Abstract
Wormlike micelles formed by amidosulfobetaine surfactants present advantage in increasing viscosity, salt-tolerance, thermal-stability and shear-resistance. In the past few years, much attention has been paid on rheology behaviours of amidosulfobetaine surfactants that normally bear C18 or shorter tails. Properties and oil displacement performances of the wormlike micelles formed by counterparts bearing the long carbon chain have not been well documented. In this paper, the various properties of C22-tailed amidosulfobetaine surfactant EHSB under high salinity (TDS = 40g/L) are investigated systematically, including solubility, rheology and interfacial activity. Moreover, its oil displacement performance is studied for the first time. These properties are first compared with those of C16-tailed counterpart HDPS. Results show that the Krafft temperature(TK) of EHSB decreases from above 100°C to 53°C with the increase of TDS to 40 g/L. Increasing concentration of EHSB in the semidilute region induces micelle growth from rod-like micelles to wormlike micelles, and then the worms become entangled or branched to form viscoelastic micelle solution, which will increase the viscosity by several orders of magnitude. The interfacial tension with oil can be reduced to ultra-low level by EHSB solution with concentration below 4.5 mM. Possessing dual functions of mobility control and reducing interfacial tension, wormlike micelles formed by EHSB present a good displacement effect as a flooding system, which is more than 10% higher than HPAM with the same viscosity. Compared with the shorter tailed surfactant, the ultra-long tailed surfactant is more efficient in enhancing viscosity and reducing interfacial tension, so as to enhance more oil recovery. Our work provides a helpful insight for comprehending surfactant-based viscoelastic fluid and provides a new viscoelastic surfactant flooding agent which is quite efficient in chemical flooding of offshore oilfield.
Keywords
Introduction
Chemical flooding is one of the main technologies to further enhance oil recovery. Surfactant flooding improves oil recovery through the reduction of oil-water interfacial tension (Pan et al., 2020), wettability alteration of reservoir rock (Esfandyari et al., 2021, 2020b) and formation of micelles and microemulsion (Jing et al., 2020; Pal et al., 2018). The surfactants used mainly include cationic, anionic, nonionic and zwitterionic surfactants. The cationic surfactants are mainly alkyl ammonium salts (Esfandyari et al., 2020a), among which CTAB is the most commonly used (Pan et al., 2020). Anionic surfactants mainly include carboxylate and sulfonate (Esfandyari et al., 2020b; Pan et al., 2020). The common type of nonionic surfactants are polyoxyethylene alkyl alcohol ethers (Hou et al., 2015). Zwitterionic surfactants can be divided into phosphate, sulfate, carboxylate and sulfonate types according to different anionic groups (Esfandyari et al., 2020a). Natrual surfactants, such as Cedar, which is extracted from Zizyphus Spina Christi trees, has abundant sources, low production cost and no adverse effects on reservoir and environment (Esfandyari et al., 2021). The mechanism of EOR by polymer flooding is to improve sweep efficiency by reducing the water-oil mobility ratio and improve oil washing efficiency by viscoelasticity of polymer (Wang, 2018). The commonly used polymers can be divided into biopolymers and synthetic polymers. Synthetic polymers includes polyacrylamide (PAM) and hydrolyzed polyacrylamide (HPAM). Biopolymers include xanthan, biologic polysaccharide and some of the natural polymers like hydroxyl ethyl cellulose (HEC), sodium carboxyl-methyl cellulose and guar gum (Davarpanah, 2020). The addition of polymer to foaming agent can increase gas viscosity and inhibit fingering and gravity segregation, so as to improve oil recovery of foam flooding (Davarpanah, 2018). The property of polymer-enhanced foam (PEF) is utterly dependent on the type of used polymer (Davarpanah et al., 2019). The nano strong reducing agent can greatly reduce the oil-water interfacial tension and change the wettability of rock surface to strongly hydrophilic, so as to reduce the injection pressure and enhance oil recovery (Zhao et al., 2019). ASP flooding utilize the synergetic effect of polymer, surfactant and alkali. The oil recovery can be increased by more than 20% on the basis of water flooding (Zhong et al., 2019).
Under the condition of high salinity, especially in the presence of high concentration of Ca2+ and Mg2+, the viscosity-enhancing property of HPAM and the ability to reduce interfacial tension of petroleum sulfonate are seriously affected (Pal et al., 2018; Wang, 2018). The alkali will react with divalent metal ions in the formation water, causing pipe scaling and reservoir corrosion (Zhong et al., 2019). Therefore, researchers have developed new types of comb-like polymer, hydrophobic associating polymer (Wang et al., 1999, Wang and Luo 2003) and amphiphilic, Gemini surfactants (Tang et al., 2011; Wang et al., 2012) as chemical flooding agents for high salinity reservoirs, and mixed them as alkali-free composite oil displacement agent. In the meantime, the development of new chemical flooding system is carried out. Among them, the self-assembly of surfactant molecules to form wormlike micelles with viscoelastic properties under certain conditions has attracted much attention.
Wormlike micelles are cylindrical aggregates assembled spontaneously by surfactant molecules, which can grow to tens of micrometres under favourable conditions (Chu et al., 2010). These aggregates entangle with each other and impart the solution viscoelasticity similar to HPAM (Zhu et al., 2013). Unlike HPAM, the self-aggregation of surfactant molecules is through intermolecular force rather than chemical bond force, which leads to the reversible and dynamic equilibrium of wormlike micelles (Acharya and Kunieda, 2006). These aggregates undergo reversible breakdown processes when passing through little pore throats then quickly reform. Thus, wormlike micelles show better shear-resistance than polymers (He, 2015). The excellent properties of wormlike micelles enable them to be used in many fields, such as templates for the synthesis of nanomaterial (Qiao et al., 2011), carriers for drug release and delivery (Afifi et al., 2011), clear fracturing fluids for increasing production (Lu et al., 2017) and mobility control agent for chemical enhanced oil recovery (Dai et al., 2015; Santvoort and Golombok, 2015). The surfactant based viscoelastic fluid exhibits excellent ability to enhance viscosity at low surfactant concentration under high salt conditions (Morvan et al., 2009).
Wormlike micelles formed by C16-tailed cationic surfactants have been most widely reported at present (Cates and Candau, 1990; Hassan et al., 1998; Maitland, 2000), the C22-tailed counterparts have recently become a research focus (Chu and Feng, 2010a; Croce et al., 2003; Raghavan and Kaler, 2001, Raghavan et al., 2002). In contrast to shorter-tailed counterparts, these C22-tailed cationic surfactants show lower overlap concentrations, stronger viscoelasticity, and better heat-resistance. Under high-salt conditions, especially when the concentration of calcium and magnesium is high, the inorganic counterions can alter surface charges of cationic and anionic micelles and compress their diffused double layer, which prevents the growth of wormlike micelles and induces the transition to branched structure. The transition reduces the viscoelasticity (Fan et al., 2014; Zhang et al., 2016). Compared with cationic and anionic micelles that are sensitive to inorganic salt, the zwitterionic micelles behave like systems with no surface charges and show better foam-stability and salt-tolerance (Gonenne and Ernst, 1987; Rosen, 2004). The self-organized aggregates formed by sulfobetaine surfactants are stable enough under high salt concentration (Hussain et al., 2018) and have potential for high salinity reservoirs (Kumar et al., 2007; Yoshimura et al., 2006). However, zwitterionic wormlike micelles have been less documented (Hashimoto and Imae, 1991; Ono and Shikata, 2005; Zhang et al., 2013). The reported sulfobetaine surfactants up to now are mostly bearing a C18 or a shorter hydrophobic tail. It appears that common sulfobetaine surfactants bearing C18 or shorter carbon chains cannot form wormlike micelles independently (Dreiss, 2007; Kumar et al., 2007; Weers et al., 1991). Fan et al. mixed the sulfobetaine surfactant N-hexadecyl-N, N-dimethyl-3-ammonio-1-propane sulfonate (HDPS) bearing a C16 hydrophobic tail with anionic surfactant sodium dodecyl sulfate (SDS) in the brine (TDS = 10g/L) to form viscoelastic surfactant solutions. The viscoelasticity, dissolution properties, reduction of oil/water interfacial tension and oil displacement efficiency of the mixed surfactant system have been studied. C22-tailed sulfobetaine surfactants such as erucyl dimethyl amidopropyl betaine (EDAB) (Kumar et al., 2007) and 3-(N-erucamidopropyl-N, N-dimethyl ammonium) propane sulfonate (EDAS) (Chu and Feng, 2010b) are documented. However, the current research on C22-tailed sulfobetaine surfactants has been mainly centered on rheology and phase behaviour, barely on oil displacing performance.
In this work, the properties of aqueous solution of wormlike micelles assembled by the long-tailed amidosulfobetaine surfactant erucoylamidopropyl hydroxylsulfobetaine (EHSB, Figure 1), were studied systematically and compared with HDPS/SDS worms. The solubility, rheology properties, interfacial activities and oil displacement efficiency under high salt concentration (Total Dissolved Salts = 40 g/L) will be presented, especially when the concentration of calcium(0.752 g/L)and magnesium(0.306 g/L)is high.
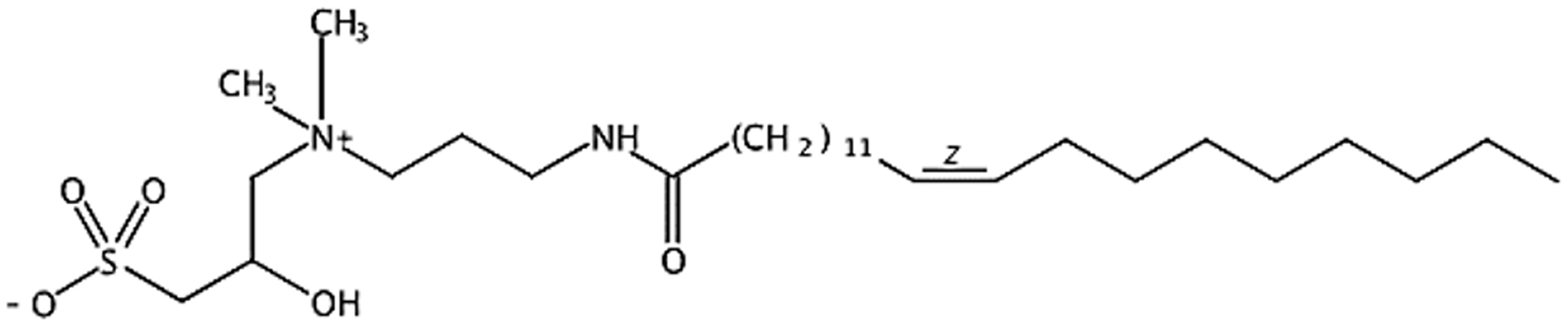
Molecular structural formula of C22-tailed sulfobetaine surfactant EHSB.
Experimental methodology
Materials
Surfactant EHSB (molecular formula C30H60N2O5S; relative molecular mass 560.85) of analytical grade was used as received. The Sodium Chloride (NaCl), Calcium Chloride (CaCl2) and Magnesium Chloride Hexahydrate (MgCl2·6H2O) used in the experiments was analytical grade. The dehydrated crude oil is taken from an offshore oilfield. The diameter and length of artificial cores are 2.5 cm and 10 cm, respectively. The main reason of choosing this size of cores is the size of core gripper in oil displacement equipment.
Sample preparation
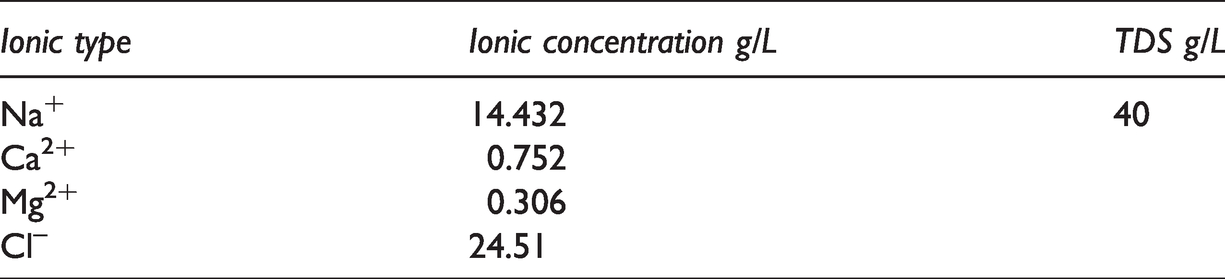
The formation water (TDS = 40g/L) is prepared by adding NaCl, MgCl2·6H2O and CaCl2 in the distilled water, in which the concentration of Na+, Ca2+, Mg2+ and Cl– are illustrated in Table 1. Desired amount of powder EHSB was added to the formation water, followed by the solution slowly heated to 65°C and gently stirred until the surfactant was completely dissolved; the solutions were then placed at 65°C in the thermal bath for more than two days before determinations. For all the samples, the TDS was fixed at 40 g/L to guarantee complete dissolution of powder EHSB, and pH value was kept at the natural value (6.2).
The ionic composition of the formation water.
Phase behaviour observation
The temperature at which the sample solution becomes clear and transparent during heating is called Krafft point (TK) as well as clearing temperature. The Krafft point is defined as the temperature where 1 wt% surfactant can be dissolved, which represents the upper limit of the Krafft point at which the solubility equals to the critical micellar concentration (cmc), because the cmc of amidosulfobetaine surfactants is generally much lower than 1 wt% (Laughlin, 1994). Surface tension of EHSB solutions at room temperature is measured with DCAT21 dynamic surface tension instrument. With the increase of EHSB concentration, the surface tension decreases gradually and then tends to be steady. The extrapolation of linear curves from above and below the break point in the curve yields the cmc, the value of which is 0.0016 wt%.
TK is generally measured by the conductivity of saturated surfactant solution. However, this method is not only time-consuming (Shinoda et al., 1976), but also not applicable to the species with little surface charge like amidosulfobetaine surfactants. Phase behaviour was recorded by the more easily accessible method, visual observation (Weers et al., 1991; Lin et al., 2005), in this work. 1 wt% EHSB solutions were prepared with distilled water or formation water, and then sealed in test tubes with capes and Teflon tapes. Slowly heated the tube to 85°C to ensure complete solubility. Then the solutions were slowly cooled and equilibrated in a constant temperature water bath from different temperatures to room temperature, resulting in the appearance of a white wax-like gel phase. The Krafft temperature, at which the last crystal dissolves and the sample becomes transparent and isotropic, was later measured by heating the tubes extreme mildly (2.4 K/h close to TK). The Krafft temperature (TK) was also utilized to qualitatively characterize the lower solubility of amidosulfobetaine surfactant in the water at room temperature. Higher TK represents lower solubility.
Rheology
Rheology were determined on a Physica MCR302 (Anton Paar, Austria) rotational rheometer with concentric cylinder geometry CC27 (ISO3219). The radii of measuring bob and measuring cup are 13.331 mm and 14.451 mm, respectively. For the steady rheological experiments, sufficient time was reserved for the viscosities corresponding to each shear rate to reach the equilibrium. Dynamic frequency spectra were conducted in the linear viscoelastic regimes at constant shear stress, according to the measurement results of dynamic stress sweep.
Cannon standard oil was used to calibrate the instrument before the experiments. Samples were kept at desired temperatures for at least 20 min before measurements. The temperature accuracy of ±0.1°C was controlled by a Peltier device, and a solution trap was utilized to reduce the solution evaporation during the experiments. All measurements are performed at 65°C without specific documented.
Interfacial tension
The IFT measurements were done by Texas-500C interfacial tension meter at 65°C. The IFT values between the provided samples prepared with EHSB solutions with different concentrations and oils of desired salinities were determined at a constant rotational speed of 5000 rpm. The values were recorded every five minutes until they reach stability.
Oil displacement
The core flood experiments aim to evaluate the properties of the wormlike micelles under reservoir conditions and ability to enhance oil recovery, which were carried out at 65°C. The experimental setup is shown in Figure 2.
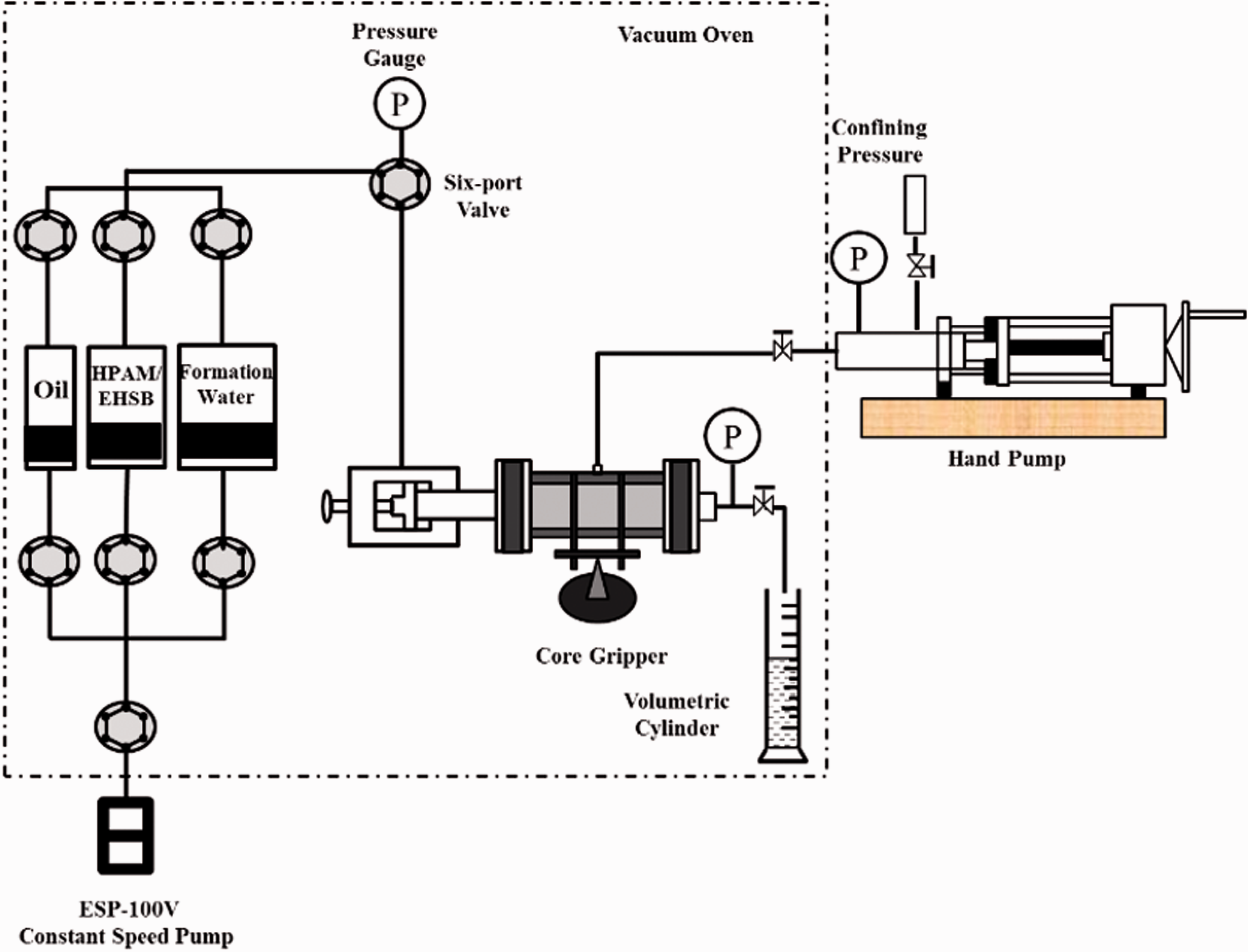
Schematic diagram of the oil displacement experiments.
Firstly, saturate the core with formation water, and the formation water was injected at three different rates to determine the absolute permeability to water. The formation water was displaced by oil until residual water saturation. After isothermal ageing, water flooding was put into effect until the water cut at the production end reached up to 98%.
The water flooding recovery E1 could be determined by the formula

Then 0.5 Vp of EHSB solution was injected into the core, which followed by injection of the formation water till the water cut at the production end reaches up to 98% again. The ultimate recovery E2 could also be determined by the formula

Then 0.5 Vp of EHSB solution was injected into the core, which followed by injection of the formation water till the water cut at the production end reaches up to 98% again. The ultimate recovery E2 could also be determined by the formula

Results and discussion
Phase behaviour observation
Long-chain amidosulfobetaine surfactants which bear a C18 or a longer tail can’t dissolve in pure water owing to their high Krafft point (TK). The Krafft temperature of the surfactant usually gets higher with increasing the length of hydrophobic tail (Domingo, 1996) and gets lower with introducing the unsaturated bond into the chain (Kumar et al., 2007). Tsujii and Mino (1978) stated that the Krafft temperature of propane sulfonate with a C18 saturated carbon chain is 73.4°C in pure water. In our work, the C22-tailed surfactant EHSB is insoluble even the temperature of pure water reaches 100°C, indicating that its Krafft temperature is above 100°C. The poor solubility of EHSB in pure water impedes its application in some industrial fields. Thus, it is important to reduce its TK to meet the needs of research and applications (Laughlin, 1994).
Chu and Feng (2012) pointed out that the amphiphilic surfactants bearing a C18 or a longer tail are insoluble in formation water when the temperature is lower than 40°C, although by which single component micellar solutions can be formed. In our work, the 1 wt% EHSB sample was taken as an instance to study the phase behaviour of the micellar solution at different temperatures. When the temperature is over 53°C, a clear and homogeneous viscoelastic solution is noted (picture A in Figure 3) and no clouding turns up, when the temperature is lower than 53°C, a white wax-like gel phase progressively turns up (picture B in Figure 3), and the gel was considered to be a hydrated surfactant phase generated due to a typical Krafft phenomenon (Zana, 2004; Zana et al., 2005). The gel system was maintained at 4–5°C for some days, resulting in a white phase on the top of the system. The range initially extended with time, then became stable after a month (picture C in Figure 3). The phenomenon shows that the gelled phase that turns up comparatively fast is metastable when cooling EHSB solutions. Hence, we endeavor to suppose that the two-phase system that forms a month later is more similar to, or symbolizes, the actual equilibrium of the system (Zana, 2004). The top white phase possibly is a hydrated surfactant phase. The generation of this phase indicates that EHSB shows the actual Krafft phenomenon only in longer time scale experiments.

The appearance of 1 wt% EHSB at different temperatures. Pictures (a), (b), and (c) denote the usual transparent micellar solution over TK, gel phase below TK, and equilibrium state at 4–5°C, respectively.
Although long-chain amidosulfobetaine surfactants are insoluble in boiling water, they can dissolve in the brine at lower temperature owing to the decrease of TK as adding inorganic salt into the surfactant solution. The addition of total amounts of inorganic salts at 40 g/L depresses the TK of EHSB from above 100°C to 53°C and significantly enhances their aqueous solubility. The mixture of 1 wt% HDPS/SDS can be dissolved with the addition of 10 g/L inorganic salts in the formation water at 35°C. The reason is that HDPS is the zwitterionic surfactant with a relatively shorter carbon chain, a saturated C16 tail, which avoids the insolubility issue of long chain zwitterionic surfactants in brine. Moreover, the addition of anionic surfactant SDS with a saturated C12 tail accelerates the formation of wormlike micelles.
All the rheological experiments, interfacial tension measurements and core flooding experiments are performed at 65°C unless stated otherwise to make sure the complete dissolution of the samples studied in the following work.
Rheology
The change of steady shear viscosity with shear rate for EHSB solutions at 65°C is shown in Figure 4. The steady shear viscosity of 1.5 mM EHSB equals to that of water and it keeps steady no matter how the shear rate changes, which accords to usual Newtonian fluid behaviour. 5.0 mM EHSB presents Newtonian behaviour at low-shear-rate regions, while shear-thinning takes place when the shear rate surpasses 2.5 s−1, which can be used as proof that the wormlike micelles are undergoing structural transitions like the parallel arrangement of flexible micelles at high shear rates (Dreiss, 2007). With increment of EHSB concentration, the wormlike micelles come to be tangled into a transient network, which enhances the viscoelasticity of the solution and shifts the critical shear rate where shear-thinning begins to lower values. This phenomenon can be regarded as a growing tendency for deformation of the tangled networks and parallel arrangement of the micelles at high shear rates (Raghavan et al., 2002). The η0 of wormlike micelles formed by 25 mM EHSB in formation water with 40 g/L salinity is 766.1mPa·s, while that formed by 25 mM HDPS/SDS in formation water with 10 g/L salinity is only 66.5mPa·s. The η0 of EHSB is much higher than that of HDPS/SDS, which indicates that wormlike micelles formed by ultra-long carbon chain surfactant possesses much better viscosity-enhancing ability.
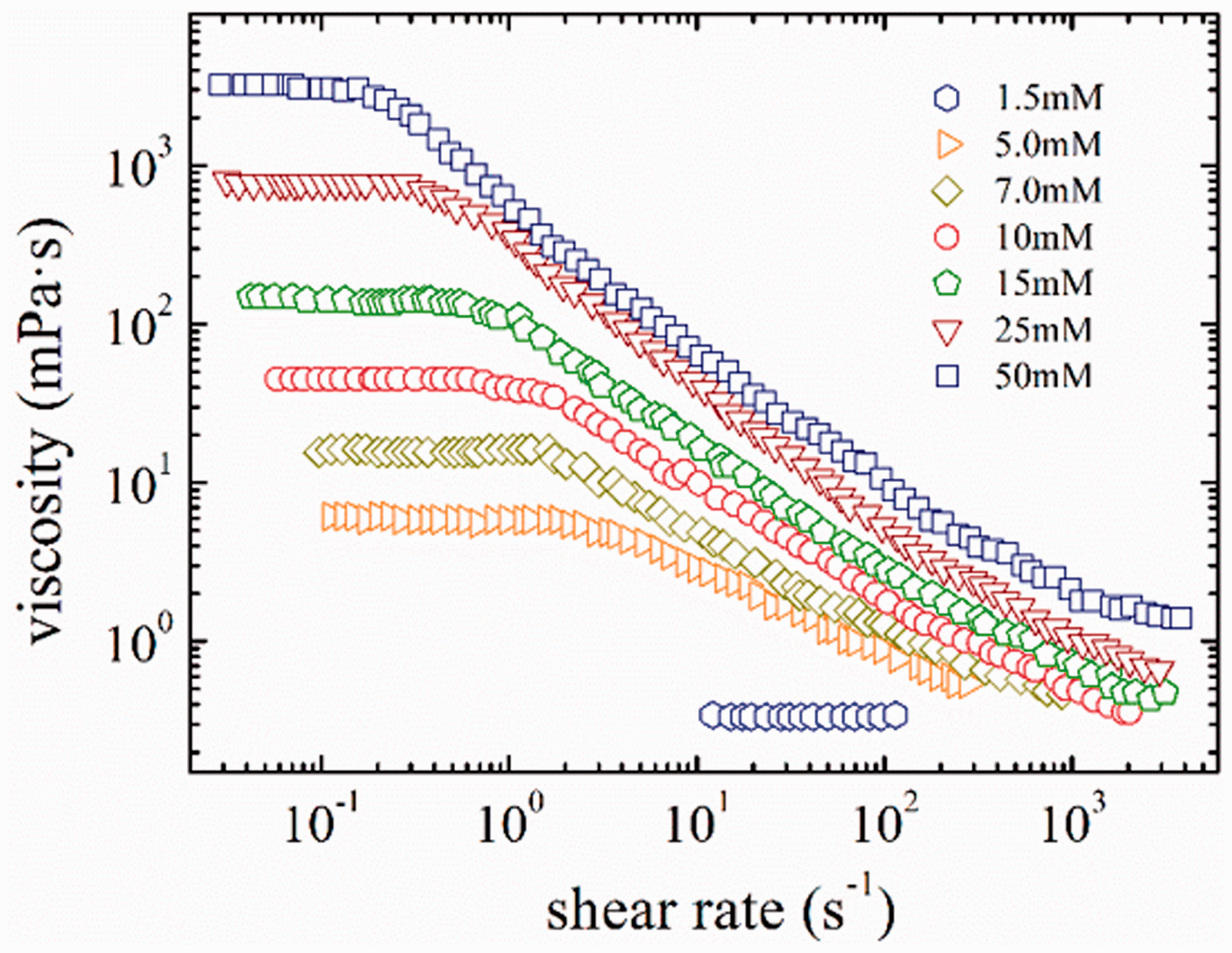
Steady shear viscosity versus shear rate for different EHSB concentrations at 65°C.
It should be noted that the viscosity enhancements because of Taylor instabilities at high shear rates are not illustrated in Figure 4. The Taylor vortex is caused by the centrifugal forces and inertia effects resulting from the solution mass in concentric cylinder geometry. The critical shear rate caused by Taylor instabilities raises with growing viscosity of the solution, depressing radius of the inner cylinder and the declined ratio of the outer to inner cylinder radius (Mezger, 2006). For instance, 1.5, 5.0, and 7.0 mM EHSB exhibit Taylor instabilities at critical shear rates of around 130, 255, and 900 s−1, respectively.
In the steady-shear measurement, the extrapolation of viscosity along the plateau in the Newtonian region to zero-shear rate obtains the zero-shear viscosity, η0. As exhibited in Figure 4, η0 raises monotonically with increment of EHSB concentration. Figure 5 illustrates the increase of zero-shear viscosity with EHSB concentration at 65°C. When the concentration is lower than the critical value(overlapping concentration, C*), zero-shear viscosity increases linearly with the concentration of EHSB in manner of the Einstein equation, η0 = ηwater(l+KC); when the concentration is higher than C*, zero-shear viscosity grows sharply according to η0∼Cn, where n is the power-law index (Berret et al., 1993). In dilute region, the average length of micelles generally grows with EHSB concentration in a power-law manner with the index of about 0.5 (Shikata et al., 1989). However, when the surfactant concentration is higher than C*, the wormlike micelles start to tangle with each other to construct bulky supramolecular aggregates, showing excellent viscoelastic behaviours (Ezrahi et al., 2006; Dreiss, 2007). As shown in Figure 5, the concentration dependence of the zero-shear viscosity is 2.51, which is less than 3. The theoretical prediction value of n for tangled linear wormlike micelles is 3.5. The discrepancy can be explained by intermicellar branching that corresponds to the structural conversion from linear wormlike micelles to branched ones (Khatory et al., 1993). For aggregates with branching, both the theoretical prediction (Shchipunov and Hoffmann, 1998) and experimental values of n are lower than 3 (Khatory et al., 1993). Under high salinity, the surface charge of anionic wormlike micelles can be screened by counterions, which will induce branching. The phenomenon even exists in the amidosulfobetaine wormlike micelles with little charge under high salinity (Rosen, 2004; Kumar et al., 2007). The value of n for wormlike micelles formed by shorter-tailed amidosulfobetaine surfactant HDPS with SDS under the salinity of 10 g/L is 3.45, which indicates that there is almost no branching structure when the salinity is 10 g/L.
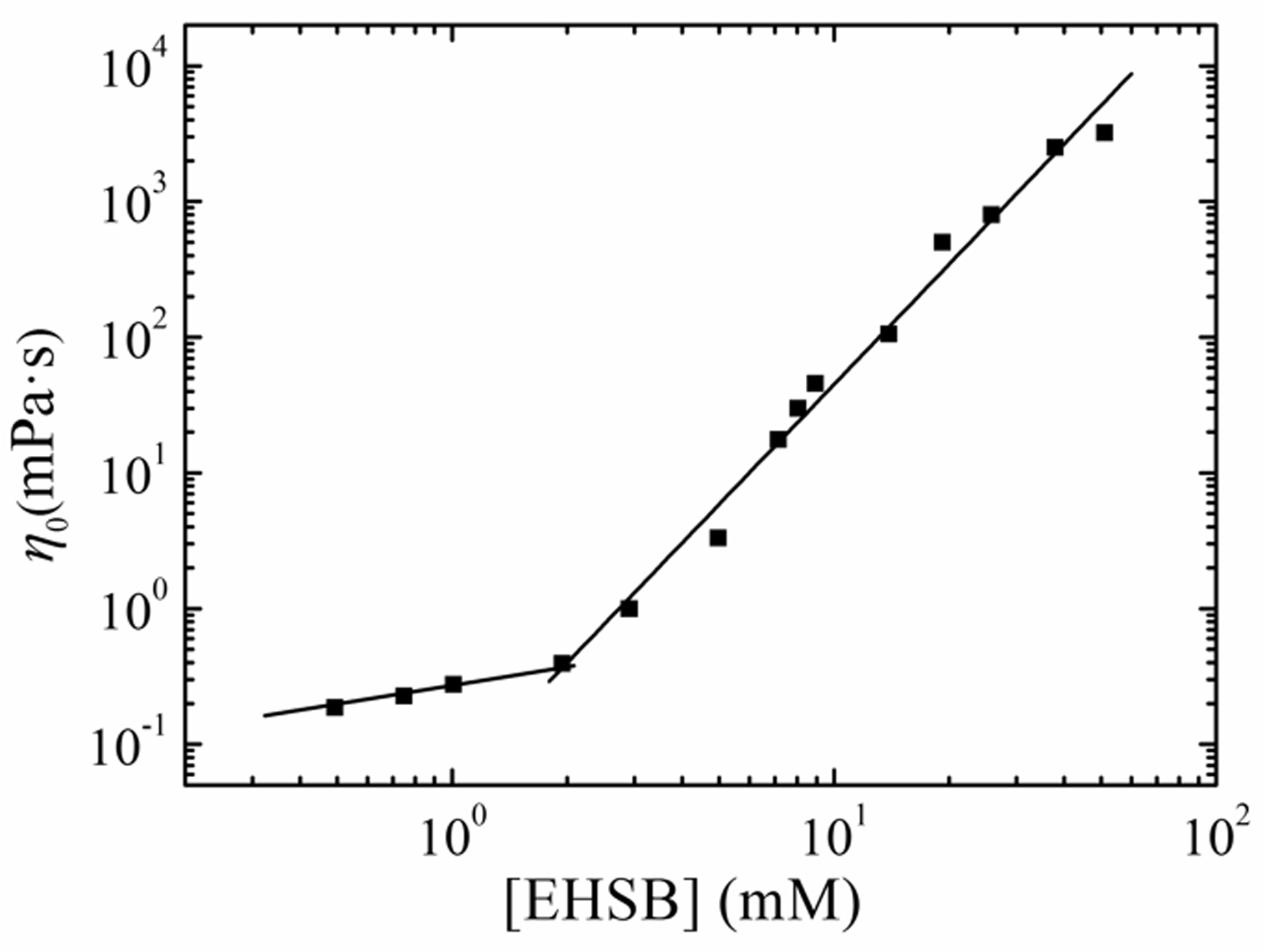
Zero-shear viscosity η0 versus EHSB concentration at 65°C.
Dynamic rheological measurements, which were determined in oscillatory-shear mode, were performed to investigate the concentration relevance of elasticity of EHSB wormlike micelles. Figure 6 illustrates the variation of elastic modulus (G’) and viscous modulus (G’’) with oscillatory-shear frequency (ω) for EHSB solutions at 65°C. The storage modulus G’ crisscrosses and falls below the loss modulus G”. Namely, the relaxation time τR (1/ωc) is limited for the solutions. The solution’s answer could be classified into two regions based on the relaxation time τR: on a timeframe more prolonged than τR (ω ≪ ωc), the answer is viscid, while for a timeframe considerably shorter than τR (ω ≫ ωc), the answer is elastic. The sample’s viscoelasticity is due to the tangle of long wormlike micelles to construct a transient network. The growing EHSB concentration, which induces the tangle, increases the plateau modulus G0 (elastic modulus G’ at high frequencies) because the plateau modulus G0 hinges upon the concentration of rod-like micelles or upon the number density of the elastic effective chains after forming networks (Rehage and Hoffmann, 1988). The entanglement also leads to an increase in the number of nodes, which impedes the reptation and extends the relaxation time τR, meanwhile, decreases ωc.
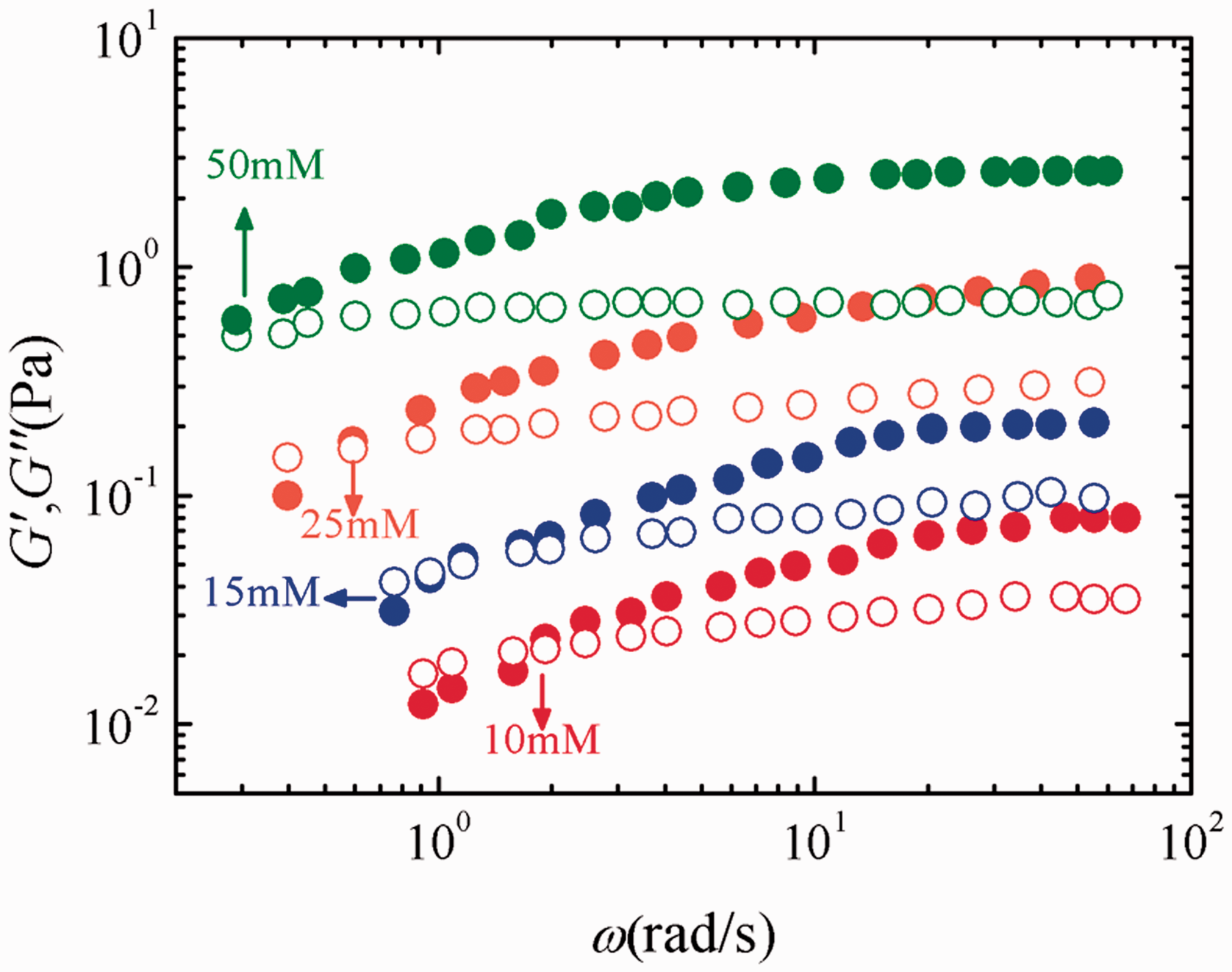
Storage modulus (G′, solid circles) and loss modulus (G″, empty circles) versus cyclic frequency (ω) for different EHSB concentrations at 65°C.
Under low shear frequencies, the dynamic rheology of the HDPS/SDS sample could be expressed by Maxwell’s equations (Fan et al., 2014)


The typical feature of the given model is that under the low shear frequencies (ω < ωc) the data fall onto a couple of loss modulus and storage modulus curves with gradients of 1 and 2, respectively. An additional noticeable characteristic is that the loss modulus has a maximal value at ωc (Fan et al., 2014). Depending on the characteristics, we could suppose with ease that the dynamic properties of EHSB does not conform to the Maxwell’s model (i.e., wormlike micellar solutions of EHSB are non-Maxwellian fluids), which is highly distinct from usual wormlike micelles assembled by shorter tailed surfactants (Cates and Candau, 1990; Candau and Oda, 2001).
Granek and Cates (1992) proposed a model of stress relaxation for wormlike micellar systems. Two dynamic regions are considered according to the characteristic relaxation time for reptation, τrep, and the reversible breaking time, τb. τrep is the time that a worm with an average length diffuses along the tube formed by tangle with other worms. τb is the average time for worms with average length to break into two parts. The Maxwellian answer of wormlike micelles takes place in the fast-breaking limit, in which τrep≫τb. The phenomenon is typical for most wormlike micelles assembled by shorter-tailed surfactants (Candau and Oda, 2001), and thus, they are classic Maxwellian fluid (Kumar et al., 2007). Yet the circumstance is quite different for long-tailed zwitterionic EHSB. Newly, Raghavan and Kaler (2001) stated that long carbon chains lead to quite prolonged breaking time, which ascribed to the low solubility (high TK) and cmc of long-tailed surfactants, which also remarkably slacken the exchange of monomers from one wormlike micelle to another in water solution. Thus, the presumption of τrep≫τb isn’t valid for long-tailed surfactant solutions, or even τrep<τb is the reality. Hence, EHSB wormlike micelles are “unbreakable” and present as a non-Maxwellian fluid.
Interfacial tension
Reducing interfacial tension between displacement fluid and crude oil to ultra-low level (10−3mN/m) is advantageous to decrease the capillary force and also improve the microscopic oil displacement efficiency.
Figure 7 shows the interfacial tension between the oils and EHSB samples as a function of EHSB concentrations at 65°C. As can be seen, when EHSB concentration is below 1.8 mM, the interfacial tension decreases with the increasing EHSB concentration, and the ultra-low can be achieved. When the concentration of EHSB is above 1.8 mM, the interfacial tension increases progressively after reaching the minimal value. When EHSB concentration is above 4.5 mM, the IFT is higher than low interfacial tension (10−2mN/m). When HDPS/SDS concentration exceeds 1 mM, the IFT also decreases first and then increases gradually with the increase of HDPS/SDS concentration. But different from EHSB, HDPS/SDS isn’t able to obtain ultra-low interfacial tension. The IFT reaches the minimal value of 2.7 × 10−2mN/m at the concentration of 5 mM.
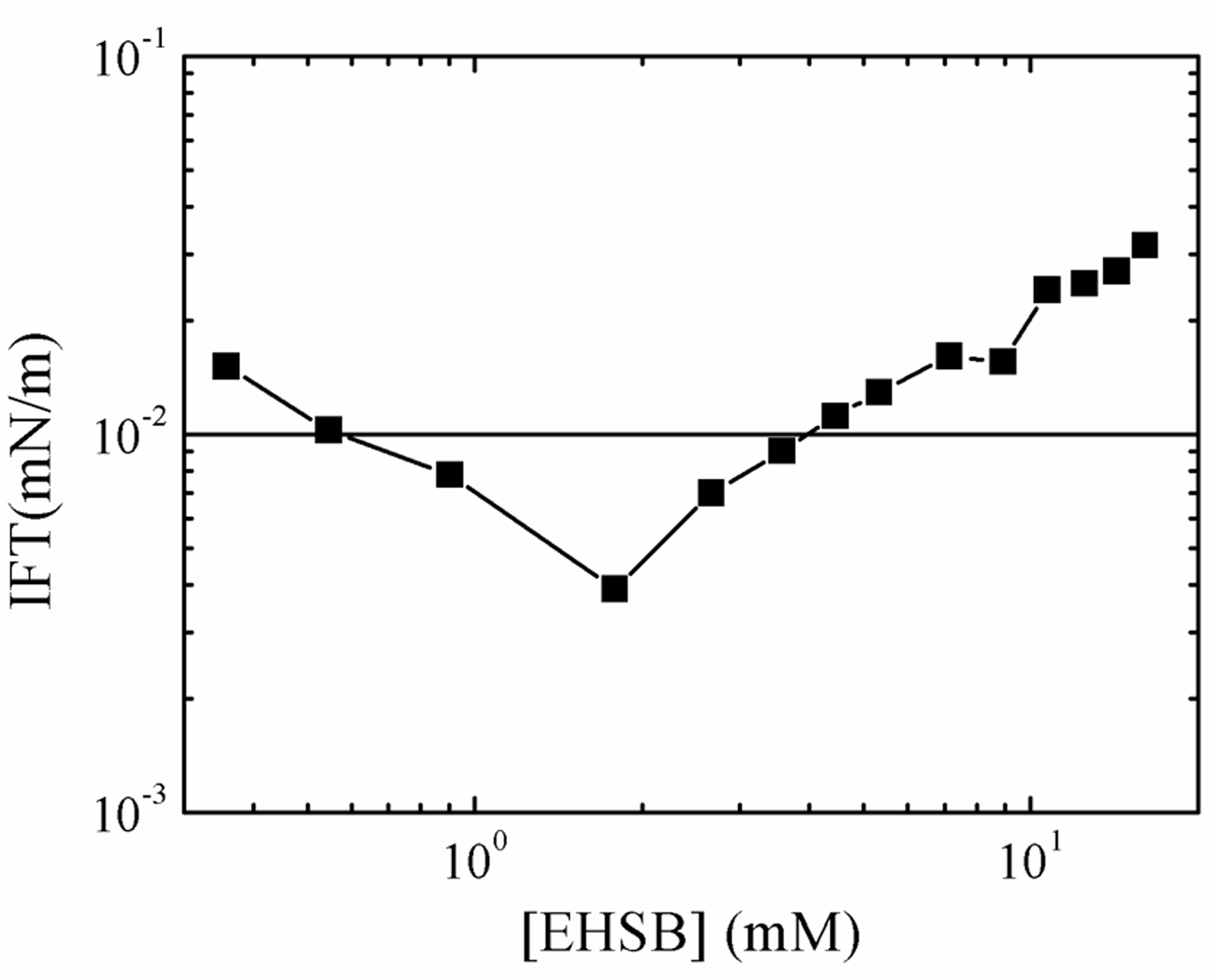
The interfacial tension between the oils and EHSB samples versus EHSB concentrations at 65°C.
The reduction of the IFT can be explained by the adsorption of surface-active substances in crude oil and the added surfactants on the oil-water interface (Aoudia et al., 2010; Tichelkamp et al., 2014; Hutin et al., 2014). To be exact, it involves the migration of surface-active substances from the oil phase and the addition of surfactants from the water phase to the oil-water interface. When EHSB concentration is low (e.g. 1 mM), it suggests that the interfacial film formed on the oil/water interface composing of surface-active substances and EHSB is close and strong enough to achieve ultra-low interfacial tension. When EHSB concentration increased beyond a certain level, the number density of wormlike micelles in the aqueous phase grows with the increasing concentration, which speeds up the desorption of crude oil surface-active compounds from oil-water interface into the aqueous phase. The desorption increases the interfacial tension.
For classic chemical-flooding systems, a high enough surfactant concentration is the precondition for the construction of wormlike micelles with heavy viscosity. However, the EHSB concentration is supposed to be over 10 mM to maintain the viscosity higher than 20mPa·s at 7.34 s−1. At the same time the oil-water interfacial tension is 2.18 × 10−2 mN/m, which is above 10−2 mN/m but below 10−1 mN/m. According to the results of oil-water interfacial tension experiments, the wormlike micelles assembled by EHSB can significantly decrease the oil-water interfacial tension.
Oil displacement efficiency
According to the results of rheology and interfacial tension, 10 mM EHSB solution is selected to estimate the oil displacement efficiency.
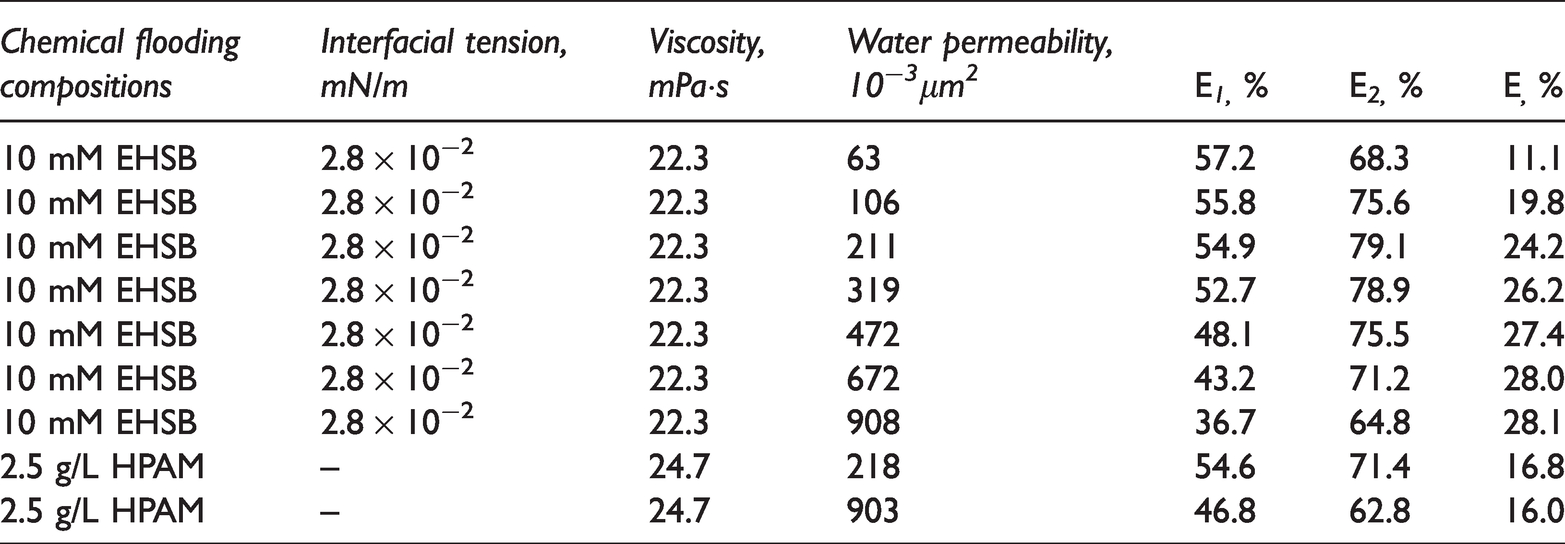
Figure 8 shows the increasing oil recovery with injection pore volume multiple at 65°C when the water permeability of the core is 211 × 10−3µm2. As can be seen, the water flooding recovery reaches up to 54.9% after injection of about 2 VP. The oil recovery increases only 4% after injection of 0.5 VP viscoelastic surfactant solution, however, which significantly increases and the ultimate recovery reaches 79.1% after the subsequent water flooding. The incremental recovery of viscoelastic surfactant flooding is up to 24.2%. When the water permeability of core is 211 × 10−3µm2, the incremental recovery of 20 mM HDPS/SDS under 10 g/L salinity is 23.1%.
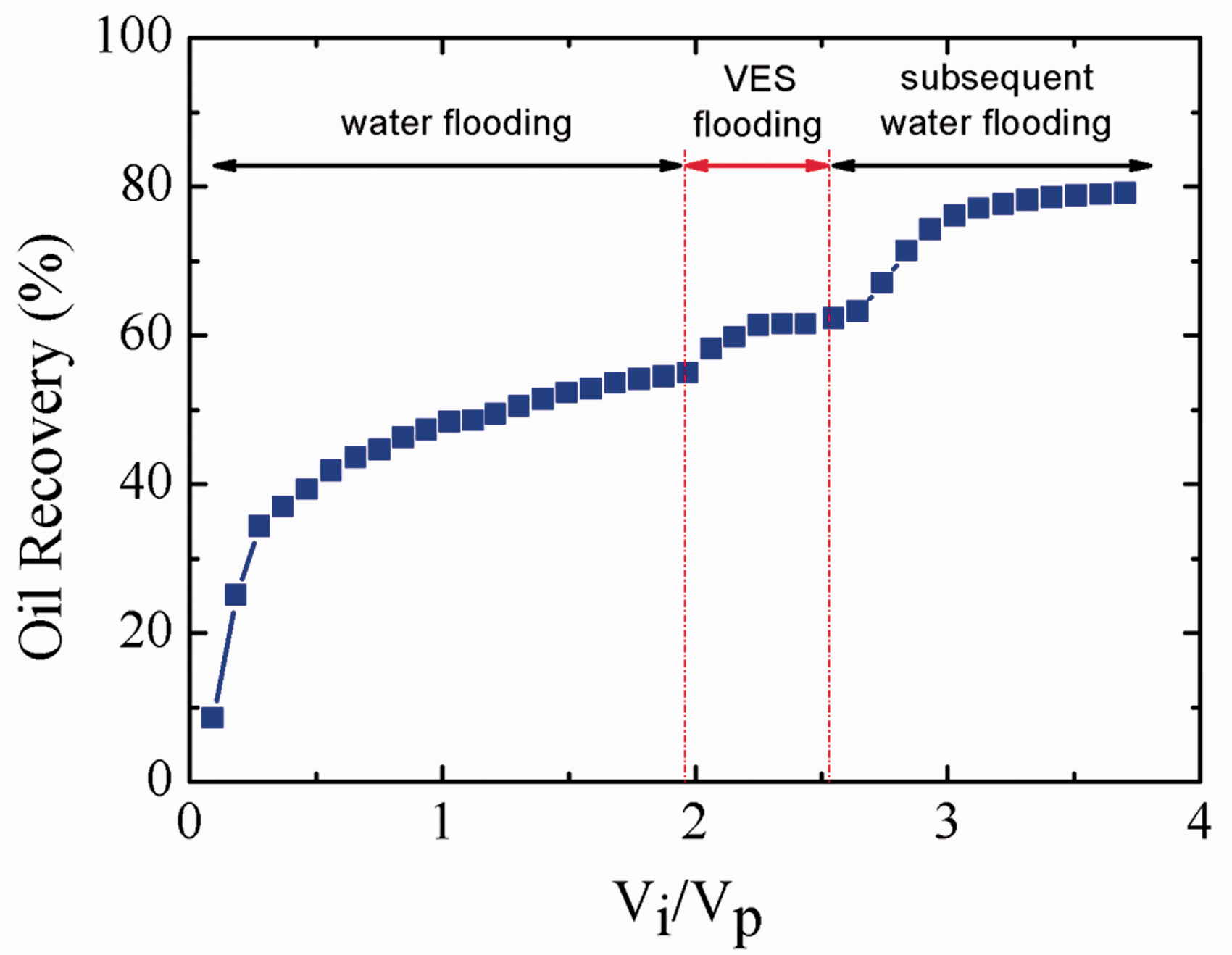
Oil recovery of 10 mM EHSB flooding versus injection pore volume multiple when the water permeability of the core is 211 × 10−3 µm2.
Figure 9 shows the incremental recovery of 10 mM EHSB flooding in the cores with different water permeability. E increases rapidly as permeability increases, and the growth rate slows down when the permeability exceeds 300 × 10−3µm2. The pore radius of the core enlarges with the increasing permeability, thus the capillary force that blocks the flow of crude oil reduces on the basis of Laplace’s formula so that more residual oil will be driven out. The EOR of various chemical-flood systems is shown in Table 2. The EOR by viscoelastic surfactant flooding is chiefly owing to the excellent capability of mobility control and reducing oil-water interfacial tension. Compared with shorter chain surfactant mixture HDPS/SDS, the ultra-long tailed EHSB has stronger viscoelasticity, salt-tolerance and interfacial active property, so as to improve the sweep efficiency and oil washing efficiency more effectively. Therefore, EHSB is better than HDPS/SDS in EOR.
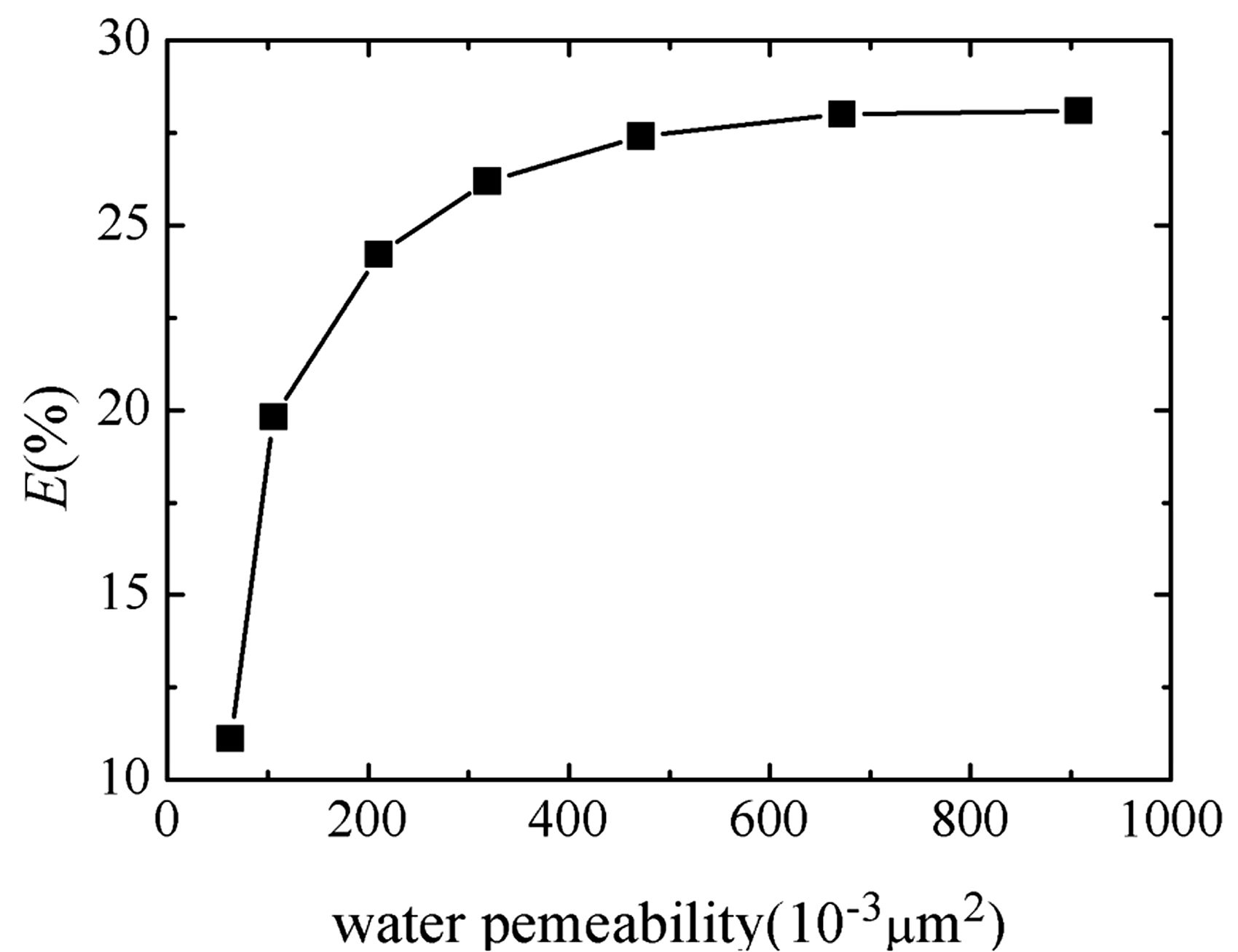
Incremental recovery of 10 mM EHSB flooding versus different water permeability of cores.
Oil displacement efficiency of various chemical-flood systems.
Conclusions
Chemical flooding plays an important role in tapping the potential of the remaining oil. However, high salinity significantly worsens the oil displacement performance of traditional EOR flooding agents. The wormlike micelles formed by zwitterionic surfactant EHSB with an ultra-long carbon chain are stable enough and exhibit excellent properties under high salt conditions. In this paper, we investigate the formation of viscoelastic wormlike micelles with C22-tailed amidosulfobetaine surfactant EHSB under the salinity of 40 g/L. The phase behavior, rheology and interfacial properties are studied systematically, and the worms are applied to oil displacement for the first time. We compare the properties with those of C16-tailed counterpart HDPS for the first time. The main conclusions of this study are as follows;
Due to the high Krafft temperature, the ultra long-tailed surfactant EHSB presents low solubility in distilled water, even in boiling water, but it can dissolve rapidly under high salinity. Because inorganic salts can significantly reduce its Krafft temperature and improve its solubility. The formation process of EHSB is as follows: With the increase of EHSB concentration, the rigid rod-like micelles first form in the solution, the steady rheological properties of which show the charateristic of Newtonian fluid. When the concentration continues to increase, the rod-like micelles are transformed into wormlike micelles, the steady rheological properties of which show Newtonian-shear-thinning charateristic. Then the wormlike micelles entangle with each other to form networks, which can increase the viscosity by several orders of magnitude. Under high salt conditions, the branching structure exists. The dynamic rheological properties show that EHSB wormlike micelles are “unbreakable” non-Maxwellian fluid. EHSB wormlike micelles have a wide ultra-low interfacial tension window and strong anti-dilution property, which can significantly reduce the oil-water interfacial tension after injection into the reservoir. Possessing dual functions of both mobility control and reducing oil-water interfacial tension, wormlike micelles formed by EHSB present a better displacement effect as a flooding system, the enhanced oil recovery is more than 10% higher than that of HPAM with the same viscosity. In comparison with HDPS/SDS wormlike micelles, EHSB worms are less sensitive to inorganic salt and exhibit better viscosification property as well as better ability to reduce the oil/water interfacial tension. These advantages of EHSB worms will enable them to be used in harsh environments, such as tertiary oil recovery in high-salinity oil reservoirs.
Footnotes
Acknowledgements
The authors would like to thank all editors and anonymous reviewers for their comments and suggestions.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed the following financial support for the research, authorship, and publication of this article: This research was funded by National Science and Technology Major Projects of China, grant number 2016ZX05031-002 and Joint Research Agreement between UC and RIPED, agreement number 19HT00001428.
