intermediate depths and resulted in the formation of fine- to medium-crystalline clean dolomite with lower Na and Sr contents, more depleted δ18O values (-5.1‰∼-6.0 ‰), andδ13C (2.5‰∼4.8 ‰) and 87Sr/86Sr values (0.70726∼0.70741) similar to those of the coeval seawater. The relatively closed hydrodynamic system during burial facilitated SO42− preservation. The whole dolomitization process enriched porewater SO42−, which have been almost exhausted by subsequent TSR, accounting for the high present-day concentrations of H2S. The output of this study shows that similar scenarios involving dolomitization driven by condensed MgSO4 seawater and H2S accumulations can occur in evaporite-free settings across a broad range and deserves special attention during deep oil/gas exploitation.
Introduction
Massive dolostone formations have been found to correlate mostly with various fluids in different environments, e.g., normal or modified sea water and deep basinal fluids (e.g. Hardie, 1987; Kaufman, 1994a; Machel, 2004; Warren, 2000). Increasing documents have reported the effects of various models of dolomitization on the quality of dolomitic oil and gas reservoirs (Enayati-Bidgoli and Navidtalab, 2020; Hu et al., 2020; Jones and Xiao, 2005; Purser et al., 1994; Saller and Henderson, 1998; Vandeginste et al., 2009; Wilson et al., 2007). However, previous studies have ignored the fact that massive dolomitization events also result in significant changes in pore water chemistry, which would be succeeded by late diagenetic fluids. The deep extension of diverse dolomitizing fluids linked with different models may generate impressive diagenetic effects. For example, hydrothermal dolomitizing fluids are capable of transporting metal ions and heat, and, therefore, has the potential to provoke mineralization and possible acidification of fluids (Biehl et al., 2016; Davies and Smith, 2006; Shelton et al., 2019).
The presence of evaporitic tidal flats could provide hydrological and hypersaline fluids conducive to the deposition of massive dolostone (Adams and Rhodes, 1960; Jones et al., 2002; Kaufman, 1994a; Shields and Brady, 1995). High sulfate levels are commonly associated with elevated Mg/Ca ratios in hypersaline waters (Hardie, 1987). Warren (2000) emphasized that unlike nonevaporitic dolomitizing fluids, hypersaline fluids flushed from evaporite sources could form anhydrite cements, which might subsequently dissolve (Vandeginste et al., 2009). These sulfate-rich dolomitizing fluids would probably be succeeded by late diagenetic fluids during the following progressive burial stage. Li et al. (2014) claimed that the early refluxing of hypersaline dolomitizing fluids from evaporite-bearing strata had brought abundant reactive sulfates for TSR, leading to gas souring in the Upper Permian reservoirs in the Puguang gasfield (see the location in Figure 1B).
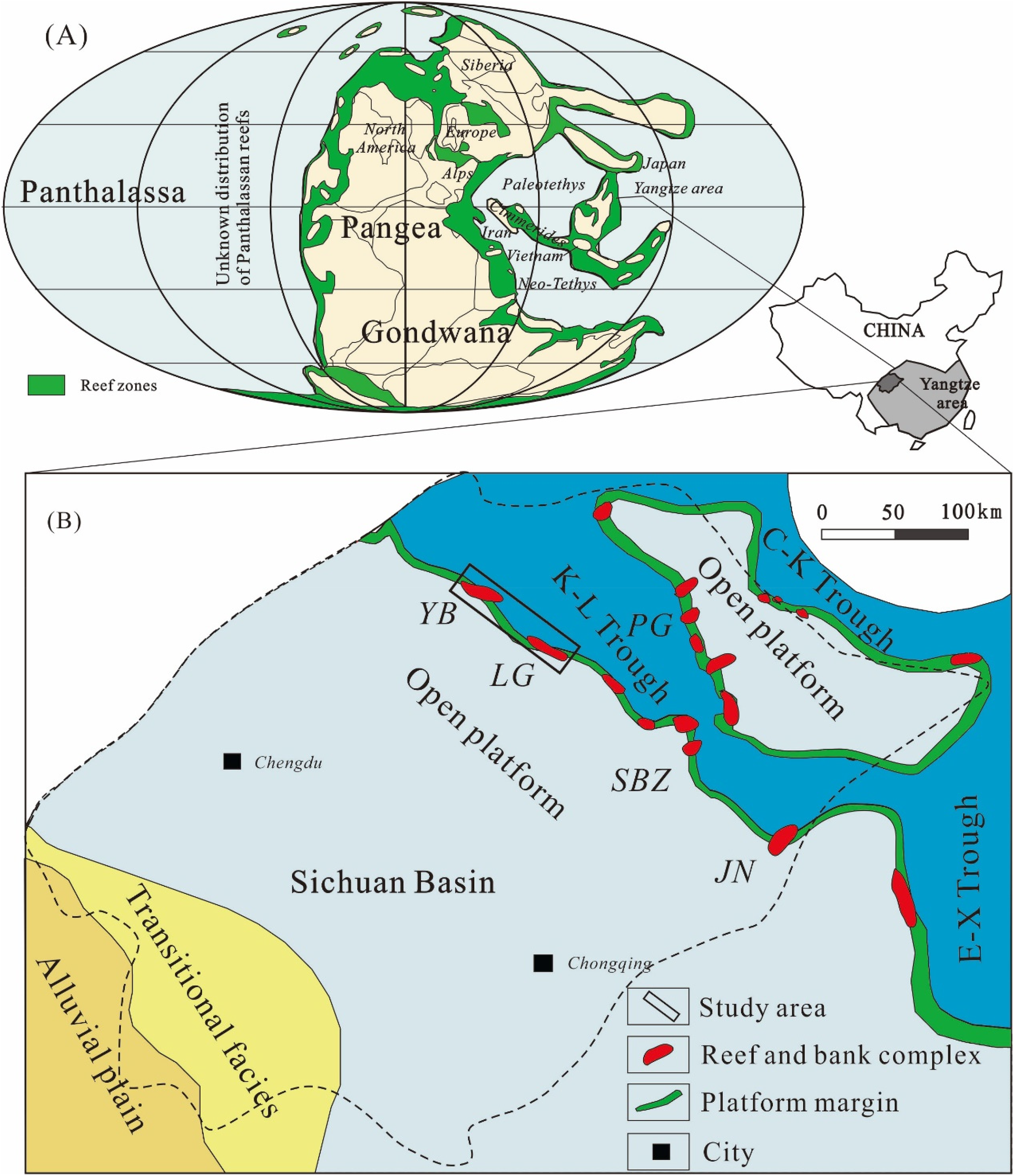
Paleogeography of (A) the earth (after Weidlich, 2002) and (B) Sichuan Basin (after He and Luo, 2010), including distributional patterns of reefs during late permian-early Triassic. The black box represents the study area. YB = Yuanba, LG = Longgang, SBZ = Shibaozhai, JN = Jiannan, PG = Puguang.
More than 1010 m3 of gas accumulations have been discovered recently in the Upper Permian dolomitized reef complexes in the Yuanba and Longgang gas fields in the northeastern (NE) Sichuan Basin (Figure 1). Elevated concentrations of H2S (mostly between 3.3% and 7.2%), of thermochemical sulfate reduction (TSR) origin (Li et al., 2014, 2016; Liu et al., 2014), is an unexpected non-hydrocarbon gas in the reservoirs. That's because rare evaporites, i.e., source of sulfate for TSR, have been found within the strata and the overlying oolitic limestone units in the Lower Triassic Feixianguan Formation (T1f) (Guo, 2011; Tian et al., 2014). The model proposed by Li et al. (2014) cannot be applied directly to the Yuanba gasfield because the absence of evaporites casts doubt on the hypersaline brine flushing model of pervasive dolomitization. It is noteworthy that the sour gas is present exclusively in dolostones rather than limestones, as indicated in previous studies (e.g. Li et al., 2016). This information yields instructive clues that there is a close relationship between the occurrence of TSR and dolomitization.
Unfortunately, the origin of the Upper Permian dolostones in the study area remains puzzled. Several dolomitization models, e.g., freshwater-seawater mixing models (e.g. Tian et al., 2014) and burial models (e.g. Cheng et al., 2013) have been proposed. Jiang et al. (2014) detected moderate-salinity fluid inclusions in dolomite crystals and claimed that these dolomites had formed from mesosaline brines. New data are therefore required to present and re-assess the hydrologic system, origin and evolution of the dolomitizing fluids, which might provide useful new insights into the source of reactive sulfate for TSR and the reason of H2S accumulation. Essentially, besides of dissolution of solid calcium sulfate, alternative sulfate sources, e.g., seawater or modified seawater, have been suggested by Machel (2001).
The main objectives of the current investigation are as follows:
To identify the origin and nature of the dolomitizing fluids in the Upper Permian bank and reef complex without extensive associated gypsum/anhydrite deposits; To account for sulfate evolution during and after dolomitization and explain why aqueous sulfate accumulated exclusively in dolostone horizons; To assess the intrinsic relation between massive dolomitization of Permian carbonates and TSR in the study area and other fields worldwide.
Geological setting
The Sichuan Basin, which is located in Southwest China, is a large intracratonic basin and hosts a 12 km-thick sedimentary succession deposited on top of the metamorphic basement of the Yangtze platform (Figure 1). Marine sedimentation dominated in the Sichuan Basin from the Upper Sinian to the Middle Triassic (Figure 2). During the late Permian to Early Triassic, the Yangtze area was an epeiric sea on the southeastern side of the Paleo-Tethys Ocean (Algeo et al., 2013). A low-latitude (Figure 1A) climate was prevalent and became increasingly arid into the Early Triassic. Continued petroleum exploration in the northeastern (NE) Sichuan Basin has resulted in the discovery of large gas fields, e.g., the Puguang gasfield on the eastern side of the Kaijiang-Liangping (K-L) Trough, and the Yuanba and Longgang gas fields on the western side (Figure 1B). High-production gas is commonly associated with deeply buried reef and shoal dolostone reservoirs in the Feixianguan (T1f) and Changxing (P3ch) Formations (Cai et al., 2004; Li et al., 2020). Geochemical signatures show that the hydrocarbons in the P3ch and T1f reservoirs were mainly sourced from the Upper Permian Longtan source rocks (e.g. Cai et al., 2017; Hao et al., 2008; Figure 2).
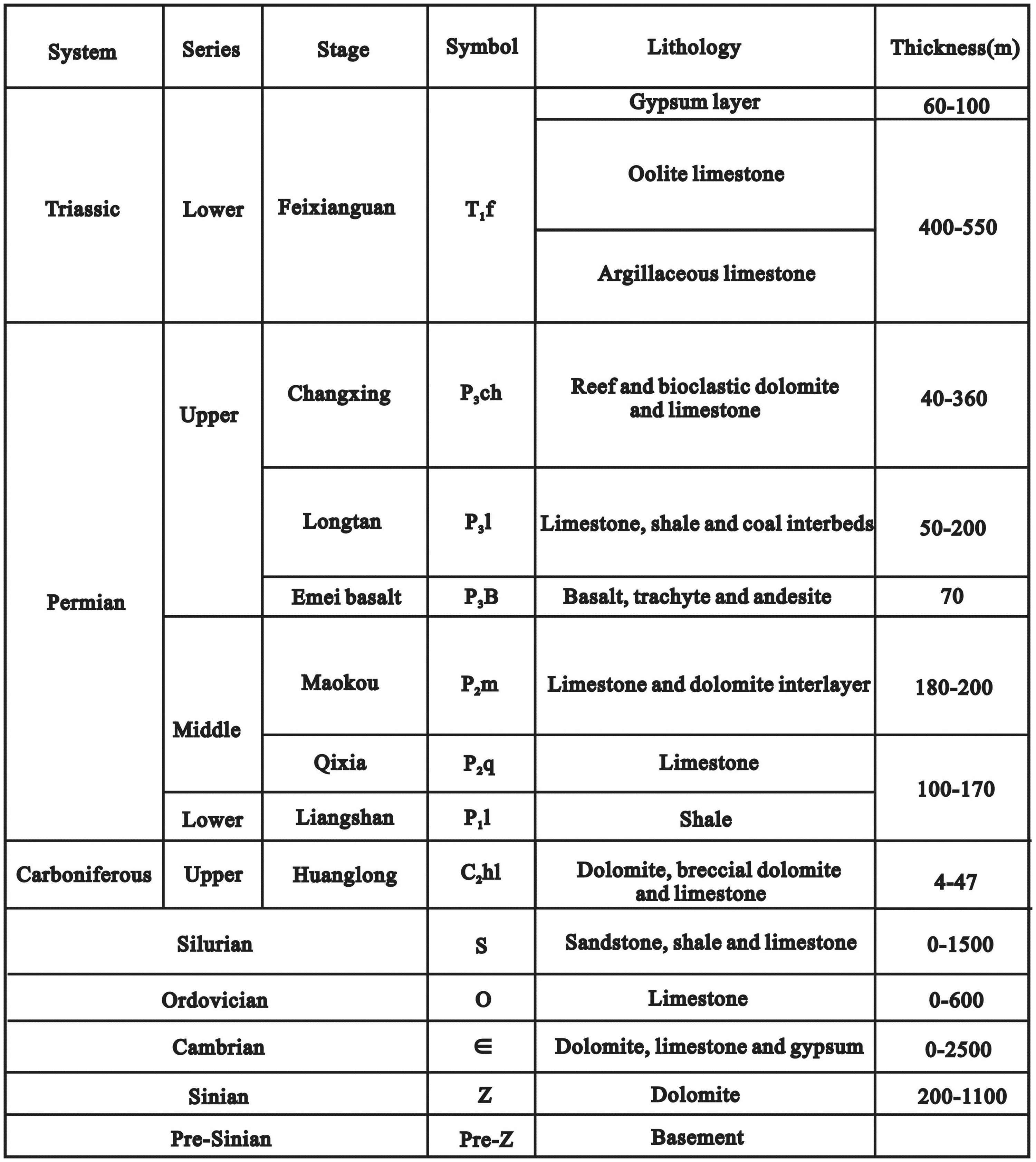
Generalized stratigraphic column of the northern sichuan basin.
The barrier reef on the western side, developed in P3ch, spreads narrowly along and parallel to the platform margin (Figure 1B), consistent with the distribution style of paleo-structural highs. Some patch reefs and bioclastic bank bodies are also scattered in the paleohighs of the backreef or inter-reef belts (Figure 3). The barrier and patch reefs are composed of gray limestones and dolostones, including bafflestone and framestone. The platform-margin bank facies in P3ch consists of thick-bedded to massive dolarenite, bioclastic dolostone and limestone (Figure 4). The platform-margin bank facies in the Lower Triassic is made up of oolitic limestone and calcarenite (Guo, 2011).
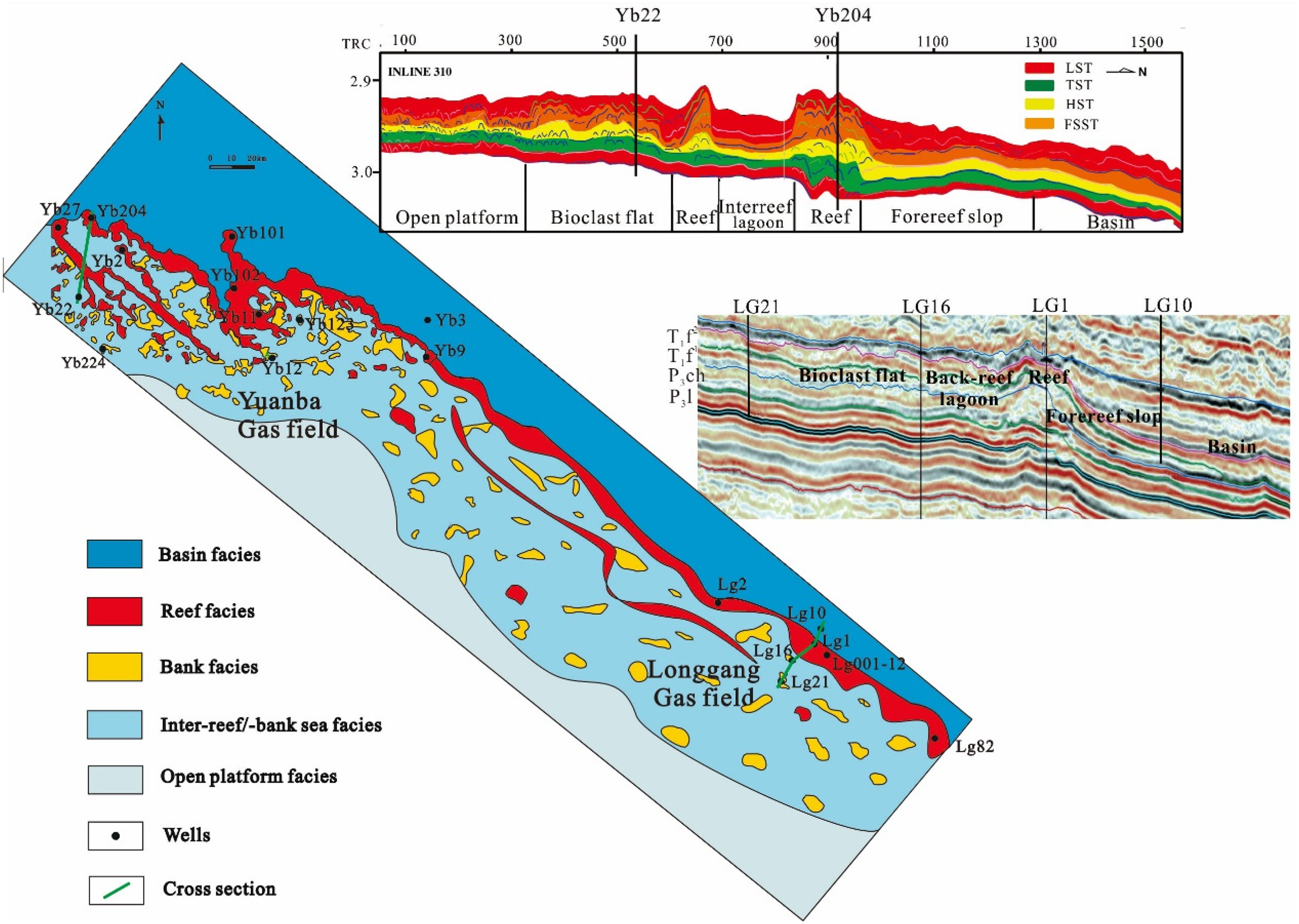
Map and seismic sections showing the sedimentary facies distribution of the late permian changxing formation (P3ch) in the Yuanba and longgang gasfields (modified from Huang et al., 2014 and Tian et al., 2014), and locations of gas wells.
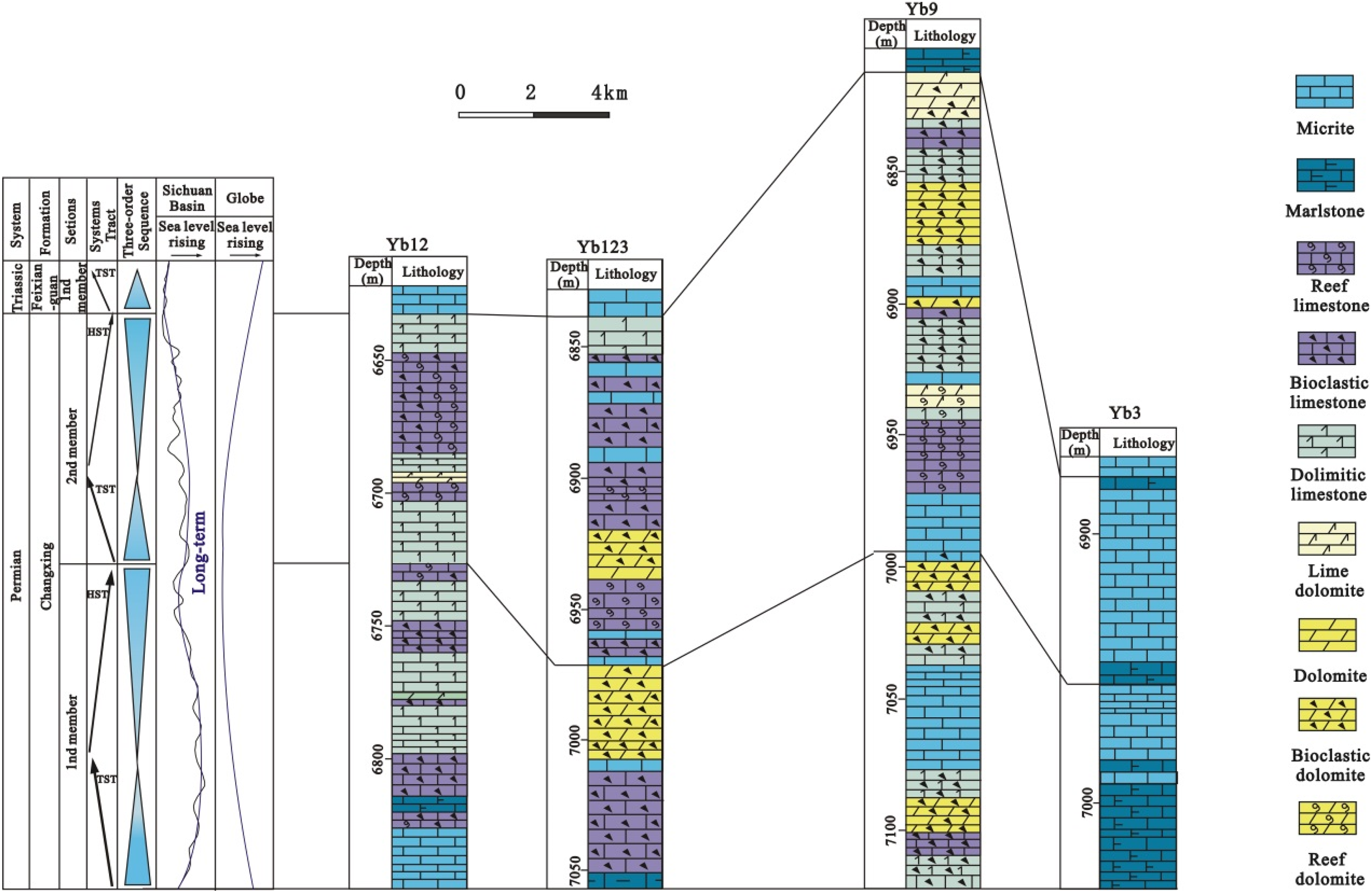
Correlation panel of wells YB12-YB123-YB9-YB3 showing lithology and sequence stratigraphy of the P3ch formation, and comparison of sea level variation during late Permian between Sichuan Basin and globe (simplified from Yin and Tong, 1996).
The Late Permian sequence stratigraphy of the reef trend in the NE Sichuan Basin has been extensively studied (e.g. Guo, 2011; Duan and Peng, 2014). Two third-order sequences have been recognized from the Changxing Formation and each sequence is composed of a transgressive systems tract (TST) and a highstand systems tract (HST; Figure 4). The TST of the first sequence is composed of interbank deposits within an open platform, and its HST comprises a set of reef-bank deposits belonging to platform-margin facies. The TST of the second sequence is characterized by a set of reef-bank deposits, and its HST is mainly a set of deposits belonging to exposed shallow-bank facies (Guo, 2011). The sea level gradually rose from the Longtanian towards Changxingian, causing reef growth, and regression did not occur until the end of the Permian; this sequence is different from the global sea level, which fell to the lowest point and later rose during the Changxingian (Figure 4). The difference might have been caused by the rapid northward migration of the Cimmerides and the approach of the Eurasian continent, which gradually closed the Paleo-Tethys from east to west (Yin and Tong, 1996).
The burial and geothermal history has been elucidated in previous studies (Cai et al., 2014). Rapid sedimentation took place during the Early Triassic and the Middle and Late Jurassic, and the strata experienced maximum burial (more than 10,000 m) and heating to about 220°C during the Early-Middle Cretaceous. Significant uplifts occurred during the Cenozoic because of the Hercynian orogeny, leading to a decrease in temperatures in the P3ch-T1f reservoirs to 120∼130°C at present.
The diagenetic history of the Upper Permian Changxing Formation limestones of the NE Sichuan Basin has been summarized in our previous work (Li et al., 2012). Submarine diagenesis led to precipitation of acicular or fibrous circumgranular calcite cement-1, which is followed by bladed and granular calcite cement-2 of meteoric origin. Subsequently, calcite cement-3 precipitated and occurred as mosaic to poikilitic crystals in the center of pores. This calcite cement shows dull red to non-luminescent under CL and posses a high range of homogenization temperatures (HTs) from about 90 to 130°C, with a distinct population at around 100°C. Post-bitumen void-filling calcite is present in low amounts as coarse mosaic to poikilotopic crystals, growing outward from bitumen-coated pore walls.
Materials and analytical methods
Late Permian cores of variable length (over 2,500 m in total, with individual cores ranging from 150 to 300 m) were examined from 14 wells, among which 11 were from the Yuanba Field and 3 were from the Longgang Field. The selected wells reach different sedimentary facies, including reef facies (barrier and patch reefs), bank facies, inter-reef or inter-bank sea facies and forereef slope facies (Figure 3). Approximately 120 samples of Upper Permian carbonates were taken from these 14 wells and examined by polished thin section petrography. Samples were collected at high resolution (sampling interval of 3∼5 m) from partially to completely dolomitized intervals. Of these, 30 bulk carbonates and 6 void-filling calcite samples were studied using cathodoluminescence (cold-CL) petrography, electron microprobe (EMP) analyzes and isotope determination. CL analyzes were performed using a Technosyn cold cathodoluminescence stage with a 15 kV beam and a current intensity of 500 μA. EMP analyzes were carried out to determine the major and trace element contents using a Cameca Camebax BX 50™ instrument equipped with three spectrometers. The precision of analyzes was better than 0.5 mol%.
Bulk dolostone samples were drilled using a dentist drill and analyzed for C, O and Sr isotopes. Approximately 50 mg of dolomite powder was reacted with anhydrous H3PO4 acid at 25°C for 24 h and for 72 h. The extracted CO2-gases were analyzed with a Finnigan MAT-252 mass spectrometer. The δ13C and δ18O values are reported as ‰ relative to the Vienna Peedee Belemnite (VPDB) standard. The precision was ± 0.1‰. For 87Sr/86Sr measurement, carbonate sample powders (50–100 mg) were dissolved in 2.5 N HCl. Then, the sample solution was loaded onto the pre-conditioned resin column with 2 mL of AG50W × 12 (200–400 mesh) for the separation of Sr from the sample matrix. After rinsing four times with 0.5 mL of 2.5 M HCl, the column was washed with 7 mL of 5 M HCl. Afterwards, the Sr fraction was stripped with 3.5 mL of 5 M HCl. The 87Sr/86Sr ratios were measured on a Finnigan MAT-261 mass spectrometer, and the precision for 87Sr/86Sr measurement was ± 0.00003–0.00007. Four samples of solid bitumen samples underwent δ34S analysis, with results reported relative to the Canyon Diablo troilite (CDT) standard.
Five post-bitumen calcites showing coarse vug/fracture-filling spar from sour reservoirs were selected for homogenization temperatures and major ion composition analysis of fluid inclusions. For crush-leach analyzes, 1 g pure giant coarse milky white calcite crystals were washed several times, and then crushed in 5 ml of ultraclean water (Vandeginste et al., 2009). After thorough crushing, the water solution was decanted/filtered to yield a clean leachate and then weighed. The solution was then analyzed by a Dionex DX500 ion chromatograph. To test the analytical procedures, triplicate and blank tests were performed for each sample. Absolute ion concentrations were calculated, taking the weight of fluid inclusion water into consideration. SO42−/Cl− are calculated using the values of SO42− and Cl− concentrations. The formation water samples from the Yuanba and Longgang gas fields were analyzed by using ion chromatography. The details of this measurement procedure followed the Oil and Gas Industry Standard of China.
Results
Dolomite distribution and petrography
Pervasive dolomitization of carbonate build-ups in the Changxing Formation occurred mainly in platform–margin reef and bank deposits, and the extent of dolomitization decreases basinward (Figures 3, 4). Reefal build-ups, reef caps and flank deposits show extensive dolomitization whereas scarce to minor dolomitization occurs in the reef cores (Lei et al., 1994; Tian et al., 2014). Bioclastic limestones in the back-reef and intraplatform flats are partially to completely dolomitized. Vertically, the distribution and abundance of the most massive dolostones are stratigraphically controlled. The regressive facies of third-order depositional sequences tend to be dominated by cycles of extensive dolomitization, whereas abundant limestones are not dolomitized in transgressive facies (Figure 4).
Five types of dolomites are distinguished based on crystal size, distribution, and geometry (Sibley and Gregg, 1987; Tucker and Wright, 1990): (1) very finely to finely crystalline non-plana replacement dolomite (RD1), (2) finely to medium crystalline cloudy non-planar replacement dolomite (RD2), (3) medium to coarsely crystalline planar-e clean replacement dolomite (RD3), (4) finely to medium crystalline planar-s dolomite cement (CD1), and (5) medium to coarsely crystalline planar-e dolomite cement (CD2).
RD1 is irregularly distributed, forming a patchy pattern in reef-bank facies where it partially to completely replaces the algal mud matrix and selectively replaces grains in packstones/grainstones. Strata-bound dolostones are also found locally in the reef caps, back-reef or inter-reef belts and restricted to supratidal facies. There is a progressive increase in abundance of RD1 with increasing ration of tidal-flat to subtidal facies in cycles. RD1 is volumetrically minor and usually accounts for less than 10% of the total rock volume. RD1 consists of 10–30 μm crystals, with nonplanar textures and a uniform orange to dull reddish appearance in CL images (Figure 5A, B). The crystals are generally rich in inclusions and retain the fabric. Various types of euryhaline organisms, such as foraminifers, tubiphytes and crinoids, can usually be identified within RD1 (Figure 5C). RD2 occurs in almost all facies and is volumetrically the most important (accounting for ∼45% by volume of all dolomite phases). It commonly exhibits a decrease in abundance downward from the supratidal facies and preferentially formed in the top reef and shoal build-ups and in the flanks rather than the lower and inner parts. RD2 crystals range from 30 to 200 μm in size and occur as nonplanar textures with a cloudy appearance (Figure 5D, E). RD2 partially preserves fabrics and commonly shows moderately bright orange–red CL colors when it is very finely crystalline, while it forms non-mimetic patches with dull red CL colors when it is coarser (Figure 5D, F). RD3 occurs as single rhombs or scattered patches of euhedral porphyrotopic dolomite crystals in slightly dolomitized reef and bioclastic limestone facies (Figure 6A, B) or as pervasive replacive dolostone in thin to massive (several m thick) dolostone intervals (Figure 6C, D). RD3 preferentially appears in close association with stylolites (Figure 6 A, B), accounting for 30∼40% of the total volume. RD3 is characterized by fine to medium crystal sizes (average sizes, 80–400 μm) and commonly shows inclusion-rich cores surrounded by more euhedral limpid dolomite rims. It occurs as completely fabric-destroyed and planar-s to planar-e mosaics, showing purple or dull red CL colors (Figure 6C, D). The massive dolostones usually exhibit abundant vuggy and intercrystalline porosities (Figure 6C). CD1 appears as bladed and granular crystals surrounding isopachous circumgranular calcite cement-1 in partially dolomitized layers in the upper part of the reef-beach bodies (Figure 7A, B). CD1 resembles calcite cement-2 in the limestone with similar crystal sizes and shapes (Figure 7A), indicating the CD1 crystals are more likely replacive rather than cements filling in void space. CD1 crystals range from 50 to 200 μm in size and occur as planar-e and planar-s textures, which resemble the precursor granular calcite cement-2 (Figure 7A, B). CD1 shows moderately bright orange–red luminescence (Figure 7C), which is similar to that of RD2 dolomite (Figure 5D). The formation of CD1 is followed by a mosaic to poikilitic calcite cement-3 with dull brown to nonluminescent staining under CL (Figure 7A, B, C). CD2 partially to completely fills mouldic, vuggy and fracture porosity in almost all facies, and is characterized by increasing crystal sizes towards the centers of cavities (Figure 7D, E). CD2 occurs as clear and fine to medium-sized crystals and shows dull red colors similar to those of RD3 (Figure 7F).
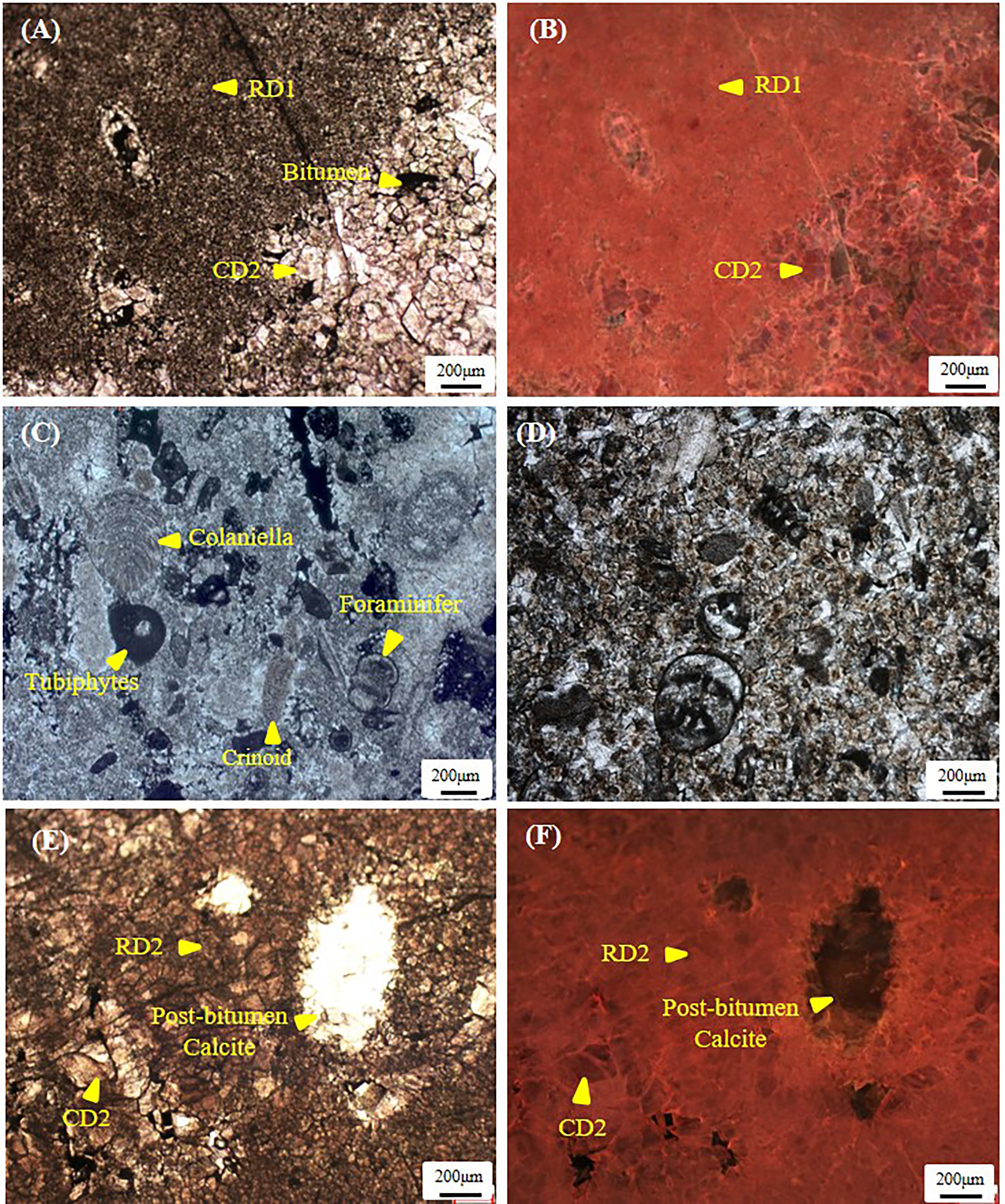
Paired polarized and cathodoluminescent photomicrographs showing RD1 and RD2 in the P3ch formation. (A) RD1 are generally microcrystalline and inclusion-rich. Local solution vugs are filled with fine-crystalline CD2 crystals, Well LG2, 5,635 m. (B) The same view as (A), but taken under cathodoluminescence (CL). RD1 show a uniform orange–dull red CL colors while CD2 dolomites show purple or dull-red CL colors. (C) Various types of euryhaline organisms, e.g., foraminiter, colaniella, tubiphytes and crinoid, can be identified, Well Yb2, 6,592 m; (D) RD2 shows a relatively wide spread in crystal sizes and a cloudy appearance, with euryhaline organisms partially preserved. (E) RD2 shows a relatively wide spread in crystal sizes and a cloudy appearance, Well Yb204, 6,548.6 m. (F) RD2 shows moderately bright orange–red CL colors.

(A) RD3 occurs as single rhombs or scattered patches of euhedral porphyrotopic dolomite, closely associated with stylolites, well Yb11, 6,912.2. (B) RD3 is dull red in color under CL. (C) RD3 occurs as pervasive dolomites, Well LG2, 6,129 m. (D) Under CL the RD3 crystals are purple or dull red in color.

Photomicrographs of two types of dolomite cements (CD1 and CD2) in the P3ch formation. (A) Stained section showing CD1 dolomite as circumgranular bladed and granular crystals and interparticle stage II calcite cements (red spars), Well Yb2. (B, C) Paired polarized and cathodoluminescent photomicrographs of CD1 dolomite, showing moderately bright orange–red luminescence, Well Yb204, 6,550.6 m. (D) CD2 dolomite occurs as clear fabric-obliterative vug-filling crystals, Well Yb123. (E, F) Paired polarized and cathodoluminescent photomicrographs of CD2 dolomite, showing purple or dull red CL colors, Well LG2, 6,118 m.
Geochemistry
Carbon, oxygen and strontium isotopes
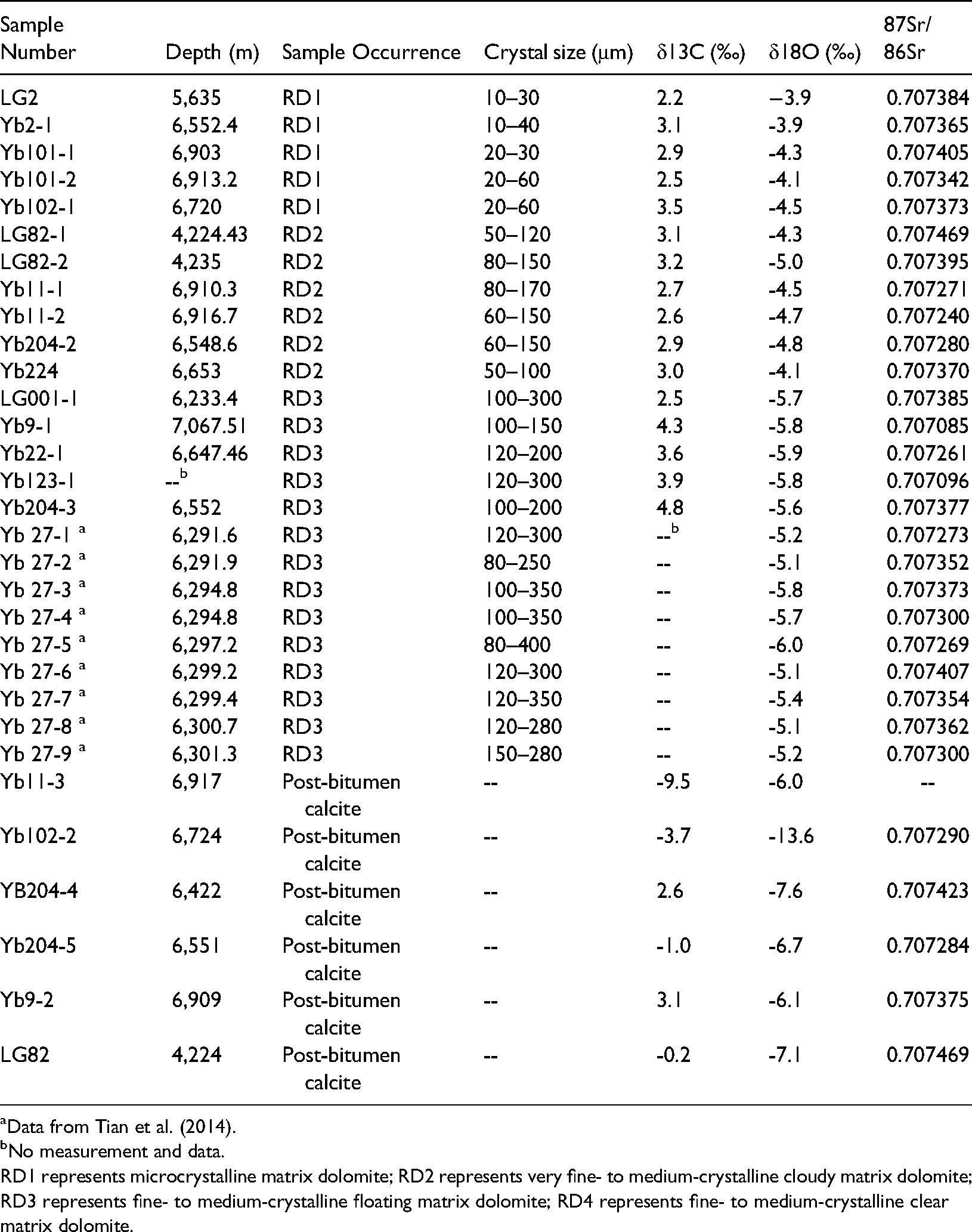
C, O and Sr isotopes of dolomites from the Upper Permian Changxing Formation are listed in Table 1 and plotted in Figure 8. Oxygen isotope values show a gradual decrease from RD1 dolomite to RD2 and RD3 dolomites. RD1 dolomite samples show the least depleted δ18O values of −3.9 and −4.5‰ (n = 5, mean −4.1‰) relative to other generations of dolomite. However, these values are lower than those of hypothetical Upper Permian marine dolomites of −2.2 to −4.3‰ (e.g. Korte et al., 2004; Tian and Zeng, 1995). RD2 dolomite samples have δ18O values ranging from −4.1 to −5.0‰ with an average of −4.6‰ (n = 6). RD3 dolomite has the lowest or most depleted δ18O values (-5.1 to −6.0‰, n = 14) with a mean of −5.5‰. RD1 and RD2 dolomite samples have similar narrow δ13C values from 2.2 to 3.5‰ (n = 11). RD3 dolomite shows a slightly wider spread at 2.5∼4.8‰ (n = 8, Figure 8A). Despite minor differences, the δ13C values of the massive dolomite are in the range of those of Late Permian seawater (Huang, 1994; Korte et al., 2004).
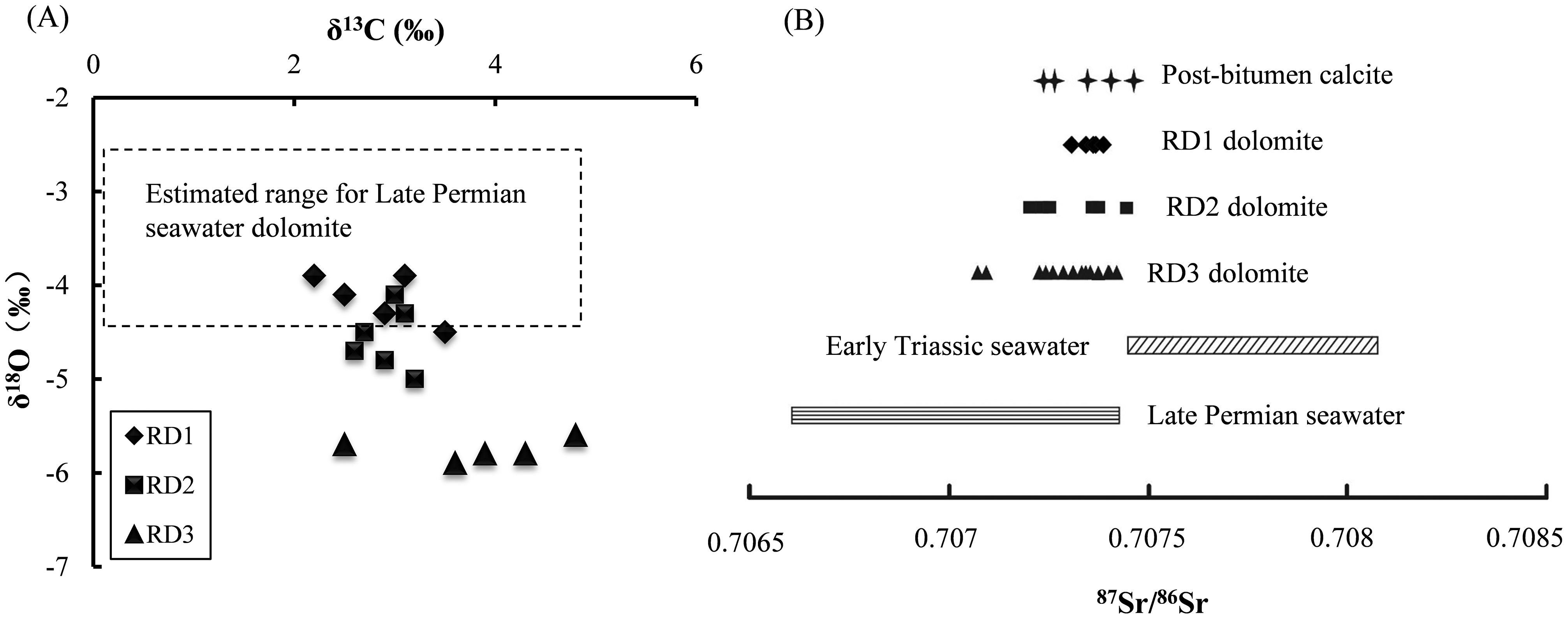
Carbon, oxygen and strontium isotopic composition of RD1, RD2 and RD3 dolomites and TSR-calcites. (A) Cross plots showing the relationships of δ13C values to δ18O values for RD1, RD2 and RD3 dolomites; (B) 87Sr/86Sr ratios of massive dolomite and post-bitumen calcite in comparison with Late Permian and Lower Triassic seawater. Data of Late Permian seawater are from Burke et al. (1982) and Korte et al. (2004) and data of Lower Triassic seawater are from Korte et al. (2003).
Sr, C and O isotopic compositions of various types of dolomite and post-bitumen calcite from the P3ch reservoirs.
Data from Tian et al. (2014).
No measurement and data.
RD1 represents microcrystalline matrix dolomite; RD2 represents very fine- to medium-crystalline cloudy matrix dolomite; RD3 represents fine- to medium-crystalline floating matrix dolomite; RD4 represents fine- to medium-crystalline clear matrix dolomite.
The replacive dolomite and the post-bitumen calcite samples have Sr isotope ratios ranging from 0.70709 to 0.70747 (n = 25, Table 1, Figure 8B), most of which are within the range of Late Permian seawater (0.7069∼0.7074, Burke et al., 1982; Korte et al., 2004). These values are much lower than those of Lower Triassic seawater (0.7074–0.7082, Korte et al., 2003).
Major and trace element contents
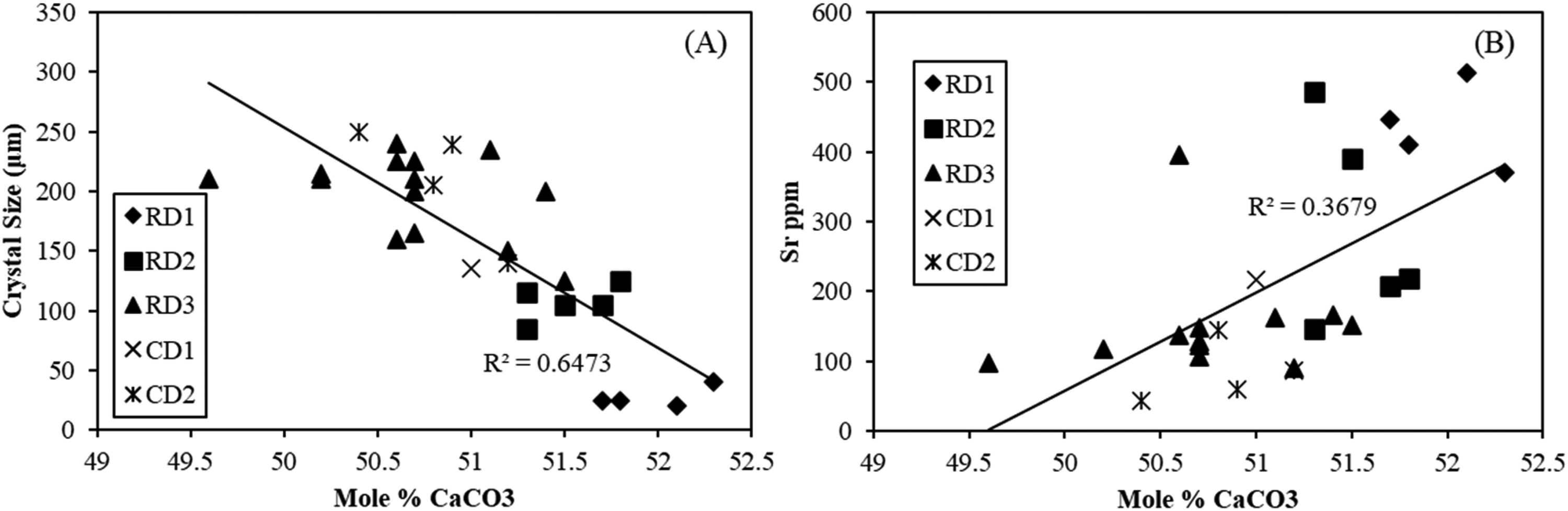
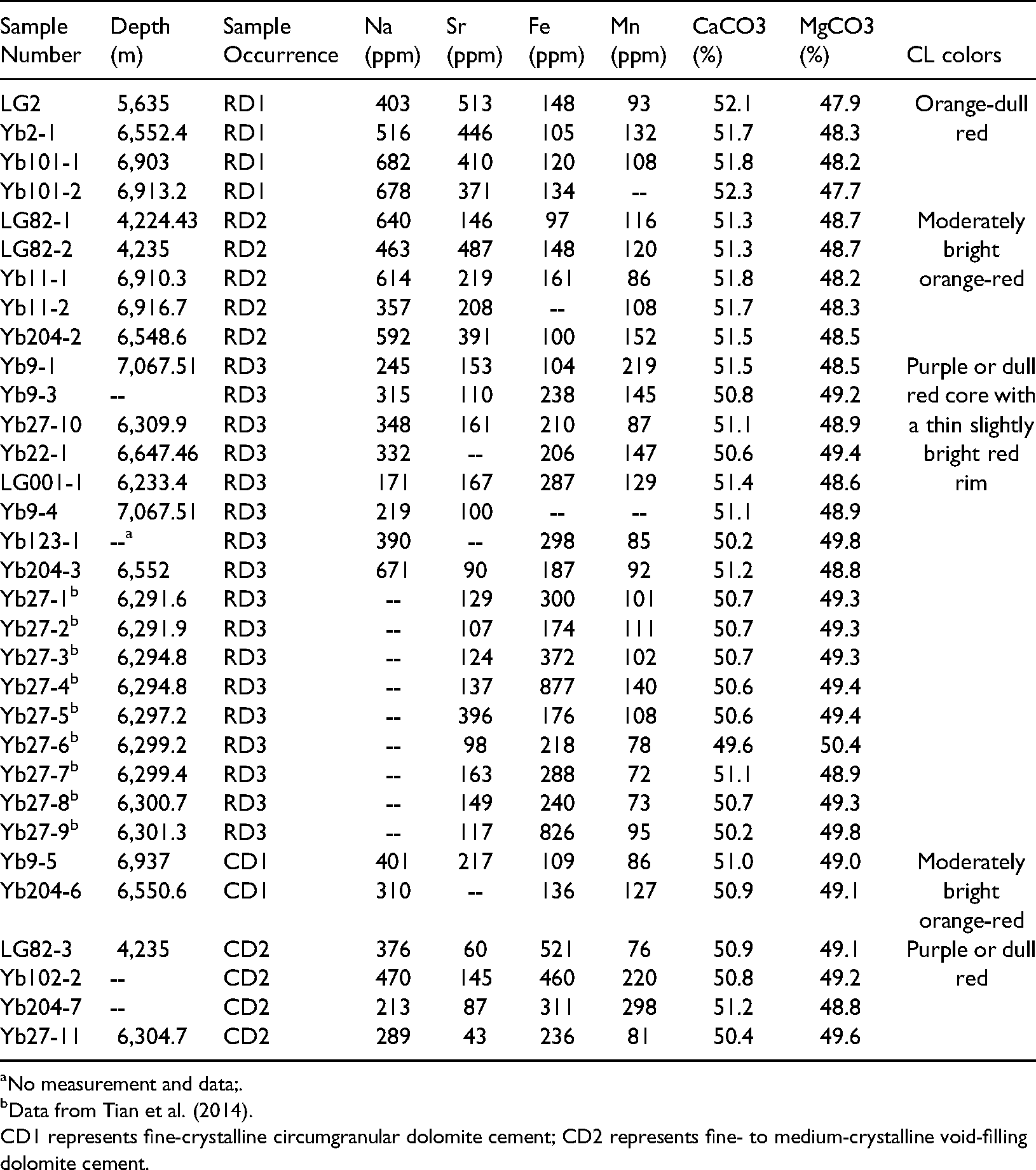
All dolomites, regardless of replacement or cement type, are near-stoichiometric to stoichiometric (49.6 to 52.3 mol% CaCO3) (n = 32, Table 2). Typically, stoichiometry increases with increasing crystal size (R2 = 0.65, Figure 9A). The mole % CaCO3 generally decreases from RD1 dolomites (51.7∼52.3 mol%, n = 4) to RD2 (51.3∼51.8 mol%, n = 5) and RD3 (49.6∼51.2 mol%, n = 17).
Cross plots showing the relationships of dolomite crystal size and CaCO3 content (A), Sr and CaCO3 content of dolomite (B).
Elemental geochemistry and CL colors of different types of dolomite.
No measurement and data;.
Data from Tian et al. (2014).
CD1 represents fine-crystalline circumgranular dolomite cement; CD2 represents fine- to medium-crystalline void-filling dolomite cement.
All dolomite types have low Sr contents (90∼513 ppm, Table 2). There is a weak correlation between the Sr contents and CaCO3 contents of the matrix dolomites and the dolomite cements, with R2 of 0.37 (Figure 9B). Strontium concentrations of RD1 and RD2 dolomites (371∼513 ppm and 146∼487 ppm, respectively) are higher than those of dolomite precipitating from normal seawater (∼125 ppm) (Machel and Anderson, 1989; Malone et al., 1996; Swart et al., 2005). RD3 and CD2 dolomites have relatively lower contents (90∼396 ppm and 43∼145 ppm, respectively). Iron concentrations of RD1 and RD2 dolomites range from 97 to 161 ppm (n = 8), and those of RD3 and CD2 are slightly enriched at 174∼877 ppm (n = 20). The replacive dolomites contain similar manganese concentrations from 72 to 219 ppm (n = 26).
The Na contents of all the dolomites show a wide range from 171 ppm to 682 ppm (n = 17). RD1 and RD2 dolomites have slightly higher Na contents (403∼682 ppm and 357∼640 ppm) than RD3 (mostly 171∼390 ppm, Table 2).
Fluid inclusion and formation water chemistry
The cation and anion compositions of fluid inclusions in post-bitumen calcite crystals and formation water samples from the Yuanba and Longgang gas fields are presented in Table 3. A progressive decrease in SO42− concentrations is accompanied by an increase in homogenization temperatures (Th) of the fluid inclusions (Figure 10A). The fluid inclusion waters have SO42−/Cl− ratios (5.4∼10.4, expressed in weight units) higher than and Na+/Cl− ratios similar to those of Late Permian seawater (0.11 and 0.54, respectively, Figure 10B). The P3ch formation waters within dolostone layers show significantly lower SO42−/Cl− ratios from 0.001 to 0.019 and lower SO42− concentrations from 0.2 g/L to 0.6 g/L. In contrast, the formation waters within limestone layers show SO42−/Cl− ratios from 0.04 to 0.12 and SO42− concentrations from 1.3 g/L to 1.9 g/L, which are similar to those of the contemporary seawater (Horita et al., 2002; Lowenstein et al., 2005). The formation waters within the limestone and dolostone layers show a wide range of Na+/Cl− ratios from 0.01 to 0.66.
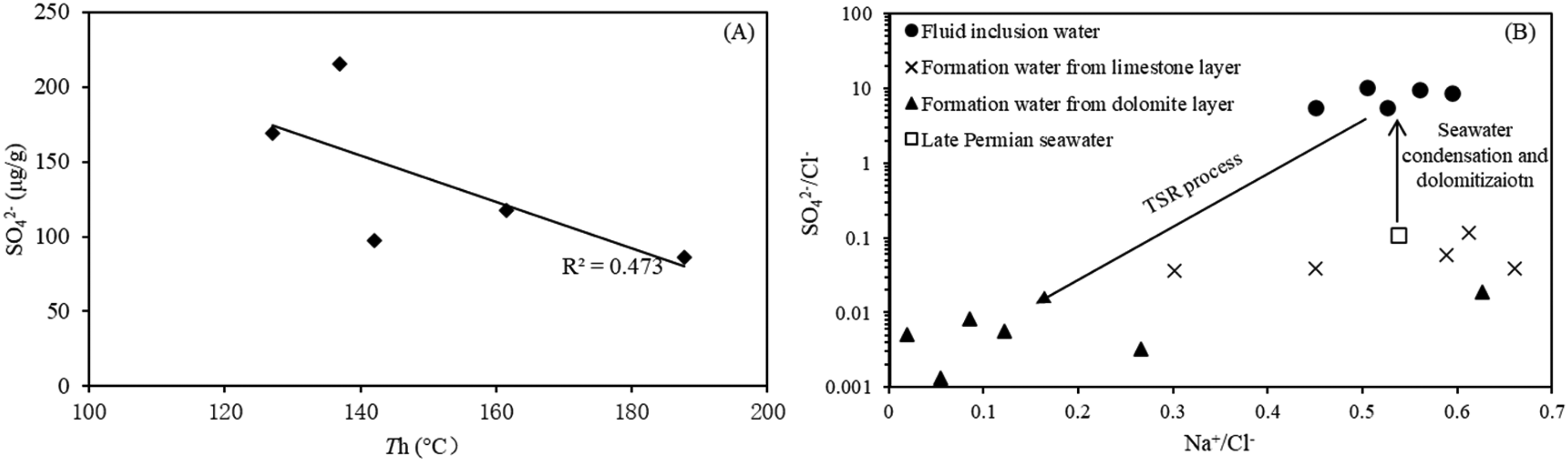
Variation of major ions concentrations of the paleo-fluids during the whole diagenesis of the Permian carbonates. (A) SO42− concentrations of fluid inclusion waters vs. Th. (B) SO42−/Cl− ratios vs Na+/Cl− in Permian seawater, fluid inclusion waters and formation waters.
Chemical compositions of fluid inclusion waters from post-bitumen calcite crystals and formation water from the P3ch Formation. The concentrations of H2S are for the associated gas reservoirs.
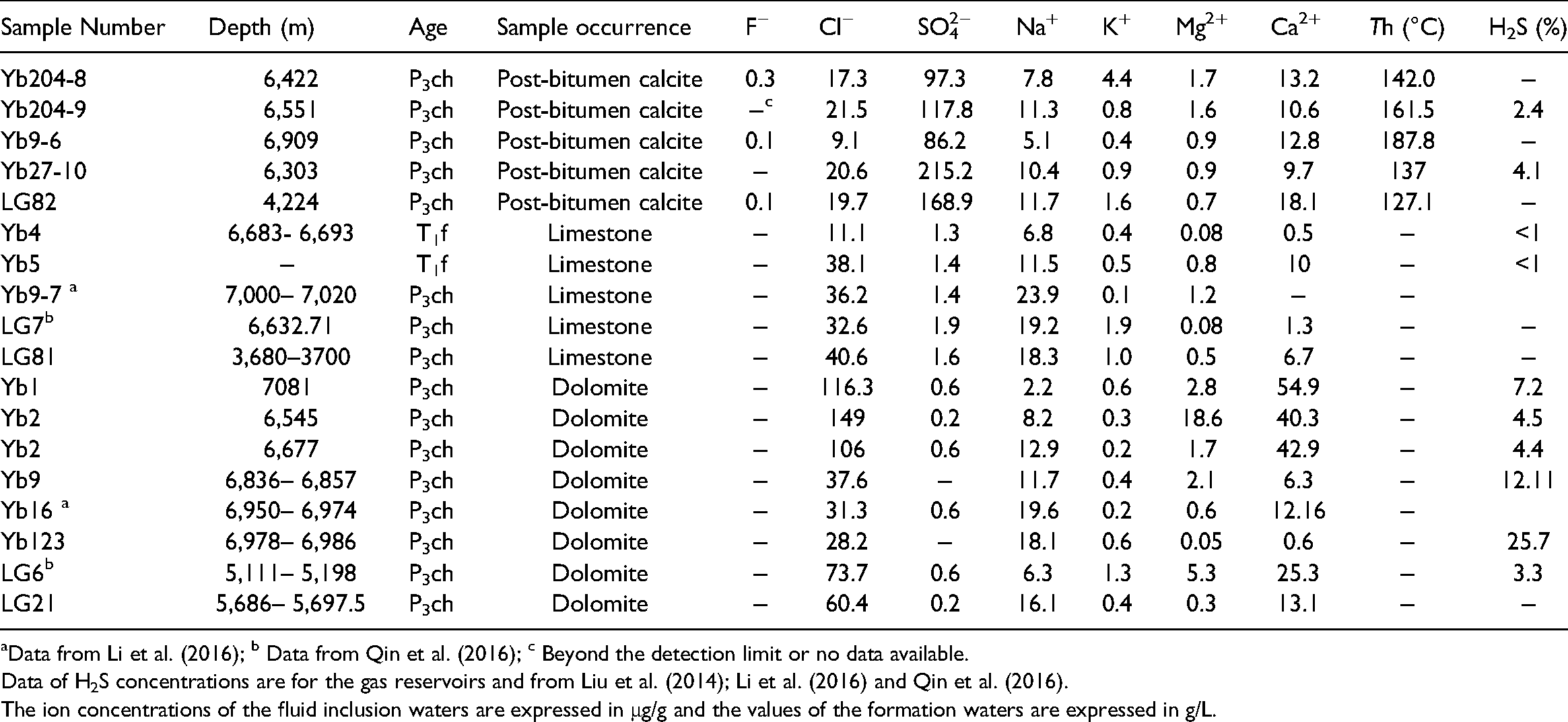
Data from Li et al. (2016); b Data from Qin et al. (2016); c Beyond the detection limit or no data available.
Data of H2S concentrations are for the gas reservoirs and from Liu et al. (2014); Li et al. (2016) and Qin et al. (2016).
The ion concentrations of the fluid inclusion waters are expressed in μg/g and the values of the formation waters are expressed in g/L.
Δ34s values of solid bitumens
Solid bitumen in reservoir rock samples associated with gas containing H2S > 4.1% shows δ34S values ranging from 17.7‰ to 24.5‰ (n = 4, Table 4), which are close to the range of Late Permian seawater that has δ34SCAS values of 11.5–26.9‰ (Newton et al., 2004).
S isotopic compositions of solid bitumen from the P3ch reservoirs. The concentrations of H2S are for the associated gas reservoirs.

Discussion
Time and condition of dolomitization
RD1 and RD2 in the extensive dolomitized zones and CD1 in the slightly dolomitized limestone lithofacies may have formed during the same period of dolomitization (Figure 11), as indicated by their identical CL colors, crystal sizes and shapes (Figure 5; Figure 7 C). Dolomite with a near-micritic to finely crystalline texture and relative fabric retention (Figure 6A, C, D) is typically formed under near-surface, low-temperature and saline conditions (Gregg and Sibley, 1984). This interpretation is further supported by the fact that the formation of CD1 dolomite in the slightly dolomitized limestone lithofacies was followed by precipitation of mosaic to poikilitic calcite cement-3 with dull brown to nonluminescent appearance under CL (Figure 7A, B). In our previous study (Li et al., 2012), this calcite cement was demonstrated to have precipitated from hot (90–130 °C) and reduced pore water in shallow to intermediate-depth burial environments. That is, early dolomitization occurred at shallow depths prior to chemical compaction.
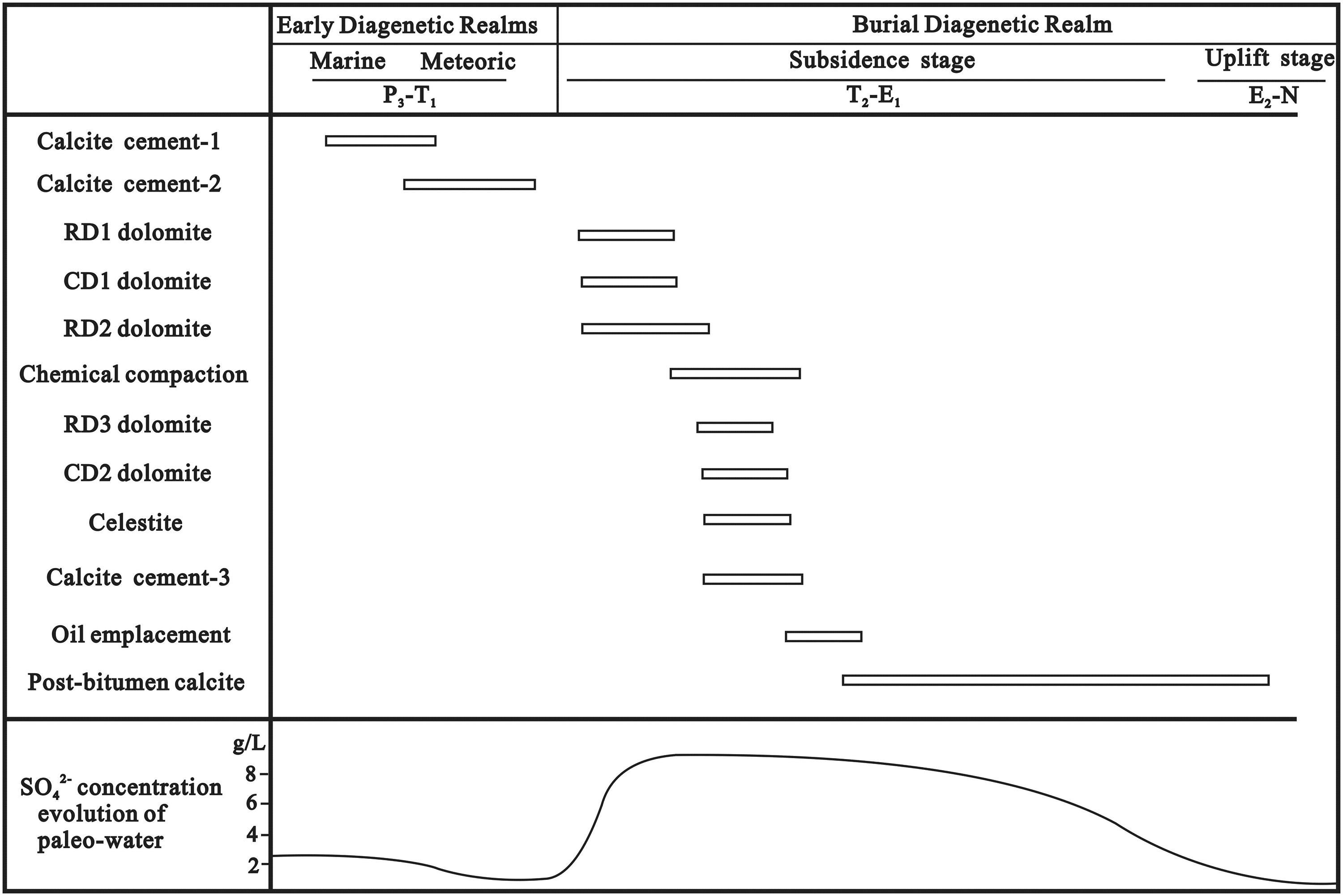
Synthetic paragenetic sequence showing main stages in the diagenetic evolution of P3ch carbonates and variation of SO42− concentrations of paleo-waters through time. Celestite was recognized by Li et al. (2012).
Most of the RD1 and RD2 samples have carbon and oxygen isotope values and strontium isotope ratios within those estimated for Late Permian seawater (Figure 8A, B; Korte et al., 2004), indicating that the dolomite was precipitated from solutions with an isotopic composition comparable to that of contemporary seawater or slightly modified seawater. With the δ18OSMOW (Vienna standard mean ocean water, SMOW) value of Late Permian seawater at −1‰ (Melim and Scholle, 2002), the precipitation temperatures of RD1 and RD2 dolomites are calculated to be 47.1–49.3°C and 49.3–53.2°C, respectively (Figure 12), using their δ18O values (Table 1) and the dolomite–water fractionation equation of Sheppard and Schwarz (1970). The corresponding dolomitization depths of RD1 and RD2 are estimated to be 479–546 m and 512–667 m, assuming a palaeotemperature of 30°C–33°C for Late Permian seawater (Joachimski et al., 2012) and a geothermal gradient of 3.17°C/100 m (Wang and Xiao, 2010). These confirm that replacement took place at shallow burial, which is consistent with the textural features of dolomites.
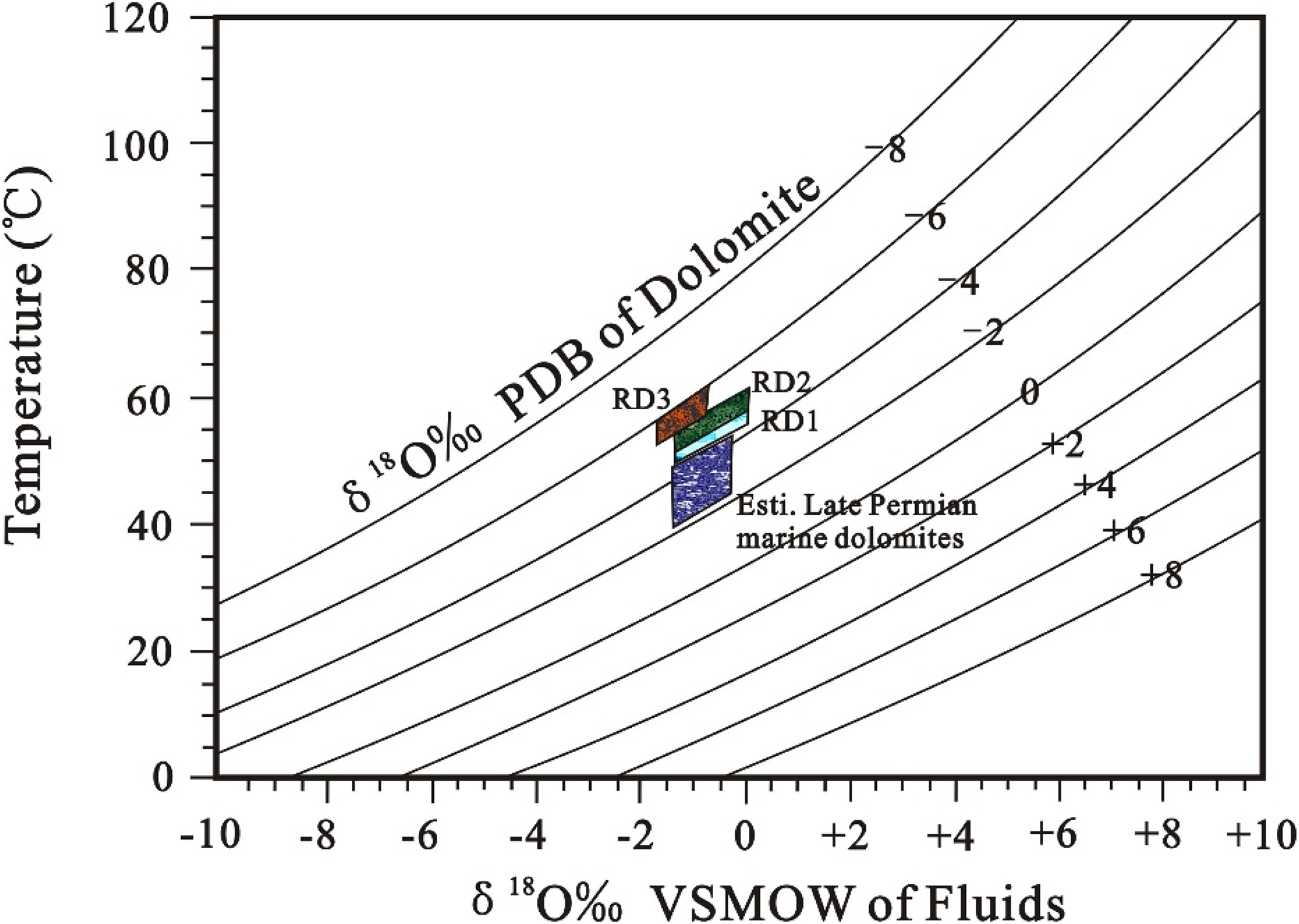
Relationship between δ18O of dolomite, δ18O of water, and temperature, according to 103 lnα = 3.2 × 106/T2-3.3 (Sheppard and Schwarz, 1970). See text for explanations.
The Na contents of RD1 and RD2 dolomites (357∼682 ppm, Table 2) are significantly higher than those of dolomites precipitated from normal seawater (∼185 ppm) (Qing and Mountjoy, 1988), suggesting a saline dolomitizing fluid (e.g. Land and Hoops, 1973; Sass and Bein, 1988). This result is consistent with the high Sr contents of RD1 and RD2 dolomites (Figure 9B, Table 2) because the precipitation rates of dolomite and the Sr distribution coefficient (KDSr) are usually high in evaporative brines (Land, 1980; Lorens, 1981; Montanez and Read, 1992). Generally, the high contents of Na and Sr in these two types of dolomites, the small crystal sizes (Table 1), and the planar-s and nonplanar fabrics (Figure 5A, C, D), are typical of brine reflux dolomites (e.g. Machel, 2004; Melim and Scholle, 2002; Rameil, 2008). However, the absence of gypsum/anhydrite (Figures 2, 4) suggests that evaporated seawater did not reach the salinities required for abundant gypsum precipitation. Therefore, the RD1 and RD2 dolomites are considered to possibly have precipitated in restricted lagoonal seawater with slightly elevated salinity, that is, under penesaline conditions. This situation has been invoked to explain extensive dolomitization in the absence of gypsum/anhydrite deposits in both experimental and field studies (e.g. Eren et al., 2007; Kaufman, 1994b; Qing et al., 2001; Rameil, 2008; Sun, 1994). Such penesaline conditions are in good accordance with the common observation of euryhaline organisms, consisting, for example, of mainly foraminifers, tubiphytes and crinoids in RD1 (Figure 5C).
Considering the paleoenvironment of the study area, seawater with elevated salinity could have developed in back-reef and inter-reef lagoons (Figure 3; Figure 13) during sea-level fall, as suggested in previous studies from other fields (e.g. Hurley and Lohmann, 1989; Mutti and Simo, 1994). Downward flux likely occurred when brines from overlying restricted lagoon facies became dense enough to pass directly through the gently sloping platform margin, resulting in early massive dolomitization (Figure 13). The frequent presence of RD1 and RD2 in the back-reef and inter-reef belts and at the top reef and shoal build-ups indicates the close relationship between dolomitization and sea-level change and therefore supports the validity of the model. The Upper Knox dolomites in the Appalachians (Montanez and Read, 1992), the middle Turonian dolomites in central Tunisia (Touir et al., 2009) and the Capitan Formation forereef dolomites (Melim and Scholle, 2002) are also good examples documenting such a relationship.
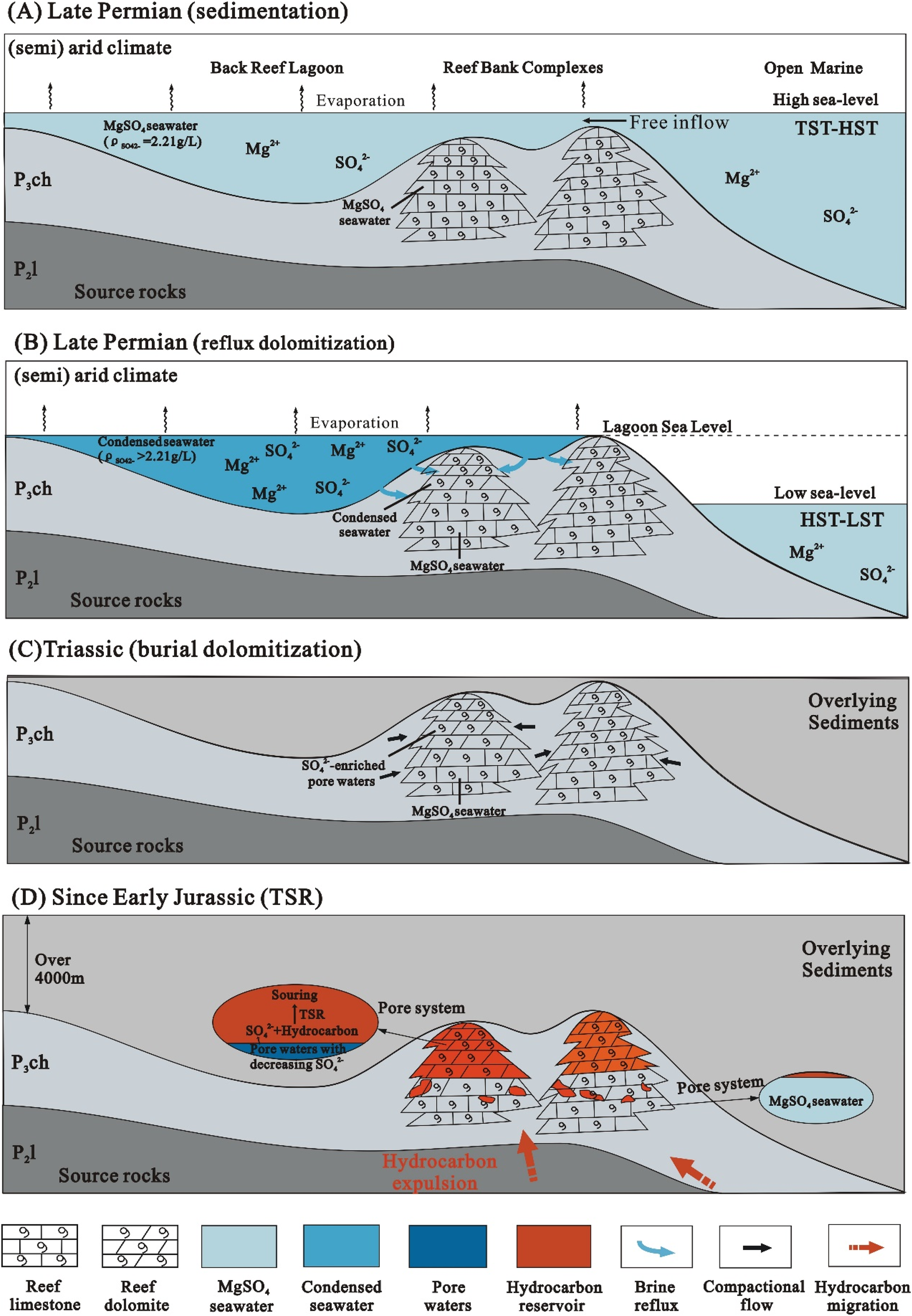
Conceptual model showing the dolomitization and TSR process and the concomitant chemical evolution of the paleo-fluids. (A) During high sea level in Late Permian the back reef lagoon was in relatively free water exchange with the open marine. The mineral deposits were mainly aragonites and the primary pores were filled with MgSO4 connate water. (B) During low sea level, limit water exchange between the lagoon and the open marine and moderate evaporation caused seawater condensation without evaporite precipitation. Reflux of the penesaline seawater initiated dolomitization of aragonite and/or calcite along the platform rim, which resulted in that dolomite layers held condensed seawater, whereas the limestone layers kept MgSO4 connate water (C) During the subsequent burial, dewatering of carbonates would liberate seawater and led to further dolomitization and SO42− accumulation. (D) The elevated concentrations of SO42− in the dolomite layers were succeeded by the deep-buried pore waters and reacted with the substantial oil/gas to produce H2S, whereas the SO42− in the limestone layers kept unchanged or slightly decreased due to minor hydrocarbons accumulated.
Further dolomitization probably occurred at shallow to intermediate depths during the subsequent onset of chemical compaction (Figure 11), as indicated by the close association of RD3 dolomites with bedding-parallel stylolites (Figure 6 A), which commonly form at burials between 500 and 800 m in limestones, some earlier, and at higher burials in dolostones due to their greater resistance to dissolution (Ebner et al., 2009; Mountjoy and Amthor, 1994; Nicolaides and Wallace, 1997). This inference is also supported by the calculated dolomitization temperatures of 53.2–58.9°C (Figure 12), using the δ18O values of RD3 dolomites, the dolomite–water fractionation equation of Sheppard and Schwarz (1970), and the corresponding depths of 668–848 m. The increasing dolomitization temperatures from RD1 and RD2 to RD3 are consistent with the covariations in stoichiometry, Sr content and crystal size of all the dolomites (Figure 9A, B). CD2 overgrows RD1, RD2 and RD3 dolomites facing intercrystalline pores, and the remaining dolomites are filled with bitumen and late diagenetic blocky calcite cement (Figures 5A, D and 6C, D). These features reveal that the precipitation of CD2 postdates the massive dolomite replacement and predates the occlusion of the intercrystalline porosity by bitumen and post-bitumen calcite (Figure 11).
The dolomitization at shallow to intermediate depths was driven by hotter and more reducing residual seawater derived from compaction. Dewatering of carbonates would liberate seawater, either pristine or evolved/concentrated, trapped within the pore system (Figure 13). This process might have provided Mg2+ for local dolomitization. The continuous stabilization of aragonite and high-Mg calcite components, as well as the clay mineral transformations during the formation of stylolites (Figure 6A), could have been an extra source of Mg2+ (Wanless, 1979). The flow of Mg2+-bearing fluids would account for progressive replacement of limestone (RD3) and precipitation of dolomite cements (CD2) within the reef and shoal build-ups (Figure 13).
Variation in SO42− concentrations during and after dolomitization
Late Permian seawaters are classified as aragonite seas (MgSO4 seas, Figure 14) and share chemical characteristics with modern seawater, including high SO42− (2.21 g/L) and Mg2+ (1.25 g/L) concentrations (Hardie, 1996; Lowenstein et al., 2005). Seawater can achieve much higher values of SO42− and Mg2+ concentrations at elevated salinities (Hardie, 1987). Similar salinities likely occurred in the condensed seawater from the restricted lagoons in the study area, where the brine reflux dolomites that formed possess elevated salinities (8∼23 wt%, Jiang et al., 2014) with respect to the original seawater (3.5% salinity). The SO42− concentrations of the pore waters in the reef and shoal rocks are supposed to be correspondingly up to 2.3∼6.6 times (change in salinity) the original values after seepage refluxing of condensed seawater (Figure 13). This result is supported by the covariation between SO42− concentration and salinity or degree of evaporation (Hardie, 1987; McCaffrey et al., 1987). The subsequent dolomitization for RD3 formation in intermediate-depth burial environments, as mentioned above, was favorable for the preservation of sulfate in the pore waters due to the relatively closed hydrodynamic system. Additionally, during the whole dolomitization process, abundant carbonate-associated sulfate (CAS) was very likely to be released and added to the pore waters due to the significant differences in CAS concentrations between dolostone and limestone (Li et al., 2014). The precipitation of pre-bitumen celestite (Figure 11; Li et al., 2012) provides compelling evidence to support the enrichment of sulfate in the pore waters during dolomitization.
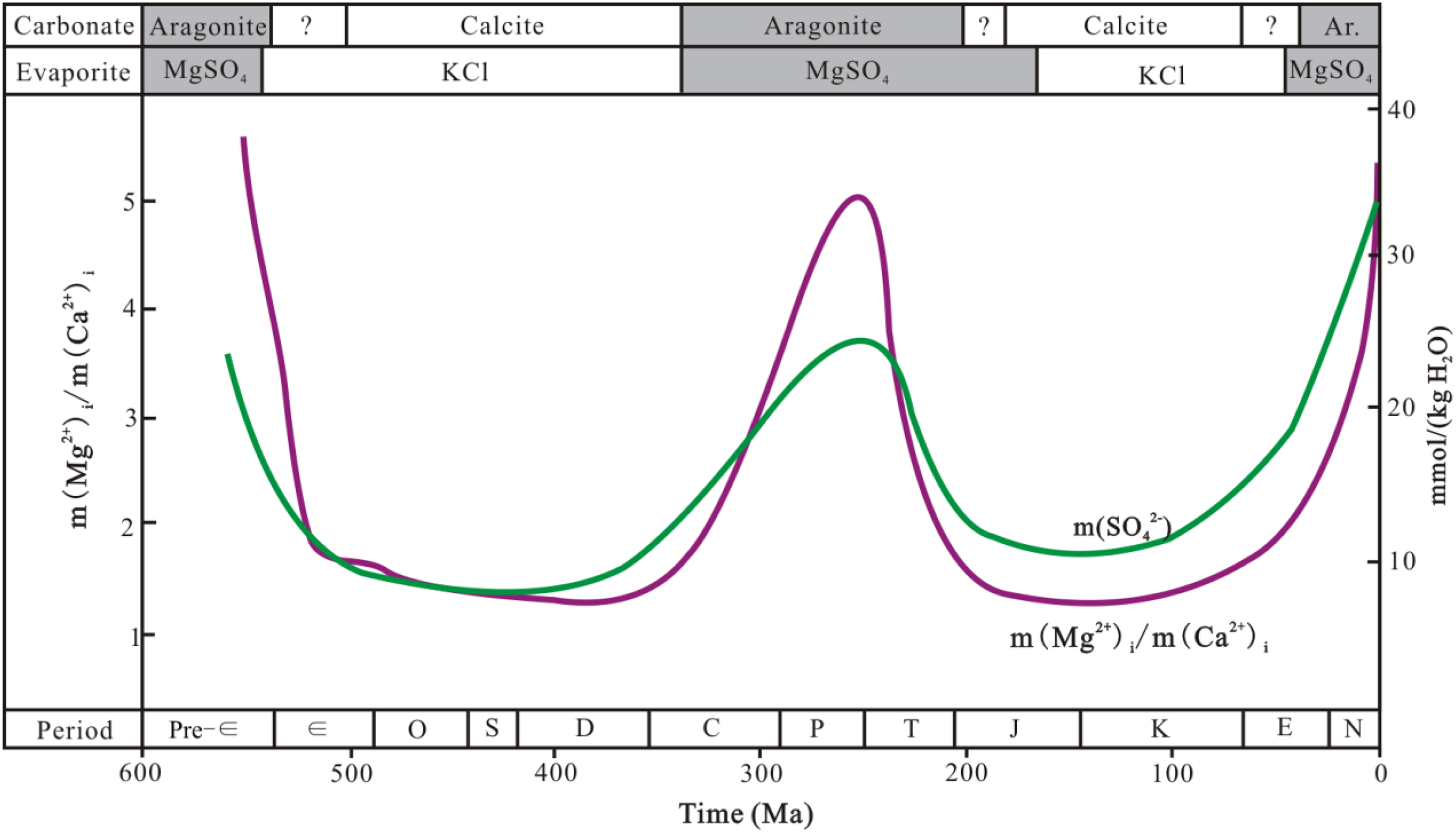
Secular variation in SO42− concentrations and Mg2+/ Ca2+ of seawater and preliminary mineralogy of marine carbonates and evaporites during Phanerozoic time, estimated from fluid inclusions in marine halites (Horita et al., 2002). The SO42− concentration and Mg2+/ Ca2+ of seawater reached the peak during Late Permian.
The geochemical signals of the pore waters seem to be succeeded by late diagenetic fluids, as indicated by 87Sr/86Sr ratios of post-bitumen calcites similar to those of dolomites (Figure 8B) and high SO42−/Cl− concentrations (Figure 10B) of the fluid inclusion water in post-bitumen calcites. These calcites, showing coarse vug/fracture-filling spar, negative shifts in carbon isotope compositions (Table 1) and high precipitation temperatures (142 to 187.8°C), are genetically correlated with TSR (Cai et al., 2003; Li et al., 2012; Machel, 1987; Worden and Smalley, 1996; Worden et al., 1995). The TSR reaction might have occurred in situ, as indicated by the exclusive presence of sour gases in the P3ch reservoirs rather than in the T1f reservoirs, and by δ34S values (17.7‰–24.5‰, Table 4) of the solid bitumen samples that are close to the range of P3ch seawater with δ34SCAS values of 11.5–26.9‰ (Newton et al., 2004).
The negative correlation (Figure 10A) of SO42− concentration with homogenization temperature (Th) hints at a continuous consumption of sulfate by the TSR reaction. The gradual decrease in SO42− concentration caused by TSR is responsible for the SO42− deficit in the present-day formation water (Figure 10B, Figure 13). A comparison of the SO42−/Cl− ratios in the fluid inclusion water (5.4∼10.4) and the formation water (0.001 to 0.019) indicates that almost all of the accumulated sulfate has been removed during TSR. The evolution of SO42− concentrations in paleowaters during diagenesis is presented in Figure 11.
Relationship between dolomitization and TSR
One of the critical factors for the occurrence of TSR could be limited amounts of dissolved sulfate in closed systems, i.e., low SO42− concentrations in pore waters and volumetrically minor residual pore spaces during deep burial (Bildstein et al., 2001; Li et al., 2019; Worden et al., 2000). Therefore, in hydrocarbon reservoirs, any improvement in these two parameters would enhance the TSR reaction and H2S accumulation.
Model mass/volume balance calculations were conducted to obtain the concentration of the generated H2S using the equation of Li et al. (2016). The calculated H2S concentration is only 2.6% based on a reservoir with 10% initial water saturation (typical for gas-filled carbonate reservoirs as suggested by Worden et al., 1996) and ∼1.0 g/L SO42− concentrations of initial pore waters (CaCl2 sea, Figure 14). In contrast, the calculated value could range up to 5.7% if the initial water was Late Permian seawater with a SO42− concentration of 2.2 g/L (Lowenstein et al., 2005), indicating that MgSO4 seawater is a superior sulfate supplier. However, the value is significantly lower than the concentrations of H2S within some P3ch reservoirs, i.e., 12.1% in well Yb9 and 25.7% in well Yb123 (Table 3). The subsequent evaporation of MgSO4 seawater and CAS released during dolomitization are more important to have multiplied (by 2.3∼6.6 times) the SO42− concentrations of the pore waters. The calculated H2S concentrations range from 13.1∼28.5%, which seems reasonable when considering the reported removal of H2S in the P3ch reservoirs, e.g., the formation of pyrite (e.g. Jiang et al., 2014) and sulfur deposits (e.g. Li et al., 2014) and the incorporation of sulfur into solid bitumen (e.g. Cai et al., 2010).
Notably, the formation waters within the Permian-Triassic limestone layers possess SO42− concentrations (1.3 g/L to 1.9 g/L, Table 3) close to or slightly lower than those of the original seawater (1.9–2.2 g/L, Horita et al., 2002; Lowenstein et al., 2005). That is, most of the SO42− was not involved in TSR throughout deep burial. This interpretation implies that there should have been another critical factor, i.e., transport of aqueous hydrocarbons (Worden et al., 2000), which inhibited the TSR reaction and H2S production (H2S% <1, Table 3). The lower porosity (2.3%) and permeability (1.2 × 10−3 μm2) of the P3ch limestone reservoirs than those of the dolostone reservoirs (5.1%∼16.7% and 5.3 × 10−3∼306 × 10−3 μm2, respectively, Guo, 2011) might have significantly reduced the chances of contact between hydrocarbons and dissolved sulfates. Zhu et al. (2007) also highlighted the importance of the workspace for the TSR reaction and found that almost all of the gas pools with high H2S concentrations in China possess porosities over 3.5%. Therefore, the occurrence of massive dolomitization in the P3ch Formation seems to be a necessary process to preserve porosities, providing more space for accommodation of both residual water and hydrocarbons. The preference for RD3 dolomites that have abundant vuggy and intercrystalline porosities further indicates that the late dolomitization is preferable to enhance the workspace for TSR.
Regional comparisons
TSR-H2S enrichment was also reported in the evaporite-free dolostone reservoirs of P3ch on the eastern side of the K-L Trough (e.g. Cai et al., 2004, 2013; Hao et al., 2015; Li et al., 2014; Liu et al., 2013). Similarly, most of the necessary sulfate was demonstrated to have been inherited from early Mg- and sulfate-rich dolomitizing fluids (Li et al., 2014). However, unlike the study area, the dolomitizing fluids on the eastern side of the K-L Trough were derived from the Early Triassic hypersaline evaporite lagoons, where laminated/nodular anhydrite and gypsum formed. The massive dolomite and the post-bitumen calcite yield radiogenic Sr isotopes (mainly from 0.70753 to 0.70785), significantly higher than those of the Upper Permian carbonates in situ (Jiang et al., 2013; Li et al., 2014) and those of RD1, RD2, RD3 and the post-bitumen calcite in this study (Figure 8B; Table 1).
The differences in dolomitizing fluids between the two sides are also responsible for the presence of different H2S concentrations, i.e., generally >10% for the eastern side (Cai et al., 2013; Liu et al., 2013; Li et al., 2014) and 3.3∼7.2% for the western side (Table 3). The brines from the Early Triassic hypersaline evaporite lagoons are obviously superior in carrying dissolved sulfate, which is favorable for the consumption of charged hydrocarbons and H2S production. This superiority is also reflected in remarkably higher H2S concentrations in the evaporite-carbonate units around the world, i.e., the Crossfield member of the Okotoks gas field in the West Canadian Basin (Dobbin, 1968), the Jurassic Smackover Formation of Black Creek Field, Mississippi (Heydari, 1997), and the Lower Carboniferous rocks in West Siberia (Belenitskaya, 2000). For the occurrence of TSR related to penesaline dolomitizing fluids, relatively limited sulfate places strong constraints on H2S production. However, the mild to moderate degrees of H2S enrichment are also worth our special concern for hydrocarbon discovery in deeply buried dolostone reservoirs.
A comparison of the sea-level trend between the Yangtze Platform in South China (Wang et al., 1999; Yin and Tong, 1996) and global eustatic changes (Figure 4; Haq and Al-Qahtani, 2005, Haq and Schutter, 2008) reveals obvious differences during the Late Permian. Regarding long-term changes, there was a distinct transgression in the Yangtze Platform during most of the period, causing reef growth instead of the global sea-level fall to the lowest point. The subsequent regression began at the end of the Permian, and the drop in sea level led to the formation of back-reefs or inter-reef lagoons, where condensed MgSO4 seawater formed in a (semi) arid climate. Such penesaline conditions are favorable for the development of dolomitizing fluids and the subsequent occurrence of TSR under deep burial. Therefore, similar scenarios involving non-evaporitic dolomitization and TSR, as well as their close relationship, have probably occurred along the platform-margin reef belts elsewhere on Yangtze platform (Figure 1B). Two case studies in point are available to support the proposal.
The Shibaozhai gas field, lying at the southern tip of the K-L Trough (Figure 1B), has high concentrations of TSR-H2S (3.7%, Cai et al., 2004) in the P3ch dolostone reservoirs compared with minor to negligible H2S in the T1f limestone reservoirs. The Jiannan gas field, located in the eastern Sichuan Basin, is the only gas field in marine carbonate strata in the middle of the Yangtze plate (Figure 1A). H2S build-ups of TSR origin (mostly from 1.7% to 3.8%) appear more frequently in the P3ch dolostone reservoirs than in the T1f reservoirs (Li et al., 2015; Zhang et al., 2010).
Interestingly, the pattern of sea-level fluctuation during the Late Permian on Yangtze Platform is representative of the Eurasian Tethys and existed in the Alps, Transcaucasia, NE Vietnam and Japan (Figure 1A, Sano, 2006; Yin and Tong, 1996). We tentatively propose that the model mentioned above might be applicable across a broader range, if hydrodynamic system stays closed during diagenesis for SO42− preservation and the temperatures of hydrocarbon reservoirs are high enough for the occurrence of TSR (Machel, 2001; Worden et al., 1995). Unfortunately, data are still scarce to support this proposal. More studies of this aspect are in progress.
Conclusions
Penesaline seawater, developed in back-reef or inter-reef lagoons during sea-level fall, dolomitized the reef-flat facies of the P3ch Formation in the western reef complex, NE Sichuan. Pervasive dolomitization was further promoted and completed by hotter residual seawater during the subsequent onset of chemical compaction. Additional Mg2+ are sourced from the stabilization of aragonite and high-Mg calcite components, as well as clay mineral transformations during the formation of stylolite.
During early reflux dolomitization, sulfate continuously accumulated in the pore waters under the combined effects of MgSO4 seawater evaporation and refluxing. The progressive dolomitization in shallow- to intermediate-depth burial environments facilitated CAS release and sulfate preservation due to the relatively closed hydrodynamic setting.
Model mass/volume balance calculations show that the sulfate accumulated in the dolomitization process was greatly depleted by the reduction of liquid and gas hydrocarbons (TSR), which was responsible for producing of the present-day high concentrations of H2S in the NE Sichuan Basin.
The occurrence of massive dolomitization driven by MgSO4 (aragonite) seawater, under the conditions of favorable sea-level fluctuation and restricted carbonate lagoon, is capable of initiating TSR in evaporite-free deep reservoirs around the world, if there is a closed hydrodynamic system during diagenesis that is favorable for sulfate preservation. This correlation has important implications for the deep exploitation of sour oil/gas in dolostone horizons, which, however, have been largely ignored to date.
Footnotes
Acknowledgements
We would like to thank two anonymous reviewers for giving us constructive suggestions, which were helpful in improving the quality of the manuscript. This work is financially supported by National Natural Science Foundation of China (Grant No. 41972154) and the Fundamental Research Funds for the Central Universities (Grant No. 2652019104).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China, the Fundamental Research Funds for the Central Universities, (grant number 41972154, 2652019104).
